Myeloid Cell Markers Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Myeloid Cell Markers Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Myeloid Cell Markers Market Forecast and Outlook 2025 to 2035
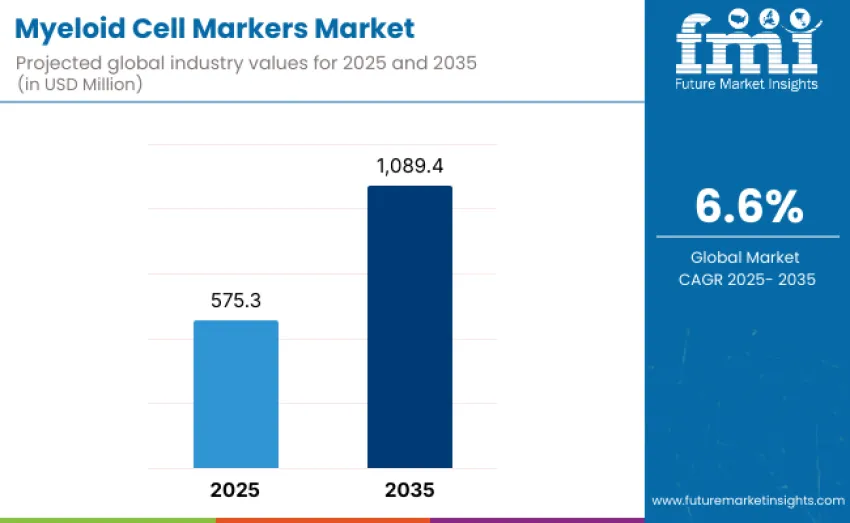
The global myeloid cell markers market is projected to reach USD 1,089.4 million by 2035, recording an absolute increase of USD 514.1 million over the forecast period. This market is valued at USD 575.3 million in 2025 and is projected to grow at a CAGR of 6.6% over the assessment period. Growth is expected to be supported by rising adoption of high-parameter immunophenotyping tools across immuno-oncology, infectious disease monitoring, and translational research programs, where detailed profiling of monocytes, macrophages, dendritic cells, and granulocyte subsets has become essential for characterizing immune system dynamics.
Technological advancements in cytometry instrumentation, fluorochrome design, and antibody validation protocols are reshaping the competitive landscape. Spectral flow cytometry systems are incorporating full-spectrum detectors and refined optical layouts that reduce signal spillover, supporting more complex myeloid marker combinations without compromising panel performance. Integration with advanced analytics platforms and bioinformatics workflows enables automated gating, high-dimensional clustering, and functional signature identification, ensuring that myeloid marker datasets can be translated more efficiently into actionable biological insights.
Quick Stats for Myeloid Cell Markers Market
- Myeloid Cell Markers Market Value (2025): USD 575.3 million
- Myeloid Cell Markers Market Forecast Value (2035): USD 1,089.4 million
- Myeloid Cell Markers Market Forecast CAGR: 6.6%
- Leading Marker Class in Myeloid Cell Markers Market: CD Antigens (Core Myeloid Markers) with 34.2% share
- Key Growth Regions in Myeloid Cell Markers Market: Asia Pacific, Europe, and North America
- Top Players in Myeloid Cell Markers Market: BD Biosciences, Beckman Coulter (Danaher), BioLegend, MiltenyiBiotec, Thermo Fisher Scientific, Bio-Rad Laboratories, R&D Systems/Bio-Techne, Abcam, Standard BioTools (Fluidigm), Others
Government research investments, large-scale immunology initiatives, and strengthening of translational infrastructure within academic medical centers are expected to accelerate expansion. Training programs in cytometry, immuno-oncology, and computational immunology are being incorporated into university and biotechnology curricula, increasing the number of skilled researchers able to operate advanced cytometry systems and standardized myeloid marker panels. Biopharmaceutical companies are prioritizing immune monitoring capabilities for early-phase trials, driving consistent demand for high-quality myeloid antibodies and conjugates.
Myeloid Cell Markers Market Year-over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the myeloid cell markers market is projected to expand from USD 575.3 million to USD 791.7 million, resulting in a value increase of USD 216.4 million, which represents 42.1% of total forecast growth for the decade. Growth during this period is expected to be driven by rising immunophenotyping demand in hematological malignancies, wider adoption of high-parameter flow cytometry systems, and increasing use of standardized myeloid marker panels in clinical diagnostics and translational research.
From 2030 to 2035, growth is forecast from USD 791.7 million to USD 1,089.4 million, adding USD 297.7 million, which constitutes 57.9% of overall ten-year expansion. This phase will be shaped by increasing clinical integration of multi-omic immune profiling, rising adoption of fully automated flow cytometry platforms, and broader use of advanced myeloid marker panels in immuno-oncology, vaccine development, and chronic inflammatory disease research. Continued standardization of diagnostic criteria for myelodysplastic syndromes, acute myeloid leukemia, and sepsis-related immune dysregulation will elevate demand.
Myeloid Cell Markers Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 575.3 million |
| Market Forecast Value (2035) | USD 1,089.4 million |
| Forecast CAGR (2025 to 2035) | 6.6% |
Why Is the Myeloid Cell Markers Market Growing Worldwide?
The myeloid cell markers market is expanding globally due to rising demand for high-precision immunophenotyping in oncology, immunology, and infectious disease research. Growth is supported by the increasing global burden of hematologic cancers such as acute myeloid leukemia, myelodysplastic syndromes, and chronic myelomonocyticleukemia, where accurate characterization of monocytes, granulocytes, dendritic cells, and myeloid-derived suppressor cells is essential for diagnosis, prognosis, and therapeutic decision-making. This clinical need drives routine use of multi-color flow cytometry panels incorporating CD11b, CD14, CD15, CD16, CD33, CD34, CD64, HLA-DR.
Biopharmaceutical companies are accelerating adoption as myeloid cell populations increasingly become central targets and biomarkers in immuno-oncology, particularly for therapies involving tumor-associated macrophages, dendritic cell vaccines, checkpoint inhibitors, and myeloid-modulating agents. Large-scale translational studies and clinical trials require standardized, reproducible myeloid marker panels that ensure data comparability across sites, thereby boosting demand for validated antibodies, companion kits, and quality-control reagents. Government and institutional investments in precision medicine, cancer immunology programs, and infectious disease surveillance reinforce expansion.
How Is the Myeloid Cell Markers Market Segmented by Marker Class, Technology, and Region?
The myeloid cell markers market is segmented by marker class, technology and region. Based on marker class, categorization includes CD antigens (core myeloid markers), checkpoint and activation markers, lineage and differentiation panels, MDSC panels, and other immune-modulatory markers. By technology, division covers flow cytometry antibodies, IHC/IF antibodies, mass cytometry (CyTOF) panels, multiplex immuno-oncology panels, and genomic/proteomic panels. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Marker Class, CD Antigens Account for Largest Share
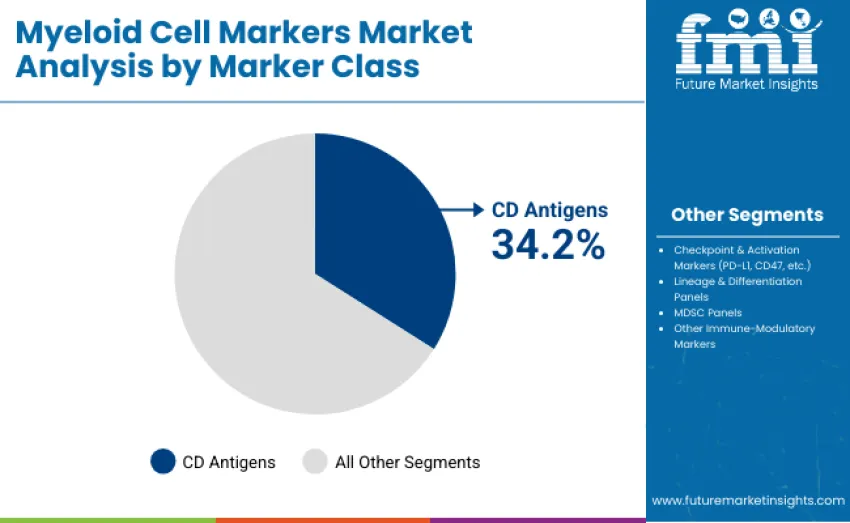
The CD antigens segment represents the dominant class, capturing 34.2% of total revenue in 2025. This category includes core myeloid phenotyping markers such as CD11b, CD14, CD15, CD33, and CD68, which are essential for baseline identification, maturation profiling, and functional assessment of myeloid populations. Leadership is reinforced by their foundational role in hematology workflows, tumor microenvironment analysis, and standardized immune profiling protocols used across both clinical and research settings.
Checkpoint and activation markers, including PD-L1, CD47, and TIM-3, hold a substantial 21.7% share, driven by their expanding relevance in immuno-oncology and therapeutic response monitoring. Lineage and differentiation panels account for 19.8%, supporting studies on myeloid development, plasticity, and disease-associated phenotypes. Myeloid-derived suppressor cell panels represent 13.6%, reflecting growing interest in tumor immune evasion and therapeutic resistance mechanisms. Other immune-modulatory markers contribute 10.7%, addressing highly specialized mechanistic and translational research needs.
Key advantages driving the CD antigens segment include:
- Central utility in routine myeloid identification enabling highly reproducible phenotyping across laboratories
- Broad compatibility with clinical diagnostics, translational oncology, and inflammatory disease research
- Ability to anchor high-parameter immunophenotyping workflows by providing stable lineage-defining markers
By Technology, Flow Cytometry Antibodies Hold Largest Share
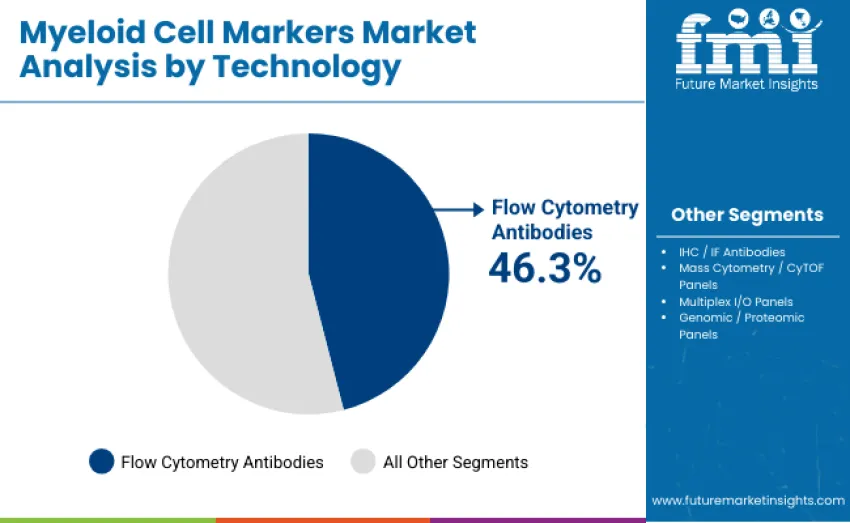
Flow cytometry antibodies dominate the technology landscape with 46.3% revenue share in 2025, driven by their central role in high-parameter immune profiling, rapid single-cell resolution, and widespread use in both clinical and research laboratories. Their scalability, multiplexing capacity, and compatibility with automated cytometry platforms reinforce leadership. IHC/IF antibodies account for 23.4%, supporting tissue-level localization and pathology-based immune profiling.
Multiplex immuno-oncology panels hold 12.7% share, reflecting expanding use in tumor microenvironment characterization. Mass cytometry/CyTOF panels contribute 9.8% as researchers adopt ultra-high-parameter analysis for deep myeloid profiling. Genomic and proteomic panels represent 7.8%, driven by emerging multi-omics workflows linking myeloid phenotype to transcriptional and proteomic signatures.
Key factors supporting technology preferences include:
- Strong demand for high-parameter flow cytometry enabling detailed myeloid subset and functional analyses
- Tissue-based IHC/IF assays providing spatial context for myeloid infiltration and activation states
- Growth in multiplex I/O and CyTOF applications supporting deep immune profiling in oncology research
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Myeloid Cell Markers Market?
The myeloid cell markers market is driven by several key factors shaping demand within the immunology and clinical research landscape. Increasing prevalence of cancer, autoimmune disorders, and infectious diseases creates growing demand for precise immunophenotyping and myeloid cell profiling solutions, with global incidence rates rising by 5-7% annually, which necessitates reliable biomarkers for diagnostic, prognostic, and therapeutic monitoring applications. Expansion of translational and clinical research programs accelerates adoption of myeloid cell markers, as 40-55% of research institutions integrate advanced flow cytometry, mass cytometry, and multiplex panels into workflows.
What Are the Major Restraints Limiting Adoption in the Myeloid Cell Markers Market?
Restraints include high reagent and instrumentation costs, which can limit adoption in smaller laboratories and emerging regions, as complete myeloid marker panels combined with flow cytometers or CyTOF instruments represent significant capital expenditure relative to standard immunoassays. Technical complexity and the need for specialized training create adoption barriers, as effective assay design, panel optimization, and data analysis require significant expertise that can take months to acquire.
What Emerging Trends Are Shaping the Myeloid Cell Markers Market?
Key trends include accelerated adoption in Asia-Pacific countries, particularly China and India, driven by government initiatives supporting biomedical research, investment in high-end laboratory infrastructure, and training programs for advanced immunology techniques. Technological innovation shapes development, with advancements in high-dimensional flow cytometry, multiplexed immunoassays, and mass cytometry panels.
How Are Leading Countries Driving Growth in the Myeloid Cell Markers Market?
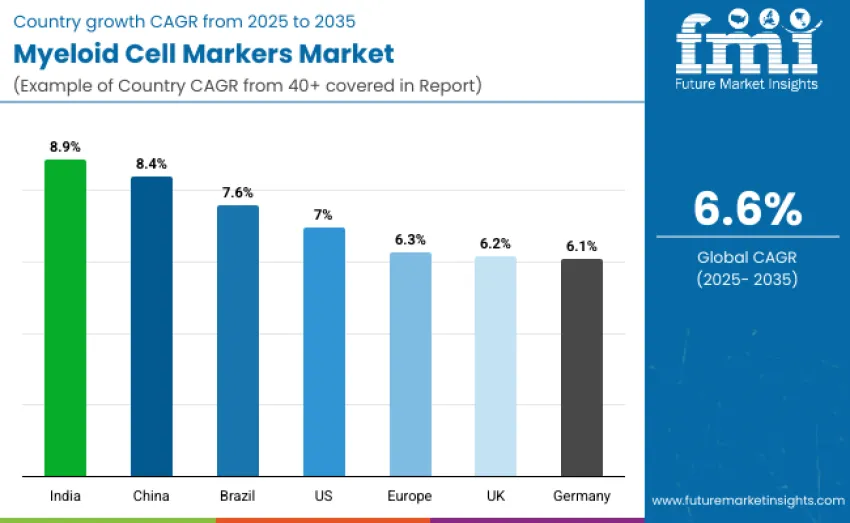
| Country/Region | CAGR (2025 to 2035)% |
|---|---|
| United States | 7.0 |
| Brazil | 7.6 |
| China | 8.4 |
| India | 8.9 |
| Europe | 6.3 |
| Germany | 6.1 |
| United Kingdom | 6.2 |
The myeloid cell markers market is expanding globally, with India leading at an 8.9% CAGR driven by government biomedical research initiatives and growing clinical and academic laboratory capacity. China follows at 8.4%, supported by rapid adoption of advanced assays and biotechnology expansion. Brazil grows at 7.6% through strengthening research ecosystems and rising immune-monitoring demand. The USA advances steadily at 7.0%, anchored by mature laboratory infrastructure and established panel usage. Europe grows at 6.3%, with Germany at 6.1% and the UK at 6.2%, supported by strong clinical research and healthcare systems.
How Are Policy Initiatives and Hospital Automation Shaping Growth in China?
China's myeloid cell markers market is projected to grow at a CAGR of 8.4% from 2025 to 2035, driven by widespread implementation in premier hospitals and specialized research centers in Beijing, Shanghai, and Guangzhou. Panels are utilized for hematologic malignancy diagnostics, immune cell profiling, and personalized treatment stratification. Expansion is propelled by government-backed biotechnology programs, precision medicine policies, and oncology research funding. Domestic manufacturers produce automated high-throughput panels and reagents, enabling rapid sample processing and high-volume testing.
Urban laboratory modernization projects, coupled with digital reporting systems, support efficient workflow management. Nationwide screening campaigns for leukemia and immune disorders encourage adoption. Training programs tailored to laboratory personnel ensure proper execution and data reliability. Integration of innovative analytical instruments enhances assay precision and reproducibility, allowing Chinese hospitals to maintain competitive standards in clinical diagnostics. Government-backed programs provide structured guidance, subsidies, and infrastructure support for biotechnology and precision medicine initiatives.
How Are Regional Laboratory Networks and Local Production Driving Expansion in India?
India's myeloid cell markers market is projected to grow at a CAGR of 8.9% from 2025 to 2035, supported by diagnostic laboratories, cancer centers, and academic institutions in Bengaluru, Hyderabad, and Mumbai. Panels are applied for leukemia classification, immune system monitoring, and clinical trial biomarker validation. Local reagent and panel manufacturing reduces procurement costs and strengthens regional supply chains, enhancing accessibility for semi-urban laboratories. Expansion is enabled by government programs aimed at increasing healthcare coverage, biotechnology research, and diagnostic capacity.
Investments in laboratory instrumentation and automated cytometry platforms accelerate testing throughput. National early detection initiatives and hematologic surveillance programs stimulate wider panel deployment. Technical skill development programs ensure consistent assay execution and reliability. Focus on affordable, scalable solutions allows broader reach into smaller hospitals and emerging healthcare facilities. Regional laboratories implement panels for leukemia classification and immune monitoring across multiple metropolitan centers throughout the country.
How Are Regulatory Standards and Sustainable Laboratory Practices Supporting Growth in Germany?
Germany's myeloid cell markers market is projected to grow at a CAGR of 6.1% from 2025 to 2035, concentrated in university hospitals and specialized diagnostic centers in Berlin, Munich, and Frankfurt. Panels are deployed for hematologic malignancy profiling, immune system characterization, and translational clinical studies. Stringent IVDR regulations ensure assay precision, reproducibility, and diagnostic credibility. Adoption is reinforced by automated cytometry instrumentation integrated with laboratory information management systems for real-time sample tracking and data integrity.
Investments in energy-efficient laboratory infrastructure, environmentally friendly equipment, and standardized assay protocols promote sustainable operations. Multi-parametric immunophenotyping technologies enable detailed cellular analysis for research and clinical purposes. Focused integration of genomics and proteomics datasets with myeloid markers allows advanced precision medicine applications. Growth is underpinned by commitment to compliance, operational efficiency, and scientific rigor. University hospitals and specialized diagnostic centers adopt panels for hematologic profiling and translational studies.
How Are Cost-Effective Solutions and Regional Networks Fueling Expansion in Brazil?
Brazil's myeloid cell markers market is projected to grow at a CAGR of 7.6% from 2025 to 2035, supported by adoption in metropolitan and regional laboratories in São Paulo, Rio de Janeiro, Brasília, and Porto Alegre. Panels are utilized for leukemia diagnostics, immune monitoring, and clinical trial biomarker evaluation. Local biotechnology companies supply affordable reagents and panels, enabling widespread access in resource-constrained facilities. Development of regional laboratory networks improves sample distribution, operational efficiency, and testing capacity.
Public health programs targeting hematologic disorders, coupled with oncology research funding, foster early detection initiatives. Investment in mid-tier laboratory instrumentation and workflow optimization facilitates high-throughput panel processing. Focus on cost-efficiency allows smaller hospitals and clinics to implement advanced diagnostic solutions without substantial financial burden. Metropolitan and regional laboratories use panels for leukemia diagnostics and immune monitoring in clinical settings.
How Are Advanced Automation Platforms and Federal Research Funding Driving Growth in United States?
The USA myeloid cell markers market is projected to grow at a CAGR of 7.0% from 2025 to 2035, driven by implementation in academic hospitals, oncology centers, and clinical research laboratories in Boston, New York, and San Francisco. Panels are applied for immunophenotyping, leukemia classification, and immune function monitoring in clinical trials. Adoption is strengthened by sophisticated automated cytometry platforms and pre-validated reagents that improve workflow speed and reproducibility.
Federal grants and research funding for immuno-oncology, hematology, and translational studies accelerate panel deployment. Investments in high-performance instrumentation and real-time analytics enhance assay precision and consistency. Early detection programs in both urban and suburban healthcare systems increase overall panel utilization. Integration of laboratory analytics platforms with clinical decision support systems provides actionable insights. Academic hospitals, oncology centers, and research labs implement panels for immunophenotyping and leukemia classification.
How Are NHS Networks and Digital Infrastructure Driving Expansion in United Kingdom?
The UK myeloid cell markers market is projected to grow at a CAGR of 6.2% from 2025 to 2035, centered on NHS hospitals, academic centers, and specialized laboratories in London, Manchester, Bristol, and Edinburgh. Panels are applied for leukemia diagnostics, immune system characterization, and translational research. Standardized digital workflows integrated with laboratory information management systems ensure operational consistency, reduce errors, and facilitate data sharing across regional centers.
Government-funded precision medicine initiatives, clinical trial support programs, and national hematologic surveillance schemes reinforce adoption. Investments in modern cytometry equipment, software analytics, and staff competency development increase assay reliability and throughput. Emphasis on scalable laboratory practices enables smaller hospitals to access advanced immunophenotyping panels efficiently. NHS hospitals, academic centers, and specialized labs implement panels for leukemia diagnostics and immune profiling.
How Are Healthcare Infrastructure Investments Advancing Expansion Across Europe?
The myeloid cell markers market in Europe is projected to grow from USD 165.1 million in 2025 to USD 299.6 million by 2035, registering a CAGR of 6.3% over the forecast period. Germany is expected to lead the region with a 27.4% share in 2025, slightly declining to 27% by 2035, supported by advanced clinical research networks and key immunology and hematologycenters in Berlin, Munich, and Hamburg.
France follows with a 21% share in 2025, reducing marginally to 20.7% by 2035, driven by expanding translational medicine programs and adoption of flow cytometry and multiplex myeloid panels in Paris and Lyon. Italy holds a 15.5% share in 2025, remaining stable at 15.4% by 2035, underpinned by strong oncology and hematology research facilities. Spain accounts for 12.1% in 2025, increasing slightly to 12.4% by 2035, supported by growing laboratory adoption of immune profiling panels. BENELUX maintains a 9% share in 2025, rising to 9.2% by 2035.
How Competitive Is the Global Myeloid Cell Markers Market and What Defines Its Structure?
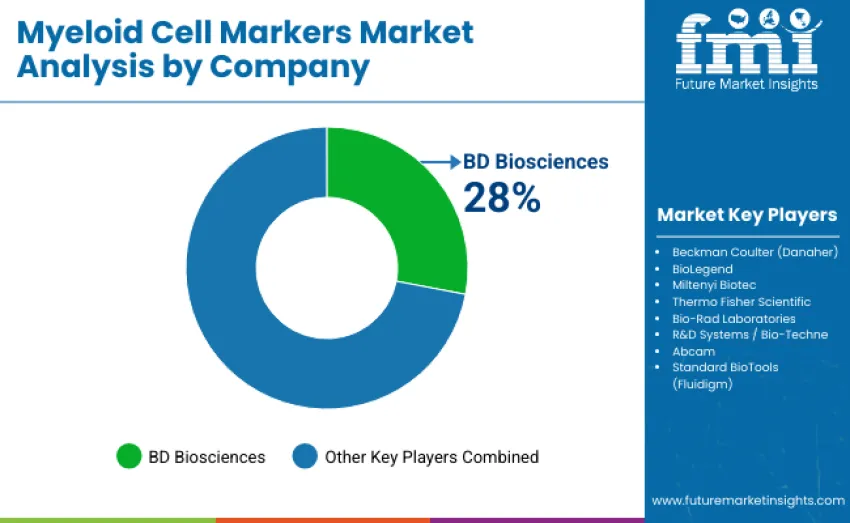
The myeloid cell markers market is moderately concentrated, with 10–12 players contributing to global competition. The top three companies collectively control an estimated 55–60% of the global share, driven by established flow cytometry panels, validated antibody sets, and widespread cytometer installed bases. BD Biosciences leads with a 28% share, supported by its extensive CD11b, CD14, CD33, and HLA-DR marker panels and a large global cytometer network. Competition is defined by panel breadth, reagent quality, assay reproducibility, and seamless workflow integration rather than price competition alone.
Key leaders, BD Biosciences, Beckman Coulter (Danaher), and BioLegend, maintain strong positions through proprietary antibody clones, validated fluorochrome combinations, high reagent consistency, and strong adoption across research and clinical environments. These companies invest heavily in high-quality reagent manufacturing, dry panel formats, and integrated cytometry workflows, expanding their influence in immune-oncology, translational research, and high-throughput immunophenotyping.
Challengers such as MiltenyiBiotec and Thermo Fisher Scientific compete through complete myeloid research workflows that integrate cell separation technologies with reagent–instrument pairing, enabling standardized sample processing and improved reproducibility. Product specialists, including Bio-Rad Laboratories, R&D Systems/Bio-Techne, Abcam, and Standard BioTools, differentiate through high-parameter CyTOF panels, novel fluorophores, recombinant proteins, and validated antibodies tailored for advanced immunology research.
Key Players in the Myeloid Cell Markers Market
- BD Biosciences
- Beckman Coulter (Danaher)
- BioLegend
- MiltenyiBiotec
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- R&D Systems / Bio-Techne
- Abcam
- Standard BioTools (Fluidigm)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 575.3 million |
| Marker Class | CD Antigens (Core Myeloid Markers), Checkpoint & Activation Markers (PD-L1, CD47, etc.), Lineage & Differentiation Panels, MDSC Panels, Other Immune-Modulatory Markers |
| Technology | Flow Cytometry Antibodies, IHC/IF Antibodies, Mass Cytometry/ CyTOF Panels, Multiplex I/O Panels, Genomic/Proteomic Panels |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | BD Biosciences, Beckman Coulter (Danaher), BioLegend , Miltenyi Biotec , Thermo Fisher Scientific, Bio-Rad Laboratories, R&D Systems/Bio- Techne , Abcam , Standard BioTools ( Fluidigm ), Others |
| Additional Attributes | Dollar sales by marker class and technology, regional trends across Asia Pacific, Europe, and North America, competitive landscape of assay providers, technical specifications, integration with cytometry and immunoassay workflows, innovations in antibody panels, detection sensitivity, reproducibility, and development of specialized panels for enhanced lineage coverage and quantitative accuracy |
Myeloid Cell Markers Market by Segments
By Marker Class:
- CD Antigens (Core Myeloid Markers)
- Checkpoint & Activation Markers (PD-L1, CD47, etc.)
- Lineage & Differentiation Panels
- MDSC Panels
- Other Immune-Modulatory Markers
By Technology:
- Flow Cytometry Antibodies
- IHC / IF Antibodies
- Mass Cytometry / CyTOF Panels
- Multiplex I/O Panels
- Genomic / Proteomic Panels
By Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the myeloid cell markers market in 2025?
The global myeloid cell markers market is valued at USD 575.3 million in 2025.
What will be the size of the myeloid cell markers market in 2035?
The market is projected to reach USD 1,089.4 million by 2035.
How fast will the myeloid cell markers market grow between 2025 and 2035?
The market will grow at a CAGR of 6.6% from 2025 to 2035.
Which marker class leads the myeloid cell markers market in 2025?
CD antigens lead the market with a 34.2% share in 2025.
Who are the major players in the myeloid cell markers market?
Key players include BD Biosciences, Beckman Coulter, BioLegend, Miltenyi Biotec, Thermo Fisher Scientific, Bio-Rad, R&D Systems/Bio-Techne, Abcam, and Standard BioTools.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Technological Advancement
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Market Dynamics
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size USD Million Analysis, 2020 to 2024
- Current and Future Market Size USD Million Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Marker Class
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Marker Class, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Marker Class, 2025 to 2035
- CD Antigens (Core Myeloid Markers)
- Checkpoint & Activation Markers (PD-L1, CD47, etc.)
- Lineage & Differentiation Panels
- MDSC Panels
- Other Immune-Modulatory Markers
- Y-o-Y Growth Trend Analysis By Marker Class, 2020 to 2024
- Absolute $ Opportunity Analysis By Marker Class, 2025 to 2035
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Technology
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Technology, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Technology, 2025 to 2035
- Flow Cytometry Antibodies
- IHC / IF Antibodies
- Mass Cytometry / CyTOF Panels
- Multiplex I/O Panels
- Genomic / Proteomic Panels
- Y-o-Y Growth Trend Analysis By Technology, 2020 to 2024
- Absolute $ Opportunity Analysis By Technology, 2025 to 2035
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Application, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Application, 2025 to 2035
- Oncology & Immuno-Oncology
- Hematology & Transplant
- Inflammation & Autoimmune
- Infectious Disease & Sepsis
- Translational Research Tools
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By End User, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By End User, 2025 to 2035
- Pharma & Biotech
- CROs & Central Labs
- Academic & Translational Centers
- Hospital / Clinical Labs
- Others
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Regional Level Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size USD Million Analysis By Region, 2020 to 2024
- Current Market Size USD Million Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- Latin America Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- East Asia Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- South Asia Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- Western Europe Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- Eastern Europe Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Markers Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Marker Class
- By Technology
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Marker Class
- By Technology
- By Application
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Marker Class
- By Technology
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- BD Biosciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Beckman Coulter (Danaher)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BioLegend
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Miltenyi Biotec
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bio-Rad Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- R&D Systems / Bio-Techne
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abcam
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Standard BioTools (Fluidigm)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BD Biosciences
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 2: Global Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 3: Global Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 4: Global Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 5: Global Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 6: Global Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 7: Global Markers Market Size (USD Million), by Technology, 2020-2035
- Table 8: Global Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 9: Global Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 10: Global Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 11: Global Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 12: Global Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 13: Global Markers Market Size (USD Million), by Application, 2020-2035
- Table 14: Global Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 15: Global Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 16: Global Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 17: Global Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 18: Global Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 19: Global Markers Market Size (USD Million), by End User, 2020-2035
- Table 20: Global Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 21: Global Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 22: Global Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 23: Global Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 24: Global Markers Market Size (USD Million), by Others, 2020-2035
- Table 25: North America Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 26: North America Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 27: North America Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 28: North America Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 29: North America Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 30: North America Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 31: North America Markers Market Size (USD Million), by Technology, 2020-2035
- Table 32: North America Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 33: North America Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 34: North America Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 35: North America Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 36: North America Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 37: North America Markers Market Size (USD Million), by Application, 2020-2035
- Table 38: North America Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 39: North America Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 40: North America Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 41: North America Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 42: North America Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 43: North America Markers Market Size (USD Million), by End User, 2020-2035
- Table 44: North America Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 45: North America Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 46: North America Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 47: North America Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 48: North America Markers Market Size (USD Million), by Others, 2020-2035
- Table 49: USA Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 50: USA Markers Market Size (USD Million), by Technology, 2020-2035
- Table 51: USA Markers Market Size (USD Million), by Application, 2020-2035
- Table 52: USA Markers Market Size (USD Million), by End User, 2020-2035
- Table 53: Canada Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 54: Canada Markers Market Size (USD Million), by Technology, 2020-2035
- Table 55: Canada Markers Market Size (USD Million), by Application, 2020-2035
- Table 56: Canada Markers Market Size (USD Million), by End User, 2020-2035
- Table 57: Mexico Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 58: Mexico Markers Market Size (USD Million), by Technology, 2020-2035
- Table 59: Mexico Markers Market Size (USD Million), by Application, 2020-2035
- Table 60: Mexico Markers Market Size (USD Million), by End User, 2020-2035
- Table 61: Latin America Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 62: Latin America Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 63: Latin America Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 64: Latin America Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 65: Latin America Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 66: Latin America Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 67: Latin America Markers Market Size (USD Million), by Technology, 2020-2035
- Table 68: Latin America Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 69: Latin America Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 70: Latin America Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 71: Latin America Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 72: Latin America Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 73: Latin America Markers Market Size (USD Million), by Application, 2020-2035
- Table 74: Latin America Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 75: Latin America Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 76: Latin America Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 77: Latin America Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 78: Latin America Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 79: Latin America Markers Market Size (USD Million), by End User, 2020-2035
- Table 80: Latin America Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 81: Latin America Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 82: Latin America Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 83: Latin America Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 84: Latin America Markers Market Size (USD Million), by Others, 2020-2035
- Table 85: Brazil Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 86: Brazil Markers Market Size (USD Million), by Technology, 2020-2035
- Table 87: Brazil Markers Market Size (USD Million), by Application, 2020-2035
- Table 88: Brazil Markers Market Size (USD Million), by End User, 2020-2035
- Table 89: Argentina Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 90: Argentina Markers Market Size (USD Million), by Technology, 2020-2035
- Table 91: Argentina Markers Market Size (USD Million), by Application, 2020-2035
- Table 92: Argentina Markers Market Size (USD Million), by End User, 2020-2035
- Table 93: Rest of Latin America Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 94: Rest of Latin America Markers Market Size (USD Million), by Technology, 2020-2035
- Table 95: Rest of Latin America Markers Market Size (USD Million), by Application, 2020-2035
- Table 96: Rest of Latin America Markers Market Size (USD Million), by End User, 2020-2035
- Table 97: East Asia Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 98: East Asia Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 99: East Asia Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 100: East Asia Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 101: East Asia Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 102: East Asia Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 103: East Asia Markers Market Size (USD Million), by Technology, 2020-2035
- Table 104: East Asia Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 105: East Asia Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 106: East Asia Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 107: East Asia Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 108: East Asia Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 109: East Asia Markers Market Size (USD Million), by Application, 2020-2035
- Table 110: East Asia Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 111: East Asia Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 112: East Asia Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 113: East Asia Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 114: East Asia Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 115: East Asia Markers Market Size (USD Million), by End User, 2020-2035
- Table 116: East Asia Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 117: East Asia Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 118: East Asia Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 119: East Asia Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 120: East Asia Markers Market Size (USD Million), by Others, 2020-2035
- Table 121: China Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 122: China Markers Market Size (USD Million), by Technology, 2020-2035
- Table 123: China Markers Market Size (USD Million), by Application, 2020-2035
- Table 124: China Markers Market Size (USD Million), by End User, 2020-2035
- Table 125: Japan Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 126: Japan Markers Market Size (USD Million), by Technology, 2020-2035
- Table 127: Japan Markers Market Size (USD Million), by Application, 2020-2035
- Table 128: Japan Markers Market Size (USD Million), by End User, 2020-2035
- Table 129: South Korea Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 130: South Korea Markers Market Size (USD Million), by Technology, 2020-2035
- Table 131: South Korea Markers Market Size (USD Million), by Application, 2020-2035
- Table 132: South Korea Markers Market Size (USD Million), by End User, 2020-2035
- Table 133: South Asia Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 134: South Asia Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 135: South Asia Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 136: South Asia Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 137: South Asia Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 138: South Asia Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 139: South Asia Markers Market Size (USD Million), by Technology, 2020-2035
- Table 140: South Asia Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 141: South Asia Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 142: South Asia Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 143: South Asia Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 144: South Asia Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 145: South Asia Markers Market Size (USD Million), by Application, 2020-2035
- Table 146: South Asia Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 147: South Asia Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 148: South Asia Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 149: South Asia Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 150: South Asia Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 151: South Asia Markers Market Size (USD Million), by End User, 2020-2035
- Table 152: South Asia Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 153: South Asia Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 154: South Asia Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 155: South Asia Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 156: South Asia Markers Market Size (USD Million), by Others, 2020-2035
- Table 157: India Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 158: India Markers Market Size (USD Million), by Technology, 2020-2035
- Table 159: India Markers Market Size (USD Million), by Application, 2020-2035
- Table 160: India Markers Market Size (USD Million), by End User, 2020-2035
- Table 161: ASEAN Countries Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 162: ASEAN Countries Markers Market Size (USD Million), by Technology, 2020-2035
- Table 163: ASEAN Countries Markers Market Size (USD Million), by Application, 2020-2035
- Table 164: ASEAN Countries Markers Market Size (USD Million), by End User, 2020-2035
- Table 165: Australia & New Zealand Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 166: Australia & New Zealand Markers Market Size (USD Million), by Technology, 2020-2035
- Table 167: Australia & New Zealand Markers Market Size (USD Million), by Application, 2020-2035
- Table 168: Australia & New Zealand Markers Market Size (USD Million), by End User, 2020-2035
- Table 169: Rest of South Asia Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 170: Rest of South Asia Markers Market Size (USD Million), by Technology, 2020-2035
- Table 171: Rest of South Asia Markers Market Size (USD Million), by Application, 2020-2035
- Table 172: Rest of South Asia Markers Market Size (USD Million), by End User, 2020-2035
- Table 173: Western Europe Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 174: Western Europe Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 175: Western Europe Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 176: Western Europe Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 177: Western Europe Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 178: Western Europe Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 179: Western Europe Markers Market Size (USD Million), by Technology, 2020-2035
- Table 180: Western Europe Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 181: Western Europe Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 182: Western Europe Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 183: Western Europe Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 184: Western Europe Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 185: Western Europe Markers Market Size (USD Million), by Application, 2020-2035
- Table 186: Western Europe Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 187: Western Europe Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 188: Western Europe Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 189: Western Europe Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 190: Western Europe Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 191: Western Europe Markers Market Size (USD Million), by End User, 2020-2035
- Table 192: Western Europe Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 193: Western Europe Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 194: Western Europe Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 195: Western Europe Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 196: Western Europe Markers Market Size (USD Million), by Others, 2020-2035
- Table 197: Germany Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 198: Germany Markers Market Size (USD Million), by Technology, 2020-2035
- Table 199: Germany Markers Market Size (USD Million), by Application, 2020-2035
- Table 200: Germany Markers Market Size (USD Million), by End User, 2020-2035
- Table 201: UK Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 202: UK Markers Market Size (USD Million), by Technology, 2020-2035
- Table 203: UK Markers Market Size (USD Million), by Application, 2020-2035
- Table 204: UK Markers Market Size (USD Million), by End User, 2020-2035
- Table 205: France Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 206: France Markers Market Size (USD Million), by Technology, 2020-2035
- Table 207: France Markers Market Size (USD Million), by Application, 2020-2035
- Table 208: France Markers Market Size (USD Million), by End User, 2020-2035
- Table 209: Italy Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 210: Italy Markers Market Size (USD Million), by Technology, 2020-2035
- Table 211: Italy Markers Market Size (USD Million), by Application, 2020-2035
- Table 212: Italy Markers Market Size (USD Million), by End User, 2020-2035
- Table 213: Spain Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 214: Spain Markers Market Size (USD Million), by Technology, 2020-2035
- Table 215: Spain Markers Market Size (USD Million), by Application, 2020-2035
- Table 216: Spain Markers Market Size (USD Million), by End User, 2020-2035
- Table 217: BENELUX Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 218: BENELUX Markers Market Size (USD Million), by Technology, 2020-2035
- Table 219: BENELUX Markers Market Size (USD Million), by Application, 2020-2035
- Table 220: BENELUX Markers Market Size (USD Million), by End User, 2020-2035
- Table 221: Nordic Countries Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 222: Nordic Countries Markers Market Size (USD Million), by Technology, 2020-2035
- Table 223: Nordic Countries Markers Market Size (USD Million), by Application, 2020-2035
- Table 224: Nordic Countries Markers Market Size (USD Million), by End User, 2020-2035
- Table 225: Rest of Western Europe Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 226: Rest of Western Europe Markers Market Size (USD Million), by Technology, 2020-2035
- Table 227: Rest of Western Europe Markers Market Size (USD Million), by Application, 2020-2035
- Table 228: Rest of Western Europe Markers Market Size (USD Million), by End User, 2020-2035
- Table 229: Eastern Europe Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 230: Eastern Europe Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 231: Eastern Europe Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 232: Eastern Europe Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 233: Eastern Europe Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 234: Eastern Europe Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 235: Eastern Europe Markers Market Size (USD Million), by Technology, 2020-2035
- Table 236: Eastern Europe Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 237: Eastern Europe Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 238: Eastern Europe Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 239: Eastern Europe Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 240: Eastern Europe Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 241: Eastern Europe Markers Market Size (USD Million), by Application, 2020-2035
- Table 242: Eastern Europe Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 243: Eastern Europe Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 244: Eastern Europe Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 245: Eastern Europe Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 246: Eastern Europe Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 247: Eastern Europe Markers Market Size (USD Million), by End User, 2020-2035
- Table 248: Eastern Europe Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 249: Eastern Europe Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 250: Eastern Europe Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 251: Eastern Europe Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 252: Eastern Europe Markers Market Size (USD Million), by Others, 2020-2035
- Table 253: Russia Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 254: Russia Markers Market Size (USD Million), by Technology, 2020-2035
- Table 255: Russia Markers Market Size (USD Million), by Application, 2020-2035
- Table 256: Russia Markers Market Size (USD Million), by End User, 2020-2035
- Table 257: Hungary Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 258: Hungary Markers Market Size (USD Million), by Technology, 2020-2035
- Table 259: Hungary Markers Market Size (USD Million), by Application, 2020-2035
- Table 260: Hungary Markers Market Size (USD Million), by End User, 2020-2035
- Table 261: Poland Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 262: Poland Markers Market Size (USD Million), by Technology, 2020-2035
- Table 263: Poland Markers Market Size (USD Million), by Application, 2020-2035
- Table 264: Poland Markers Market Size (USD Million), by End User, 2020-2035
- Table 265: Rest of Eastern Europe Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 266: Rest of Eastern Europe Markers Market Size (USD Million), by Technology, 2020-2035
- Table 267: Rest of Eastern Europe Markers Market Size (USD Million), by Application, 2020-2035
- Table 268: Rest of Eastern Europe Markers Market Size (USD Million), by End User, 2020-2035
- Table 269: Middle East & Africa Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 270: Middle East & Africa Markers Market Size (USD Million), by CD Antigens (Core Myeloid Markers), 2020-2035
- Table 271: Middle East & Africa Markers Market Size (USD Million), by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Table 272: Middle East & Africa Markers Market Size (USD Million), by Lineage & Differentiation Panels, 2020-2035
- Table 273: Middle East & Africa Markers Market Size (USD Million), by MDSC Panels, 2020-2035
- Table 274: Middle East & Africa Markers Market Size (USD Million), by Other Immune-Modulatory Markers, 2020-2035
- Table 275: Middle East & Africa Markers Market Size (USD Million), by Technology, 2020-2035
- Table 276: Middle East & Africa Markers Market Size (USD Million), by Flow Cytometry Antibodies, 2020-2035
- Table 277: Middle East & Africa Markers Market Size (USD Million), by IHC / IF Antibodies, 2020-2035
- Table 278: Middle East & Africa Markers Market Size (USD Million), by Mass Cytometry / CyTOF Panels, 2020-2035
- Table 279: Middle East & Africa Markers Market Size (USD Million), by Multiplex I/O Panels, 2020-2035
- Table 280: Middle East & Africa Markers Market Size (USD Million), by Genomic / Proteomic Panels, 2020-2035
- Table 281: Middle East & Africa Markers Market Size (USD Million), by Application, 2020-2035
- Table 282: Middle East & Africa Markers Market Size (USD Million), by Oncology & Immuno-Oncology, 2020-2035
- Table 283: Middle East & Africa Markers Market Size (USD Million), by Hematology & Transplant, 2020-2035
- Table 284: Middle East & Africa Markers Market Size (USD Million), by Inflammation & Autoimmune, 2020-2035
- Table 285: Middle East & Africa Markers Market Size (USD Million), by Infectious Disease & Sepsis, 2020-2035
- Table 286: Middle East & Africa Markers Market Size (USD Million), by Translational Research Tools, 2020-2035
- Table 287: Middle East & Africa Markers Market Size (USD Million), by End User, 2020-2035
- Table 288: Middle East & Africa Markers Market Size (USD Million), by Pharma & Biotech, 2020-2035
- Table 289: Middle East & Africa Markers Market Size (USD Million), by CROs & Central Labs, 2020-2035
- Table 290: Middle East & Africa Markers Market Size (USD Million), by Academic & Translational Centers, 2020-2035
- Table 291: Middle East & Africa Markers Market Size (USD Million), by Hospital / Clinical Labs, 2020-2035
- Table 292: Middle East & Africa Markers Market Size (USD Million), by Others, 2020-2035
- Table 293: Saudi Arabia Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 294: Saudi Arabia Markers Market Size (USD Million), by Technology, 2020-2035
- Table 295: Saudi Arabia Markers Market Size (USD Million), by Application, 2020-2035
- Table 296: Saudi Arabia Markers Market Size (USD Million), by End User, 2020-2035
- Table 297: Other GCC Countries Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 298: Other GCC Countries Markers Market Size (USD Million), by Technology, 2020-2035
- Table 299: Other GCC Countries Markers Market Size (USD Million), by Application, 2020-2035
- Table 300: Other GCC Countries Markers Market Size (USD Million), by End User, 2020-2035
- Table 301: Türkiye Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 302: Türkiye Markers Market Size (USD Million), by Technology, 2020-2035
- Table 303: Türkiye Markers Market Size (USD Million), by Application, 2020-2035
- Table 304: Türkiye Markers Market Size (USD Million), by End User, 2020-2035
- Table 305: South Africa Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 306: South Africa Markers Market Size (USD Million), by Technology, 2020-2035
- Table 307: South Africa Markers Market Size (USD Million), by Application, 2020-2035
- Table 308: South Africa Markers Market Size (USD Million), by End User, 2020-2035
- Table 309: Rest of MEA Markers Market Size (USD Million), by Marker Class, 2020-2035
- Table 310: Rest of MEA Markers Market Size (USD Million), by Technology, 2020-2035
- Table 311: Rest of MEA Markers Market Size (USD Million), by Application, 2020-2035
- Table 312: Rest of MEA Markers Market Size (USD Million), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 2: Global Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 3: Global Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 4: Global Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 5: Global Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 6: Global Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 7: Global Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 8: Global Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 9: Global Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 10: Global Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 11: Global Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 12: Global Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 13: Global Market (USD Million) Forecast, by Application, 2020-2035
- Figure 14: Global Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 15: Global Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 16: Global Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 17: Global Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 18: Global Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 19: Global Market (USD Million) Forecast, by End User, 2020-2035
- Figure 20: Global Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 21: Global Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 22: Global Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 23: Global Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 24: Global Market (USD Million) Forecast, by Others, 2020-2035
- Figure 25: North America Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 26: North America Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 27: North America Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 28: North America Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 29: North America Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 30: North America Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 31: North America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 32: North America Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 33: North America Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 34: North America Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 35: North America Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 36: North America Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 37: North America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 38: North America Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 39: North America Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 40: North America Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 41: North America Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 42: North America Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 43: North America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 44: North America Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 45: North America Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 46: North America Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 47: North America Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 48: North America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 49: USA Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 50: USA Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 51: USA Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 52: USA Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 53: USA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 54: USA Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 55: USA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 56: USA Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 57: Canada Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 58: Canada Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 59: Canada Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 60: Canada Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 61: Canada Market (USD Million) Forecast, by Application, 2020-2035
- Figure 62: Canada Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 63: Canada Market (USD Million) Forecast, by End User, 2020-2035
- Figure 64: Canada Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 65: Mexico Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 66: Mexico Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 67: Mexico Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 68: Mexico Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 69: Mexico Market (USD Million) Forecast, by Application, 2020-2035
- Figure 70: Mexico Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 71: Mexico Market (USD Million) Forecast, by End User, 2020-2035
- Figure 72: Mexico Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 73: Latin America Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 74: Latin America Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 75: Latin America Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 76: Latin America Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 77: Latin America Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 78: Latin America Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 79: Latin America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 80: Latin America Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 81: Latin America Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 82: Latin America Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 83: Latin America Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 84: Latin America Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 85: Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 86: Latin America Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 87: Latin America Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 88: Latin America Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 89: Latin America Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 90: Latin America Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 91: Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 92: Latin America Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 93: Latin America Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 94: Latin America Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 95: Latin America Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 96: Latin America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 97: Brazil Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 98: Brazil Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 99: Brazil Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 100: Brazil Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 101: Brazil Market (USD Million) Forecast, by Application, 2020-2035
- Figure 102: Brazil Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 103: Brazil Market (USD Million) Forecast, by End User, 2020-2035
- Figure 104: Brazil Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 105: Argentina Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 106: Argentina Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 107: Argentina Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 108: Argentina Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 109: Argentina Market (USD Million) Forecast, by Application, 2020-2035
- Figure 110: Argentina Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 111: Argentina Market (USD Million) Forecast, by End User, 2020-2035
- Figure 112: Argentina Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 113: Rest of Latin America Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 114: Rest of Latin America Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 115: Rest of Latin America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 116: Rest of Latin America Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 117: Rest of Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 118: Rest of Latin America Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 119: Rest of Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 120: Rest of Latin America Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 121: East Asia Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 122: East Asia Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 123: East Asia Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 124: East Asia Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 125: East Asia Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 126: East Asia Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 127: East Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 128: East Asia Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 129: East Asia Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 130: East Asia Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 131: East Asia Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 132: East Asia Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 133: East Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 134: East Asia Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 135: East Asia Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 136: East Asia Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 137: East Asia Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 138: East Asia Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 139: East Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 140: East Asia Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 141: East Asia Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 142: East Asia Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 143: East Asia Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 144: East Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 145: China Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 146: China Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 147: China Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 148: China Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 149: China Market (USD Million) Forecast, by Application, 2020-2035
- Figure 150: China Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 151: China Market (USD Million) Forecast, by End User, 2020-2035
- Figure 152: China Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 153: Japan Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 154: Japan Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 155: Japan Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 156: Japan Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 157: Japan Market (USD Million) Forecast, by Application, 2020-2035
- Figure 158: Japan Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 159: Japan Market (USD Million) Forecast, by End User, 2020-2035
- Figure 160: Japan Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 161: South Korea Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 162: South Korea Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 163: South Korea Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 164: South Korea Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 165: South Korea Market (USD Million) Forecast, by Application, 2020-2035
- Figure 166: South Korea Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 167: South Korea Market (USD Million) Forecast, by End User, 2020-2035
- Figure 168: South Korea Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 169: South Asia Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 170: South Asia Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 171: South Asia Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 172: South Asia Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 173: South Asia Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 174: South Asia Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 175: South Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 176: South Asia Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 177: South Asia Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 178: South Asia Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 179: South Asia Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 180: South Asia Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 181: South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 182: South Asia Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 183: South Asia Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 184: South Asia Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 185: South Asia Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 186: South Asia Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 187: South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 188: South Asia Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 189: South Asia Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 190: South Asia Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 191: South Asia Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 192: South Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 193: India Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 194: India Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 195: India Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 196: India Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 197: India Market (USD Million) Forecast, by Application, 2020-2035
- Figure 198: India Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 199: India Market (USD Million) Forecast, by End User, 2020-2035
- Figure 200: India Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 201: ASEAN Countries Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 202: ASEAN Countries Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 203: ASEAN Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 204: ASEAN Countries Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 205: ASEAN Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 206: ASEAN Countries Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 207: ASEAN Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 208: ASEAN Countries Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 209: Australia & New Zealand Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 210: Australia & New Zealand Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 211: Australia & New Zealand Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 212: Australia & New Zealand Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 213: Australia & New Zealand Market (USD Million) Forecast, by Application, 2020-2035
- Figure 214: Australia & New Zealand Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 215: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020-2035
- Figure 216: Australia & New Zealand Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 217: Rest of South Asia Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 218: Rest of South Asia Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 219: Rest of South Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 220: Rest of South Asia Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 221: Rest of South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 222: Rest of South Asia Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 223: Rest of South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 224: Rest of South Asia Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 225: Western Europe Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 226: Western Europe Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 227: Western Europe Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 228: Western Europe Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 229: Western Europe Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 230: Western Europe Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 231: Western Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 232: Western Europe Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 233: Western Europe Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 234: Western Europe Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 235: Western Europe Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 236: Western Europe Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 237: Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 238: Western Europe Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 239: Western Europe Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 240: Western Europe Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 241: Western Europe Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 242: Western Europe Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 243: Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 244: Western Europe Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 245: Western Europe Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 246: Western Europe Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 247: Western Europe Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 248: Western Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 249: Germany Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 250: Germany Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 251: Germany Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 252: Germany Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 253: Germany Market (USD Million) Forecast, by Application, 2020-2035
- Figure 254: Germany Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 255: Germany Market (USD Million) Forecast, by End User, 2020-2035
- Figure 256: Germany Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 257: UK Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 258: UK Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 259: UK Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 260: UK Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 261: UK Market (USD Million) Forecast, by Application, 2020-2035
- Figure 262: UK Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 263: UK Market (USD Million) Forecast, by End User, 2020-2035
- Figure 264: UK Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 265: France Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 266: France Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 267: France Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 268: France Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 269: France Market (USD Million) Forecast, by Application, 2020-2035
- Figure 270: France Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 271: France Market (USD Million) Forecast, by End User, 2020-2035
- Figure 272: France Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 273: Italy Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 274: Italy Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 275: Italy Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 276: Italy Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 277: Italy Market (USD Million) Forecast, by Application, 2020-2035
- Figure 278: Italy Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 279: Italy Market (USD Million) Forecast, by End User, 2020-2035
- Figure 280: Italy Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 281: Spain Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 282: Spain Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 283: Spain Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 284: Spain Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 285: Spain Market (USD Million) Forecast, by Application, 2020-2035
- Figure 286: Spain Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 287: Spain Market (USD Million) Forecast, by End User, 2020-2035
- Figure 288: Spain Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 289: BENELUX Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 290: BENELUX Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 291: BENELUX Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 292: BENELUX Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 293: BENELUX Market (USD Million) Forecast, by Application, 2020-2035
- Figure 294: BENELUX Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 295: BENELUX Market (USD Million) Forecast, by End User, 2020-2035
- Figure 296: BENELUX Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 297: Nordic Countries Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 298: Nordic Countries Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 299: Nordic Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 300: Nordic Countries Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 301: Nordic Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 302: Nordic Countries Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 303: Nordic Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 304: Nordic Countries Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 305: Rest of Western Europe Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 306: Rest of Western Europe Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 307: Rest of Western Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 308: Rest of Western Europe Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 309: Rest of Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 310: Rest of Western Europe Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 311: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 312: Rest of Western Europe Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 313: Eastern Europe Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 314: Eastern Europe Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 315: Eastern Europe Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 316: Eastern Europe Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 317: Eastern Europe Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 318: Eastern Europe Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 319: Eastern Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 320: Eastern Europe Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 321: Eastern Europe Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 322: Eastern Europe Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 323: Eastern Europe Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 324: Eastern Europe Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 325: Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 326: Eastern Europe Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 327: Eastern Europe Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 328: Eastern Europe Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 329: Eastern Europe Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 330: Eastern Europe Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 331: Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 332: Eastern Europe Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 333: Eastern Europe Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 334: Eastern Europe Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 335: Eastern Europe Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 336: Eastern Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 337: Russia Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 338: Russia Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 339: Russia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 340: Russia Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 341: Russia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 342: Russia Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 343: Russia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 344: Russia Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 345: Hungary Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 346: Hungary Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 347: Hungary Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 348: Hungary Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 349: Hungary Market (USD Million) Forecast, by Application, 2020-2035
- Figure 350: Hungary Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 351: Hungary Market (USD Million) Forecast, by End User, 2020-2035
- Figure 352: Hungary Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 353: Poland Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 354: Poland Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 355: Poland Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 356: Poland Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 357: Poland Market (USD Million) Forecast, by Application, 2020-2035
- Figure 358: Poland Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 359: Poland Market (USD Million) Forecast, by End User, 2020-2035
- Figure 360: Poland Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 361: Rest of Eastern Europe Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 362: Rest of Eastern Europe Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 363: Rest of Eastern Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 364: Rest of Eastern Europe Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 365: Rest of Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 366: Rest of Eastern Europe Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 367: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 368: Rest of Eastern Europe Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 369: Middle East & Africa Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 370: Middle East & Africa Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 371: Middle East & Africa Market (USD Million) Forecast, by Checkpoint & Activation Markers (PD-L1, CD47, etc.), 2020-2035
- Figure 372: Middle East & Africa Market (USD Million) Forecast, by Lineage & Differentiation Panels, 2020-2035
- Figure 373: Middle East & Africa Market (USD Million) Forecast, by MDSC Panels, 2020-2035
- Figure 374: Middle East & Africa Market (USD Million) Forecast, by Other Immune-Modulatory Markers, 2020-2035
- Figure 375: Middle East & Africa Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 376: Middle East & Africa Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 377: Middle East & Africa Market (USD Million) Forecast, by IHC / IF Antibodies, 2020-2035
- Figure 378: Middle East & Africa Market (USD Million) Forecast, by Mass Cytometry / CyTOF Panels, 2020-2035
- Figure 379: Middle East & Africa Market (USD Million) Forecast, by Multiplex I/O Panels, 2020-2035
- Figure 380: Middle East & Africa Market (USD Million) Forecast, by Genomic / Proteomic Panels, 2020-2035
- Figure 381: Middle East & Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 382: Middle East & Africa Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 383: Middle East & Africa Market (USD Million) Forecast, by Hematology & Transplant, 2020-2035
- Figure 384: Middle East & Africa Market (USD Million) Forecast, by Inflammation & Autoimmune, 2020-2035
- Figure 385: Middle East & Africa Market (USD Million) Forecast, by Infectious Disease & Sepsis, 2020-2035
- Figure 386: Middle East & Africa Market (USD Million) Forecast, by Translational Research Tools, 2020-2035
- Figure 387: Middle East & Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 388: Middle East & Africa Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 389: Middle East & Africa Market (USD Million) Forecast, by CROs & Central Labs, 2020-2035
- Figure 390: Middle East & Africa Market (USD Million) Forecast, by Academic & Translational Centers, 2020-2035
- Figure 391: Middle East & Africa Market (USD Million) Forecast, by Hospital / Clinical Labs, 2020-2035
- Figure 392: Middle East & Africa Market (USD Million) Forecast, by Others, 2020-2035
- Figure 393: Saudi Arabia Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 394: Saudi Arabia Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 395: Saudi Arabia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 396: Saudi Arabia Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 397: Saudi Arabia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 398: Saudi Arabia Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 399: Saudi Arabia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 400: Saudi Arabia Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 401: Other GCC Countries Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 402: Other GCC Countries Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 403: Other GCC Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 404: Other GCC Countries Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 405: Other GCC Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 406: Other GCC Countries Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 407: Other GCC Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 408: Other GCC Countries Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 409: Türkiye Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 410: Türkiye Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 411: Türkiye Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 412: Türkiye Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 413: Türkiye Market (USD Million) Forecast, by Application, 2020-2035
- Figure 414: Türkiye Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 415: Türkiye Market (USD Million) Forecast, by End User, 2020-2035
- Figure 416: Türkiye Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 417: South Africa Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 418: South Africa Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 419: South Africa Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 420: South Africa Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 421: South Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 422: South Africa Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 423: South Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 424: South Africa Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 425: Rest of MEA Market (USD Million) Forecast, by Marker Class, 2020-2035
- Figure 426: Rest of MEA Market (USD Million) Forecast, by CD Antigens (Core Myeloid Markers), 2020-2035
- Figure 427: Rest of MEA Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 428: Rest of MEA Market (USD Million) Forecast, by Flow Cytometry Antibodies, 2020-2035
- Figure 429: Rest of MEA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 430: Rest of MEA Market (USD Million) Forecast, by Oncology & Immuno-Oncology, 2020-2035
- Figure 431: Rest of MEA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 432: Rest of MEA Market (USD Million) Forecast, by Pharma & Biotech, 2020-2035
- Figure 433: BD Biosciences - Company Snapshot
- Figure 434: BD Biosciences - SWOT Analysis
- Figure 435: Beckman Coulter (Danaher) - Company Snapshot
- Figure 436: Beckman Coulter (Danaher) - SWOT Analysis
- Figure 437: BioLegend - Company Snapshot
- Figure 438: BioLegend - SWOT Analysis
- Figure 439: Miltenyi Biotec - Company Snapshot
- Figure 440: Miltenyi Biotec - SWOT Analysis
- Figure 441: Thermo Fisher Scientific - Company Snapshot
- Figure 442: Thermo Fisher Scientific - SWOT Analysis
- Figure 443: Bio-Rad Laboratories - Company Snapshot
- Figure 444: Bio-Rad Laboratories - SWOT Analysis
- Figure 445: R&D Systems / Bio-Techne - Company Snapshot
- Figure 446: R&D Systems / Bio-Techne - SWOT Analysis
- Figure 447: Abcam - Company Snapshot
- Figure 448: Abcam - SWOT Analysis
- Figure 449: Standard BioTools (Fluidigm) - Company Snapshot
- Figure 450: Standard BioTools (Fluidigm) - SWOT Analysis

