Nail Disorder Treatment Product Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Nail Disorder Treatment Product Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Nail Disorder Treatment Product Market Forecast and Outlook 2026 to 2036
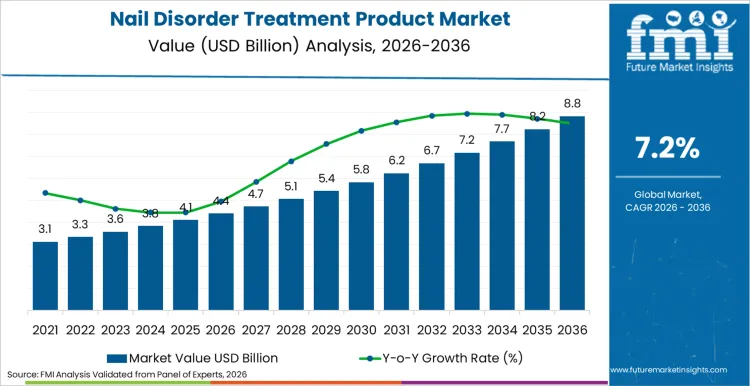
Nail disorder treatment product market is expected to reach USD 4.4 billion in 2026, driven by rising prevalence of onychomycosis and psoriasis-related nail deformities. With a steady CAGR of 7.2% extending through 2036, total sector revenue is set to exceed USD 8.8 billion by the conclusion of this assessment period. Increasing geriatric population, which is significantly more prone to fungal infections and structural nail anomalies, acts as a primary accelerator for this upward financial trajectory. Pharmaceutical entities are directing substantial capital into developing novel transungual delivery systems, aiming to enhance penetration rates for stubborn pathogens.
A transition from concealing nail issues with cosmetic solutions to seeking active medical management is redefining consumer spending habits across major economic zones. Patients are actively seeking professional-grade prescriptions, pushing manufacturers to innovate beyond standard over-the-counter options. This growth pattern reflects a broader necessity for specialized dermatological interventions in maintaining appendage health effectively over the coming decade.
Escalating incidence of lifestyle-induced nail disorders significantly influences industry expansion. Healthcare providers are emphasizing early diagnosis, utilizing various therapeutic classes to restore nail plate integrity before permanent dystrophy occurs. Clinical trials exploring advanced biologics for psoriatic nails are yielding promising results, fostering confidence among investors and dermatologists. Rising awareness regarding diabetic foot care continues to fuel demand for preventative and curative treatments, creating sustained opportunities for efficacious medical solutions.
Government health campaigns aimed at raising hygiene standards are facilitating faster approval processes for next-generation antifungal lacquers. Consequently, companies are witnessing robust opportunities in developing regions where personal grooming standards are rapidly evolving to align with medical health awareness. High unmet needs in treating brittle nail syndrome further drive research initiatives, ensuring that product pipelines remain robust and responsive to diverse patient requirements.
Quick Stats for Nail Disorder Treatment Product Market
- Nail Disorder Treatment Product Industry Value (2026): USD 4.4 Billion
- Nail Disorder Treatment Product Industry Value (2036): USD 8.8 Billion
- Nail Disorder Treatment Product Industry CAGR (2026 to 2036): 7.2%
- Nail Disorder Treatment Product Industry Leading Segment: Topical antifungal drugs
- Nail Disorder Treatment Product Industry Key Growth Countries: India, Brazil, China
- Nail Disorder Treatment Product Industry Key Players: Pfizer, Bausch Health, Novartis
Innovations in formulation science are transforming how active ingredients penetrate the keratinized nail plate. Researchers are integrating onychomycosis treatment agents with advanced lacquers to offer sustained release profiles, reducing application frequency. Development of nail strengtheners infused with biogenic polymers is gaining traction, addressing fragility barriers that often hinder patient satisfaction. Focus is also shifting towards foot care product lines that incorporate specialized applicators for precise dosage to infected areas. Advancements in biotechnology allow for targeted delivery of antimycotics, enhancing efficacy of treatment protocols while minimizing systemic side effects. Such technological strides are pivotal in shaping future product landscapes, ensuring that diverse clinical needs are met with precision and safety.
Nail Disorder Treatment Product Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 4.4 Billion |
| Industry Value (2036) | USD 8.8 Billion |
| CAGR (2026 to 2036) | 7.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Which Innovations Are Propelling Therapeutic Efficacy?
Adoption of laser-compatible topical agents is serving as a significant catalyst for broader acceptance of combination therapies. Recent breakthroughs in anti-fungal therapy have revolutionized treatment protocols by offering synergistic effects when used alongside device-based interventions. This dual-action approach appeals strongly to healthcare providers managing patients with resistant fungal strains. Clinical success of fungal testing kits in enabling rapid pathogen identification has expanded prescribing precision beyond empirical approaches.
Continuous education programs regarding long-term risks of untreated infections are also driving patient proactivity. Pharmaceutical companies are capitalizing on these trends by launching nutricosmetics that improve nail quality from within, thereby improving holistic adherence rates. Integrating biotin supplements into treatment regimens is becoming standard practice, supporting structural recovery alongside pharmaceutical intervention.
How Is the Nail Disorder Treatment Product Market Segmented Across Key Categories?
The nail disorder treatment product market is segmented across several key categories to effectively address the diverse needs of consumers. Primary segments include product type, which encompasses antifungal treatments, nail growth enhancers, and moisturizers, each targeting specific conditions such as nail fungus, brittleness, or ingrown nails. Distribution channels further divide the market, comprising pharmacies, online retailers, and dermatology clinics, influencing accessibility and consumer choice.
Why Do Topical Antifungal Drugs Retain Prominence?

Topical antifungal drugs account for 34% of share in 2026, driven by their direct application capabilities and minimal systemic absorption. Dominance is underpinned by clinical guidelines that recommend lacquers and solutions as first-line therapies for mild-to-moderate fungal infections. This segment benefits significantly from formulation versatility, allowing incorporation of penetration enhancers that navigate the dense keratin matrix. Ongoing research into reducing volatile solvents in this class is renewing interest among health-conscious consumers. Furthermore, their role in managing foot fungus treatments remains vital for preventing cross-contamination between skin and nails. As healthcare systems strive for cost-effective disease management, demand for proven topical solutions is expected to remain robust, sustaining their leadership position throughout the forecast period.
Why Is Onychomycosis the Primary Indication?
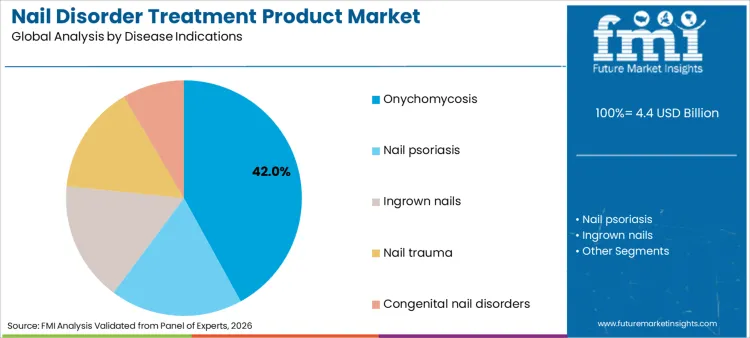
Onychomycosis applications command a 42% share of the sector in 2026, reflecting the massive global prevalence of fungal nail infections. Dominance is inextricably linked to the stubborn and recurrent nature of the disease, which often requires prolonged treatment courses. Sector relies heavily on specialized nail care products to manage fungal loads effectively and restore aesthetic appearance. These therapies are essential for preventing secondary bacterial infections in immunocompromised patients, which drives healthcare priorities. Consequently, relentless drive to eradicate fungal pathogens ensures that products designed to treat onychomycosis remain highest priority for funding and procurement in both clinical and home-care settings. High recurrence rates further necessitate continuous usage of prophylactic products, bolstering long-term revenue streams for manufacturers.
How Will Digital Health Integration Alter Future Revenues?
Digital health integration is set to fundamentally reshape sales landscapes by facilitating remote diagnosis and treatment monitoring. As teledermatology platforms gain traction, patients can receive prescriptions for nail disorders without physical clinic visits, expanding access in underserved areas. This shift towards virtual care is creating demand for high-quality, easy-to-apply products that require minimal physician supervision. Manufacturers who fail to align their marketing strategies with digital health ecosystems risk losing visibility to tech-savvy consumers. Furthermore, availability of AI-driven diagnostic apps drives consumption volume, as users can track their treatment progress accurately. Rise of digital therapeutics also stimulates innovation in supportive care, as companies seek to differentiate their offerings by bundling products with app-based adherence tools.
How Are Regulatory Frameworks Impacting Development?
Regulatory bodies globally are increasingly advocating for rigorous clinical endpoints in nail disorder trials, directly impacting development timelines and costs. This regulatory shift forces pharmaceutical companies to demonstrate not just mycological cure but also complete clinical clearance, increasing barriers to entry for generic players. However, it also introduces opportunities for differentiation; agents that achieve faster cosmetic improvement gain rapid prescriber endorsement. Strict safety standards are impacting production of brittle nails treatment formulations. Manufacturers must now ensure rigorous stability testing to prevent degradation of active ingredients. While these regulations drive development of more effective products, they also impose higher compliance costs. Companies that proactively align with FDA and EMA guidelines regarding patient-reported outcomes will gain a competitive edge, positioning their products as gold standards for efficacy.
How Will the Nail Disorder Treatment Industry Evolve in Major Nations?
Europe is projected to demonstrate steady expansion from 2026 to 2036, while emerging economies like India and China are demonstrating even faster acceleration. Regional evolution is characterized by distinct growth drivers: established nations in the West are pivoting towards biologic therapies for nail psoriasis, whereas Asian nations are expanding access to affordable antifungals and skincare products for hands and feet. Disparity in growth rates highlights a shifting center of gravity, where rising disposable incomes in Asia-Pacific are creating massive volume demand. Conversely, North American and European sectors are focusing on innovation and specialized applications, such as post-trauma nail repair. Government grants and private funding in these major nations are heavily slanted towards dermatology research, ensuring sustained capital influx.
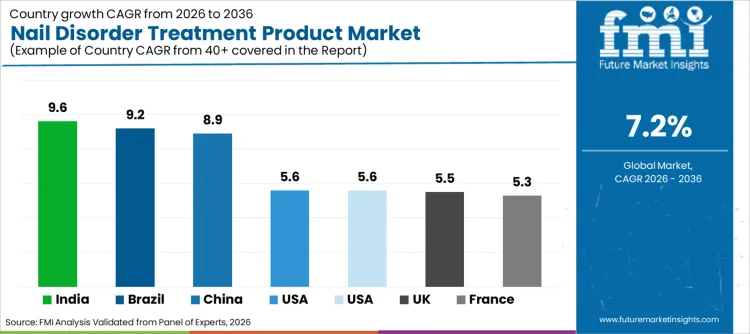
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 5.6% |
| Brazil | 9.2% |
| China | 8.9% |
| India | 9.6% |
| Germany | 5.6% |
| France | 5.3% |
| UK | 5.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Nail Disorder Treatment Industry in the USA Driving Rapid Growth?
Sales of nail disorder treatment products in the USA are set to grow at a CAGR of 5.6% through 2036. This expansion is underpinned by high awareness of podiatric health and a healthcare system that supports regular screenings for diabetic complications. Growth is fueled by substantial insurance coverage for prescription antifungals, which translates into high revenue generation per unit. Furthermore, rapid adoption of advanced laser therapies in clinics has created a follow-up market for topical recovery products. Presence of key industry players headquartered within the country also streamlines supply chains, ensuring rapid access to latest launches. Emphasis on aesthetic restoration alongside medical cure continues to be a primary driver, maintaining USA position as a critical revenue generator.
Why is the Nail Disorder Treatment Sector in China Expanding?
Demand for nail treatment solutions in China is projected to rise at an 8.9% CAGR during the assessment period. "Healthy China 2030" initiatives emphasize personal hygiene and chronic disease management, fostering a favorable environment for domestic and international pharmaceutical brands. Increasing investments in aesthetic dermatology clinics are creating massive supply of premium recovery kits to support vast patient populations. Rising prevalence of onychomycosis in humid southern regions is prompting local pharmacies to stock advanced antifungal protocols. This shift is further supported by e-commerce platforms promoting niche medical beauty brands, thereby accelerating uptake of specialized products across provinces and fueling sector expansion.
Why is the Nail Disorder Treatment Industry in India Showing High Potential?
Nail disorder treatment industry in India is poised to expand at a CAGR of 9.6%, representing highest growth rate among analyzed nations. Surge is largely attributed to growing urbanization and increasing footfall in dermatology centers for cosmetic concerns. Government's focus on improving sanitation standards is encouraging population to upgrade from home remedies to clinically proven treatments. Furthermore, increasing number of partnerships between Indian pharma giants and global skincare innovators necessitates production of high-quality formulations. Cost advantage of manufacturing in India, combined with rising middle-class purchasing power, is attracting significant investment into the domestic personal care ecosystem. Generic availability of high-potency antifungals further democratizes access, boosting volume sales significantly.
What is the Competitive Outlook for the Nail Disorder Treatment Product Market?
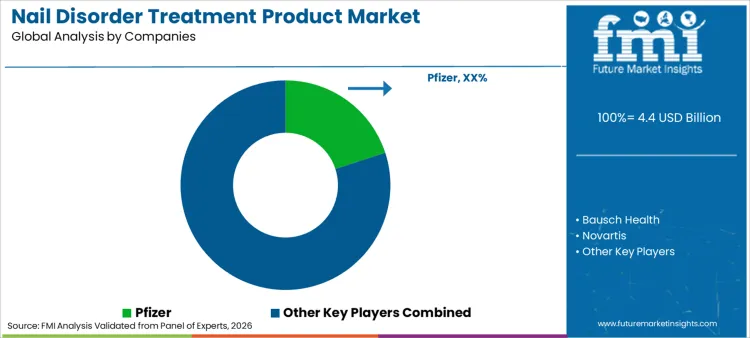
Competition in this industry is intensifying as major players leverage mergers and acquisitions to consolidate their portfolios and expand geographic footprints. Leading companies like Pfizer and Bausch Health are actively developing patent-protected delivery systems to distinguish their antifungals from generic competitors. Strategies are increasingly focused on bundling therapeutics with educational apps to offer comprehensive disease management platforms. "Rx-to-OTC" switch strategies ensure broader consumer access and retention, allowing brands to capture patients earlier in the disease progression. Additionally, strategic partnerships with podiatry associations allow companies to co-develop products that address specific clinical pain points. Competitive landscape is also witnessing a rise in influencer marketing by dermatologists, where brands are differentiating themselves by offering science-backed validation to appeal to skeptical consumers.
Key Players in Nail Disorder Treatment Product Market
- Pfizer
- Bausch Health
- Novartis
- GSK
- Sanofi
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Million, USD Billion |
| Treatment Types | Topical antifungal drugs, Systemic antifungal drugs, Anti-inflammatory nail therapies, Nail repair biologics, Supportive nail care products |
| Indications | Onychomycosis, Nail psoriasis, Ingrown nails, Nail trauma, Congenital nail disorders |
| End Users | Dermatology clinics, Hospitals, Retail pharmacies, Online pharmacies |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
Nail Disorder Treatment Product Market by Segments
Nail Disorder Treatment Types:
- Topical antifungal drugs
- Systemic antifungal drugs
- Anti-inflammatory nail therapies
- Nail repair biologics
- Supportive nail care products
Disease Indications:
- Onychomycosis
- Nail psoriasis
- Ingrown nails
- Nail trauma
- Congenital nail disorders
End Users:
- Dermatology clinics
- Hospitals
- Retail pharmacies
- Online pharmacies
Regions:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- American Academy of Dermatology. (2026, January 15). Nail fungus: Diagnosis and treatment. American Academy of Dermatology, United States.
- Centers for Disease Control and Prevention. (2024, May 20). Treatment of ringworm and fungal nail infections. Centers for Disease Control and Prevention, United States.
- USA Food and Drug Administration. (2025, February 8). Latest news on OTC monographs at FDA. USA Food and Drug Administration, United States.
- World Health Organization. (2025, May 27). Skin diseases as a global public health priority (WHA78.15). World Health Organization.
- European Medicines Agency. (2024, June 26). Cresemba: EPAR assessment report (H-C-002734-X-0042-G). European Medicines Agency.
Frequently Asked Questions
How large is the demand for Nail Disorder Treatment Products in the global market in 2026?
The demand for Nail Disorder Treatment Products in the global market is estimated to be valued at USD 4.4 billion in 2026.
What will be the size of Nail Disorder Treatment Products in the global market by 2036?What will be the size of Nail Disorder Treatment Products in the global market by 2036?
The market size for Nail Disorder Treatment Products in the global market is projected to reach USD 8.8 billion by 2036.
What is the expected demand growth for Nail Disorder Treatment Products in the global market between 2026 and 2036?
The demand for Nail Disorder Treatment Products in the global market is expected to grow at a CAGR of 7.2% between 2026 and 2036.
Which product type in Nail Disorder Treatment Products is poised to lead global sales by 2026?
Topical antifungal drugs are expected to be the dominant product category, capturing approximately 34% of the global market share in 2026 due to high safety and efficacy.
How significant is the role of Dermatology Clinics in driving Nail Disorder Treatment Product adoption in 2026?
Dermatology Clinics represent a critical end-user segment, projected to hold a substantial 40% share of the total market in 2026 as patients seek specialist care for persistent conditions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Treatment Types
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment Types , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment Types , 2026 to 2036
- Topical antifungal drugs
- Systemic antifungal drugs
- Anti-inflammatory nail therapies
- Nail repair biologics
- Supportive nail care products
- Topical antifungal drugs
- Y-o-Y Growth Trend Analysis By Treatment Types , 2021 to 2025
- Absolute $ Opportunity Analysis By Treatment Types , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indications, 2026 to 2036
- Onychomycosis
- Nail psoriasis
- Ingrown nails
- Nail trauma
- Congenital nail disorders
- Onychomycosis
- Y-o-Y Growth Trend Analysis By Disease Indications, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users, 2026 to 2036
- Dermatology clinics
- Retail pharmacies
- Hospitals
- Online pharmacies
- Dermatology clinics
- Y-o-Y Growth Trend Analysis By End Users, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Treatment Types
- By Disease Indications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Types
- By Disease Indications
- By End Users
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Types
- By Disease Indications
- By End Users
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Treatment Types
- By Disease Indications
- By End Users
- Competition Analysis
- Competition Deep Dive
- Pfizer
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bausch Health
- Novartis
- GSK
- Sanofi
- Pfizer
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Treatment Types , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Disease Indications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Treatment Types
- Figure 6: Global Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Disease Indications
- Figure 9: Global Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Users
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Treatment Types
- Figure 26: North America Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Disease Indications
- Figure 29: North America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Users
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Treatment Types
- Figure 36: Latin America Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Disease Indications
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Treatment Types
- Figure 46: Western Europe Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Disease Indications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Treatment Types
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Disease Indications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Treatment Types
- Figure 66: East Asia Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Disease Indications
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Treatment Types
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Disease Indications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Treatment Types , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Treatment Types , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Treatment Types
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Disease Indications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Indications, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Disease Indications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

