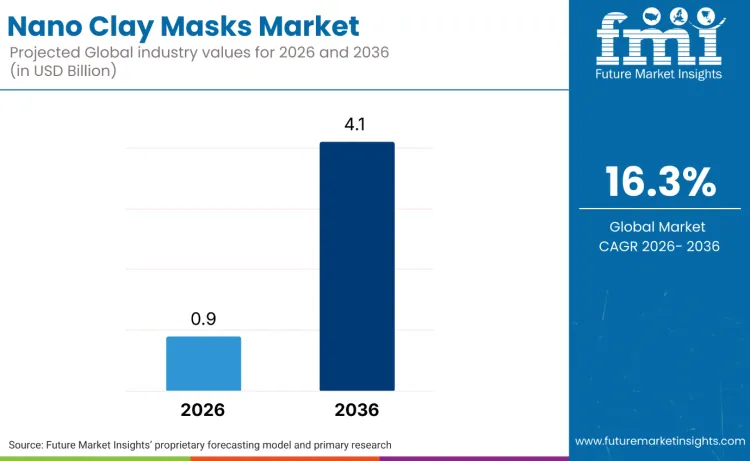
The global nano clay masks market is valued at USD 0.9 billion in 2026 and is projected to reach USD 4.1 billion by 2036, expanding at a 16.3% CAGR. Growth will concentrate in oil-control and pore-focused masks that can be positioned with measurable outcomes and reordered in short usage cycles. Regulatory governance will shape the addressable formulation set. In the EU, cosmetic products containing nanomaterials face a specific notification regime, including a lead-time requirement before market placement, which raises the value of suppliers that can maintain complete nanomaterial characterisation and safety files across frequent line extensions.
China will remain a high-velocity scaling engine because online retail infrastructure supports fast replenishment loops, while efficacy-claim substantiation requirements shift brands toward documented performance positioning. The NMPA’s efficacy-claim evaluation standard formalises how cosmetic efficacy claims must be supported and disclosed, which structurally favours clay systems that can be defended through repeatable skin outcomes rather than sensory narratives.
Portfolio investment by large beauty groups will keep pulling the market toward science-led masks and clinical positioning. L’Oréal’s move to acquire a majority stake in Medik8 is framed as a scale play in science-driven skincare, with L’Oréal Luxe leadership stating: ‘We are delighted to welcome Medik8 to the L’Oréal family.’
| Metrics | Values |
|---|---|
| Market Value (2026) | USD 0.9 Billion |
| Market Value (2036) | USD 4.1 Billion |
| CAGR (2026 to 2036) | 16.3% |
Future Market Insights projects the nano clay masks market to grow from USD 0.9 billion in 2026 to USD 4.1 billion by 2036, at a 16.3% CAGR.
FMI Research Approach: Forecasting anchored in nanomaterial governance, sebum-management clinical signals, and DTC replenishment behaviour.
FMI sees the market shifting toward substantiated performance masks where clay selection, particle engineering, and safety documentation determine how fast brands can scale across regulated channels.
FMI Research Approach: Mapping EU and UK nanomaterial notification lead-times and country-level compliance costs into portfolio velocity.
China will remain the fastest growth engine, supported by claims substantiation governance and a large online retail base that sustains repeat mask consumption.
FMI Research Approach: Country-level CAGR modelling aligned with NMPA efficacy disclosure systems and online retail throughput.
Purifying nano clay masks lead with a 39% share, reflecting demand concentration in oil-control and pore-management routines.
FMI Research Approach: Segment modelling based on acne and sebum-management usage frequency and retailer replenishment data.
Montmorillonite nano clay leads with a 45.3% share due to adsorption-led performance logic and wide formulation compatibility in rinse-off masks.
FMI Research Approach: Ingredient system mapping using adsorption function and compliance feasibility under nanomaterial safety assessment frameworks.
Nano clay masks will scale because regulatory systems are tightening claim discipline while consumers and clinics are normalising repeatable oil-control routines. In China, NMPA’s efficacy-claim evaluation standard increases the commercial value of masks that can be supported through structured substantiation rather than broad detox language. In Europe, cosmetics containing nanomaterials face mandatory notification and safety expectations that raise the barrier for undisciplined entrants and concentrate growth into compliance-ready portfolios. Clinical-use logic reinforces demand because clay-based masks have been evaluated for acne and oiliness management in controlled settings, validating their positioning as functional skin-management tools rather than seasonal pampering products.
The nano clay masks market is segmented by product type, clay type, skin type, distribution channel, end user, and region to reflect how performance claims, ingredient systems, and access channels determine repeat purchase behaviour. Product type captures outcome-led positioning such as purifying or hydrating masks. Clay type reflects adsorption and tolerability trade-offs across montmorillonite, kaolin, bentonite, and multimineral systems. Skin type segmentation reflects differential sebum burden and sensitivity constraints. Distribution channel segmentation captures the role of direct digital replenishment versus clinic and pharmacy trust infrastructure. End user segmentation reflects routine intensity differences across women, men, and unisex protocols.
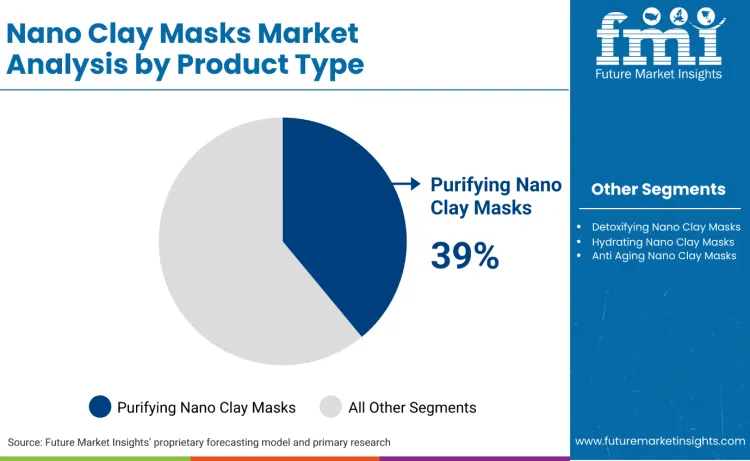
Purifying nano clay masks lead with a 39% market share because the category’s highest-frequency use case is oil-load reduction and pore management, where consumers repeat purchase within short cycles. This creates a structural advantage versus detox and anti-ageing mask narratives that rely more on episodic usage. Clinical and dermatology literature supports the functional logic of clay systems for acne-prone and oily presentations, reinforcing purifying formats as the default entry point for first-time users and as the maintenance protocol for repeat buyers. Governance amplifies this dominance because performance claims in regulated markets increasingly need substantiation. In China, the NMPA efficacy-claim evaluation standard pushes brands toward measurable outcomes and disclosure discipline, which aligns more naturally with sebum-management and pore-appearance claims than with ambiguous detox framing. Scaled groups also reinforce the structure by allocating R&D and marketing resources to science-led skincare platforms, which tends to prioritise high-repeat, high-proof product stories that can travel across channels and geographies.
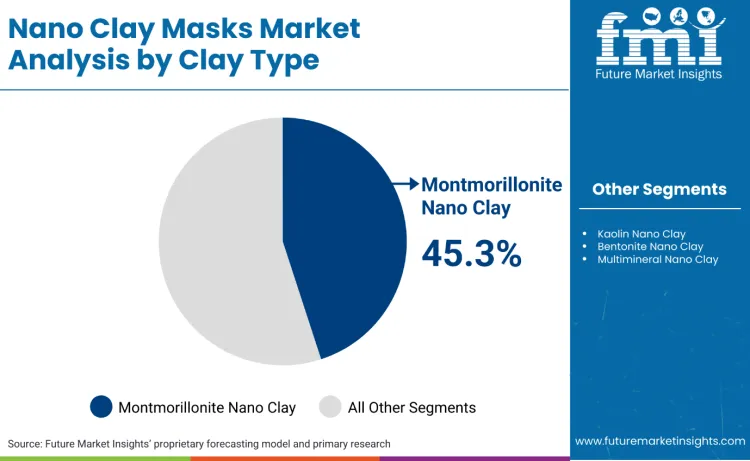
Montmorillonite nano clay leads the clay type segment with a 45.3% share because adsorption-driven performance and high surface interaction translate into visible outcomes that support repeat purchase and premium positioning. Montmorillonite is widely documented as an adsorptive clay mineral used for binding and removal of compounds, which directly maps to consumer expectations for oil-control and deep-clean narratives in rinse-off masks. The dominance is reinforced by compliance reality. Where nanomaterial scrutiny is high, brands prefer ingredient systems that can be characterised, standardised, and supported through stable supplier documentation across multiple SKUs. EU frameworks explicitly require notification lead-times and safety assessment expectations for nanomaterials, so portfolio teams tend to concentrate on clay systems with established characterisation pathways rather than fragmenting across too many novel particulate variants. This concentrates supplier qualification, reduces reformulation churn, and enables multi-market rollouts without repeated claim resets.
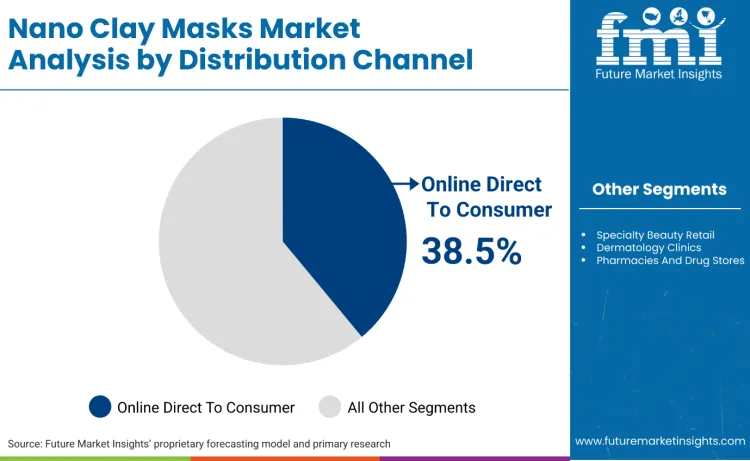
Online direct to consumer leads the distribution mix with a 38.5% share because masks are replenishment-led products that benefit from subscription, bundling, and rapid repeat ordering rather than one-time store browsing. China’s macro retail data shows online retail sales of physical goods at scale, supporting the conversion of skincare masks into routine replenishment items that are reordered with high predictability once users identify a tolerable clay system. Regulatory mechanics also shape channel concentration. Where products contain nanomaterials, notification lead-times and documentation requirements reward brands that can control product information flows, update dossiers fast, and maintain traceability across frequent marketing refresh cycles. In Great Britain, products containing certain nanomaterials require notification processes with explicit timing rules before market placement, which further strengthens the strategic value of controlled, authorised online selling environments. The net effect is a market where DTC becomes the scaling engine while clinics and pharmacies provide credibility in selected geographies.
Nano clay masks will shift toward compliance-led performance positioning as regulators formalise nanomaterial safety expectations and claims substantiation. In the EU, nanomaterial notification requirements and SCCS-linked safety assessment expectations create a predictable compliance gate that favours structured dossiers and stable particle characterisation. In the US, FDA has issued guidance addressing safety considerations for nanomaterials in cosmetics, which reinforces the need for risk-based substantiation and supplier traceability in particulate systems. These governance signals will push portfolios toward fewer, better-documented nano clay systems and will strengthen premiumisation where documentation supports higher price points.
The same governance that enables trust will slow SKU velocity and raise operating cost, limiting fast followers. Nanomaterial notification lead-times in Europe and Great Britain introduce time-to-market friction for new particulate variants, constraining rapid iteration and making reformulation cycles more expensive. In the US, MoCRA increases compliance overhead through facility registration, product listing, and adverse event reporting expectations, which shifts resources away from rapid launch cadence toward regulatory operations. Smaller brands without quality systems will face constrained scaling, even when consumer demand exists.
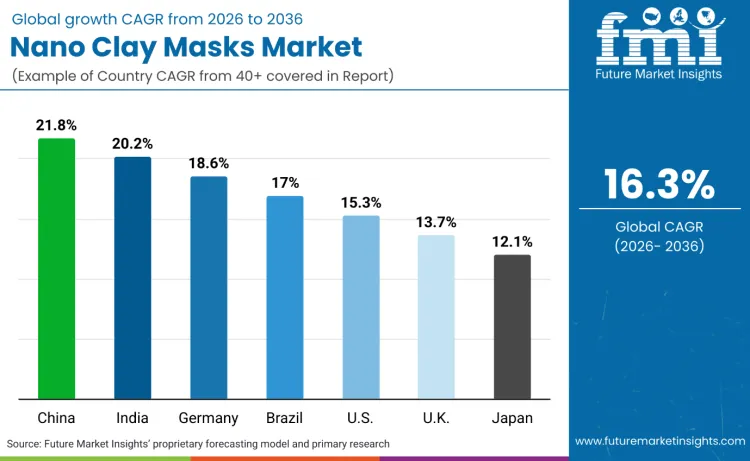
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 21.8% |
| India | 20.2% |
| Germany | 18.6% |
| Brazil | 17.0% |
| USA | 15.3% |
| UK | 13.7% |
| Japan | 12.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
China’s 21.8% CAGR is anchored in an online retail system that industrialises replenishment and in governance that rewards substantiated efficacy narratives. National Bureau of Statistics data shows online retail sales of physical goods at very large scale, supporting rapid reorder loops once users settle on a clay system that fits oily or combination skin needs. The regulatory mechanism reinforces performance framing. NMPA’s efficacy-claim evaluation standard formalises how efficacy claims must be supported and disclosed, pushing brands toward documented oil-control and pore-appearance outcomes that map directly to purifying nano clay masks. This combination favours scale players that can fund testing, maintain documentation across frequent SKU refresh cycles, and deploy DTC conversion infrastructure to convert trial into subscription-like repeat usage through 2036.
India’s 20.2% CAGR will be driven by regulatory formalisation and expanding digital beauty fulfilment beyond tier one cities. The Cosmetics Rules, 2020 establish licensing, import registration, and inspection expectations that increase accountability for labelling and product governance, which matters for nano clay masks because particulate claims and tolerability narratives must remain consistent across batches and suppliers. As compliance discipline improves, premium brands gain confidence to scale performance-led masks through authorised online channels and dermatology-adjacent retail, converting periodic acne and oiliness management into routine purchases. The growth mechanism is not general skincare expansion. It is reduced friction for compliant portfolios and improved consumer trust in traceable ingredient stories through the forecast period.
Germany’s 18.6% CAGR will be supported by high consumer spend capacity in beauty and home care and by EU compliance structures that funnel premium purchase into trusted retail and pharmacy-adjacent ecosystems. IKW reports record consumer spending on beauty and home care products in 2024, confirming demand headroom for higher-value skincare routines. EU Cosmetics Regulation requirements around product safety documentation and the specific notification regime for nanomaterials raise the barrier for low-discipline entrants, concentrating shelf space and clinic recommendation into compliance-ready brands. This environment rewards nano clay masks positioned for oily and combination skin management with conservative claims architecture, supporting sustained growth without relying on procedure-led dynamics.
Brazil’s 17.0% CAGR will be driven by regulated market access pathways that stabilise premium import and local manufacturing decisions, enabling broader distribution through pharmacies, clinics, and authorised digital channels. ANVISA’s guidance frames RDC 907/2024 as the governing structure for marketing authorisation logic, clarifying which products require pre-marketing approval versus notification and strengthening compliance discipline for companies placing cosmetics on the market. For nano clay masks, this matters because particulate systems and claims architecture benefit from consistent governance and traceability expectations, reducing disruption from enforcement uncertainty. As compliance stabilises, brands can scale repeat-use masks as part of acne and oil-control routines through 2036, with pharmacies and clinic-adjacent channels acting as trust infrastructure.
The USA will expand at a 15.3% CAGR because regulatory uplift and nanomaterial safety guidance increase trust, favouring scaled operators that can industrialise repeat purchase through premium retail and pharmacy channels. FDA guidance explicitly addresses safety considerations for nanomaterials in cosmetic products, reinforcing the need for structured assessment frameworks in particulate systems. MoCRA further increases operational requirements via facility registration, product listing, and adverse event reporting expectations, shifting the competitive balance toward compliance-ready brands with mature quality systems. These governance mechanisms will support premiumisation and consolidation, keeping nano clay masks positioned as routine skin management tools rather than novelty wellness items.
The UK’s 13.7% CAGR will be shaped by post-Brexit product governance that rewards brands capable of running Great Britain compliance processes with disciplined documentation control. Companies must notify cosmetics through the UK notification system before products are made available in Great Britain, and products containing certain nanomaterials face specific guidance on when they can be placed on the market after notification. For nano clay masks, this reinforces conservative claim architecture, controlled portfolio expansion, and authorised channel concentration, because traceability and documentation speed become commercial differentiators. DTC will still scale the category, but growth will concentrate in brands that can manage compliance timelines while maintaining repeat purchase through online replenishment loops.
Japan’s 12.1% CAGR will reflect strict ingredient governance and category discipline that constrain aggressive performance positioning in particulate skincare. The MHLW Standards for Cosmetic Products specify restricted and prohibited ingredients and define compliance expectations, reinforcing conservative formulation and labelling practice for imported and domestic brands. Classification discipline further shapes go-to-market because products that drift toward functional drug-like claims can trigger quasi-drug regulatory pathways, favouring established operators with local regulatory capability. These mechanisms support steady adoption of nano clay masks in oily and combination skin routines while moderating the pace of claim-led acceleration through 2036.
Competition in nano clay masks is structured around compliance readiness for nanomaterials, claims substantiation capability, and distribution control in DTC and pharmacy-adjacent channels. Scope includes rinse-off facial masks using nano-engineered clay systems for purifying, detoxifying, hydrating, and anti-ageing positioning. Scope excludes LED masks, sheet masks without clay actives, peel-off polymer masks, and professional-only in-clinic chemical peels.
L’Oréal Advanced Research is positioned as the largest global player in this category, supported by scale in science-led skincare portfolio building and repeated investment in dermatological and clinical positioning platforms, including acquisition-led expansion in science-driven skincare. In North America, compliance uplift under MoCRA and FDA nanomaterial safety guidance favours large multi-brand operators and prestige platforms with regulatory infrastructure. In Europe, EU nanomaterial notification regimes and SCCS safety assessment expectations concentrate share into brands that can maintain stable dossiers and control authorised channel execution. In Asia, China’s efficacy-claim governance and online retail scale accelerate portfolio velocity, while Japan’s ingredient standards and classification discipline mean global leaders do not automatically translate into domestic leadership, where Shiseido and Kao maintain structural advantages through local governance capability and IP-driven skin science programs.
Recent Developments
Nano clay masks are rinse-off facial mask products formulated with nano-engineered clay systems designed to manage sebum, impurities, pore appearance, and skin comfort through adsorption-led mechanisms. The market covers consumer and dermatology-adjacent retail products positioned for purifying, detoxifying, hydrating, and anti-ageing outcomes where the primary functional active is a clay system, including montmorillonite, kaolin, bentonite, and multimineral clay variants in nano or fine-particle formats. Value capture is driven by compliance-ready formulation, substantiated claims, and repeat purchase cycles enabled by digital replenishment and trusted health-adjacent distribution.
Included products comprise rinse-off nano clay masks sold via online direct to consumer, specialty beauty retail, dermatology clinics, and pharmacies or drug stores, across women, men, and unisex end users. Included formulations use nano or fine-particle clays as the core functional system and are marketed for oily skin, dry skin, combination skin, or sensitive skin routines. Included activity spans at-home maintenance masks linked to acne-prone or oil-control regimens, pore-appearance management, and hydration balancing, where brands maintain documentation for ingredient safety and claims substantiation under relevant national cosmetics governance.
Excluded products include LED or device-based facial masks, sheet masks without clay actives, peel-off polymer masks, exfoliating chemical peel systems, professional-only clinical masks not sold as consumer products, and clay-free mud masks that do not use nano or engineered particulate clays. Also excluded are cleansers and scrubs where clay is secondary and not positioned as the primary functional active, and topical leave-on creams or serums using unrelated nanomaterials without a clay mask format. Revenue from broader skincare platforms is excluded unless it is directly attributable to nano clay mask SKUs.
| Attributes | Details |
|---|---|
| Market Size (2026) | USD 0.9 billion |
| Market Size (2036) | USD 4.1 billion |
| CAGR (2026 to 2036) | 16.3% |
| Forecast Period | 2026 to 2036 |
| Historical Data Available for | 2021 to 2025 |
| Quantitative Units | USD billion for value and CAGR for growth |
| Report Coverage | Market sizing, forecast, segment analysis, country analysis, competitive landscape, and regulatory signals |
| Segments Covered | Product type, clay type, skin type, distribution channel, end user, and region |
| Regional Scope | North America, Latin America, Europe, South Asia, East Asia |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan |
| Key Companies Profiled | L’Oréal Advanced Research, Amorepacific Skin Science, Shiseido Professional, Dr G Skincare, Innisfree Advanced Labs, COSRX Laboratories, Dermalogica, Murad Skincare, Peter Thomas Roth, Glow Recipe |
| Additional Attributes | Nanomaterial governance mapping including EU and UK notification lead-times, SCCS-linked safety assessment expectations, and FDA nanomaterial safety guidance. Claims substantiation and disclosure assessment with emphasis on China’s efficacy-claim evaluation standard. Channel concentration analysis across online DTC, pharmacy and drug retail, and dermatology clinic dispensing. Ingredient system risk mapping focused on particle characterisation, traceability readiness, and dossier maintenance across frequent SKU refresh cycles. Country-specific compliance friction analysis, including MoCRA-driven operational uplift in the USA and ANVISA regularisation pathways in Brazil. Competitive strategy signals based on acquisition-led science-skincare scaling, R&D emphasis, and IP-led skin science programs disclosed in company filings. |
Purifying Nano Clay Masks; Detoxifying Nano Clay Masks; Hydrating Nano Clay Masks; Anti Aging Nano Clay Masks
Montmorillonite Nano Clay; Kaolin Nano Clay; Bentonite Nano Clay; Multimineral Nano Clay
Oily Skin; Dry Skin; Combination Skin; Sensitive Skin
Online Direct To Consumer; Specialty Beauty Retail; Dermatology Clinics; Pharmacies And Drug Stores
Women; Men; Unisex
North America; Latin America; Europe; South Asia; East Asia
What will be the nano clay masks market size in 2036?
The market is projected to reach USD 4.1 billion by 2036, from USD 0.9 billion in 2026, at a 16.3% CAGR.
Which product type leads the nano clay masks market in 2026?
Purifying nano clay masks lead with a 39% share due to repeatable oil-control routines and substantiated performance positioning.
Which clay type dominates formulation choice in the nano clay masks market?
Montmorillonite nano clay leads with a 45.3% share, supported by adsorption-led performance logic and compliance-ready dossier feasibility.
Which distribution channel is most important for scale in nano clay masks?
Online direct to consumer leads with a 38.5% share, driven by replenishment loops and authorised channel control under nanomaterial governance.
Which country grows fastest in nano clay masks through 2036?
China is the fastest growth market at a 21.87% CAGR, supported by efficacy-claim governance and large-scale online retail throughput.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.