The peripheral intravascular lithotripsy market is expected to grow from USD 1.8 billion in 2026 to USD 5.5 billion by 2036, reflecting a compound annual growth rate (CAGR) of 11.5%. Peripheral intravascular lithotripsy (PIVL) is a medical procedure used to treat calcified peripheral artery disease (PAD) by using sonic waves to break down calcium deposits in the arteries, allowing for better blood flow. The market’s growth is driven by the increasing prevalence of PAD, advancements in endovascular technologies, and the rising demand for minimally invasive procedures. As the healthcare sector continues to shift toward less invasive treatments that offer faster recovery times and lower risks, PIVL is gaining widespread adoption.
The growth of this market is further supported by the increasing number of patients with cardiovascular diseases, an aging population, and the growing availability of advanced medical technologies for treating complex vascular conditions. The adoption of PIVL is also accelerating as it is shown to improve procedural success rates and patient outcomes in peripheral artery interventions.
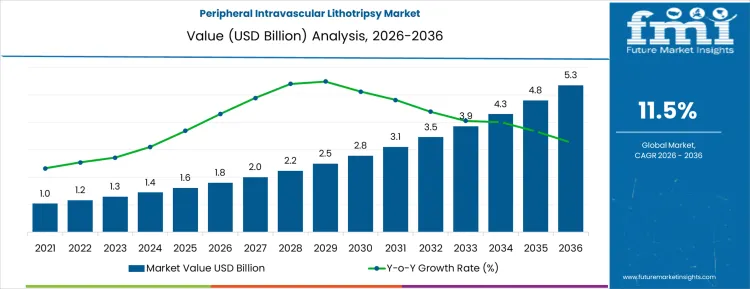
The year-on-year (YoY) growth analysis for the peripheral intravascular lithotripsy market indicates steady progress, with noticeable acceleration in the later years of the forecast period. Starting at USD 1.8 billion in 2026, the market sees gradual growth to USD 2.1 billion in 2027 and USD 2.3 billion in 2028. These early increases reflect early adoption and integration of PIVL technologies into clinical settings as healthcare providers become familiar with its benefits in treating calcified arterial blockages.
From 2028 to 2030, the market continues to expand at an increasing pace, reaching USD 2.6 billion in 2029 and USD 2.8 billion in 2030, marking a period of faster adoption as clinical evidence supporting PIVL’s effectiveness grows. Between 2030 and 2035, the market shows more substantial acceleration, reaching USD 3.2 billion in 2031, USD 3.5 billion in 2032, and USD 3.9 billion in 2033. The final phase, from 2033 to 2036, is expected to see continued growth, with the market reaching USD 4.4 billion in 2035 and USD 5.5 billion by 2036. The YoY growth analysis highlights how the market benefits from greater adoption, driven by increased awareness, improved procedural outcomes, and expanding clinical applications of PIVL in vascular treatments.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 1.8 billion |
| Industry Forecast Value (2036) | USD 5.5 billion |
| Industry Forecast CAGR (2026-2036) | 11.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
The global demand for peripheral intravascular lithotripsy (IVL) is driven by rising prevalence of peripheral artery disease (PAD) and the need for minimally invasive solutions to treat calcified lesions in peripheral vessels. PAD affects arteries outside the heart and brain, most commonly in the legs, reducing blood flow and causing pain, disability, and risk of limb loss. Traditional endovascular tools such as angioplasty and atherectomy can be less effective or carry higher complication risks when severe calcification is present.
IVL uses acoustic pressure waves to fracture calcium within the vessel wall, facilitating vessel expansion with lower procedural force and reducing trauma to surrounding tissues. Clinicians and interventional specialists increasingly adopt IVL because it can improve procedural success, reduce vessel injury, and expand treatment options for patients who might otherwise face more invasive surgery. Growth in diagnostic imaging and screening for PAD, increasing clinician familiarity with advanced endovascular tools, and demographic trends such as ageing populations and rising diabetes rates all contribute to IVL uptake.
Future demand for peripheral IVL is expected to grow as evidence of clinical benefit and long term safety accumulates and as device technology continues to improve. Continued innovation in catheter design, energy delivery, and compatibility with adjunctive therapies will enhance usability and expand the range of treatable lesion types. Broader adoption may be supported by guidelines that recognise IVL as a viable option for heavily calcified lesions and by reimbursement frameworks that reflect value from reduced complications and shorter hospital stays.
Expansion of vascular care infrastructure in emerging markets will also support demand as access to advanced endovascular technologies improves. Education and training for interventionalists in optimal IVL use, integration with multimodality imaging, and real world evidence demonstrating improved outcomes will further shape market growth. As healthcare systems prioritise less invasive, durable treatments for peripheral vascular disease, the peripheral IVL market is expected to expand steadily. Future demand will be influenced by how quickly newer indications and evidence translate into wider clinical practice globally.
The peripheral intravascular lithotripsy (IVL) market is driven by several key indications and device components, each contributing to its overall growth. Among the indications, peripheral artery disease (PAD) stands out with a substantial share, accounting for 62% of the market, while dialysis access lesions and coronary indications also play significant roles. In terms of device components, IVL catheters are the most prominent, comprising 68% of the market. The market is also supported by generator/console systems and accessories/connectors. These segments are shaped by regional demand across various healthcare settings, including hospitals and specialized treatment centers, with North America and Europe leading in adoption. Understanding the dynamics of these key segments reveals insights into the growth potential and direction of the market, highlighting the role of each component and indication in addressing vascular challenges.
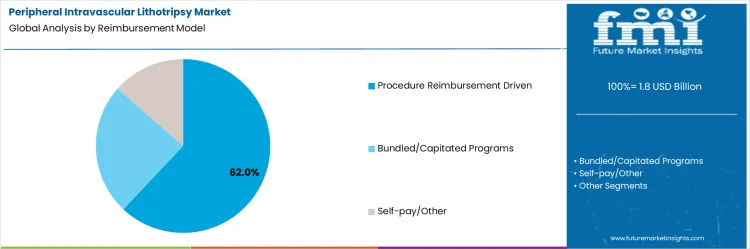
The peripheral artery disease (PAD) segment remains the largest and most dominant within the peripheral intravascular lithotripsy market, accounting for 62% of the total market share. PAD is one of the leading causes of morbidity, especially among older populations, making it a significant driver of demand for IVL technologies. The increasing prevalence of PAD, particularly in developed regions such as North America and Europe, has led to a growing need for effective treatment options. IVL, with its ability to safely treat calcified plaques in peripheral arteries, offers a compelling alternative to traditional interventions like balloon angioplasty and stenting.
This is especially true for patients with complex, heavily calcified lesions where conventional methods may fall short. The growth of the PAD segment is also supported by advancements in IVL technology, which have enhanced the precision and safety of treatments. Moreover, as healthcare providers focus on improving patient outcomes and reducing complications, the demand for non-invasive, minimally disruptive treatments like IVL is expected to rise. PAD’s high prevalence, combined with the benefits of IVL, positions this segment for continued growth, particularly as awareness and adoption of the technology increase in regions with aging populations.
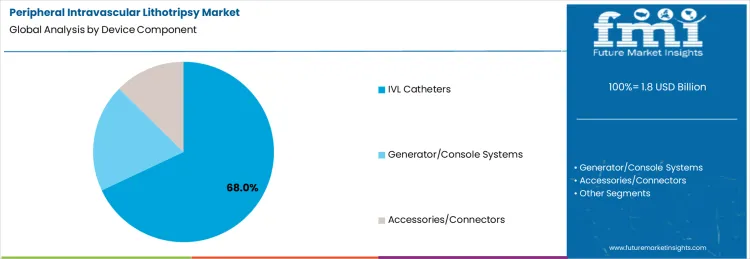
IVL catheters are the most critical device component in the peripheral intravascular lithotripsy market, comprising 68% of the market share. The demand for IVL catheters is largely driven by their pivotal role in the successful treatment of calcified vascular lesions, a common condition in patients with peripheral artery disease (PAD), dialysis access lesions, and other vascular conditions. These catheters deliver localized lithotripsy therapy to break up calcium deposits in the arterial walls, facilitating improved blood flow and reducing the need for more invasive procedures. The adoption of IVL catheters is growing rapidly due to their ability to treat complex lesions with minimal trauma and faster recovery times compared to traditional methods.
The advanced design and functionality of IVL catheters contribute to their widespread use, particularly in interventional cardiology and vascular surgery. Additionally, as healthcare systems continue to focus on reducing healthcare costs and improving patient outcomes, IVL catheters provide an effective solution for managing challenging vascular diseases. The ongoing development of more user-friendly catheter designs and increased clinician familiarity with the technology are expected to further drive the adoption of IVL catheters, securing their position as the leading device component in the peripheral intravascular lithotripsy market. The growth of this segment will be critical in expanding the overall reach and effectiveness of IVL therapies across a wide range of indications.
The global peripheral intravascular lithotripsy market is expanding as clinicians adopt new technologies to treat calcified peripheral arterial disease in the legs, pelvis and other extremities. Intravascular lithotripsy uses sonic pressure waves to fracture hardened plaque, improving vessel compliance and enabling safer dilation. Rising incidence of peripheral artery disease, greater screening efforts and preference for minimally disruptive interventions support demand. Hospitals, vascular centres and specialty clinics are incorporating these systems into treatment offerings to enhance procedural outcomes and patient comfort across diverse geographies.
Wider recognition of the clinical challenge posed by heavily calcified peripheral arteries motivates adoption of technologies that facilitate effective revascularisation. The ageing global population and increasing rates of diabetes and chronic cardiovascular conditions contribute to higher peripheral artery disease caseloads, spurring interest in advanced treatment platforms. Clinicians value intravascular lithotripsy for improved vessel expansion with reduced trauma relative to traditional atherectomy or high pressure ballooning. Investments in vascular care infrastructure and rising access to interventional procedures in outpatient and hospital settings further support uptake. Evidence emerging from clinical studies that highlight procedural safety and performance assists specialists in selecting appropriate therapies for complex lesions.
Uptake is constrained in some settings by the capital expenditure required for acquisition of specialised lithotripsy generators and compatible catheters. Hospitals and clinics with budget limitations may prioritise conventional treatment options, which have lower initial cost. Training requirements for interventional teams and inclusion of new procedural protocols into established practice routines can be resource intensive. Variation in reimbursement frameworks across countries and uncertainty around payment for advanced vascular therapies influences decision making among providers. Supply chain limitations for consumable catheters and regulatory clearance timelines in certain regions also affect availability and speed of market entry.
Innovation is focusing on enhanced catheter designs that improve deliverability, lesion crossing and sonic wave transmission to target calcified segments with precision. Integration of imaging guidance tools such as intravascular ultrasound and optical coherence tomography with lithotripsy planning supports more tailored interventions. Expansion of evidence from prospective registries encourages confidence among vascular specialists and informs best practice guidelines. Collaboration between device makers and clinical research centres is increasing, with an emphasis on long term outcomes and broader lesion subsets. Programmes offering practitioner education and procedural training are growing, helping to accelerate adoption and optimisation of intravascular lithotripsy in peripheral artery disease management.
The peripheral intravascular lithotripsy (PIL) market analysis reveals significant growth prospects across key regions, with varying growth rates based on local demand, technological adoption, and regulatory factors. The compound annual growth rates (CAGRs) for each country demonstrate the market’s expansion potential in different regions. By understanding these forecasts, businesses can optimize strategies for each country, targeting high-growth markets while considering regional challenges. Factors such as increasing cardiovascular diseases, the need for minimally invasive procedures, and technological advancements are contributing to the market's expansion. By understanding the country-specific trends and forecasts, businesses can position themselves to meet the increasing demand for innovative medical solutions.
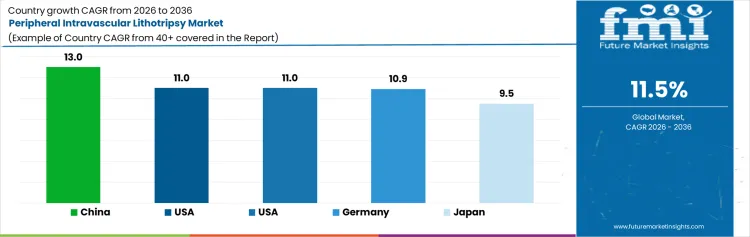
| Country | CAGR (2026-2036) |
|---|---|
| China | 13% |
| United States | 11% |
| United Kingdom | 11% |
| Germany | 10.9% |
| Japan | 9.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
The peripheral intravascular lithotripsy market in China is projected to experience the highest growth, with a CAGR of 13%. This robust expansion is driven by China’s growing healthcare infrastructure, rising cardiovascular disease rates, and increasing demand for minimally invasive procedures. The market benefits from government policies that promote innovation in medical technology and healthcare services, along with an expanding middle class that is increasingly seeking advanced medical treatments. Moreover, China’s active investment in healthcare modernization and the adoption of cutting-edge technologies support the rapid growth of the PIL market. The country’s significant population, combined with a focus on healthcare improvement, makes China an attractive market for companies in the medical device industry.
The market for peripheral intravascular lithotripsy in the United States is expected to grow at a steady pace, with a projected CAGR of 11%. This growth is driven by the increasing prevalence of cardiovascular diseases and the rising demand for less invasive treatment options. The USA remains a leader in healthcare innovation, and advancements in PIL technology are expected to contribute to further market growth. Regulatory support and widespread access to cutting-edge medical treatments also provide a conducive environment for market expansion. The well-established healthcare system and a high level of awareness among patients are expected to drive adoption of PIL devices, further solidifying the USA as a key market in the sector.
The peripheral intravascular lithotripsy market in the United Kingdom is forecasted to grow at a CAGR of 11%. The UK's healthcare system, which is among the most advanced globally, supports the adoption of innovative medical technologies, including PIL. The rise in cardiovascular conditions and the country’s focus on improving patient outcomes through minimally invasive techniques are driving market demand. Furthermore, the National Health Service (NHS) plays a vital role in the distribution of medical technologies, ensuring accessibility and widespread adoption. Regulatory standards and guidelines also foster an environment conducive to market growth. As a result, the UK is expected to see a steady increase in the adoption of PIL treatments.
The market for peripheral intravascular lithotripsy in Germany is anticipated to grow at a moderate pace, with a projected CAGR of 10.9%. Germany’s strong healthcare system, combined with its reputation for medical innovation, provides a favorable environment for the adoption of PIL technologies. Cardiovascular diseases remain a significant health concern in the country, driving the demand for effective treatment options like PIL. As healthcare facilities in Germany continue to integrate advanced medical devices, the market for PIL is expected to grow. The country's regulatory environment supports the introduction of innovative healthcare solutions, and the demand for less invasive treatment alternatives further contributes to market growth.
The peripheral intravascular lithotripsy market in Japan is expected to grow at a CAGR of 9.5%. Japan’s aging population and the rising incidence of cardiovascular diseases are key factors contributing to the market’s growth. The country has a well-established healthcare infrastructure that facilitates the adoption of advanced medical technologies like PIL. Additionally, Japan’s strong focus on minimally invasive medical procedures, along with the increasing demand for better patient outcomes, drives the need for innovative solutions. As technological advancements in medical devices continue to evolve, Japan is expected to see gradual growth in the PIL market, supported by a highly regulated and efficient healthcare environment.
The peripheral intravascular lithotripsy market is growing rapidly, with companies focusing on advanced technologies to treat peripheral artery disease (PAD). Shockwave Medical (Johnson & Johnson) leads the market with its innovative intravascular lithotripsy system, which uses sonic pressure waves to treat calcified lesions in peripheral arteries. Their focus on improving patient outcomes with minimally invasive procedures has positioned them at the forefront of this market. Boston Scientific, Medtronic, and Abbott are strong competitors, each offering advanced technologies in vascular intervention. Boston Scientific provides a range of peripheral artery disease treatment options, while Medtronic offers intravascular lithotripsy devices designed to treat complex arterial conditions. Abbott also offers peripheral vascular interventions, combining their established reputation in cardiology with advanced lithotripsy systems.
Cook Medical, Terumo, BD, B. Braun, Cardinal Health (devices), and Merit Medical further contribute to the competitive dynamics. Cook Medical offers a variety of devices for vascular procedures, including treatments for calcified lesions. Terumo and BD provide solutions focused on improving vascular access and interventional therapies. B. Braun and Cardinal Health offer a broad portfolio of devices for peripheral interventions, while Merit Medical specializes in providing high-quality vascular products. These companies compete through the development of cutting-edge, minimally invasive devices that improve clinical outcomes and expand treatment options for patients suffering from peripheral artery disease. Their strategies are centered on advancing technological capabilities, enhancing safety, and providing effective solutions for complex vascular conditions.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Billion |
| Indication | Peripheral Artery Disease (PAD), Dialysis Access (AVF/AVG) Lesions, Coronary (adjacent adoption), Other Indications |
| Device Component | IVL Catheters, Generator/Console Systems, Accessories/Connectors |
| Care Setting | Hospital Cath Labs, Ambulatory / OBL Centers, Specialty Vascular Centers |
| Reimbursement/Payment Model | Procedure Reimbursement Driven, Bundled/Capitated Programs, Self-pay/Other |
| Companies | Shockwave Medical (Johnson & Johnson), Boston Scientific, Medtronic, Abbott, Cook Medical, Terumo, BD, B. Braun, Cardinal Health (devices), Merit Medical |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar by sales by indication, device component, care setting, reimbursement/payment model, and region. Includes market trends in peripheral intravascular lithotripsy, focusing on the role of IVL catheters, generator/console systems, and accessories/connectors in treating peripheral artery disease (PAD), dialysis access lesions, and coronary applications. Focus on reimbursement models, regulatory compliance, cost-effectiveness, market share, and competitive positioning of key companies. |
How big is the peripheral intravascular lithotripsy market in 2026?
The global peripheral intravascular lithotripsy market is estimated to be valued at USD 1.8 billion in 2026.
What will be the size of peripheral intravascular lithotripsy market in 2036?
The market size for the peripheral intravascular lithotripsy market is projected to reach USD 5.3 billion by 2036.
How much will be the peripheral intravascular lithotripsy market growth between 2026 and 2036?
The peripheral intravascular lithotripsy market is expected to grow at a 11.5% CAGR between 2026 and 2036.
What are the key product types in the peripheral intravascular lithotripsy market?
The key product types in peripheral intravascular lithotripsy market are peripheral artery disease (pad), dialysis access (avf/avg) lesions, coronary (adjacent adoption) and other indications.
Which device component segment to contribute significant share in the peripheral intravascular lithotripsy market in 2026?
In terms of device component, ivl catheters segment to command 68.0% share in the peripheral intravascular lithotripsy market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.