The global pharma grade synthetic camphor market is expected to show steady and dependable growth over the coming decade. Valued at USD 484.1 million in 2026, the industry is projected to reach USD 703.0 million by 2036, expanding at a value based CAGR of 3.8%. This growth is mainly supported by the continued use of synthetic camphor in pharmaceutical formulations such as pain relief balms, topical ointments, inhalants, and respiratory products. Compared with natural camphor, the synthetic variant is widely preferred because it offers consistent purity, reliable quality, and stable availability, all of which are essential for pharmaceutical grade applications. Asia Pacific plays a central role in both production and consumption, driven by strong pharmaceutical manufacturing activity in China and India, while North America and Europe contribute steady demand through regulated healthcare markets.
Rising demand is closely linked to the broad therapeutic use of pharma grade synthetic camphor as a counterirritant, mild analgesic, and decongestant. Its effectiveness in providing quick, localized relief keeps it relevant in both prescription medicines and over the counter healthcare products. Pharmaceutical manufacturers also favor synthetic camphor because it meets pharmacopeia standards more consistently, supporting quality assurance and regulatory compliance. Growing consumer preference for topical treatments that offer fast and convenient relief further reinforces long term demand.
The industry faces challenges related to regulatory oversight, quality control, and cost pressures. Manufacturers must comply with strict purity and documentation requirements, which adds to production complexity. Fluctuations in raw material and energy costs can also affect margins. In response, producers are focusing on process efficiency, stronger quality systems, and more sustainable manufacturing practices to maintain competitiveness and ensure reliable supply across global pharmaceutical markets.
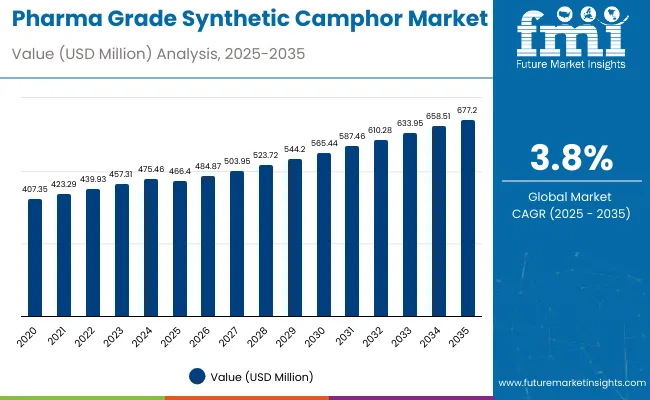
| Metric | Value |
|---|---|
| Market Value (2026) | USD 484.1 million |
| Market Forecast Value (2036) | USD 703.0 million |
| Forecast CAGR (2026-2036) | 3.8% |
The pharma grade synthetic camphor market is segmented based on formulation type, application, distribution channel, and region. By formulation type, the industry is divided into topical and inhalation forms. Based on application, demand is categorized into respiratory disorders, muscular rheumatism, counterirritant and antipruritic uses, and other applications such as pain relief, skin care, and miscellaneous therapeutic uses. In terms of distribution channel, the industry includes conventional B2B channels such as tenders, direct procurement, and wholesalers and distributors, as well as online B2B channels through internal and external platforms. Regionally, the industry spans North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, and the Middle East and Africa.
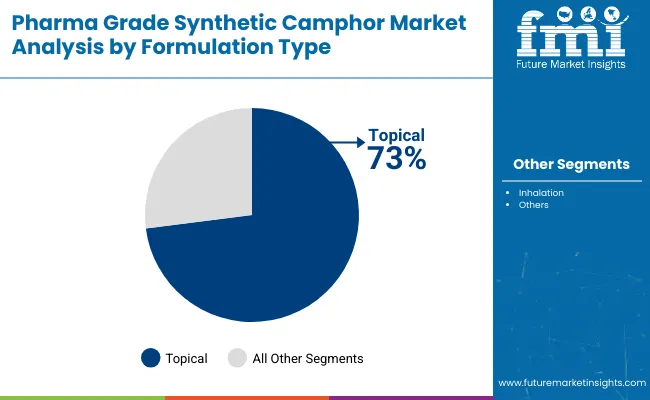
Topical formulations account for around 73% of global demand, making them the dominant formulation type in the pharma grade synthetic camphor market in 2026. This leadership is driven by the widespread use of synthetic camphor in creams, ointments, balms, and gels designed for pain relief, muscle relaxation, and anti-inflammatory support. Camphor’s ability to penetrate the skin and act as a counterirritant enhances blood circulation and provides a cooling sensation, making it highly effective for conditions such as arthritis, sprains, back pain, and muscular fatigue. Its antiseptic and antipruritic properties further expand its use in dermatological products for skin irritation, rashes, and insect bites. Growing consumer preference for over-the-counter, non-invasive, and fast-acting topical therapies continues to reinforce the dominance of this segment across developed and emerging markets.
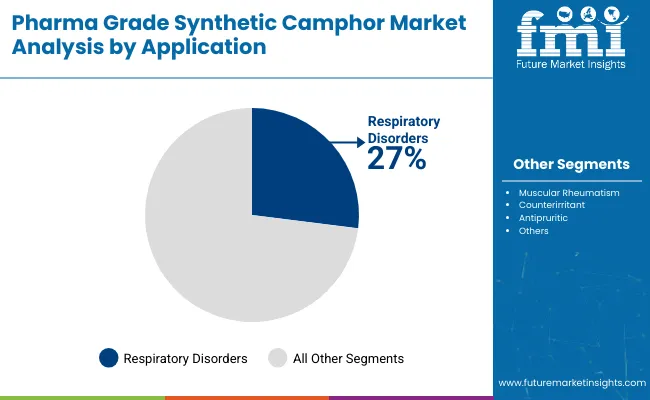
Respiratory disorders represent the largest application segment, accounting for approximately 27% of market demand in 2026. Synthetic camphor is widely used in inhalants, vapor rubs, and topical decongestants to relieve symptoms associated with coughs, colds, nasal congestion, bronchitis, and asthma. Its ability to open airways and provide rapid symptomatic relief supports strong adoption in both prescription and OTC respiratory products. Rising incidence of respiratory conditions linked to pollution, allergies, and seasonal infections has further increased demand. In addition, strong consumer acceptance of camphor-based remedies in self-care routines, particularly across North America, Europe, and Asia Pacific, continues to support steady growth of this application segment.
The pharma grade synthetic camphor market is driven by steady demand from pharmaceutical manufacturers that rely on camphor for a variety of therapeutic applications. It is widely used in topical pain relief products, medicated balms, inhalers, and cold and cough formulations, where high purity and predictable performance are essential. Synthetic camphor offers manufacturers a reliable substitute for natural camphor, ensuring consistent availability, uniform quality, and easier adherence to regulatory standards. Rising consumer preference for non opioid pain management options and convenient over the counter healthcare solutions continues to support sustained demand for pharma grade synthetic camphor.
How Are Therapeutic Uses and Healthcare Trends Shaping Demand?
Topical applications remain the main area of use, as camphor is valued for its cooling and counterirritant effects in products aimed at relieving muscle pain, joint discomfort, and minor skin irritation. Respiratory care also remains important, with camphor widely used in vapor rubs and inhalation products for congestion and cough relief. Rising awareness of self care, an aging population, and frequent respiratory issues are helping sustain demand across both developed and emerging markets.
How Do Regulations, Production Standards, and Supply Conditions Affect the industry?
Regulatory requirements strongly influence the industry, with manufacturers required to meet strict standards for purity, safety, and documentation. While this limits new entrants, it builds confidence among pharmaceutical buyers. Production also depends on reliable access to chemical intermediates, and changes in raw material availability can affect costs and output. To stay competitive, producers emphasize efficient manufacturing processes, strong quality control systems, and long term sourcing arrangements. Market stability depends on maintaining regulatory compliance, managing costs, and ensuring consistent supply to meet pharmaceutical industry needs.
Demand for pharma grade synthetic camphor continues to rise as pharmaceutical companies increase its use in topical pain relief, respiratory care, and widely used over the counter medicines. Manufacturers favor synthetic camphor for its consistent purity, predictable quality, and dependable supply compared with natural alternatives. Country level trends differ based on pharmaceutical production capacity, healthcare spending, aging demographics, and growing preference for non opioid and self care treatments. Emerging manufacturing centers show faster momentum, while developed markets sustain stable demand driven by quality standards, regulatory compliance, and established therapeutic use.
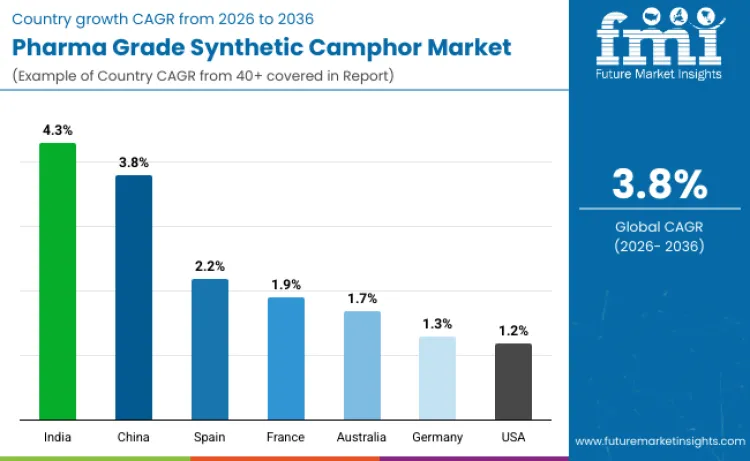
| Country | CAGR (%) |
|---|---|
| India | 4.3% |
| China | 3.8% |
| Spain | 2.2% |
| France | 1.9% |
| Australia | 1.7% |
| Germany | 1.3% |
| United States | 1.2% |
India records the fastest growth with a CAGR of 4.3%, supported by a well established pharmaceutical manufacturing base and rising domestic as well as export demand. Synthetic camphor is widely used in affordable topical analgesics and respiratory products that cater to large population needs. Expanding healthcare access, growing self medication awareness, and strong demand from export markets support consumption. Government policies encouraging pharmaceutical manufacturing, combined with cost efficient production and adherence to quality standards, allow Indian producers to strengthen their position in both regional and global supply chains.
China shows steady growth at a CAGR of 3.8%, supported by large scale manufacturing capacity and rising healthcare consumption. Pharma grade synthetic camphor is increasingly used in pain relief and respiratory therapies as healthcare access improves. Investments in modern synthesis techniques and tighter quality controls have enhanced product consistency and regulatory acceptance. China’s ability to produce at scale while meeting international quality expectations reinforces its role as both a major domestic consumer and a key exporter serving pharmaceutical markets across Asia, Europe, and other regions.
Spain experiences moderate growth with a CAGR of 2.2%, shaped largely by an aging population and steady demand for topical pain management products. Synthetic camphor is commonly used in formulations addressing muscle aches, joint discomfort, and respiratory issues. The country’s pharmaceutical industry operates under strict regulatory supervision, supporting high safety and quality benchmarks. Ongoing collaboration between local producers and multinational pharmaceutical companies sustains demand, particularly for formulations that emphasize reliability, clinical validation, and compliance with European healthcare standards.
France records a CAGR of 1.9%, reflecting demand from a well developed healthcare system that prioritizes high quality pharmaceutical ingredients. Synthetic camphor continues to be used in trusted topical and inhalation therapies, supported by strong research activity and consistent prescribing practices. Strict regulatory oversight ensures stable usage levels rather than rapid expansion. Consumer confidence in established therapeutic products and continued focus on safety, efficacy, and formulation integrity help maintain steady demand across both prescription and over the counter segments.
Australia shows growth at a CAGR of 1.7%, driven by advanced healthcare infrastructure and sustained use of camphor based pain relief and respiratory products. Rising consumer interest in wellness oriented and natural inspired remedies supports ongoing demand. Pharmaceutical companies focus on improving formulations, ensuring quality assurance, and meeting regulatory expectations. While overall market expansion remains measured, consistent investment in product development and strong consumer trust in established therapies help reinforce long term demand for pharma grade synthetic camphor.
Germany grows at a CAGR of 1.3%, supported by its emphasis on pharmaceutical quality, safety, and research driven development. Demand is linked to aging demographics and ongoing need for treatments addressing chronic pain and respiratory conditions. Manufacturers prioritize strict regulatory compliance and controlled production processes, which support consistent but gradual growth. Sustainability considerations and precision manufacturing also influence market behavior, keeping demand stable rather than expansion focused within the highly regulated German healthcare environment.
The United States records a CAGR of 1.2%, reflecting a mature market where growth is driven more by formulation improvement than volume increase. Synthetic camphor remains an important ingredient in non opioid pain relief and respiratory care products. Strong regulatory oversight, advanced healthcare infrastructure, and continued investment in improved delivery systems support steady consumption. Demand is reinforced by ongoing interest in effective, trusted over the counter therapies rather than rapid expansion in new applications.
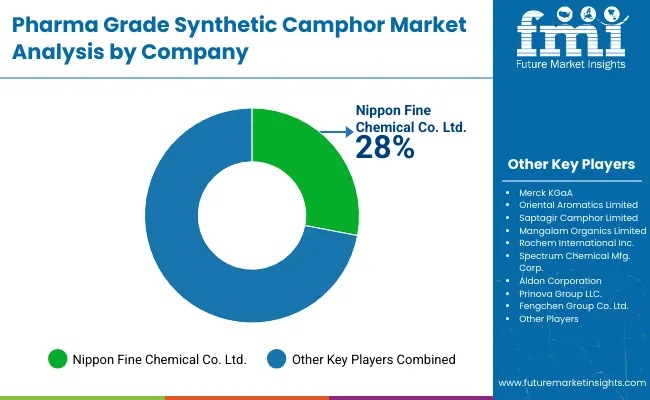
The competitive landscape of the pharma grade synthetic camphor market is shaped less by aggressive price competition and more by reliability, purity standards, and long term trust with pharmaceutical buyers. The industry remains moderately consolidated, with a small group of established manufacturers accounting for a significant share of global supply. Companies that lead this space are those capable of consistently delivering pharmaceutical grade camphor that meets strict pharmacopoeia requirements across multiple regions. Buyers prioritize suppliers with proven quality systems, regulatory transparency, and stable production processes, as synthetic camphor is widely used in sensitive applications such as topical analgesics, inhalants, and respiratory formulations.
Leading players such as Nippon Fine Chemical Co. Ltd. and Merck KGaA maintain strong competitive positions by investing heavily in advanced synthesis and purification technologies. Their strength lies in their ability to ensure batch to batch consistency, high purity levels, and compliance with global regulatory frameworks. These companies typically serve multinational pharmaceutical clients that demand dependable supply, detailed documentation, and technical support for formulation development. Their global footprint and strong compliance culture allow them to navigate regulatory differences across North America, Europe, and Asia with relative ease.
Asian manufacturers including Oriental Aromatics Ltd., Saptagir Camphor Ltd., and Mangalam Organics Ltd. play an increasingly important role, particularly in serving cost sensitive and fast growing markets. These companies benefit from vertical integration into turpentine based intermediates, which helps stabilize raw material supply and manage costs. Many are expanding capacity and refining crystallization and odor control techniques to align more closely with pharmaceutical grade expectations. Across the industry, competition is gradually shifting toward cleaner production methods, sustainability focused operations, and tighter quality controls, as regulatory scrutiny increases and pharmaceutical customers demand greater assurance on safety, traceability, and long term supply stability.
Key Players in the Pharma Grade Synthetic Camphor Market
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Formulation Types Analyzed | Topical, Inhalation |
| Applications Covered | Respiratory Disorders, Muscular Rheumatism, Counterirritant and Antipruritic, Others including cosmetic and personal care uses |
| Distribution Channels | Conventional B2B including tenders, direct procurement, wholesalers and distributors; Online B2B including internal and external channels |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, China, India, Japan, Australia, South Korea, Brazil, and 40+ other countries |
| Key Companies Profiled | Nippon Fine Chemical Co. Ltd., Oriental Aromatics Ltd., Saptagir Camphor Ltd., Mangalam Organics Ltd., Merck KGaA |
| Additional Attributes | Revenue analysis by formulation type and application, country wise demand assessment, focus on high purity and specialty products, synthesis and formulation innovation trends, expansion across pharmaceutical and personal care sectors, supply chain efficiency and quality assurance analysis |
How big is the pharma grade synthetic camphor market in 2026?
The global pharma grade synthetic camphor market is valued at USD 484.1 million in 2026.
What will be the size of the pharma grade synthetic camphor market in 2036?
The global pharma grade synthetic camphor market is valued at USD 484.1 million in 2026.
How fast will the pharma grade synthetic camphor market grow between 2026 and 2036?
The industry is expected to expand at a CAGR of 3.8% during the forecast period.
Which formulation type leads the pharma grade synthetic camphor market in 2026?
Topical formulations dominate the industry with a 73% share in 2026.
Which application segment holds a significant share in the pharma grade synthetic camphor market?
Respiratory disorders account for around 27% share of the industry in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.