Preservative-Rich Skincare Products Market
Preservative-Rich Skincare Products Market
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Preservative-Rich Skincare Products Market Forecast and Outlook 2026 to 2036
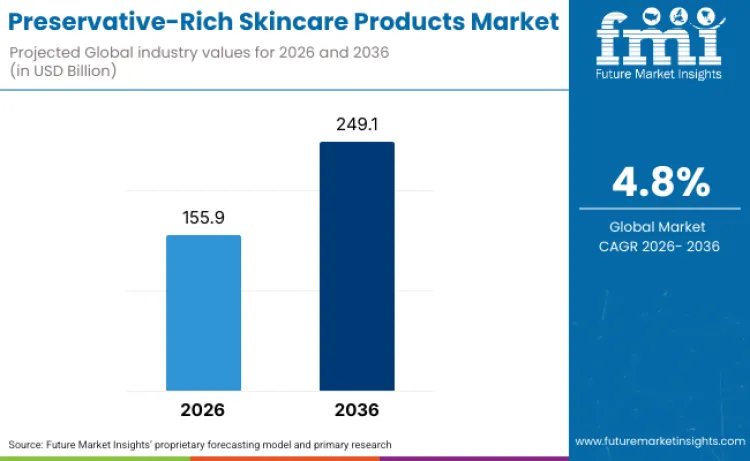
Future Market Insights projects the global preservative-rich skincare products market to reach USD 155.9 billion in 2026 and expand to USD 249.1 billion by 2036, advancing at a 4.8% CAGR over the forecast period. This sustained growth is driven by the essential role of preservatives in preventing microbial contamination across water-based formulations, where regulatory compliance with pharmacopeial standards and consumer safety requirements mandate effective antimicrobial protection throughout product shelf life, according to FMI's analysis.
The FDA’s longitudinal analysis of cosmetic recalls through 2023 confirms that microbial contamination accounts for 76.8% of all safety-related market withdrawals, underscoring the vital necessity for the stabilized, bio-active preservation systems required in next-generation BioCircuit serums. Shifting consumer preferences toward clean beauty formulations are creating demand for naturally derived and multifunctional preservatives that balance efficacy with environmental compatibility, particularly in face creams and moisturizers that dominate 36.70% of product type demand.
FMI highlights regulatory evolution as a defining transformation. The European Commission's Scientific Committee on Consumer Safety maintains a positive list of 59 authorized preservatives for cosmetic use, while China's National Medical Products Administration implemented revised cosmetics supervision regulations in January 2021, mandating comprehensive preservation efficacy testing according to ISO 11930:2019 methodology. These frameworks are standardizing preservation requirements globally while accelerating innovation in low-concentration, broad-spectrum preservative combinations that meet stringent residual limits. Female consumers representing 71.50% of the demographic maintain higher usage frequencies.
Summary of the Preservative-Rich Skincare Products Market
What Is Growth Outlook for Preservative-Rich Skincare Products Market as per Future Market Insights Projection?
The market will grow at a 4.8% CAGR, from USD 155.9 Billion (2026) to USD 249.1 Billion (2036).
FMI Research Approach: Based on cosmetic production trends and preservative usage across emulsion formats.
How Do FMI Analysts Perceive Preservative-Rich Skincare Products Market to Evolve?
Preservatives are shifting to multifunctional systems with added skin benefits like antioxidants and pH balancing.
FMI Research Approach: Regulatory analysis and ingredient substitution tracking.
Which Country Holds Largest Share in Global Preservative-Rich Skincare Products Market?
China leads, driven by strict NMPA preservation testing norms and local brand compliance.
FMI Research Approach: Country-level modeling by regulatory enforcement and clean beauty adoption.
How Large Will Preservative-Rich Skincare Products Market Be by 2036?
It is projected to reach USD 249.1 Billion by 2036.
FMI Research Approach: Modeled using emulsion types, preservative loads, and packaging impacts.
What Is Definition of Preservative-Rich Skincare Products Market?
Products with antimicrobial agents that ensure shelf life and safety in water-rich skincare.
FMI Research Approach: Inclusion based on ISO 11930 and emulsion-based risk profiles.
What Are Globally Unique Trends Shaping Preservative-Rich Skincare Products Market?
Trends include bio-based preservatives, airless packaging, and microbiome-safe formulas.
FMI Research Approach: ESG mandates, formulation innovation, and clean label consumer data.
Preservative-Rich Skincare Products Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 155.9 Billion |
| Industry Value (2036) | USD 249.1 Billion |
| CAGR (2026-2036) | 4.80% |
Why is the Demand for Preservative-Rich Skincare Products Growing?
Future Market Insights analysts perceive the preservative-rich skincare products market evolving toward scientifically optimized, environmentally compatible, and consumer-transparent preservation ecosystems. Analysts highlight transitions from traditional single-preservative approaches relying on parabens or formaldehyde releasers toward combination systems utilizing phenoxyethanol, organic acids, and multifunctional ingredients that achieve broad-spectrum protection at reduced total preservative concentrations.
Examples include preservation platforms engineered for specific pH ranges and emulsion types, closed-loop challenge testing protocols that validate efficacy under real-use conditions, and transparency initiatives where brands provide detailed preservative rationale addressing both safety and necessity. Demand for preservative systems with established safety profiles and regulatory acceptance is growing faster than novel antimicrobials facing lengthy approval processes, while formulation strategies incorporating hurdle technologies, combining preservatives with pH control, water activity reduction, and packaging barriers, are being integrated into product development workflows to optimize both safety and consumer acceptance.
How is the Preservative-Rich Skincare Products Market Segmented?
The preservative-rich skincare products market is segmented by product type, gender, and distribution channel, reflecting how formulation risk, usage intensity, and retail exposure determine preservation requirements. By product type, the market spans face creams and moisturizers, cleansers and face wash, sunscreen, body creams and moisturizers, shaving lotions and creams, and other water-based formats, with emulsion-heavy face creams and moisturizers forming the core demand base due to their high water content and daily usage frequency. By gender, demand is concentrated among female consumers, whose multi-step skincare routines and higher application frequency necessitate robust, broad-spectrum preservation systems capable of maintaining microbiological stability throughout extended use. By distribution channel, sales are led by supermarkets and hypermarkets, followed by pharmacy and drugstore outlets and online platforms, each imposing different shelf-life, storage, and stability expectations on preservative performance.
FMI analysis suggests that while clean-label narratives are reshaping ingredient selection, the market structure remains anchored in regulatory compliance and microbiological risk management. This segmentation highlights a safety-led market where preservation strategy is dictated less by marketing preference and more by formulation science, regulatory thresholds, and real-world consumer use conditions.
How Do Face Creams and Moisturizers Dominate Product Type Demand?
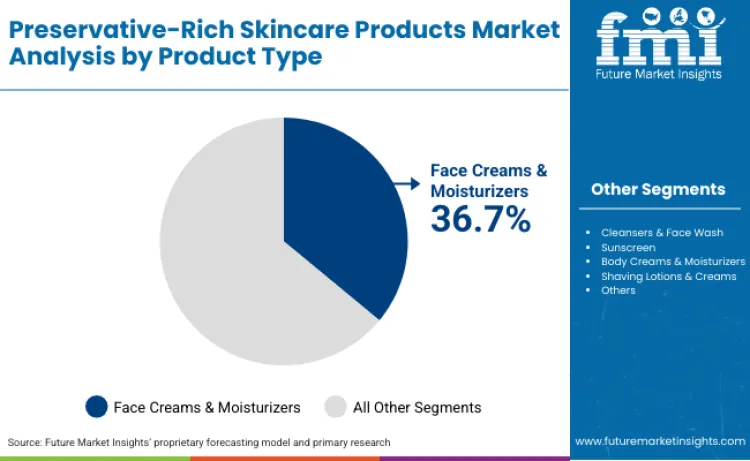
According to Future Market Insights, face creams and moisturizers lead the global preservative-rich skincare products market due to their emulsion-based formulations containing 40-80% water content that creates conditions conducive to microbial growth requiring effective preservation systems. With an estimated 36.70% share of product type revenue, these formulations demand broad-spectrum preservatives effective against bacteria, yeast, and mold that can proliferate in nutrient-rich cream bases.
Their dominance is further reinforced by high usage frequency among consumers who apply facial moisturizers daily, creating repeated product exposure to environmental contaminants and requiring preservation systems that maintain efficacy throughout extended use periods. Research published in the International Journal of Cosmetic Science demonstrated that oil-in-water emulsions require minimum preservative concentrations of 0.6-1.0% phenoxyethanol equivalents to pass ISO 11930 challenge testing, establishing baseline preservation requirements that shape formulation strategies. The premium pricing sustainable in facial skincare categories enables incorporation of advanced preservative systems including combination approaches and multifunctional ingredients that optimize both efficacy and consumer perception.
How Do Supermarkets and Hypermarkets Lead Distribution Channel Demand?
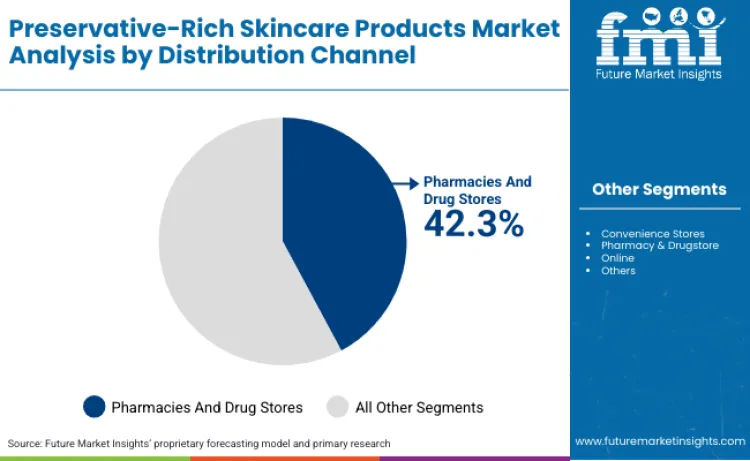
Future Market Insights notes that supermarkets and hypermarkets capture 42.30% of distribution volume, offering controlled temperature and humidity environments that support preservative stability and product integrity throughout retail storage. These channels provide consumers with ability to physically examine products, read ingredient labels, and compare preservation systems across brands, creating transparency pressures that influence preservative selection toward systems with established safety profiles and consumer recognition.
Mass-market positioning in these channels requires preservation systems that balance efficacy with cost optimization, favoring proven preservative platforms including phenoxyethanol, sodium benzoate, and organic acid combinations rather than premium preservation technologies that increase formulation costs. However, channel requirements for extended shelf life, typically 36 months from manufacture, necessitate preservation systems that maintain efficacy under potential temperature stress during distribution and storage, driving specifications that ensure microbiological stability across diverse retail conditions.
How Are Regulatory Frameworks Shaping Preservative Selection and Innovation?
Industry evolution is being shaped by harmonization of global regulatory frameworks that establish preservative positive lists, use concentration limits, and safety assessment requirements. The European Commission's Scientific Committee on Consumer Safety maintains authorization for 59 preservatives in EU Cosmetics Regulation Annex V, with each preservative subjected to rigorous safety evaluation including systemic exposure assessment, skin sensitization potential, and reproductive toxicity evaluation.
This framework provides regulatory certainty for manufacturers while ensuring consumer protection through evidence-based safety thresholds. Similar positive list approaches implemented in Japan, South Korea, and ASEAN countries are creating regulatory convergence that enables multinational brands to develop preservation systems acceptable across major markets, reducing formulation complexity and accelerating product launch timelines. However, regional variations in concentration limits and labeling requirements continue to necessitate market-specific adjustments.
Why Is Consumer Clean Beauty Pressure Driving Preservative Reformulation?
The preference is driving brands to reformulate away from preservatives with negative consumer perception, particularly parabens, formaldehyde releasers, and methylisothiazolinone, toward alternatives including phenoxyethanol, benzyl alcohol, and organic acid combinations that consumers perceive as safer despite limited comparative clinical evidence.
Brand communication strategies increasingly emphasize preservation necessity while providing transparent rationale for preservative selection, acknowledging consumer concerns while maintaining scientific commitment to product safety. Premium brands are investing in education initiatives that explain microbial contamination risks and preservation science, attempting to shift consumer focus from preservative absence toward preservation efficacy and safety validation.
How Are Multifunctional Ingredients Transforming Preservation Strategies?
Innovation focus has shifted toward multifunctional ingredients that provide antimicrobial protection alongside additional formulation benefits, enabling brands to reduce dedicated preservative concentrations while maintaining microbiological stability. Examples include ingredients with combined preservative-antioxidant properties that address both microbial contamination and oxidative degradation, humectants with antimicrobial activity that contribute to preservation while providing skin hydration benefits, and pH adjusters that create unfavorable conditions for microbial growth while optimizing active ingredient stability.
This approach enables ingredient list simplification that resonates with clean beauty preferences while maintaining comprehensive product protection. Research published in the Journal of Cosmetic Science demonstrated that formulations utilizing multifunctional approaches can reduce primary preservative concentrations by 30-40% while achieving equivalent ISO 11930 challenge test performance, validating the technical viability of these strategies.
How Will Preservative-Rich Skincare Products Market Expansion Unfold Across Key Global Regions?
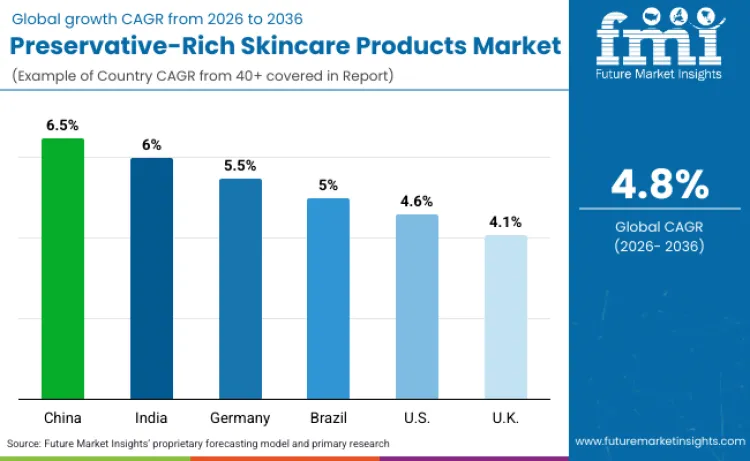
Future Market Insights identifies China (CAGR 6.5%), India (6.0%), Germany (5.5%), Brazil (5.0%), United States (4.6%), and United Kingdom (4.1%) as the six core demand drivers. China leads through regulatory modernization mandating preservation efficacy testing and domestic brand quality upgrades. India drives growth through organized retail expansion and rising safety awareness among urban consumers. Germany anchors demand for premium preservation systems meeting European Pharmacopoeia standards.
Brazil's tropical climate necessitates preservation systems effective under thermal and humidity stress. The US market is shaped by FDA regulatory oversight and consumer clean beauty preferences. The UK combines regulatory rigor with consumer ingredient scrutiny. FMI emphasizes that each country combines distinct regulatory frameworks with unique consumer expectations, creating differentiated preservation system requirements through 2036.
| Country | CAGR (2026-2036) |
|---|---|
| China | 6.5% |
| India | 6.0% |
| Germany | 5.5% |
| Brazil | 5.0% |
| United States | 4.6% |
| United Kingdom | 4.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is India Formalizing Preservative Oversight Through Quality Standardization?
India is expanding at a 6.0% CAGR, driven by a decisive shift from fragmented, informal manufacturing toward regulated, audit-ready cosmetics production. The Cosmetics (Amendment) Rules, 2025, issued by the Ministry of Health and Family Welfare, mandate batch-wise traceability, clearer shelf-life definitions, and stricter documentation of raw materials and finished products. These measures directly impact preservative systems, as brands must now demonstrate stability, expiry accuracy, and microbial safety over the declared product life.
Complementing this, Bureau of Indian Standards guidelines on restricted preservatives are aligning domestic formulations with international safety expectations, particularly for water-based creams and lotions sold through organized retail. FMI analysis indicates that this regulatory tightening is acting as a market filter, accelerating the shift toward branded, compliant products while marginalizing unregulated local players. Over the forecast period, preservation strategy in India is becoming a prerequisite for retail access, export eligibility, and long-term brand credibility rather than a back-end formulation choice.
How is China Enforcing High-Risk Ingredient Governance Under CSAR?
China is growing at a 6.5% CAGR, anchored in a compliance-first transformation under the Cosmetics Supervision and Administration Regulation. By 2026, preservatives are formally treated as high-risk cosmetic ingredients, triggering registration, annual safety reporting, and post-market surveillance requirements under the oversight of the National Medical Products Administration. This marks a structural shift away from ingredient-list compliance toward performance- and risk-based regulation, where chemical preservatives, UV filters, and colorants face heightened scrutiny.
FMI observes that this framework is forcing both domestic and international brands to rationalize preservative selection, favoring globally validated systems with robust toxicological dossiers. The result is a rapid upgrade cycle in formulation practices, contract testing demand, and regulatory consulting activity. As China’s skincare market matures, preservation efficacy and documentation are becoming central to brand survival, positioning compliance capability as a competitive advantage rather than a cost burden.
Why is Germany Doubling Down on Scientific Risk Assessment?
Germany’s 5.5% CAGR reflects a market defined by precautionary science and regulatory precision rather than volume growth. The country’s preservative-rich skincare segment is shaped by evaluations from the German Federal Institute for Risk Assessment, which continuously assesses long-term exposure risks associated with commonly used preservatives such as parabens and phenoxyethanol. This evidence-led approach reinforces consumer trust in “preservative-rich” products that meet stringent safety thresholds, countering simplistic preservative-free narratives.
FMI analysis indicates that German consumers prioritize toxicological validation and regulatory endorsement over marketing claims, sustaining demand for pharmacy-grade and dermo-cosmetic products. Retailers and pharmacies reinforce this bias by requiring documented safety assessments and EU-compliant preservative systems as conditions for listing. As a result, Germany functions as a benchmark market where preservation strategies must withstand scientific, regulatory, and professional scrutiny simultaneously, shaping formulation standards well beyond its national borders.
How is Brazil Aligning Preservation Rules With Global Safety Lists?
Brazil is advancing at a 5.0% CAGR, driven by aggressive regulatory alignment led by the ANVISA. By early 2026, updates to RDC frameworks have prohibited specific chemical precursors and tightened the national positive list of cosmetic substances, directly affecting preservative systems. This regulatory clean-up reduces ambiguity for manufacturers by harmonizing Brazilian requirements with international safety standards, particularly those used in the EU and North America.
FMI analysis highlights that Brazil’s tropical climate amplifies microbial risk, making robust, multi-hurdle preservation non-negotiable for market access. As enforcement strengthens, multinational brands are accelerating the rollout of globally standardized preservative platforms, while local players are reformulating to remain compliant. Brazil is consequently emerging as a stable, regulation-driven market where preservation strategy underpins product safety, consumer confidence, and cross-border scalability.
Why is the United States Maturing Under MoCRA Enforcement?
The United States is growing at a 4.6% CAGR, reflecting a transition from voluntary compliance to statutory accountability under the Modernization of Cosmetics Regulation Act. MoCRA requires responsible persons to register products with the U.S. Food and Drug Administration, disclose ingredient compositions, and maintain records substantiating cosmetic safety, including preservative efficacy. FMI notes that this shift is elevating preservation from a formulation assumption to a documented safety obligation, particularly for water-based skincare products with higher contamination risk. Annual updates and inspection readiness are compelling brands to standardize preservative systems and invest in microbial challenge testing. Over time, this framework is stabilizing the market by discouraging under-preserved formulations and reducing recall risk. The U.S. market is therefore maturing into one where regulatory discipline, not clean-label rhetoric alone, defines trust and long-term competitiveness.
What Strategic Moves Are Defining Competitive Landscape?
Future Market Insights analysts observe that competition in the preservative-rich skincare products market is increasingly defined by sustainable sourcing certification, preservation system transparency, and formulation innovation that balances efficacy with consumer acceptance, rather than traditional brand positioning strategies. Leading cosmetic manufacturers are investing in green chemistry platforms that develop bio-derived and nature-identical preservatives meeting the same efficacy standards as synthetic systems while addressing consumer clean beauty preferences. Companies achieving third-party sustainability certifications including ISCC+ and COSMOS-approved preservatives are gaining competitive advantages in premium and natural beauty segments where certification provides differentiation and supports sustainability claims.
Another defining move is the adoption of transparent communication strategies that educate consumers about preservation necessity while acknowledging safety concerns. Brands including CeraVe and La Roche-Posay provide detailed preservative rationale on product packaging and digital platforms, explaining specific preservative selection based on formulation requirements and safety validation. This approach builds consumer trust through transparency rather than avoiding preservative discussions. Strategic investments in multifunctional ingredient platforms are enabling brands to reduce dedicated preservative concentrations while maintaining comprehensive product protection, addressing consumer preference for simplified ingredient lists without compromising microbiological safety.
FMI also highlights growing emphasis on packaging innovation as a preservation strategy, with brands adopting airless dispensing systems, dose-controlled pumps, and single-use formats that reduce contamination risk and enable lower preservative concentrations. These packaging technologies command premium pricing while addressing preservation challenges inherent in jar packaging that exposes product to repeated air and hand contact. Collectively, these moves are shifting competition toward holistic preservation strategies that integrate formulation science, consumer communication, and packaging technology, favoring companies with comprehensive capabilities over those relying solely on traditional preservative chemistry.
Recent Developments:
- In July 2024, BASF SE secured ISCC+ certification to supply over 60 sustainable solvent and preservative variants, strengthening competitiveness in eco-aligned cosmetic production by offering bio-derived preservative alternatives with equivalent efficacy to synthetic systems.
- In March 2024, Beiersdorf AG disclosed in its Sustainability Report that the company reformulated 43% of its skincare portfolio between 2021-2024 to incorporate next-generation preservation systems with improved environmental profiles and reduced use concentrations.
Key Players in Preservative-Rich Skincare Products Market
- L'Oréal S.A.
- Beiersdorf AG
- Shiseido Co., Ltd.
- Procter & Gamble
- Unilever
- Johnson & Johnson Services, Inc.
- Avon Products, Inc.
- Coty Inc.
- Colgate-Palmolive Company
- Revlon
Market Definition
The preservative-rich skincare products market comprises finished cosmetic and personal care formulations that intentionally contain antimicrobial preservative systems to ensure microbiological safety, product stability, and regulatory compliance across the declared shelf life. These products are primarily water-based or emulsion-based formats where microbial growth risk is inherent due to moisture, nutrients, and repeated consumer exposure. The market includes facial skincare, body care, cleansing, and sun care products formulated with approved preservative systems such as alcohols, organic acids, parabens, isothiazolinones, and multifunctional preservative blends. Market value is measured at the retail sales level and reflects consumer demand for safe, stable, and regulation-compliant skincare products across mass, premium, and dermo-cosmetic segments distributed through physical and digital channels.
Market Inclusion
Market inclusion covers consumer-facing skincare products that incorporate validated preservative systems to prevent bacterial, yeast, and mold contamination under normal use conditions. Included categories span face creams and moisturizers, cleansers, sunscreens, body lotions, shaving creams, and similar water-containing formulations that must comply with preservative efficacy standards such as ISO 11930 and regional cosmetic regulations. Products using synthetic, nature-identical, or bio-derived preservatives listed on regulatory positive lists are included, provided they are marketed as finished skincare goods. The scope also includes products sold through pharmacies, supermarkets, specialty beauty retailers, online platforms, and direct-to-consumer channels, targeting both female and male consumers in household and personal use contexts.
Market Exclusion
The market excludes anhydrous skincare products such as pure oils, balms, wax-based sticks, and powders that do not require antimicrobial preservation due to the absence of free water. Raw preservative ingredients, bulk antimicrobial chemicals, and formulation aids sold exclusively to manufacturers or laboratories are excluded, as the market focuses on finished consumer products rather than inputs. Products marketed explicitly as preservative-free and relying solely on packaging barriers or single-use formats are not included. Prescription dermatological treatments, medical devices, injectables, and professional-only formulations used under controlled clinical conditions are excluded. Functional foods, oral supplements, hair dyes, fragrances, and non-skin cosmetic categories also fall outside the defined market scope.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Face Creams & Moisturizers; Cleansers & Face Wash; Sunscreen; Body Creams & Moisturizers; Shaving Lotions & Creams; Others |
| Gender | Female; Male |
| Distribution Channel | Supermarkets & Hypermarkets; Convenience Stores; Pharmacy & Drugstore; Online; Others |
| Preservation System Focus | Broad-Spectrum Antimicrobial Blends; Nature-Identical & Bio-Derived Preservatives; Multifunctional Preservation Ingredients; Low-Concentration High-Efficacy Systems; Packaging-Integrated Preservation Solutions |
| Regulatory & Compliance Framework | ISO 11930 Challenge Testing Standards; EU Cosmetics Regulation Annex V; China CSAR Preservation Governance; U.S. MoCRA Safety Documentation; Pharmacopoeia-Grade Raw Material Adoption |
| Regions Covered | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Countries Covered | China; India; Germany; Brazil; United States; United Kingdom; Japan; South Korea; France; and 40+ additional countries |
| Key Companies Profiled | L’Oréal S.A.; Beiersdorf AG; Shiseido Co., Ltd.; Procter & Gamble; Unilever; Johnson & Johnson; Avon Products; Coty Inc.; Colgate-Palmolive Company; Revlon |
| Additional Attributes | Dollar sales by product type, gender, and channel; preservation efficacy benchmarking across microbial challenge tests and shelf-life stability; adoption trends for bio-derived and multifunctional preservative systems; regulatory impact on formulation strategy; packaging-driven contamination risk reduction; clean beauty reformulation dynamics and sustainability certification influence |
Preservative-Rich Skincare Products Market by Segments
By Product Type:
- Face Creams & Moisturizers
- Cleansers & Face Wash
- Sunscreen
- Body Creams & Moisturizers
- Shaving Lotions & Creams
- Others
By Gender:
- Female
- Male
By Distribution Channel:
- Supermarkets & Hypermarkets
- Convenience Stores
- Pharmacy & Drugstore
- Online
- Others
By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Bibliography
- National Medical Products Administration of China. (2021). Cosmetics Supervision and Administration Regulation. Order of the State Council No. 727.
- International Organization for Standardization. (2019). ISO 11930:2019 - Cosmetics - Microbiology - Evaluation of the Antimicrobial Protection of a Cosmetic Product.
- Beiersdorf AG. (2024). Sustainability Report 2024 - Product Safety and Innovation. Retrieved from https://www.beiersdorf.com/sustainability/reporting
- Bureau of Indian Standards. (2020). IS 17526:2020 - Cosmetics - Microbiology - Microbiological Limits. Retrieved from https://www.bis.gov.in
- International Journal of Cosmetic Science. (2023). Impact of Preservative Systems on Emulsion Stability and Microbiological Protection. Volume 45, Issue 3.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Face Creams & Moisturizers
- Cleansers & Face Wash
- Sunscreen
- Body Creams & Moisturizers
- Shaving Lotions & Creams
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Gender
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Gender, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Gender, 2026 to 2036
- Female
- Male
- Y-o-Y Growth Trend Analysis By Gender, 2021 to 2025
- Absolute $ Opportunity Analysis By Gender, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Supermarkets & Hypermarkets
- Convenience Stores
- Pharmacy & Drugstore
- Online
- Others
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Gender
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Gender
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Gender
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Gender
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- L'Oréal S.A.
- Beiersdorf AG
- Shiseido Co., Ltd.
- Procter & Gamble
- Unilever
- Johnson & Johnson Services, Inc.
- Avon Products, Inc.
- Coty Inc.
- Colgate-Palmolive Company
- Revlon
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Preservative-Rich Skincare Products Market Key Takeaways
- Global Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Global Preservative-Rich Skincare Products Market Y-o-Y Growth (%), 2021-2036
- Global Preservative-Rich Skincare Products Market Absolute $ Opportunity Analysis (USD Million), 2026-2036
- Global Preservative-Rich Skincare Products Market Pricing Analysis (USD/Unit), 2021-2036
- Global Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- Global Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- Global Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- Global Preservative-Rich Skincare Products Market Size (USD Million) By Region, 2021-2036
- North America Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- North America Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- North America Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- North America Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Latin America Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- Latin America Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- Latin America Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- Latin America Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Western Europe Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- Western Europe Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- Western Europe Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- Western Europe Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- Eastern Europe Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- Eastern Europe Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- Eastern Europe Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- East Asia Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- East Asia Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- East Asia Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- East Asia Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Preservative-Rich Skincare Products Market Size (USD Million) By Country, 2021-2036
- Middle East & Africa Preservative-Rich Skincare Products Market Size (USD Million) By Product Type, 2021-2036
- Middle East & Africa Preservative-Rich Skincare Products Market Size (USD Million) By Gender, 2021-2036
- Middle East & Africa Preservative-Rich Skincare Products Market Size (USD Million) By Distribution Channel, 2021-2036
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- USA Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- USA Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- USA Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- USA Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Canada Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Canada Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Canada Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Canada Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Mexico Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Mexico Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Mexico Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Mexico Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Brazil Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Brazil Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Brazil Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Chile Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Chile Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Chile Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Chile Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Germany Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Germany Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Germany Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Germany Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- UK Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- UK Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- UK Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- UK Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Italy Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Italy Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Italy Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Italy Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Spain Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Spain Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Spain Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Spain Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- France Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- France Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- France Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- France Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- India Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- India Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- India Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- India Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- ASEAN Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- ASEAN Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- ASEAN Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Australia & New Zealand Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Australia & New Zealand Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Australia & New Zealand Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- China Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- China Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- China Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- China Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Japan Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Japan Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Japan Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Japan Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- South Korea Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- South Korea Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- South Korea Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- South Korea Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Russia Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Russia Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Russia Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Russia Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Poland Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Poland Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Poland Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Poland Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Hungary Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Hungary Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Hungary Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Hungary Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Kingdom of Saudi Arabia Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Kingdom of Saudi Arabia Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Kingdom of Saudi Arabia Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Turkiye Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- Turkiye Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- Turkiye Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- South Africa Preservative-Rich Skincare Products Pricing Analysis (USD/Unit), 2021-2036
- South Africa Preservative-Rich Skincare Products Market Share Analysis (%) By Product Type, 2025
- South Africa Preservative-Rich Skincare Products Market Share Analysis (%) By Gender, 2025
- South Africa Preservative-Rich Skincare Products Market Share Analysis (%) By Distribution Channel, 2025
- Preservative-Rich Skincare Products Market Competition Dashboard
- Preservative-Rich Skincare Products Market Competition Benchmarking Analysis
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Regional, 2025
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Product Type, 2025
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Gender, 2025
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Distribution Channel, 2025
- L'Oréal S.A. - Company Profile
- Beiersdorf AG - Company Profile
- Shiseido Co., Ltd. - Company Profile
- Procter & Gamble - Company Profile
- Unilever - Company Profile
- Johnson & Johnson Services, Inc. - Company Profile
- Avon Products, Inc. - Company Profile
- Coty Inc. - Company Profile
- Colgate-Palmolive Company - Company Profile
- Revlon - Company Profile
- Assumptions & Acronyms Used in Preservative-Rich Skincare Products Market Report
List of Figures
- Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Preservative-Rich Skincare Products Market Y-o-Y Growth (%), 2021-2036
- Preservative-Rich Skincare Products Market Absolute $ Opportunity Analysis (USD Million), 2026-2036
- Preservative-Rich Skincare Products Market Value Chain Analysis
- Preservative-Rich Skincare Products Market Supply Chain Analysis
- Preservative-Rich Skincare Products Market Investment Feasibility Matrix
- Preservative-Rich Skincare Products Market PESTLE Analysis
- Preservative-Rich Skincare Products Market Porter's Five Forces Analysis
- Preservative-Rich Skincare Products Market Product Life Cycle Analysis
- Preservative-Rich Skincare Products Market Opportunity Map Analysis
- Preservative-Rich Skincare Products Market Scenario Forecast Analysis
- Preservative-Rich Skincare Products Market Production and Consumption Statistics
- Preservative-Rich Skincare Products Market Import and Export Statistics
- Global Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- Global Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- Global Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- Global Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- Global Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- Global Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- Global Preservative-Rich Skincare Products Market Share (%) By Region, 2025
- Global Preservative-Rich Skincare Products Market Share (%) By Region, 2036
- Global Preservative-Rich Skincare Products Market Attractiveness Analysis By Region
- North America Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- North America Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- North America Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- North America Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- North America Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- North America Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- North America Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- North America Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- North America Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- North America Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Latin America Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Latin America Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- Latin America Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- Latin America Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- Latin America Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- Latin America Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- Latin America Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- Latin America Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- Latin America Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Latin America Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Western Europe Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- Western Europe Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Western Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- Eastern Europe Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Eastern Europe Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- East Asia Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- East Asia Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- East Asia Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- East Asia Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- East Asia Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- East Asia Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- East Asia Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- East Asia Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- East Asia Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- East Asia Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- South Asia and Pacific Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- South Asia and Pacific Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Preservative-Rich Skincare Products Market Size (USD Million), 2021-2036
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Country, 2025
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Country, 2036
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Product Type, 2025
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Product Type, 2036
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Gender, 2025
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Gender, 2036
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Preservative-Rich Skincare Products Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Country
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Product Type
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Gender
- Middle East & Africa Preservative-Rich Skincare Products Market Attractiveness Analysis By Distribution Channel
- USA Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- France Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- India Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- China Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Preservative-Rich Skincare Products Pricing Trend Analysis (USD/Unit), 2021-2036
- Preservative-Rich Skincare Products Market Competitive Landscape
- Preservative-Rich Skincare Products Market Structure Analysis
- Preservative-Rich Skincare Products Market Competition Dashboard
- Preservative-Rich Skincare Products Market Competition Benchmarking Analysis
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Regional
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Product Type
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Gender
- Preservative-Rich Skincare Products Market Share Analysis of Top Players By Distribution Channel
- L'Oréal S.A. - Revenue Analysis (USD Million), 2019-2025
- Beiersdorf AG - Revenue Analysis (USD Million), 2019-2025
- Shiseido Co., Ltd. - Revenue Analysis (USD Million), 2019-2025
- Procter & Gamble - Revenue Analysis (USD Million), 2019-2025
- Unilever - Revenue Analysis (USD Million), 2019-2025
- L'Oréal S.A. - Market Position Analysis
- Beiersdorf AG - Market Position Analysis
- Shiseido Co., Ltd. - Market Position Analysis
- Procter & Gamble - Market Position Analysis
- Unilever - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

