Steroid Measurements Reagents Market
Steroid Measurements Reagents Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Steroid Measurements Reagents Market Forecast and Outlook 2026 to 2036
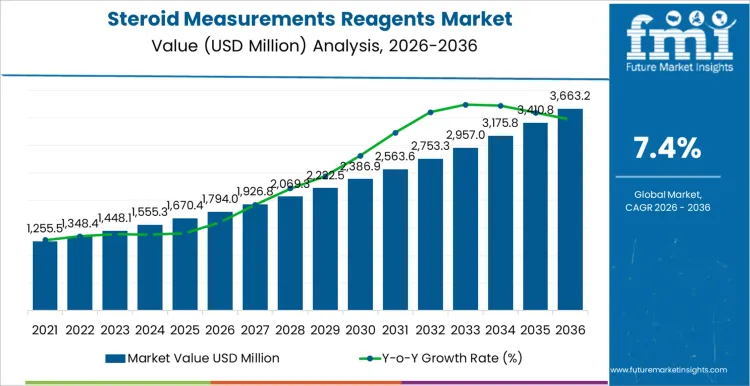
The steroid measurements reagents market is expected to reach USD 1,794.0 million in 2026 and expand to USD 3,663.2 million by 2036, at a CAGR of 7.4%. Value behavior reflects the central role of hormone quantification in endocrine diagnostics, fertility management, oncology assessment, and long-term chronic disease monitoring. Demand remains structurally linked to repeat testing volumes rather than episodic diagnostics, as steroid hormones require longitudinal measurement to guide therapy adjustment. Spending concentrates on reagents that deliver analytical specificity, low cross-reactivity, and calibration stability across automated immunoassay and LC-MS workflows deployed in hospital and reference laboratories.
Utilization patterns are shaped by laboratory accreditation requirements, assay validation obligations, and integration with standardized diagnostic platforms. Testing intensity rises where endocrine and reproductive protocols mandate serial hormone measurement within narrow clinical decision thresholds. Transition toward mass spectrometry improves specificity, though it introduces higher technical and cost barriers that moderate unrestricted expansion. Reagent selection is influenced by lot consistency, traceability, and uninterrupted supply to support continuous patient monitoring. Market structure remains governed by compliance discipline, analytical rigor, and institutional testing protocols, sustaining predictable growth anchored to regulated diagnostic pathways rather than discretionary screening expansion.
Quick Stats for Steroid Measurements Reagents Market
- Steroid Measurements Reagents Market Value (2026): USD 1,794.0 million
- Steroid Measurements Reagents Market Forecast Value (2036): USD 3,663.2 million
- Steroid Measurements Reagents Market Forecast CAGR 2026 to 2036: 7.4%
- Leading Reagent Category by Demand Share: Calibrators & sample preparation reagents
- Fastest-Growing Countries: India, Brazil, China, USA, Germany
- Top Players in Global Demand: Roche Diagnostics, Abbott, Siemens Healthineers, Beckman Coulter, DiaSorin
Steroid Measurements Reagents Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1,794.0 million |
| Market Forecast Value (2036) | USD 3,663.2 million |
| Forecast CAGR 2026 to 2036 | 7.4% |
Why is the Demand for Steroid Measurements Reagents Growing?
Demand for steroid measurements reagents is increasing as clinical laboratories expand diagnostic capabilities to quantify endogenous and exogenous steroids across endocrinology, oncology, and sports medicine applications. Accurate measurement of cortisol, testosterone, estradiol, and related metabolites supports assessment of adrenal and gonadal function, monitoring of hormone replacement therapy, and investigation of endocrine disorders where subtle concentration changes have significant clinical implications.
Laboratories specify reagents validated for high specificity and minimal cross-reactivity because steroid analytes often occur at low concentrations and within complex biological matrices. Analytical performance, lot-to-lot consistency, and compatibility with immunoassay platforms and mass spectrometry workflows are key procurement criteria because reproducible results influence diagnostic confidence, clinical decision pathways, and longitudinal patient monitoring.
Growth in personalized medicine and increased attention to chronic conditions such as polycystic ovary syndrome, osteoporosis, and adrenal insufficiency reinforces adoption of steroid measurement solutions that align with evidence-based practice. Reference laboratories and hospital systems integrate steroid panels into metabolic assessments and wellness evaluations where hormone balance informs treatment planning.
Regulatory emphasis on assay performance and laboratory accreditation encourages use of reagents with well-documented traceability and quality control materials. Technical improvements in reagent stability, calibration protocols, and multiplexing support efficient laboratory throughput and reduce manual handling errors. These clinical, operational, and quality management factors are contributing to sustained demand growth in the steroid measurements reagents market.
How Is the Steroid Measurements Reagents Industry Segmented by Reagent Category, Clinical Application, and End User?
Demand for steroid measurement reagents is driven by precision hormone quantification needs, expanding LC-MS/MS adoption, and tighter diagnostic accuracy expectations. Laboratories emphasize analytical specificity, traceability, and low cross-reactivity across diverse steroid panels. Utilization reflects broader endocrine testing volumes, fertility program growth, and oncology-related hormone assessment. Selection priorities include calibration robustness, workflow compatibility, and regulatory alignment. Segmentation explains how reagent focus, clinical purpose, and testing setting shape utilization patterns across endocrine and hormone analytics.
Which Reagent Category Holds the Largest Share in Steroid Measurement Workflows?
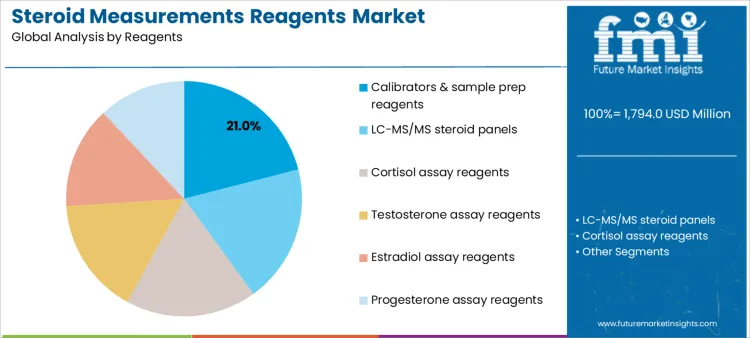
Calibrators and sample preparation reagents account for 21.0%, reflecting their foundational role in ensuring accuracy and comparability across analytical runs. These reagents support matrix normalization and method validation within routine and advanced workflows. LC-MS/MS steroid panels at 19.0% enable multiplexed measurement with high specificity across multiple hormones. Cortisol assay reagents at 18.0% address high-volume testing linked to stress and adrenal evaluation. Testosterone reagents at 16.0%, estradiol at 14.0%, and progesterone at 12.0% support targeted endocrine and reproductive testing. Reagent segmentation indicates emphasis on accuracy enablers alongside high-throughput hormone assays.
Key Points
- Calibrators underpin accuracy and method consistency.
- LC-MS/MS panels enable multiplexed steroid analysis.
- Core hormones drive sustained reagent consumption.
How Do Clinical Applications Influence Steroid Measurement Reagent Demand?

Endocrine disorder diagnosis represents 28.0%, driven by evaluation of hormonal imbalance and metabolic regulation. Fertility and IVF monitoring at 22.0% relies on precise estradiol and progesterone tracking across treatment cycles. Adrenal disorder workups at 18.0% focus on cortisol and related steroids for functional assessment. Hormone-dependent oncology at 18.0% applies steroid measurement to disease monitoring and therapy planning. Pediatric endocrinology at 14.0% requires sensitive assays suited to low concentration ranges. Application-based segmentation highlights higher utilization where clinical decisions depend on precise hormonal profiling.
Key Points
- Endocrine diagnostics generate consistent testing demand.
- Fertility programs require cycle-specific hormone tracking.
- Oncology and adrenal care depend on precise steroid profiling.
Which End User Settings Drive Utilization of Steroid Measurement Reagents?
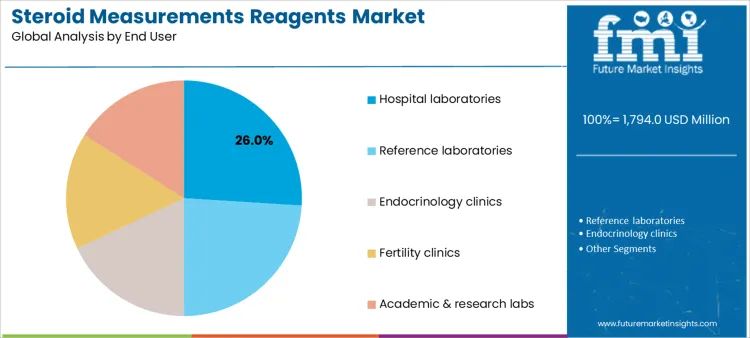
Hospital laboratories account for 26.0%, reflecting integrated diagnostic services supporting inpatient and outpatient care. Reference laboratories at 24.0% provide standardized testing at scale with advanced analytical platforms. Endocrinology clinics at 18.0% focus on targeted hormone assessment within specialty care. Fertility clinics at 16.0% apply reagents in time-sensitive reproductive protocols. Academic and research laboratories at 16.0% support method development and clinical studies. End-user segmentation shows concentration where testing volume, analytical capability, and clinical specialization determine reagent adoption.
Key Points
- Hospital labs integrate steroid testing into routine diagnostics.
- Reference labs deliver scale and method standardization.
- Specialty clinics focus on application-specific hormone analysis.
What are the Key Dynamics in the Steroid Measurements Reagents Market?
Demand for steroid measurement reagents reflects analytical requirements to quantify endogenous and exogenous steroid hormones for clinical diagnosis, therapeutic monitoring, and research. Adoption concentrates in hospital laboratories, endocrinology centers, fertility clinics, anti-doping laboratories, and pharmaceutical research units. Global scope aligns with rising endocrine disorder diagnosis and hormone-based therapy use. Usage centers on immunoassay and mass spectrometry-compatible reagents designed for accurate detection of cortisol, testosterone, estradiol, progesterone, and related steroids.
How do diagnostic accuracy requirements and clinical decision dependence shape adoption?
Steroid hormones influence multiple physiological systems, making precise measurement critical for diagnosis and treatment adjustment. Demand increases where clinical decisions depend on narrow concentration ranges and diurnal variation patterns. Endocrinology practices require reliable reagents to distinguish pathological states from normal hormonal fluctuation. Fertility and reproductive medicine rely on serial hormone measurements to guide treatment timing and dosing. Mass spectrometry-compatible reagents gain relevance where immunoassay cross-reactivity limits specificity. Laboratories prioritize reagents with consistent lot performance and matrix compatibility across serum, plasma, and urine. Adoption reflects dependence on analytically robust inputs to support high-stakes clinical interpretation and longitudinal patient monitoring.
How do analytical interference, regulatory oversight, and cost exposure influence market scalability?
Steroid measurement faces interference from structurally similar compounds and metabolites. Demand sensitivity rises where reagent cross-reactivity compromises result reliability. Regulatory requirements mandate validation, calibration traceability, and ongoing quality control, increasing laboratory workload. Transition toward mass spectrometry increases technical complexity and reagent qualification effort. Reagent cost affects testing frequency in routine screening contexts. Sample preparation requirements add labor and turnaround time pressure. Standardization challenges persist across platforms and laboratories. Scalability remains constrained by specificity expectations, compliance burden, and economic tradeoffs between analytical rigor and testing volume across clinical and research settings.
How Is Demand for Steroid Measurement Reagents Evolving Globally?
Demand for steroid measurement reagents is expanding globally as endocrine diagnostics, reproductive health testing, and chronic disease management intensify. Clinical laboratories prioritize accurate quantification of cortisol, testosterone, estradiol, and aldosterone to support diagnosis and therapy monitoring. Growth of automated immunoassay and LC-MS workflows improves testing throughput and reproducibility. Guideline-driven testing frequency and reimbursement clarity influence adoption. Growth rates in India at 9.9%, Brazil at 9.4%, China at 8.9%, the USA at 5.9%, and Germany at 5.8% indicate sustained expansion driven by endocrine care standardization, laboratory modernization, and protocol-led monitoring rather than population-wide screening expansion.
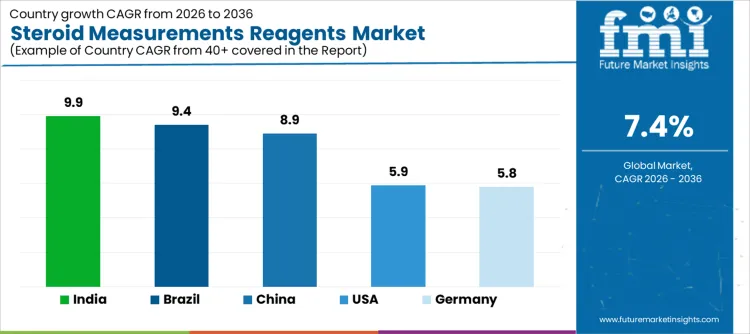
| Country | CAGR (%) |
|---|---|
| India | 9.9% |
| Brazil | 9.4% |
| China | 8.9% |
| USA | 5.9% |
| Germany | 5.8% |
What Is Driving Accelerated Growth of Steroid Measurement Reagent Demand in India?
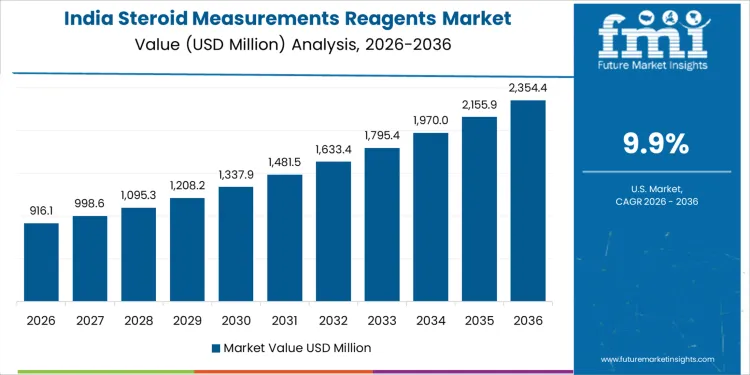
Steroid measurement reagent demand in India is growing at a CAGR of 9.9%, supported by rising diagnosis of endocrine and metabolic disorders. Fertility clinics and obstetric centers expand hormone testing volumes for assisted reproduction monitoring. Tertiary hospitals increase cortisol and adrenal hormone testing for chronic disease management. Expansion of private diagnostic chains accelerates reagent consumption through standardized platforms. Improved access to automated immunoassays reduces turnaround times. Growth reflects healthcare access expansion, protocol adoption, and laboratory scaling rather than changes in disease prevalence.
- Expansion of fertility and reproductive health testing
- Increased endocrine disorder diagnosis rates
- Growth of private diagnostic laboratory networks
- Wider adoption of automated immunoassay platforms
Why Is Brazil Experiencing Strong Uptake of Steroid Measurement Reagents?
Steroid measurement reagent demand in Brazil is expanding at a CAGR of 9.4%, driven by public healthcare diagnostics and women’s health programs. Hospitals integrate hormone testing within endocrine and reproductive care pathways. Public laboratories increase reagent procurement to support chronic disease monitoring. Academic medical centers apply LC-MS-based steroid profiling for complex cases. Regional laboratories expand service coverage beyond major cities. Growth reflects institutional investment and standardized diagnostic protocols rather than episodic testing demand.
- Public healthcare hormone testing programs
- Women’s health and reproductive diagnostics
- Adoption of LC-MS for complex profiling
- Regional laboratory service expansion
How Is China Sustaining Momentum in Steroid Measurement Reagent Utilization?
Steroid measurement reagent demand in China is growing at a CAGR of 8.9%, aligned with large-scale hospital diagnostics and chronic disease management. High patient volumes increase routine hormone testing across endocrinology departments. Public insurance coverage supports clinically indicated assays. Domestic reagent manufacturers supply cost-competitive kits compatible with automated analyzers. Central laboratory models enable high-throughput testing. Growth reflects healthcare system scale, reimbursement alignment, and laboratory capacity expansion rather than discretionary testing growth.
- High-volume hospital endocrine testing
- Insurance-backed diagnostic assay coverage
- Cost-competitive domestic reagent supply
- Centralized high-throughput laboratory models
What Factors Are Supporting Steroid Measurement Reagent Demand in United States?
Steroid measurement reagent demand in the United States is expanding at a CAGR of 5.9%, supported by guideline-driven endocrine care and laboratory standardization. Hormone testing remains integral to adrenal, reproductive, and metabolic disorder management. Strong penetration of automated immunoassay and LC-MS platforms sustains reagent usage. Reimbursement stability supports routine testing volumes. Replacement demand and assay refinement drive incremental growth. Expansion reflects protocol adherence and technology refresh cycles rather than increased testing indications.
- Guideline-mandated endocrine testing protocols
- Broad use of automated and LC-MS platforms
- Stable reimbursement for diagnostic assays
- Replacement-driven reagent demand
Why Is Germany Showing Steady Expansion in Steroid Measurement Reagents?
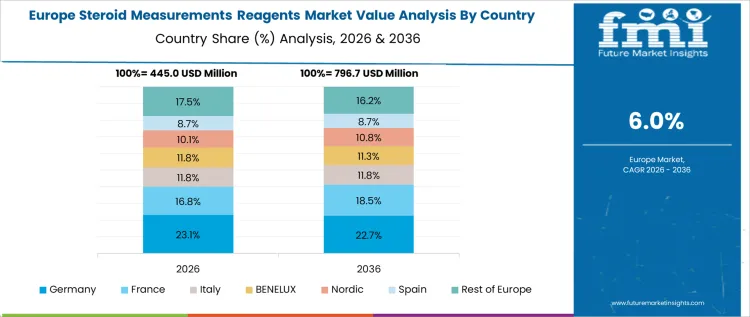
Steroid measurement reagent demand in Germany is growing at a CAGR of 5.8%, shaped by evidence-based diagnostics and laboratory quality standards. University hospitals and certified laboratories apply hormone testing within defined clinical pathways. Statutory insurance supports reimbursement for approved assays. Emphasis on analytical accuracy promotes use of validated reagents. Centralized laboratory networks ensure consistency and cost control. Growth remains steady, reflecting structured diagnostic utilization and governance rather than rapid volume escalation.
- Evidence-based endocrine diagnostic pathways
- Statutory insurance reimbursement coverage
- Emphasis on analytical accuracy and validation
- Central laboratory network standardization
What is the competitive landscape of demand for the steroid measurements reagents market globally?
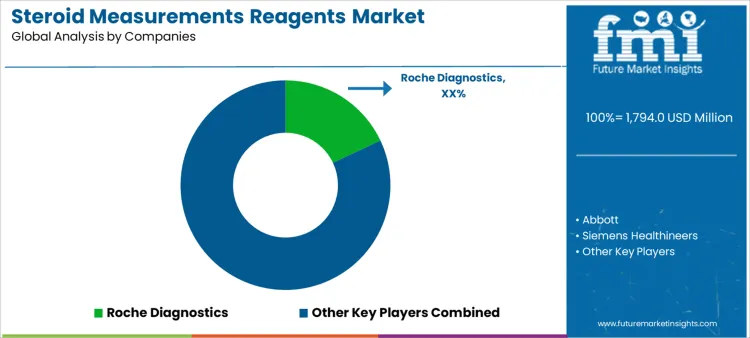
Demand for steroid measurement reagents is driven by endocrine disorder diagnosis, fertility assessment, and therapeutic monitoring across hospital and reference laboratories. Laboratories evaluate assay sensitivity at low concentration ranges, specificity across structurally similar steroids, lot-to-lot consistency, and turnaround reliability. Buyer assessment includes compatibility with automated immunoassay platforms, calibration stability, regulatory approvals, and integration with laboratory information systems. Procurement behavior reflects analyzer standardization, long-term reagent supply agreements, and preference for validated clinical performance. Trend in the steroid measurements reagents market shows sustained testing volumes supported by chronic disease monitoring and expanded endocrine screening protocols.
Roche Diagnostics leads competitive positioning through automated steroid immunoassays embedded within widely installed clinical laboratory systems. Abbott competes with high-throughput reagent menus designed for routine endocrine testing in hospital laboratories. Siemens Healthineers supports demand via steroid assays integrated with scalable automation and data management platforms. Beckman Coulter maintains relevance by supplying reagents aligned with core laboratory workflows and consistent analytical performance. DiaSorin participates through specialty endocrine reagents positioned for accuracy and reliability in targeted testing environments. Competitive differentiation centers on analytical specificity, platform integration, regulatory breadth, and continuity of reagent supply.
Key Players in the Steroid Measurements Reagents Market
- Roche Diagnostics
- Abbott
- Siemens Healthineers
- Beckman Coulter
- DiaSorin
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Reagents | Calibrators & sample prep reagents; LC-MS/MS steroid panels; Cortisol assay reagents; Testosterone assay reagents; Estradiol assay reagents; Progesterone assay reagents |
| Applications | Endocrine disorder diagnosis; Fertility & IVF monitoring; Adrenal disorder workups; Pediatric endocrinology; Hormone-dependent oncology |
| End User | Hospital laboratories; Reference laboratories; Endocrinology clinics; Fertility clinics; Academic & research labs |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Roche Diagnostics; Abbott; Siemens Healthineers; Beckman Coulter; DiaSorin; Others |
| Additional Attributes | Dollar sales by reagent type, application, and end user; analytical sensitivity and specificity requirements for low-level steroid quantification across LC-MS/MS and immunoassay workflows; role of calibrators and sample preparation in accuracy and inter-laboratory comparability; testing demand driven by endocrine, fertility, pediatric, and oncology diagnostics; procurement patterns shaped by centralized reference labs and hospital testing volumes. |
Steroid Measurements Reagents Market by Segment
Reagents:
- Calibrators & sample prep reagents
- LC-MS/MS steroid panels
- Cortisol assay reagents
- Testosterone assay reagents
- Estradiol assay reagents
- Progesterone assay reagents
Applications:
- Endocrine disorder diagnosis
- Fertility & IVF monitoring
- Adrenal disorder workups
- Pediatric endocrinology
- Hormone-dependent oncology
End User:
- Hospital laboratories
- Reference laboratories
- Endocrinology clinics
- Fertility clinics
- Academic & research labs
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Organization for Standardization. (2023). ISO 15189: Medical laboratories-Requirements for quality and competence (Latest consolidated edition). ISO.
- United States Food and Drug Administration. (2023). Bioanalytical method validation: Guidance for industry (Updated guidance). USA. Department of Health and Human Services.
- European Medicines Agency. (2024). Guideline on bioanalytical method validation for endogenous compounds, including hormones and steroids (Current revision). EMA.
- Journal of Clinical Endocrinology & Metabolism Editorial Board. (2023). Analytical considerations for accurate steroid hormone measurement in clinical practice. Endocrine Society.
- World Health Organization. (2023). WHO technical guidance on laboratory assessment of steroid hormones for endocrine and reproductive health. WHO.
Frequently Asked Questions
How big is the steroid measurements reagents market in 2026?
The global steroid measurements reagents market is estimated to be valued at USD 1,794.0 million in 2026.
What will be the size of steroid measurements reagents market in 2036?
The market size for the steroid measurements reagents market is projected to reach USD 3,663.2 million by 2036.
How much will be the steroid measurements reagents market growth between 2026 and 2036?
The steroid measurements reagents market is expected to grow at a 7.4% CAGR between 2026 and 2036.
What are the key product types in the steroid measurements reagents market?
The key product types in steroid measurements reagents market are calibrators & sample prep reagents, lc-ms/ms steroid panels, cortisol assay reagents, testosterone assay reagents, estradiol assay reagents and progesterone assay reagents.
Which applications segment to contribute significant share in the steroid measurements reagents market in 2026?
In terms of applications, endocrine disorder diagnosis segment to command 28.0% share in the steroid measurements reagents market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Reagents
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Reagents , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Reagents , 2026 to 2036
- Calibrators & sample prep reagents
- LC-MS/MS steroid panels
- Cortisol assay reagents
- Testosterone assay reagents
- Estradiol assay reagents
- Progesterone assay reagents
- Calibrators & sample prep reagents
- Y to o to Y Growth Trend Analysis By Reagents , 2021 to 2025
- Absolute $ Opportunity Analysis By Reagents , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Endocrine disorder diagnosis
- Fertility & IVF monitoring
- Adrenal disorder workups
- Pediatric endocrinology
- Hormone-dependent oncology
- Endocrine disorder diagnosis
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital laboratories
- Reference laboratories
- Endocrinology clinics
- Fertility clinics
- Academic & research labs
- Hospital laboratories
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Reagents
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagents
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Reagents
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Reagents
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Roche Diagnostics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott
- Siemens Healthineers
- Beckman Coulter
- DiaSorin
- Others
- Roche Diagnostics
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Reagents , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Reagents
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Reagents
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Reagents
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Reagents
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Reagents
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Reagents
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Reagents
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Reagents , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Reagents , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Reagents
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

