The global tissue protectors market is set to record a valuation of USD 2.3 billion in 2026, advancing toward USD 4.7 billion by 2036, representing a 7.6% CAGR. High volume of surgeries necessitate the use of advanced sterile barriers to mitigate hospital-acquired conditions, a steady demand environment validated when Medtronic plc reported FY25 revenue of USD 33.5 billion, noting consistent performance where the Medical Surgical Portfolio includes the Surgical & Endoscopy (SE) and the Acute Care & Monitoring (ACM) divisions.
Expansion is further driven by a fundamental shift where secure, ring-based retraction systems are replacing legacy cloth drapes to enable cleaner operative fields. The implementation of strict infection control protocols is now standardizing across emerging economies, validating the immense scalability of these frameworks in preventing postsurgical complications.
Clinical guidelines are increasingly mandating the use of protection devices to ensure patient safety standards are met globally. The World Health Organization (WHO) explicitly advises to "consider the use of wound protector devices in clean-contaminated, contaminated and dirty abdominal surgical procedures for the purpose of reducing the rate of SSI."
Based on Future Market Insights’ analysis, companies that align with these global protocols will dominate the abdominal & general surgery segment, which currently accounts for 28.1% of applications. This regulatory pressure compels hospitals to abandon inconsistent manual retraction methods, a shift formalized when health authorities began linking reimbursement penalties directly to postsurgical infection rates. The adoption of these mandated protocols ensures that verified wound protection becomes a non-negotiable component of the modern surgical tray rather than an optional accessory.
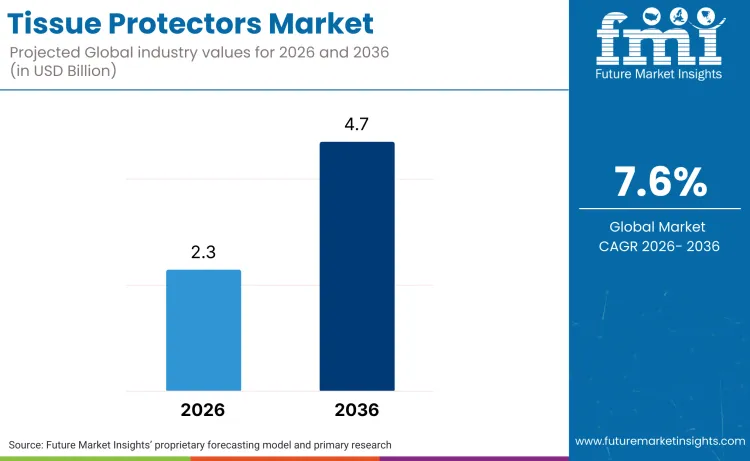
Future Market Insights projects the global tissue protectors market to expand at a CAGR of 7.6% from 2026 to 2036, rising from USD 2.3 billion in 2026 to USD 4.7 billion by 2036.
FMI Research Approach: Based on FMI’s proprietary bottom‑up revenue forecasting model, procedure volume modeling across abdominal and general surgeries, SSI incidence normalization using CDC inpatient surgery data, and scenario analysis aligned with tightening infection control enforcement.
FMI analysts expect the market to evolve from traditional passive incision barriers toward standardized, guideline‑mandated wound protection systems integrated into surgical trays and hospital infection‑prevention protocols.
FMI Research Approach: Supported by WHO recommendations for wound protector use, hospital procurement transitions from cloth drapes to ring‑based protectors, strengthened SSI‑linked reimbursement penalties, and the expansion of protocol‑driven infection prevention bundles.
The United States holds the largest share of the global tissue protectors market by value.
FMI Research Approach: Based on country‑level revenue modeling across acute care hospitals, high surgical procedure volumes, intensive SSI surveillance, early adoption of WHO/CDC‑aligned standards, and strong penetration of premium wound edge protectors.
The global tissue protectors market is projected to reach USD 4.7 billion by 2036.
FMI Research Approach: Based on long‑term revenue forecasts by product type and end‑user mix, scaling assumptions for wound edge protectors in abdominal surgery, exclusion of reusable retractors and post‑closure dressings, and cross‑validation with historical growth in surgical consumables.
The tissue protectors market includes sterile, single‑use devices designed to retract incisions and protect wound edges from contamination in clean‑contaminated and contaminated surgical procedures.
FMI Research Approach: Based on FMI’s taxonomy defining product scope across wound edge protectors and incision drapes, mapping across abdominal, colorectal, OB/GYN, and cardiac surgeries, and excluding reusable retractors and pharmaceutical‑only products.
Globally unique trends include mandatory infection control harmonization, distribution channel consolidation through vertical integration, and sustainability‑driven procurement requirements across surgical consumables.
FMI Research Approach: Supported by WHO‑aligned regulatory tightening, consolidation of surgical distribution channels, EU tenders with environmental weighting, and increasing preference for single‑use, protocol‑compliant wound protection systems.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.3 billion |
| Industry Value (2036) | USD 4.7 billion |
| CAGR (2026-2036) | 7.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
Rising infection rates in specific high-volume procedures are fundamentally reshaping the tissue protectors landscape by creating an urgent clinical imperative for better barrier devices. The CDC reported a notable 8% increase in abdominal hysterectomy SSIs in 2024, highlighting a critical unmet need in OB/GYN surgeries and complex pelvic reconstruction. This data point is significant because it forces hospitals to re-evaluate their standard surgical trays and invest in upgraded prophylaxis tools to avoid readmission penalties. Becton, Dickinson and Company (BD) validated this trend in their FY2024 report, stating that "surgery performance reflects double-digit growth in Infection Prevention and Phasix hernia resorbable scaffold that was partially offset by a tough prior-year comparison in Advanced Repair & Reconstruction." This drives demand not just for solitary protectors but for a holistic prevention suite, often integrating tissue sealants and tissue adhesive technologies, as the WHO also recommends "Triclosan-coated sutures" alongside physical barriers to create a multi-layered defense against contamination.
The tissue protectors industry is segmented by product type, application, and end user. By product, the market includes wound edge protectors, surgical retractors, and incision drapes, reflecting the varying needs for retraction versus pure barrier protection. By application, the sector covers general surgery, obstetrics & gynecology, colorectal, and cardiac procedures, driven by procedure-specific infection risks. By end user, the market is categorized into hospitals, ambulatory surgical centers, and specialty clinics. FMI analysis suggests that segmentation is evolving from simple physical barriers toward integrated infection control systems, with product choices increasingly dictated by the specific "clean-contaminated" classification of the surgery being performed.
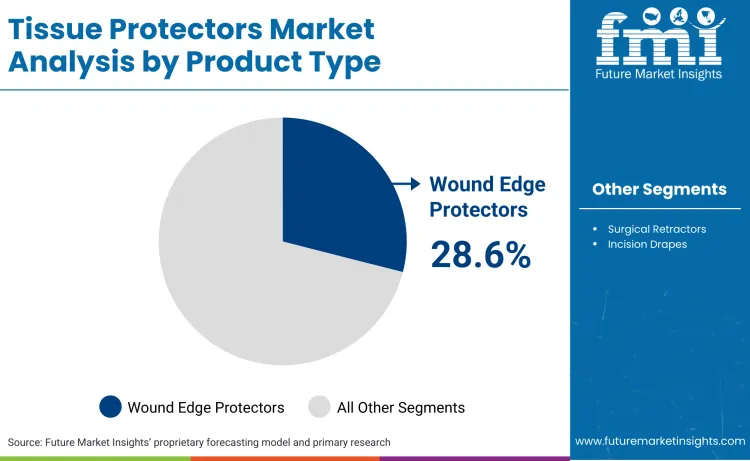
Wound edge protectors command 28.6% market share, driven by their dual capability to provide atraumatic retraction while simultaneously isolating the incision from visceral bacteria. The WHO guidelines specifically cite these devices for "clean-contaminated" procedures, making them indispensable during bowel resections and hernia repair devices applications where the risk of fecal contamination is elevated. The widespread reliance on these systems is evident as Applied Medical marked a major milestone, noting that "Applied Medical is commemorating 20 years of wound protection since the first Alexis wound protector-retractor was introduced. Over the last two decades, Alexis devices have protected 17 million wounds in over 75 countries." The segment's dominance is further reinforced by the shift toward minimally invasive techniques, where maintaining a clean port site is critical for the deployment of surgical clips and other laparoscopic instruments. Manufacturers are responding by expanding size ranges to accommodate diverse incision lengths, ensuring that standard of care adoption in laparoscopic and open surgeries remains the primary volume driver for the foreseeable future.
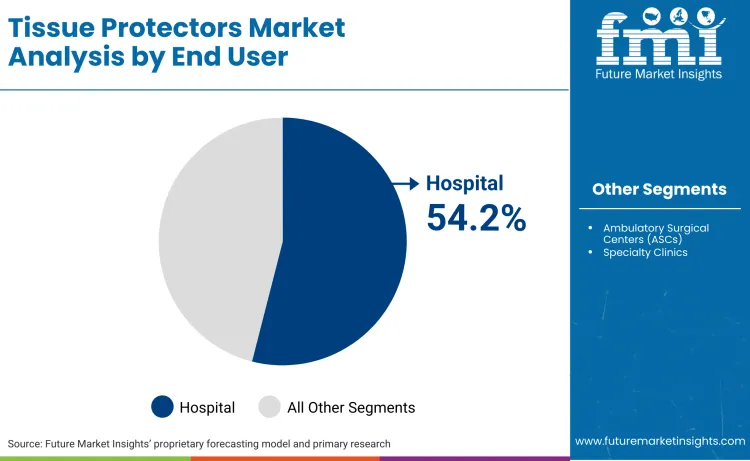
Hospitals remain the dominant end-user, holding 54.2% of the market, as they handle the vast majority of major surgeries requiring deep cavity access. Economic motivation is crucial because it shifts purchasing decisions from pure cost-minimization to value-based care, where the upfront cost of a protector is justified by the avoidance of costly infection treatments. Hospitals are increasingly signing long-term bulk contracts for these devices to standardize care protocols across their surgical departments.
Emerging economies are aggressively aligning with global safety standards, raising entry barriers for generic manufacturers while favoring established multinationals. ANVISA (Brazil) enforced RDC 848/2024 in September 2024, mandating clinical data for high-risk devices, which effectively forces non-compliant local players out of the market. ANVISA officially stated that "by incorporating global best practices and leveraging technological advances, RDC 848/2024 marks a significant step toward improving the safety and efficacy of medical devices in Brazil." The implication for the market is a consolidation of share toward global leaders like Medtronic and Applied Medical, who already possess extensive clinical dossiers and can easily meet these new rigorous requirements, leaving smaller regional competitors struggling to fund the necessary validation studies required to compete with established general surgery devices.
Competitors are shifting toward vertical integration to control the surgical service point, fundamentally altering how devices are marketed and sold to healthcare facilities. Cardinal Health acquired a majority stake in GI Alliance, the leading gastroenterology MSO in the US, for approximately USD 2.8 billion in August 2025, effectively securing a captive channel for its products. Jason Hollar, CEO of Cardinal Health, commented on their strategy, noting "we continued to accelerate our momentum with a relentless focus on driving simplification and operational efficiencies in our core, while investing for long-term growth." By owning the GI surgery channel, distributors can preferentially position their own or partnered wound protection portfolios alongside complementary surgical wound care solutions, squeezing out independent vendors and suggesting that future market share will be won on the ability to integrate into large-scale managed services organizations.
With EU tenders increasingly weighing environmental impact, sustainable manufacturing has evolved from a corporate social responsibility metric to a "right to play" requirement for market access. Mölnlycke Health Care achieved 100% renewable electricity sourcing in 2024, setting a new benchmark that competitors must match to remain viable in European bidding processes. This commitment was highlighted in the Mölnlycke Health Care annual report, which stated "in 2024, Mölnlycke reached its SBTi-validated near-term target of sourcing 100% renewable electricity for all manufacturing sites and HQ." This trend impacts the entire supply chain, forcing changes in everything from packaging materials to the production of United kingdom absorbable tissue spacer products, as failing to decarbonize production lines could result in exclusion from lucrative public health contracts regardless of clinical performance.
The global tissue protectors landscape is witnessing a divergence where emerging markets like India and Brazil are outpacing developed regions, with India projected at 10.1% CAGR versus Germany's 2.4%. This shift is driven by rapidly expanding surgical volume and hospital construction in Asia versus the replacement-driven nature of the European market, where demand for noninvasive surgical wound closure and similar niche products is stabilizing. While Western markets focus on premium, specialized devices for complex procedures, developing nations are scaling up basic infection control infrastructure. Consequently, the volume growth for the next decade will be heavily concentrated in the Global South, forcing multinational corporations to tailor their pricing and distribution strategies to capture these high-growth but price-sensitive opportunities.
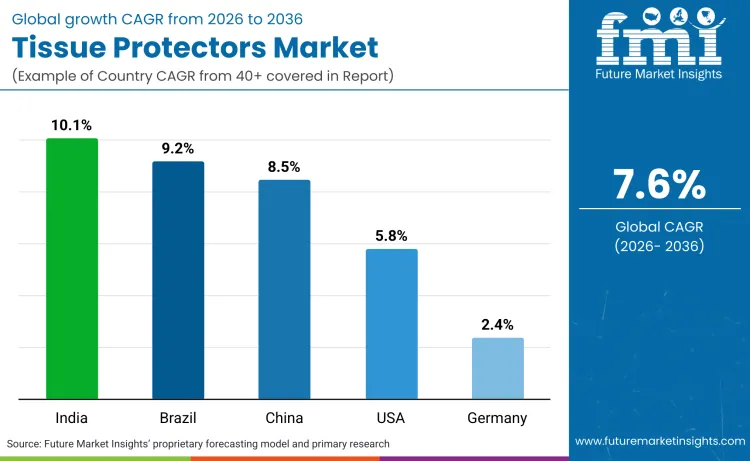
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.1% |
| Brazil | 9.2% |
| China | 8.5% |
| USA | 5.8% |
| Germany | 2.4% |
Source: FMI historical analysis and forecast data.
Sales of tissue protectors in India are growing at a rapid 10.1% CAGR, fueled by a massive expansion in private healthcare infrastructure and increased access to elective surgeries. Global surgical volume growth, particularly in lower-middle income countries, drives demand for basic infection control and postoperative panniculus retractor adoption as standard protocols. This region represents the volume engine for the next decade as private healthcare chains expand into Tier 2 cities, necessitating reliable, standardized surgical supplies to maintain accreditation and patient trust in these new facilities. The focus is shifting from simple access to comprehensive safety, with hospitals increasingly adopting single-use sterile protectors to mitigate the high costs associated with hospital-acquired infections in a price-sensitive reimbursement environment.
The market in Brazil is projected to grow at 9.2% CAGR, driven by a rigorous overhaul of medical device regulations that favors high-quality imports. Harmonization with IMDRF standards via ANVISA RDC 848/2024, implemented in September 2024, allows easier entry for premium US and EU devices that have already cleared strict safety hurdles. This regulation forces consolidation as smaller, non-compliant manufacturers exit, creating space for established players in the hernia mesh devices and protection space to expand their market share. The new rules effectively raise the floor for quality, ensuring that Brazilian patients receive the same standard of care as those in developed nations, and compelling hospitals to invest in verified, clinical-grade protection systems rather than cheaper, unverified alternatives.
China is poised to register an 8.5% CAGR, characterized by a surge in domestic manufacturing and a regulatory preference for locally produced medical technologies. The NMPA approved 13,133 medical device registrations in 2024, a 7.5% increase, signaling a robust environment for local innovation and product launches. Data from the NMPA indicates that "Domestic Class III applications rose 7% while imports fell 5.9%," highlighting a clear shift where global players face stiff competition from local manufacturers in the commoditized wound protector segment. This trend mirrors developments in the powered surgical staplers sector, where Chinese companies are rapidly closing the quality gap. To succeed, international firms must now localize their production or form strategic joint ventures, as the "Made in China" policy continues to reshape procurement priorities in public hospitals.
The competitive landscape is heavily concentrated around established leaders like Applied Medical, Medtronic, and BD, who leverage their massive scale and distribution networks to maintain dominance. This market structure is reinforced by extensive clinical evidence and a massive installed base, creating a high barrier to trust any new entrant attempting to displace them. Mölnlycke Health Care highlighted their own competitive strength in their financial report, noting that "Wound Care continued to outperform the market demonstrated by an 8% organic growth ending the year with more than EUR 1.2 billion in sales." These giants use their portfolio breadth to bundle protectors with other essential consumables, making it operationally difficult for hospitals to switch to standalone suppliers.
Medtronic validated this steady demand environment when they explicitly stated in their May 2025 report that "FY25 revenue of $8.407 decreased 0.1% reported and increased 0.8% organic," signaling that growth in mature categories requires strategic innovation rather than just volume expansion. Competitors are responding by shifting toward vertical integration and sustainability to differentiate themselves in a crowded market. Cardinal Health's USD 2.8 billion acquisition of GI Alliance signals a move to control the surgical service point, while competitors differentiate through sustainability, achieving renewable energy targets to appeal to European tenders. Medtronic remains focused on portfolio optimization, with new leadership in its Cardiovascular portfolio likely to influence future investments in the minimally invasive neurosurgery devices and general surgery sectors.
Recent Developments:
In September 2024, Solventum launched the V.A.C. Peel and Place Dressing, an all-in-one dressing and drape for negative pressure wound therapy. This innovation simplifies application, reduces dressing change time, and features a non-adherent layer to prevent tissue ingrowth, improving patient comfort in wound protection.
The tissue protectors market represents revenue generated from sterile, single-use devices designed to retract incisions and protect wound edges from contamination during surgery. As operationally defined in this article, the market measures commercial sales of wound edge protectors, retractors, and surgical retractors solutions used in hospitals and ambulatory centers. Market sizing reflects the value of these consumable devices sold to healthcare providers, analyzed by product type, application, and end-user, and expressed in USD billion. The market includes devices explicitly used for "clean-contaminated" and "contaminated" procedures as defined by the CDC, including abdominal, colorectal, and Caesarean sections.
It covers both active and passive protection systems but excludes pharmaceutical agents unless integrated into the device matrix. The scope counts adoption by hospitals, ambulatory surgical centers, and specialty clinics across key global geographies. It includes revenue from ring-based retractors and incision drapes but excludes general surgical drapes that do not provide specific wound edge retraction and protection functions. The market analysis excludes revenue from surgical capital equipment, reusable retractors, and general wound care dressings applied post-closure. Government healthcare spending data and general hospital budget allocations are referenced for context but are not counted as direct market revenue.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.3 billion |
| Product Type | Wound Edge Protectors, Surgical Retractors, Incision Drapes, Ring-Based Retraction Systems |
| Application Category | General Surgery, Obstetrics & Gynecology (OB/GYN), Colorectal Surgery, Cardiac Surgery, Abdominal Surgery, Caesarean Sections |
| End User | Hospitals, Ambulatory Surgical Centers (ASCs), Specialty Clinics, Acute Care Facilities, Maternity Wards |
| Procedure Classification | Clean-Contaminated Procedures, Contaminated Surgeries, Dirty Abdominal Surgical Procedures, Bowel Resections, Hernia Repair, Pelvic Reconstruction |
| Device Characteristics | Sterile Single-Use Devices, Ring-Based Protection Systems, Integrated Barrier Technologies, Atraumatic Retraction Mechanisms, Multi-Size Formats |
| Clinical Focus | Surgical Site Infection (SSI) Prevention, Wound Edge Protection, Incision Site Isolation, Visceral Bacteria Containment, Hospital-Acquired Infection Control |
| Compliance Framework | WHO Infection Control Guidelines, CDC SSI Surveillance Standards, Protocol-Driven Infection Prevention Bundles, Reimbursement-Linked Quality Metrics |
| Distribution Strategy | Vertical Integration Models, Surgical Service Point Control, Managed Services Organizations (MSO) Integration, Bulk Contract Procurement, Standardized Surgical Tray Inclusion |
| Regulatory Alignment | IMDRF Standards Harmonization, ANVISA RDC 848/2024 (Brazil), NMPA Registration Pathways (China), FDA 510(k) Clearance, European Public Tender Compliance |
| Sustainability Requirements | Renewable Electricity Sourcing, Circular Material Compatibility, Environmental Impact Weighting in Procurement, Decarbonized Production Lines |
| Technology Integration | Minimally Invasive Surgery Compatibility, Laparoscopic Port Site Protection, Complementary Surgical Consumable Bundling, Multi-Layered Defense Systems |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, China, India, Brazil, and 20+ countries |
| Key Companies Profiled | Applied Medical, Medtronic, Becton Dickinson and Company (BD), Cardinal Health, Mölnlycke Health Care, 3M, Prescient Surgical, Ethicon (Johnson & Johnson) |
| Additional Attributes | Dollar sales by product type and application, regional demand trends, competitive landscape analysis, wound edge protector adoption trajectories, hospital procurement standardization, infection control protocol enforcement, regulatory harmonization impact, strategic channel consolidation, sustainability-driven procurement criteria, emerging market infrastructure scaling, clinical evidence requirements, reimbursement penalty linkage, value-based care purchasing, domestic manufacturing localization, guideline-mandated adoption patterns, surgical tray integration strategies, and vertical MSO acquisition trends |
Source: FMI historical analysis and forecast data.
What is the current global market size for tissue protectors?
The global industry is valued at USD 2.3 billion in 2026, driven by strict infection control mandates from the CDC and WHO.
What is the projected CAGR for the tissue protectors market?
Market revenues are projected to expand at a 7.6% CAGR from 2026 to 2036.
Which segment currently dominates the industry?
Wound edge protectors hold the dominant share of 28.6%, serving as the standard of care in abdominal surgeries.
What are the primary drivers for adoption?
Adoption is forced by the need to reduce Surgical Site Infections (SSIs), as hospital reimbursements are increasingly tied to infection rates.
Who are the leading players in the tissue protectors space?
Applied Medical, Medtronic, and BD lead the sector, with Applied Medical's Alexis device protecting over 17 million wounds globally.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.