Demand for Intelligent Prosthetics in UK
Demand for Intelligent Prosthetics in UK Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Intelligent Prosthetics Industry Forecast and Outlook in the UK 2026 to 2036
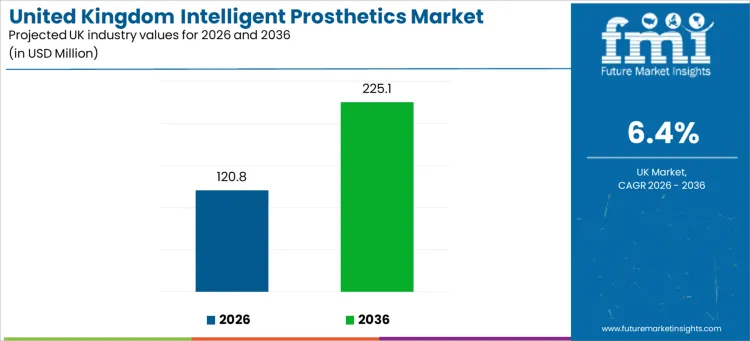
Demand for intelligent prosthetics in the UK is projected to reach a valuation of USD 120.81 million in 2026. This sector is anticipated to expand to USD 225.11 million by 2036, advancing at a 6.4% CAGR.
This expansion reflects a clear shift in limb replacement priorities across the UK healthcare ecosystem. Patients, clinicians, and procurement teams are looking beyond basic mobility support toward sensor-enabled prosthetic limbs, robotic actuation, and AI-assisted control that improve function in daily living.
Device capability is no longer defined only by mechanical motion. It is increasingly shaped by software calibration, myoelectric signal processing, comfort-driven socket design, and continuous rehabilitation alignment that keeps outcomes stable over time.
For hospital leaders and prosthetic service heads, the focus sits on practical performance indicators: safe ambulation, reduced fatigue, fewer fit-related complications, and faster return to independence.
For technology providers, the value pathway is defined by clinical acceptance, evidence-backed outcomes, device uptime, and seamless integration into prosthetic care pathways that span assessment, fitting, training, and long-term follow-up.
Quick Stats for Intelligent Prosthetics Demand in the UK
- Intelligent Prosthetics Valuation in the UK (2026): USD 120.81 million
- Intelligent Prosthetics Forecast Valuation in the UK (2036): USD 225.11 million
- Intelligent Prosthetics Forecast CAGR (2026 to 2036): 6.4%
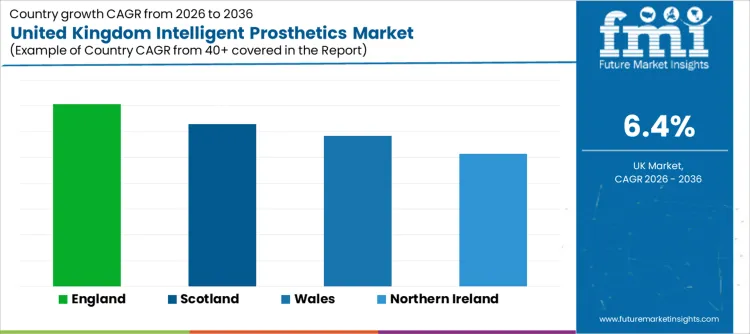
- Leading Regional Growth: England (CAGR 7.1%)
- Leading Product Type: Upper Extremity (53%)
- Leading Distribution Channel: Hospitals (46.1%)
- Leading Players: Sarcos Technology and Robotics Corporation; Ktwo Healthcare Pvt. Ltd.; Touch Bionics Inc.; HDT Global; SynTouch, Inc.
Why is the UK Emerging as a High-Impact Adoption Hub for Intelligent Prosthetic Care?
The UK stands out as a strong adoption environment because prosthetic care is closely tied to structured clinical services, specialist teams, and formal referral pathways. This creates a clear route for advanced devices to move from evaluation to real-world use, especially when functional outcomes can be demonstrated in therapy settings and daily living activities. NHS England’s updated service specification for amputee rehabilitation and prosthetic services places strong emphasis on structured assessment, multidisciplinary input, and ensuring each person receives a clinically appropriate prosthesis aligned with capability and safety.
Demand is also shaped by broader population needs. The UK has a sizable disabled population base, with parliamentary research citing 16.8 million people living with disability in 2023/24, representing 25% of the total population. While limb loss is only one part of this landscape, the statistic reinforces why assistive technology and functional restoration remain national priorities across healthcare planning and social participation outcomes.
Innovation pull comes from rapid gains in myoelectric control, multi-articulating hands, adaptive ankles, and sensory feedback concepts. Procurement teams increasingly benchmark clinical direction using global intelligence from the intelligent prosthetics outlook, while also tracking adjacent device ecosystems such as prosthetics and orthotics outlook and medical rehabilitation robotics outlook for longer-term capability planning.
How is the Intelligent Prosthetics Landscape Segmented by Product Type and Distribution Channel?
Why do upper extremity solutions lead with a 53% share in the UK?
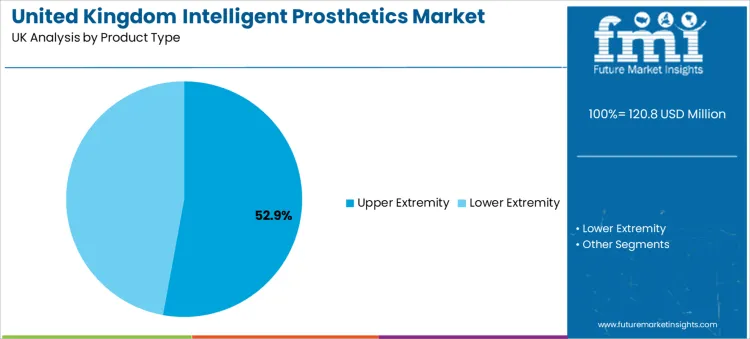
Upper extremity intelligent prosthetics hold a 53% share, reflecting the high functional value of restoring hand and arm capability across daily activities. For many users, regaining grip patterns, pinch strength, and stable object handling directly impacts independence, employability, and quality of life. These devices often rely on myoelectric sensing, intuitive control algorithms, and ergonomic design that supports longer wear time without discomfort.
Clinical teams view upper limb performance through practical outcome filters: ease of training, predictability of movement, comfort during prolonged use, and the ability to complete real tasks such as dressing, cooking, device handling, or workplace duties. Solutions that reduce cognitive load and improve motion confidence tend to gain stronger patient adherence, especially when combined with structured rehabilitation and ongoing calibration support.
Why do hospitals account for 46.1% share across distribution channels?
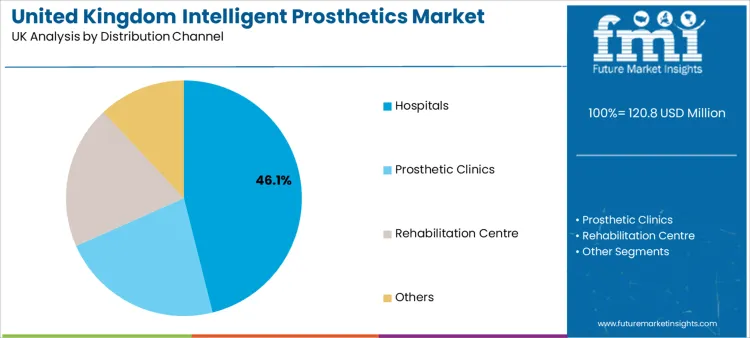
Hospitals represent a 46.1% share, showing that acute and specialist care settings remain central to evaluation, prescription decisions, and complex patient management. Hospitals are often where limb loss pathways intersect with trauma care, vascular disease management, post-surgical rehabilitation, and multidisciplinary review. This environment supports adoption of advanced prosthetic technologies that require assessment, structured training, and monitoring during early use.
For stakeholders planning scale-up, hospital-led distribution also signals the importance of procurement governance, clinical evidence packages, and service support frameworks. Technology partners that can align device integration with specialist teams, therapy protocols, and patient follow-up routines typically gain stronger placement opportunities.
What are the Dynamics, Restraints, Opportunities, and Threats Shaping this Sector?
How are clinical pathway upgrades and functional expectations driving sustained demand?
Demand is driven by an outcomes-first approach to rehabilitation. Clinicians and commissioners want solutions that improve mobility, reduce complications, and support stable long-term function.
NHS England’s revised specification for amputee rehabilitation and prosthetic services highlights structured assessment and appropriate device provision based on the person’s ability to use a prosthesis safely and effectively. This supports intelligent devices when they demonstrate clear functional benefits and predictable patient progress.
Alongside clinical drivers, national service guidance is evolving toward stronger consistency of care delivery, including suitability of consultation modes such as face-to-face or remote options when appropriate. This creates room for connected device support models, including digital adjustments, usage monitoring, and rehabilitation-aligned optimisation.
Do cost pressure, training demands, and service capacity slow adoption?
High-performance prosthetic solutions can require significant upfront investment, specialist fitting time, and therapy support for training. Adoption can slow when local service capacity is constrained or when procurement models prioritise unit cost over long-term outcomes.
Workforce availability in prosthetic services also affects rollout speed, especially when advanced devices require repeated calibration, troubleshooting, or user coaching. Service leaders often evaluate whether the full care pathway can sustain higher technology intensity without compromising patient access or clinical quality.
How are AI-enabled control, sensory feedback, and connected support creating new expansion opportunities?
Opportunities are strongest where intelligent devices reduce the gap between clinic performance and real-world use. Improvements in signal processing, adaptive gait logic, and sensor-driven stability support better function on uneven terrain, stairs, and long walking sessions.
Sensory feedback concepts, including touch perception and pressure awareness, strengthen user confidence and reduce dependency on visual monitoring of movement. This theme is also reinforced by innovation tracking across the orthopedic prosthetic device and the orthopedic prosthetic accessories domains, where liners, suspension systems, and comfort interfaces improve long-term mobility outcomes.
What regulatory and safety factors shape implementation confidence for intelligent devices?
Software and AI-driven functionality adds a compliance layer for device developers and hospital governance teams. The UK government notes that software, including AI, can be regulated as a medical device, reinforcing the need for safety, performance, and lifecycle oversight.
Clinical stakeholders also monitor procedural innovation pathways such as osseointegration approaches for selected patients, which have dedicated evidence reviews and assessment structures. These pressures increase emphasis on clinical validation, risk management, cybersecurity preparedness, and robust post-deployment monitoring.
How is Intelligent Prosthetics Demand Evolving across Key Regions in the UK?
Specialist service availability, prosthetic clinic networks, rehabilitation capacity, and procurement maturity influence regional demand patterns. The projected compound annual growth rates from 2026 to 2036 illustrate these geographic distinctions.
| Region | CAGR (2026-2036) |
|---|---|
| England | 7.1% |
| Scotland | 6.3% |
| Wales | 5.8% |
| Northern Ireland | 5.1% |
Why is England leading adoption with stronger specialist scale and service coordination?
England leads with a projected CAGR of 7.1%, supported by the concentration of specialist services, larger hospital networks, and higher system capacity for structured assessment and follow-up.
The updated national specification for amputee rehabilitation and prosthetic services also shapes care consistency, helping advanced devices gain placement when clinical value is clear. Technology suppliers often prioritise England for pilot programmes because therapy infrastructure is better positioned to support training intensity and iterative device optimisation.
How is Scotland building steady growth through outcome-driven rehabilitation delivery?
Scotland records a CAGR of 6.3%, reflecting stable adoption anchored in rehabilitation focus and a strong emphasis on functional outcomes. Service leaders often evaluate intelligent devices based on wear time, comfort stability, and measurable daily function improvements.
Procurement teams also pay attention to serviceability and support logistics, ensuring device uptime remains high even when catchment areas are spread across urban and remote communities.
What is supporting Wales as it scales intelligent limb solutions through practical care pathway fit?
Wales shows a CAGR of 5.8%, driven by the need for reliable functional restoration aligned with real service capacity. Adoption expands when devices simplify training demands and reduce complications related to socket fit, skin irritation, or fatigue. Hospitals and clinics also prioritise solutions with clear support structures, ensuring users receive consistent education and follow-up, especially during the early adaptation phase.
Why is Northern Ireland progressing with a value-assurance approach to advanced prosthetic upgrades?
Northern Ireland grows at 5.1%, reflecting measured adoption where clinical value and long-term service viability guide purchasing decisions. Providers tend to focus on robust evaluation protocols and repeatable rehabilitation workflows that protect patient confidence and safety. Leasing, shared service models, and staged upgrades can play a role when stakeholders need to balance technology advancement with predictable cost planning and support availability.
What Defines the Competitive Landscape for Intelligent Prosthetics in the UK?
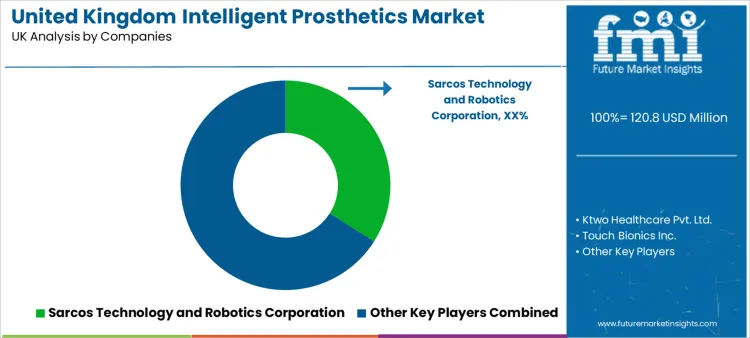
Device intelligence, comfort engineering, service readiness, and clinical integration depth shape competition. Buyers compare control responsiveness, durability, battery performance, and the realism of movement, especially during repeated daily use. Hospitals and prosthetic clinics also consider how quickly a solution can be fitted, trained, and supported over time, since patient adherence depends on reliable function and comfort stability.
Technology-forward suppliers differentiate through AI-driven calibration tools, sensor fusion for smoother motion, and refined user interfaces that reduce cognitive burden. Some competitors also prioritise tactile sensing and haptic feedback innovations that improve confidence in object handling. Decision makers assessing portfolio direction often map these capabilities against the broader medical bionic implant and exoskeleton outlook, since exoskeletons and advanced limb systems share overlapping innovation pathways in robotics, sensing, and assistive control design.
In the UK context, supplier success is closely tied to stakeholder alignment across hospitals, prosthetic clinics, and rehabilitation centres. Winning strategies typically include strong training support, clinician education, documentation readiness, and a service model that maintains device performance long after initial fitting.
Key Industry Participants
- Sarcos Technology and Robotics Corporation
- Ktwo Healthcare Pvt. Ltd.
- Touch Bionics Inc.
- HDT Global
- SynTouch, Inc.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Upper Extremity; Lower Extremity |
| Distribution Channel | Hospitals; Prosthetic Clinics; Rehabilitation Centre; Others |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Sarcos Technology and Robotics Corporation; Ktwo Healthcare Pvt. Ltd.; Touch Bionics Inc.; HDT Global; SynTouch, Inc. |
Intelligent Prosthetics Demand in the UK by Segment
By Product Type:
- Upper Extremity
- Lower Extremity
By Distribution Channel:
- Hospitals
- Prosthetic Clinics
- Rehabilitation Centre
- Others
By Region:
- England
- Scotland
- Wales
- Northern Ireland
Bibliography
- British Association of Prosthetists and Orthotists. (2025). Service Provision Guidance for Prosthetic & Orthotic Services.
- HM Government. (2025). Software and artificial intelligence (AI) as a medical device.
- National Health Service (NHS) England. (2025). Amputee rehabilitation and prosthetics services for people of all ages with limb loss and limb difference.
- National Health Service (NHS) England. (2025). Service specification: amputee rehabilitation and prosthetics services for people of all ages with limb loss and limb difference.
- National Institute for Health and Care Excellence (NICE). (2024). Osseointegrated prosthesis for rehabilitation of amputees: Overview of evidence.
- UK Parliament, House of Commons Library. (2025). UK disability statistics: Prevalence and life experiences.
Frequently Asked Questions
How big is the demand for intelligent prosthetics in uk in 2026?
The demand for intelligent prosthetics in uk is estimated to be valued at USD 120.8 million in 2026.
What will be the size of intelligent prosthetics in uk in 2036?
The market size for the intelligent prosthetics in uk is projected to reach USD 225.1 million by 2036.
How much will be the demand for intelligent prosthetics in uk growth between 2026 and 2036?
The demand for intelligent prosthetics in uk is expected to grow at a 6.4% CAGR between 2026 and 2036.
What are the key product types in the intelligent prosthetics in uk?
The key product types in intelligent prosthetics in uk are upper extremity and lower extremity.
Which distribution channel segment is expected to contribute significant share in the intelligent prosthetics in uk in 2026?
In terms of distribution channel, hospitals segment is expected to command 46.1% share in the intelligent prosthetics in uk in 2026.
Table of Content
- Executive Summary
- UK Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- UK Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- UK Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Upper Extremity
- Lower Extremity
- Upper Extremity
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospitals
- Prosthetic Clinics
- Rehabilitation Centre
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Sarcos Technology and Robotics Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Ktwo Healthcare Pvt. Ltd.
- Touch Bionics Inc.
- HDT Global
- SynTouch, Inc.
- Sarcos Technology and Robotics Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: UK Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: UK Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: UK Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 4: UK Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: UK Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: UK Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: UK Market Pricing Analysis
- Figure 2: UK Market Value (USD Million) Forecast 2021-2036
- Figure 3: UK Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: UK Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: UK Market Attractiveness Analysis by Product Type
- Figure 6: UK Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 7: UK Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 8: UK Market Attractiveness Analysis by Distribution Channel
- Figure 9: UK Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: UK Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: UK Market Attractiveness Analysis by Region
- Figure 12: UK Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: UK Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 14: UK Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 15: UK Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 16: UK Market Attractiveness Analysis by Product Type
- Figure 17: UK Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 18: UK Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 19: UK Market Attractiveness Analysis by Distribution Channel
- Figure 20: UK Market - Tier Structure Analysis
- Figure 21: UK Market - Company Share Analysis

