Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market Forecast and Outlook 2026 to 2036
The anti-static PCR resin solutions for cleanroom manufacturing market is projected to grow from USD 840 million in 2026 to USD 2,679.6 million by 2036, registering a CAGR of 12.3%. This market matters commercially because it sits at the intersection of sustainability mandates and zero-defect manufacturing economics. Cleanroom operators are under pressure to reduce virgin plastic use, yet procurement decisions remain dominated by risk containment, qualification certainty, and long-term process stability.
Buyer intent is shaped less by recycled content targets and more by assurance that electrostatic performance and contamination control will not introduce yield loss or regulatory exposure. As a result, purchasing cycles are deliberate, specification-driven, and concentrated among a limited pool of validated suppliers. Decision-making authority often rests with quality, EHS, and operations teams rather than sustainability functions, elevating the importance of documented performance and traceability. Between 2026 and 2036, economic value is expected to accrue to suppliers that reduce buyer hesitation, support lengthy validation timelines, and embed themselves early in cleanroom material standards, rather than those competing on price or recycled content claims alone.
Quick Stats for Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market
- Anti-Static PCR Resin Solutions Market Value (2026): USD 840 Million
- Anti-Static PCR Resin Solutions Market Forecast Value (2036): USD 2,679.6 Million
- Anti-Static PCR Resin Solutions Market Forecast CAGR 2026 to 2036: 12.3%
- Leading Resin Solution Type: Permanent Anti-Static Resins (41% share)
- Key Growth Regions: Asia Pacific, North America, Europe
- Top Key Players in Anti-Static PCR Resin Solutions Market: SABIC, BASF SE, Avient Corporation, Celanese Corporation, Toray Industries, Inc., Asahi Kasei Corporation, Kingfa Sci. & Tech. Co., Ltd.

Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market Key Takeaways
| Metric | Value |
|---|---|
| Anti-Static PCR Resin Solutions Market Value (2026) | USD 840 Million |
| Anti-Static PCR Resin Solutions Market Forecast Value (2036) | USD 2,679.6 Million |
| Anti-Static PCR Resin Solutions Market Forecast CAGR 2026 to 2036 | 12.3% |
How Are Contamination Risk Management and Cleanroom Standards Driving Demand for Anti-Static PCR Resin Solutions?
Demand for anti-static PCR resin solutions in cleanroom manufacturing is rising as industries seek to combine recycled content adoption with strict electrostatic discharge (ESD) and contamination-control requirements. Cleanroom environments in semiconductor fabrication, pharmaceutical manufacturing, medical devices, and precision electronics are highly sensitive to static charge buildup, which can attract particulates, damage components, or disrupt processes. This is driving demand for PCR resin formulations that deliver reliable anti-static performance while meeting cleanroom compatibility standards.
Buyer evaluation criteria are increasingly centered on performance stability rather than recycled content percentage alone. Cleanroom operators and packaging manufacturers require anti-static PCR resins with controlled surface resistivity, low ionic contamination, and minimal additive migration over time. Suppliers capable of delivering compounded solutions with consistent ESD performance across molding, extrusion, and thermoforming processes are gaining preference. Validation data, long-term performance testing, and compatibility with ISO cleanroom classifications are becoming critical differentiators during supplier qualification.
Scale-up execution and material consistency are shaping commercial success. Variability in PCR feedstock can affect electrical properties, making formulation expertise and inline quality control essential. As cleanroom manufacturing expands in Asia Pacific and North America, demand is increasing for regionally available, pre-qualified anti-static PCR materials supported by technical service teams. For compounders and additive suppliers, competitive advantage in this market depends on balancing ESD performance, cleanliness requirements, and supply reliability positioning anti-static PCR resin solutions as a strategic enabler for sustainable cleanroom manufacturing growth.
How is the Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market Structured by End Use and Resin Type?
The anti-static PCR resin solutions for cleanroom manufacturing market is segmented by end use and resin solution, reflecting how electrostatic control and contamination sensitivity influence material selection. By end use, semiconductor and medical cleanrooms represent the leading segment, supported by strict requirements for particle control and electrostatic discharge management. Other end uses include pharma and electronics manufacturing, precision cleanrooms, electronics fabs, display fabs, and pharma manufacturing environments with varying cleanliness classes. By resin solution, permanent anti-static resins form the dominant category, while ESD-safe compounds, low-outgassing resins, and anti-static ABS grades address application-specific performance and processing needs.
Why Do Semiconductor And Medical Cleanrooms Lead Demand By End Use?
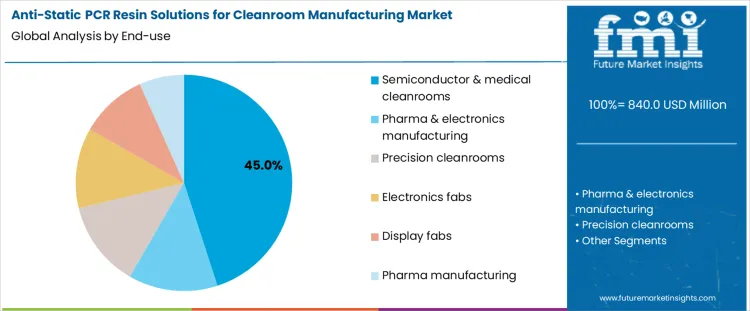
Semiconductor and medical cleanrooms account for 45% share of the anti-static PCR resin solutions for cleanroom manufacturing market because electrostatic discharge poses direct risks to product integrity and process yield. Semiconductor wafers, medical devices, and sterile components are highly sensitive to static-induced particle attraction and micro-damage. Cleanroom operators specify anti-static materials for trays, housings, panels, and handling components to maintain controlled environments. High capital intensity and continuous production cycles drive consistent material consumption. Equipment standardization across fabs and medical facilities supports repeat procurement. These operational and quality-critical factors explain why semiconductor and medical cleanrooms remain the largest end-use segment.
Why Do Permanent Anti-Static Resins Dominate Resin Solution Selection?
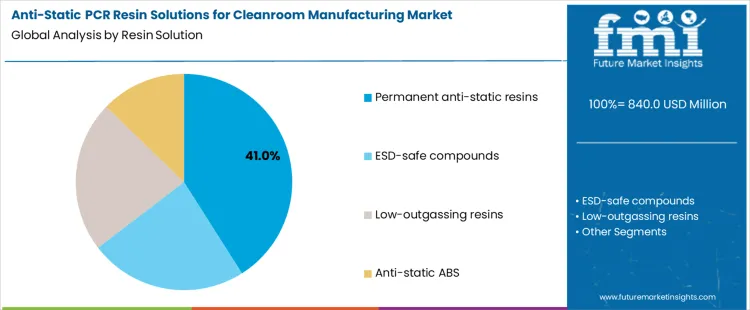
Permanent anti-static resins hold 41% share of the anti-static PCR resin solutions for cleanroom manufacturing market because they provide long-lasting electrostatic control without reliance on surface coatings or humidity levels. These resins embed conductive or dissipative properties throughout the polymer matrix, ensuring consistent performance over extended service life. Cleanroom manufacturers prefer permanent solutions to avoid degradation during cleaning cycles and repeated handling. Compatibility with injection molding and extrusion supports scalable component production. For resin suppliers, permanent anti-static grades enable standardized formulations across multiple cleanroom applications. This durability, processing compatibility, and reliability explain their dominance among resin solutions.
What Are The Key Dynamics Of The Anti-Static PCR Resin Solutions For Cleanroom Manufacturing Market?
The anti-static PCR resin solutions for cleanroom manufacturing market is driven by the growing use of post-consumer recycled (PCR) resins in precision-controlled environments where electrostatic discharge (ESD) can compromise product quality and safety. Cleanroom applications such as semiconductor handling trays, medical device components, and electronic packaging require both consistent PCR resin performance and effective static dissipation. For resin compounders, additive suppliers, and converters, balancing anti-static performance with mechanical properties (tensile strength, impact resistance) and processing reliability (melt flow, dimensional stability) is central to product acceptance. Consistent supply of high-quality PCR feedstock and technical support for cleanroom validation protocols are key competitive factors.
How Are Technical Requirements And Cleanroom Standards Shaping The Market?
Technical requirements and cleanroom standards are shaping the anti-static PCR resin market by establishing strict thresholds for surface resistivity, charge decay time, and contamination control. Manufacturers of cleanroom consumables and equipment must use materials that meet specified ESD performance ratings while ensuring no particulate generation, extractables, or chemical incompatibilities that could affect sensitive processes. Resin formulations must be optimized for cleanroom class ratings (ISO Class 5-8), meaning anti-static additives must deliver consistent performance without leaching or interfering with processing conditions. For suppliers, documentation packages including test data and conformity reports are often prerequisites for approval.
Why Do Feedstock Variability And Qualification Challenges Restrain Market Adoption?
Feedstock variability and qualification challenges restrain wider adoption of anti-static PCR resin solutions for cleanroom manufacturing. Post-consumer recycled resin streams vary in color, residual contaminants, and melt flow characteristics, complicating formulation of anti-static compounds that deliver reliable ESD control and dimensional precision. Cleanroom applications often require long qualification cycles with extensive validation testing, traceability records, and evidence that materials meet both electrical performance and cleanroom compatibility criteria. These requirements increase development time, documentation burden, and total cost of ownership for converters and end users, particularly in industries with zero-defect tolerance.
What is the Country-Wise Outlook for Anti-Static PCR Resin Solutions in Cleanroom Manufacturing?
The anti-static PCR resin solutions market for cleanroom manufacturing is shaped by rising adoption of recycled polymers in electronics, pharmaceutical, medical device, and semiconductor environments where electrostatic discharge (ESD) control is critical. Growth varies by country based on cleanroom density, regulatory acceptance of PCR materials, and performance expectations around particle generation, conductivity stability, and contamination control. Mature markets prioritize validation, consistency, and compliance, while high-growth regions focus on scaling PCR usage without compromising cleanroom integrity. Suppliers offering engineered anti-static performance, low outgassing, and documented cleanroom compatibility are best positioned across global markets.
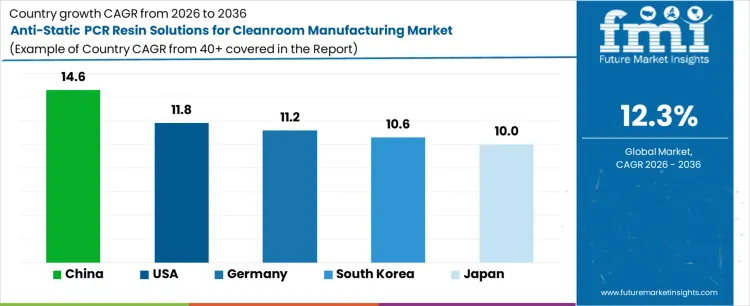
| Country | CAGR (%) |
|---|---|
| China | 14.6 |
| United States | 11.8 |
| Germany | 11.2 |
| South Korea | 10.6 |
| Japan | 10.0 |
How is Germany Advancing Validated Anti-Static PCR Resin Use in Cleanroom Environments?
Demand for anti-static PCR resin solutions for cleanroom manufacturing in Germany, growing at a CAGR of 11.2% during 2026-2036, is driven by strict contamination control standards and sustainability mandates across electronics, medical devices, and pharmaceutical production. Cleanroom operators require PCR-based resins that deliver stable surface resistivity while minimizing particle shedding and ionic contamination. Adoption is largely upgrade-led, with converters replacing virgin anti-static plastics in trays, carriers, housings, and packaging components. Buyers emphasize material traceability, repeatable ESD performance, and compliance with ISO cleanroom classifications. Qualification cycles are long and validation-intensive, often involving particle testing, aging studies, and electrostatic decay verification. Procurement favors suppliers offering compounded PCR resins with embedded anti-static functionality rather than surface coatings. Volumes are moderate, but margins are attractive due to high specification requirements.
What is Driving Strong Demand for Anti-Static PCR Resin Solutions in the United States?
The anti-static PCR resin solutions market for cleanroom manufacturing in the United States, expanding at a CAGR of 11.8% during 2026-2036, is shaped by sustainability targets in semiconductor, pharmaceutical, and advanced electronics production. Manufacturers seek PCR resins that maintain ESD control without affecting cleanliness, mechanical strength, or processability. Buyers prioritize FDA and ISO compliance, conductivity stability across humidity ranges, and compatibility with injection molding and thermoforming. Adoption is supported by strong demand from semiconductor fabs and life sciences cleanrooms upgrading material inputs. Procurement decisions emphasize supply security, technical collaboration, and scalability across multi-site operations. Qualification timelines are structured but faster than in Europe, allowing broader adoption. The US is a value-driven market where suppliers offering engineered anti-static PCR compounds, testing support, and reliable logistics gain preferred status.
How Does Japan Maintain Precision Standards for Anti-Static PCR Resins in Cleanrooms?
Sale of anti-static PCR resin solutions for cleanroom manufacturing in Japan, growing at a CAGR of 10.0%, reflects an extremely precision-oriented adoption model. Cleanroom users demand ultra-low particle generation, stable resistivity, and long-term performance consistency. PCR adoption is incremental, often starting with secondary cleanroom components such as trays, bins, and transport carriers. Buyers emphasize extensive pilot testing, aging validation, and tight process control. Procurement favors suppliers with advanced compounding expertise, strong documentation, and long-standing reliability. Volumes remain measured, but performance thresholds are among the highest globally. Japan rewards suppliers that deliver predictable anti-static behavior, minimal contamination risk, and long qualification support rather than rapid volume expansion.
Why is China Emerging as the Fastest-Growing Market for Anti-Static PCR Resin Solutions?
The anti-static PCR resin solutions market for cleanroom manufacturing in China, expanding at a CAGR of 14.6% during 2026-2036, is driven by rapid growth in semiconductor fabs, electronics assembly, and pharmaceutical cleanrooms. Manufacturers seek cost-effective PCR resins that provide basic ESD control while supporting high-volume production. Buyers prioritize scalability, affordability, and integration with domestic molding operations. Domestic compounders compete aggressively, while international suppliers target higher-end applications requiring tighter ESD and cleanliness control. Qualification cycles are shorter, enabling faster deployment. China represents a scale-led market where supplier success depends on throughput, localization, and ability to progressively improve material performance as cleanroom standards tighten.
How is South Korea Supporting Specialized Growth in Anti-Static PCR Resin Applications?
Demand for anti-static PCR resin solutions for cleanroom manufacturing in South Korea, expanding at a CAGR of 10.6% during 2026-2036, is closely linked to semiconductor, display panel, and battery manufacturing. Cleanroom users require materials that deliver reliable electrostatic discharge control while supporting automated, high-precision handling systems. Anti-static PCR resins are increasingly used in wafer carriers, component trays, reels, and protective packaging inside Class 100 to Class 1000 environments. Buyers emphasize consistency, dimensional stability, and resistance to performance drift under continuous use. Qualification processes are structured and technical, with strong emphasis on compatibility with robotic handling and conveyor systems. Procurement favors suppliers offering semiconductor-grade material control, fast iteration cycles, and close collaboration with equipment manufacturers.
How Are Companies Competing in the Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market?

Competition in the anti-static PCR resin solutions for cleanroom manufacturing market is driven by verified electrostatic discharge (ESD) performance, cleanroom compatibility, and documented compliance with industry standards that matter to OEMs, processors, and quality engineers. SABIC (Saudi Basic Industries Corporation) positions its anti-static PCR resin solutions with technical materials that highlight consistent surface resistivity control, low ionic contamination, and stability under repeated sterilization cycles. Brochures emphasize optimized formulations tailored for high PCR content while meeting ISO and ANSI/ESD-S standards relevant to semiconductor, medical, and microelectronics cleanroom assembly.
BASF SE competes by offering PCR-compatible anti-static masterbatches and engineered compounds documented for superior dispersion of ESD additives. Official product literature notes surface resistivity ranges, moisture uptake profiles, and cleanroom suitability classifications essential for precision assembly environments. Avient Corporation differentiates through customizable anti-static PCR compounds with brochures that detail controlled triboelectric charging and low particle generation, attributes critical to controlled environments.
Material science leaders extend competitive differentiation through base resin performance and additive integration. Celanese Corporation positions its PCR resin solutions around balanced mechanical strength and static control with datasheets specifying processing windows compatible with precision molding. Toray Industries, Inc. and Asahi Kasei Corporation focus on high-performance PCR polymers with documented cleanroom particle emission data and ESD stability over thermal cycles, appealing to semiconductor and medical device customers. Kingfa Sci. & Tech. Co., Ltd. competes through cost-effective anti-static PCR solutions with technical materials that outline surface resistivity control and processability across extrusion and injection applications. Across suppliers, competitive value is defined by brochure-verified ESD performance, contaminant control suitability, and cleanroom compliance rather than basic pricing alone.
Key Players in Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market
- SABIC (Saudi Basic Industries Corporation)
- BASF SE
- Avient Corporation
- Celanese Corporation
- Toray Industries, Inc.
- Asahi Kasei Corporation
- Kingfa Sci. & Tech. Co., Ltd.
Scope of Report
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Million |
| End-use | Semiconductor & Medical Cleanrooms, Pharma & Electronics Manufacturing, Precision Cleanrooms, Electronics Fabs, Display Fabs, Pharma Manufacturing |
| Resin Solution | Permanent Anti-Static Resins, ESD-Safe Compounds, Low-Outgassing Resins, Anti-Static ABS |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | SABIC (Saudi Basic Industries Corporation), BASF SE, Avient Corporation, Celanese Corporation, Toray Industries, Inc., Asahi Kasei Corporation, Kingfa Sci. & Tech. Co., Ltd. |
| Additional Attributes | Dollar sales by end-use and resin solution are evaluated across contamination-sensitive cleanroom manufacturing environments. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of ESD protection and outgassing control requirements, evaluation of PCR resin compatibility with cleanroom standards, competitive benchmarking of material suppliers, and assessment of adoption trends in semiconductor, pharmaceutical, and electronics fabrication facilities. |
Anti-Static PCR Resin Solutions for Cleanroom Manufacturing Market by Key Segments
End-use:
- Semiconductor & medical cleanrooms
- Pharma & electronics manufacturing
- Precision cleanrooms
- Electronics fabs
- Display fabs
- Pharma manufacturing
Resin Solution:
- Permanent anti-static resins
- ESD-safe compounds
- Low-outgassing resins
- Anti-static ABS
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Organization for Standardization. (2024). ISO 14644-4: Cleanrooms and associated controlled environments — Part 4: Design, construction and start-up. ISO.
- Institute of Environmental Sciences and Technology. (2025). Recommended Practice for Electrostatic Discharge (ESD) Control in Cleanrooms. IEST.
- European Committee for Standardization. (2024). EN IEC 61340-5-1: Electrostatics — Protection of electronic devices from electrostatic phenomena. CEN/IEC.
Frequently Asked Questions
How big is the anti-static PCR resin solutions for cleanroom manufacturing market in 2026?
The global anti-static PCR resin solutions for cleanroom manufacturing market is estimated to be valued at USD 840.0 million in 2026.
What will be the size of anti-static PCR resin solutions for cleanroom manufacturing market in 2036?
The market size for the anti-static PCR resin solutions for cleanroom manufacturing market is projected to reach USD 2,679.6 million by 2036.
How much will be the anti-static PCR resin solutions for cleanroom manufacturing market growth between 2026 and 2036?
The anti-static PCR resin solutions for cleanroom manufacturing market is expected to grow at a 12.3% CAGR between 2026 and 2036.
What are the key product types in the anti-static PCR resin solutions for cleanroom manufacturing market?
The key product types in anti-static PCR resin solutions for cleanroom manufacturing market are semiconductor & medical cleanrooms, pharma & electronics manufacturing, precision cleanrooms, electronics fabs, display fabs and pharma manufacturing.
Which resin solution segment to contribute significant share in the anti-static PCR resin solutions for cleanroom manufacturing market in 2026?
In terms of resin solution, permanent anti-static resins segment to command 41.0% share in the anti-static PCR resin solutions for cleanroom manufacturing market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use , 2026 to 2036
- Semiconductor & medical cleanrooms
- Pharma & electronics manufacturing
- Precision cleanrooms
- Electronics fabs
- Display fabs
- Pharma manufacturing
- Semiconductor & medical cleanrooms
- Y to o to Y Growth Trend Analysis By End-use , 2021 to 2025
- Absolute $ Opportunity Analysis By End-use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Resin Solution
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Resin Solution, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Resin Solution, 2026 to 2036
- Permanent anti-static resins
- ESD-safe compounds
- Low-outgassing resins
- Anti-static ABS
- Permanent anti-static resins
- Y to o to Y Growth Trend Analysis By Resin Solution, 2021 to 2025
- Absolute $ Opportunity Analysis By Resin Solution, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-use
- By Resin Solution
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Resin Solution
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Resin Solution
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-use
- By Resin Solution
- Competition Analysis
- Competition Deep Dive
- SABIC (Saudi Basic Industries Corporation)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BASF SE
- Avient Corporation
- Celanese Corporation
- Toray Industries, Inc.
- Asahi Kasei Corporation
- Kingfa Sci. & Tech. Co., Ltd.
- SABIC (Saudi Basic Industries Corporation)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Resin Solution, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by End-use
- Figure 6: Global Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Resin Solution
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by End-use
- Figure 23: North America Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Resin Solution
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by End-use
- Figure 30: Latin America Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Resin Solution
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by End-use
- Figure 37: Western Europe Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Resin Solution
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by End-use
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Resin Solution
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by End-use
- Figure 51: East Asia Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Resin Solution
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by End-use
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Resin Solution
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by End-use , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by End-use
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Resin Solution, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Resin Solution, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Resin Solution
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

