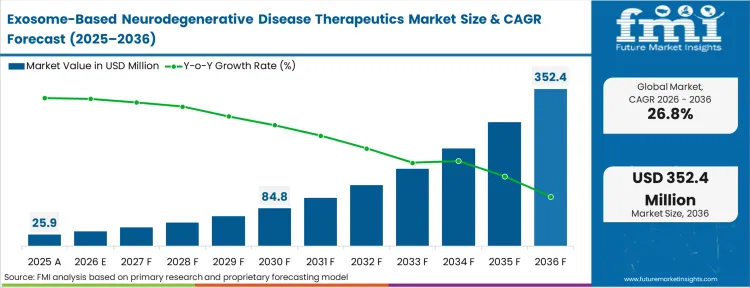
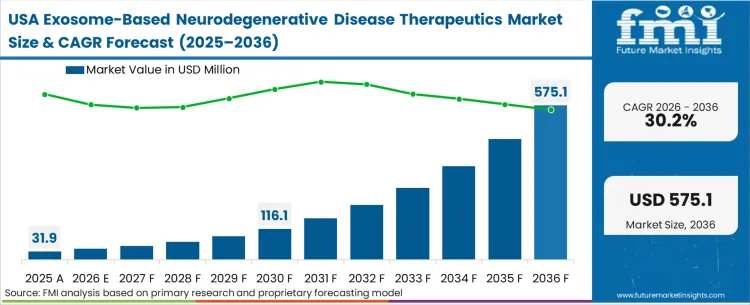
The Exosome-Based Neurodegenerative Disease Therapeutics Market was valued at USD 25.9 million in 2025. The sector is poised to reach USD 32.8 million in 2026 at a CAGR of 26.80% during the forecast period. Continued investment is driving the market’s valuation upward to USD 352.5 million through 2036 as the consistent clinical failure of traditional small molecules to cross the blood-brain barrier without systemic toxicity forces neurology pipelines toward natural lipid-bilayer delivery mechanisms.
Biopharmaceutical companies targeting the central nervous system are shifting away from proprietary synthetic lipid nanoparticles toward licensed biological extracellular platforms. This pivot acknowledges that overcoming the blood-brain barrier requires leveraging inherent cellular transport rather than brute-force synthetic chemistry, a dynamic thoroughly covered in the exosome diagnostic and therapeutics space. Companies that delay this transition risk stranding advanced nucleic acid assets in preclinical purgatory. As developers integrate these mechanisms, the landscape increasingly resembles the early days of gene therapy in cns disorder development, where holding foundational delivery IP dictated the commercial winners long before clinical efficacy was proven.
For growth to become self‑sustaining, the industry must first establish standardised isolation and characterisation metrics for clinical-grade production. Regulatory agencies and leading contract manufacturers are currently determining these baseline parameters to eliminate empirical batch-to-batch variations. Once this analytical foundation is codified, biopharma can confidently scale neurodegenerative disease pipelines, clearing the primary bottleneck of early-phase trial qualification and moving assets into late-stage evaluation.
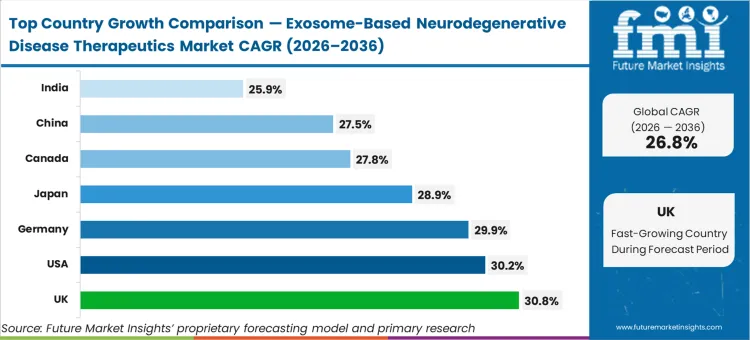
The United Kingdom leads the growth trajectory at 30.8%, followed closely by the United States at 30.2% and Germany at 29.9%. Japan tracks at 28.9%, while Canada and China are estimated to advance at 27.8% and 27.5%, respectively. India registers a 25.9% compound rate. This geographic variance reflects how quickly specific national regulators, particularly the MHRA in the UK, have established adaptive licensing pathways specifically tailored for advanced therapy medicinal products, distinct from standard biologic frameworks.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 32.8 Million |
| Industry Value (2036) | USD 352.5 Million |
| CAGR (2026–2036) | 26.80% |
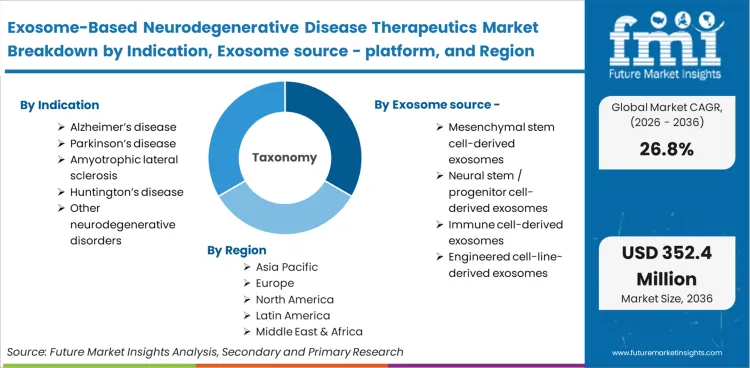
The Exosome-Based Neurodegenerative Disease Therapeutics Market encompasses the development and commercialization of cell-derived extracellular vesicles engineered or utilized to deliver therapeutic payloads to the central nervous system. This boundary explicitly focuses on biological vesicle treatments aimed at halting or reversing neuronal degradation, functionally distinct from generic drug delivery systems or purely synthetic nanoparticle platforms.
Scope includes native and engineered exosomes derived from mesenchymal, neural, or immune cell lines, loaded with nucleic acids, proteins, or small molecules. It incorporates the alzheimers therapeutics applications that utilize these specifically sourced biological vesicles for targeted central nervous system delivery and localized cellular repair.
Standard synthetic lipid nanoparticles, viral vectors, and direct stem cell transplants are explicitly excluded. These modalities, while targeting similar neurological indications, operate on fundamentally different pharmacokinetic principles and require entirely separate manufacturing workflows, purification protocols, and regulatory compliance infrastructures.
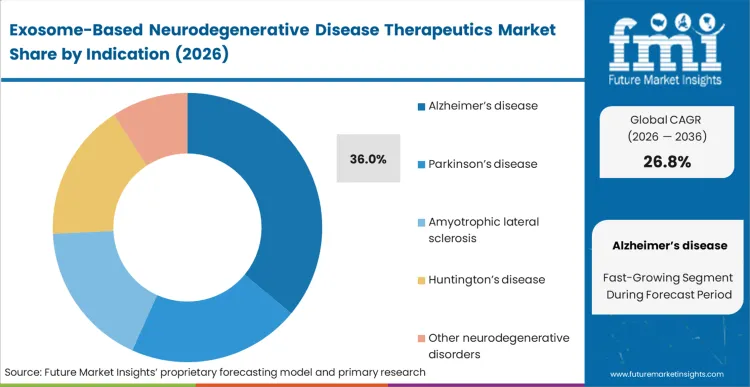
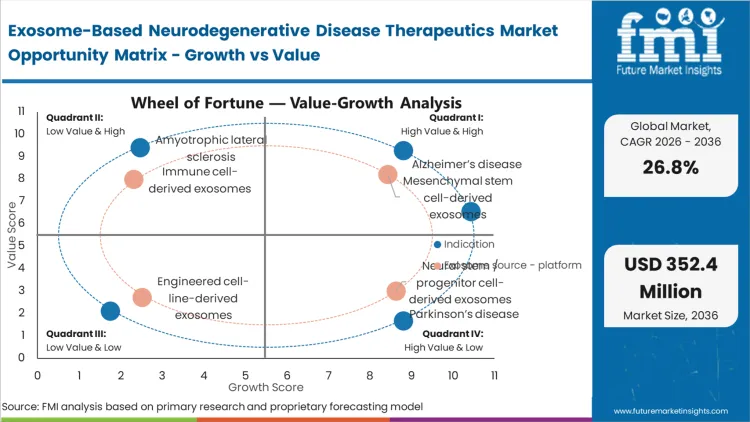
The exact decision clinical sponsors face right now is whether to deploy unproven delivery technology against established amyloid targets or risk entirely novel mechanisms. Alzheimer’s disease holds a dominant 36.0% share because the massive historical sunk costs in failed CNS trials compel sponsors to test new delivery vectors on known disease pathways first.
According to FMI's estimates, this concentration of capital creates a proving ground where exosome platforms must demonstrate safety at scale before expanding into orphan indications. Sponsors operating in the cell and gene therapy clinical trial space use Alzheimer's as the baseline for payload capacity and biodistribution metrics. Early validation here establishes the pharmacokinetics necessary to approach more aggressive pathologies. Firms that attempt to validate their platforms on obscure targets first struggle to secure the late-stage funding required for commercial scale-up.
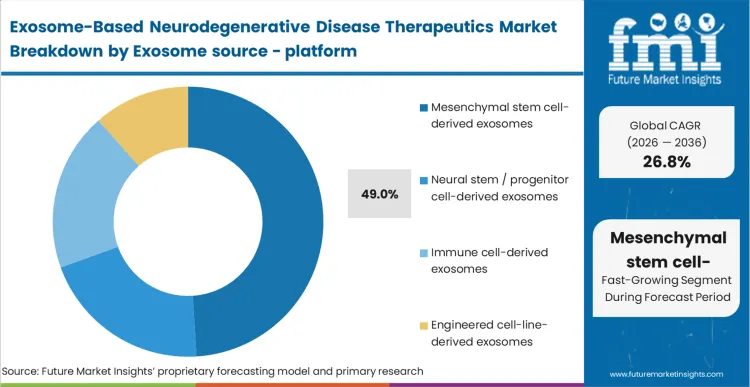
The reason mesenchymal stem cell-derived exosomes hold 49.0% of this market comes down to a single reality: isolation and culturing protocols for these specific cells are already regulatory-approved and globally scaled.
In FMI's view, manufacturing directors are not choosing this source for superior theoretical payload capacity; they choose it because the chemistry, manufacturing, and controls (CMC) documentation can be adapted from legacy stem cell therapies. The infrastructure to grow these cells in 3D bioreactors exists today, mitigating the massive capital expenditure required to establish new cell lines. Sourcing from non-standardized platforms forces developers to build dedicated purification cascades that often yield inconsistent vesicle counts. Attempting to navigate Phase II trials without a secure, scalable mesenchymal supply chain guarantees clinical delays.
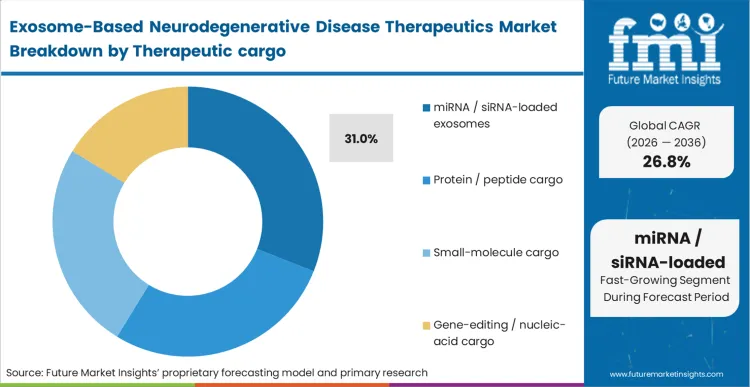
What legacy synthetic nanoparticles failed to deliver, stable transport of fragile genetic material, dictates the current investment landscape. The miRNA / siRNA-loaded exosomes sub-segment captures 31.0% share because these specific payloads require the lipid bilayer protection that exosomes naturally provide to survive systemic circulation.
As per FMI's projection, pharmacology teams evaluating platforms for parkinsons disease therapies prioritize vesicle systems that demonstrate high loading efficiency without disrupting the integrity of the genetic payload. The operational change involves moving from empirical co-incubation loading methods to active electroporation techniques that guarantee uniform therapeutic density. A miscalculation in loading efficiency results in sub-therapeutic dosing at the target tissue, rendering the entire clinical protocol invalid.
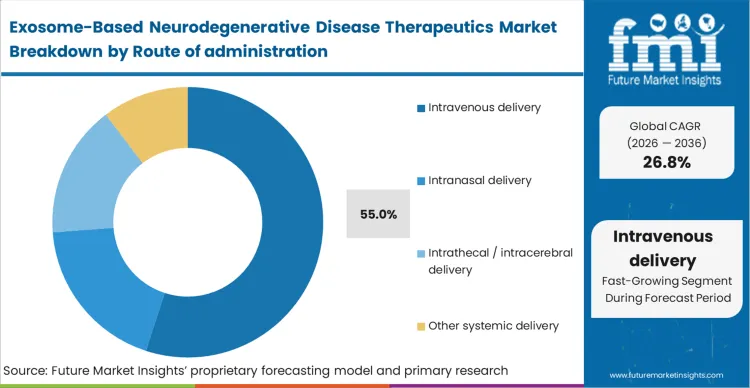
The commercial consequence of requiring specialised neurosurgical intervention severely limits patient addressability, compelling the need for systemic approaches. Intravenous delivery leads with 55.0% share because it leverages the natural homing capabilities of exosomes to cross the blood-brain barrier without the clinical friction of intrathecal ports.
Based on FMI's assessment, outpatient infusion centers can administer these therapies using standard infrastructure, drastically reducing the total cost of care. The pharmacokinetic advantage relies entirely on surface engineering that directs the intravenously administered vesicles specifically to neurological targets, bypassing peripheral tissue sinks. Failing to achieve high targeting efficiency via this route forces developers back to invasive administration methods, severely compressing the peak revenue potential of the asset.
The high incidence of adverse immune responses to synthetic lipid nanoparticles compels translational medicine directors to qualify natural biological vectors for chronic central nervous system indications. In neurodegenerative diseases, the pressure is especially intense because patients often need ongoing treatments for many years, not just a one‑time cure. Directors cannot advance legacy cell therapy manufacturing frameworks that trigger neutralizing antibodies upon secondary administration. The commercial stakes are absolute: assets that provoke an immune response during Phase II dosing escalation are systematically abandoned, forcing companies to secure exosome-based licensing agreements to salvage their proprietary payloads.
The primary operational friction slowing immediate commercialization is the severe lack of standardized, scalable purification technologies capable of separating therapeutic exosomes from host-cell proteins and non-therapeutic vesicles. This is a fundamental downstream processing bottleneck, not a theoretical biological limitation. Current ultracentrifugation and size-exclusion chromatography methods fail to yield the purity and volume required for late-stage multi-center trials. While tangential flow filtration offers a partial scaling solution, the lack of universally accepted characterization assays means sponsors spend excessive time validating their batches to regulators rather than advancing their clinical protocols.
Based on the regional analysis, the Exosome-Based Neurodegenerative Disease Therapeutics Market is segmented into North America, Europe, Asia Pacific, and other regions across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United Kingdom | 30.8% |
| United States | 30.2% |
| Germany | 29.9% |
| Japan | 28.9% |
| Canada | 27.8% |
| China | 27.5% |
| India | 25.9% |
The regulatory environment in Europe specifically shapes adoption through targeted pathways designed for advanced therapy medicinal products that do not fit traditional biologic frameworks. Rather than forcing exosome platforms through standard pharmacokinetic evaluations designed for small molecules, regulators like the MHRA and EMA are establishing bespoke analytical requirements. This clarity allows translational teams to design their preclinical programs with precise knowledge of what downstream processing data will be demanded. FMI analysts opine that this alignment accelerates the transition from academic spinouts into commercially viable clinical sponsors. It establishes a distinct operational rhythm where quality-by-design principles are embedded into the purification process from day one, rather than retrofitted ahead of Phase II.
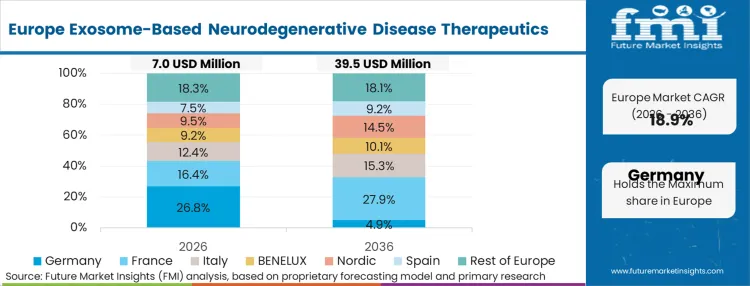
FMI's report includes detailed analysis of France, Italy, and the Nordics. These adjacent markets are aggressively harmonizing their clinical trial requirements to ensure they can participate in the multi-center efficacy trials that will originate from the UK and Germany.
The physical and digital infrastructure condition in North America heavily dictates the speed of commercialization, specifically regarding specialized cold-chain logistics and highly centralized contract development and manufacturing organizations (CDMOs). Sponsors in this region are not building proprietary manufacturing suites; they are relying entirely on a concentrated network of specialized facilities capable of executing complex ultracentrifugation and tangential flow filtration at clinical scale. As per FMI's projection, this infrastructure concentration forces developers to lock in manufacturing slots years in advance. The operational reality is that access to dedicated bioprocessing capacity, rather than access to capital, serves as the ultimate governor on pipeline progression across the continent.
FMI's report includes broader North American partnership metrics and cross-border licensing data. The pattern reveals a total reliance on US-based analytical testing firms to validate Canadian-originating vesicle platforms.
Cost structures and aggressive government funding initiatives define the adoption pattern across the Asia Pacific region, separating it from Western regulatory-led dynamics. State-sponsored investments in regenerative medicine infrastructure allow regional biotechs to subsidize the exorbitant costs of downstream processing and donor cell culturing. In FMI's view, this economics-led environment enables companies to run massive parallel preclinical programs testing various cellular sources, a capital-intensive strategy that Western venture models rarely support. The focus here is on achieving rapid domestic approvals through accelerated pathways, leveraging localized clinical data to establish market dominance before Western platforms can navigate the region's diverse import regulations.
FMI's report includes evaluation of South Korea and Australia. These nations are acting as critical clinical trial bridges, offering Western-compliant data integrity while operating within the broader Asian economic cost.
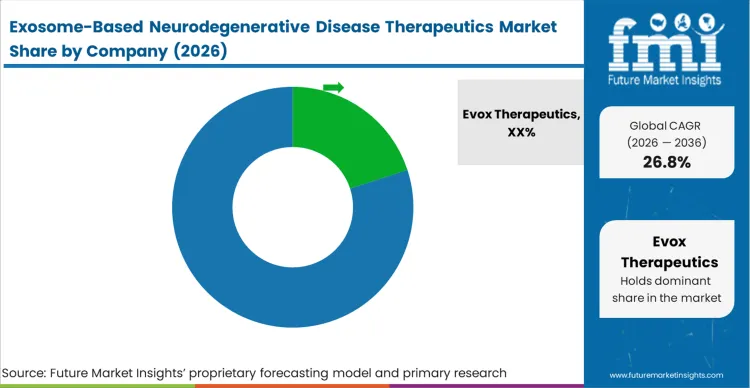
The competitive situation of this market is exceptionally concentrated because the barrier to entry is not identifying a therapeutic payload, but possessing the proprietary intellectual property required to load it into a vesicle and scale the purification. Buyers in this context, large pharmaceutical sponsors distinguish qualified vendors based entirely on their demonstrated chemistry, manufacturing, and controls (CMC) documentation. Companies like Evox Therapeutics and Coya Therapeutics do not compete on theoretical biology; they compete on their ability to prove that batch 100 will perform identically to batch 1. A firm with a mediocre targeting peptide but a flawless, scalable manufacturing process will consistently beat a firm with superior biology but empirical, unscalable isolation methods.
Incumbents holding foundational patents on specific electroporation loading techniques or surface-display engineering, such as ArunA Bio and ILIAS Biologics, maintain a distinct advantage. Their advantage persists because regulatory bodies treat the exosome vector and its manufacturing process as intrinsically linked; changing the loading method requires restarting the safety evaluation. To replicate this position, a challenger must build a fundamentally novel mechanism for embedding genetic material into the lipid bilayer without triggering intellectual property tripwires or degrading the vesicle. They must engineer a completely orthogonal engineered cell therapy platform that achieves high-density loading at commercial scale.
Large biopharma buyers inherently resist this vendor lock-in by aggressively pursuing internal development of synthetic alternatives while simultaneously licensing biological platforms as a hedge. The tension lies between pharmaceutical companies demanding complete technology transfer and biotechnology firms guarding their proprietary manufacturing cell lines as their sole source of enterprise value. As the decade progresses toward 2036, the market will naturally stratify: a few dominant players will provide commoditized, empty vesicle chassis for broad delivery, while specialized firms will maintain high-margin monopolies over highly engineered vectors designed exclusively for complex brain tumor treatment and targeted neuronal repair.
| Metric | Value |
|---|---|
| Quantitative Units | USD 32.8 Million to USD 352.5 Million, at a CAGR of 26.80% |
| Market Definition | This market tracks the commercial progression of cell-derived lipid vesicles deployed specifically to transport therapeutic agents across the blood-brain barrier. It focuses entirely on biological cellular secretion systems rather than synthetic chemistry to achieve targeted neurological intervention. |
| Indication Segmentation | Alzheimer’s disease, Parkinson’s disease, Amyotrophic lateral sclerosis, Huntington’s disease, Other neurodegenerative disorders |
| Exosome source / platform Segmentation | Mesenchymal stem cell-derived exosomes, Neural stem / progenitor cell-derived exosomes, Immune cell-derived exosomes, Engineered cell-line-derived exosomes |
| Therapeutic cargo Segmentation | miRNA / siRNA-loaded exosomes, Protein / peptide cargo, Small-molecule cargo, Gene-editing / nucleic-acid cargo |
| Route of administration Segmentation | Intravenous delivery, Intranasal delivery, Intrathecal / intracerebral delivery, Other systemic delivery |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East, Africa, Oceania |
| Countries Covered | United Kingdom, United States, Germany, Japan, Canada, China, India, and 40 plus countries |
| Key Companies Profiled | Evox Therapeutics, ArunA Bio, Coya Therapeutics, ILIAS Biologics, NurExone Biologic, ReNeuron Group, Vesalic Limited |
| Forecast Period | 2026 to 2036 |
| Approach | FMI interviewed translational medicine directors and chief scientific officers at neuro-focused biotechs. The baseline anchors to current R&D expenditure on active exosome CNS pipelines and projected milestone licensing payments. Forecasts were cross-validated against historical adoption curves of early CNS monoclonal antibodies. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Exosome-Based Neurodegenerative Disease Therapeutics Market in 2026?
The industry is expected to reach USD 32.8 million in 2026. This initial valuation reflects early-stage licensing milestones and contract manufacturing expenditures rather than mass commercial product sales, signaling a market entirely in its clinical development phase.
What will it be valued at by 2036?
The valuation advances to USD 352.5 million through 2036. This cumulative buildup indicates the expected timeline for current Phase I/II neurological assets to secure final regulatory clearance and initiate broad commercial dosing.
What CAGR is projected?
The sector advances at a 26.80% compound annual growth rate. This rate is strictly constrained by the time required to build specialized bioprocessing infrastructure and validate clinical-grade purification cascades, rather than theoretical biological demand.
Which Indication segment leads?
Alzheimer’s disease captures a dominant 36.0% share. Biopharma sponsors are unwilling to risk unproven delivery vectors on obscure orphan targets, forcing them to validate exosome safety profiles on established amyloid and tau pathways where historical clinical endpoints are well understood.
Which Exosome source / platform segment leads?
Mesenchymal stem cell-derived exosomes hold 49.0% of the platform market. Manufacturing directors choose this source because the culturing protocols and master cell bank regulatory frameworks are already fully established from legacy regenerative medicine pipelines.
Which Therapeutic cargo segment leads?
miRNA / siRNA-loaded exosomes represent 31.0% of active payloads. These specific genetic materials are acutely vulnerable to enzymatic degradation in human serum, absolutely requiring the protective lipid bilayer of an exosome to survive transit to the brain.
What drives rapid growth?
Translational medicine teams are compelled to abandon synthetic lipid nanoparticles due to their adverse immune profiles during chronic administration. The operational requirement for a biologically tolerated, repeated-dosing vector forces capital directly into natural extracellular platforms.
What is the primary restraint?
The fundamental downstream processing bottleneck of scalable purification limits commercialization. Current ultracentrifugation techniques cannot yield the volume or purity required for multi-center Phase III trials without exorbitant batch validation costs.
Which country grows fastest?
The United Kingdom outpaces the United States, 30.8% to 30.2%. The difference lies in the MHRA's highly adaptive Innovative Licensing and Access Pathway, which allows UK-based sponsors to transition from preclinical models to human trials significantly faster than the rigid sequential processes mandated elsewhere.
Why do biopharma companies outsource exosome production instead of building internal capacity?
The capital expenditure required to establish continuous tangential flow filtration suites specifically for exosomes is difficult to justify for a single pipeline asset. Sponsors rely on centralized contract manufacturers to absorb this infrastructure risk until their asset clears Phase II efficacy endpoints.
What makes intravenous delivery the preferred route of administration?
It bypasses the requirement for specialized neurosurgical intervention, drastically lowering the clinical friction of patient enrollment. Developers engineer specific surface tropisms to ensure these intravenously administered vesicles cross the blood-brain barrier rather than accumulating in the liver.
How does the Japanese regulatory framework alter the commercial timeline?
Japan's PMDA permits conditional approval for regenerative therapies based entirely on early-phase safety data. This allows biotechs to generate domestic revenue while conducting pivotal efficacy trials, completely removing the late-stage funding bottleneck that strangles Western developers.
What operational metric determines if a sponsor renews a platform licensing agreement?
Sponsors measure the loading efficiency consistency across multiple commercial-scale batches. If the platform requires constant empirical adjustments to achieve therapeutic payload density, the sponsor will abandon the partnership before entering Phase III.
Why are engineered cell-line exosomes gaining traction against primary donor sources?
Relying on primary mesenchymal donors introduces unavoidable biological variability into the manufacturing process. Immortalized cell lines offer a highly standardized, regulatory-compliant master bank that drastically reduces the hidden costs of ongoing donor qualification.
What role does analytical assay development play in this market?
Regulators demand rigorous proof that the therapeutic exosome is separate from host-cell debris. Vendors who develop the industry-standard assays for exact vesicle quantification become indispensable, as their tools are embedded directly into the sponsor's FDA submissions.
Why do synthetic lipid nanoparticles fail in chronic neurological applications?
Synthetic vectors often trigger neutralizing antibodies upon secondary or tertiary administration. Because neurodegenerative diseases require lifelong therapeutic intervention, this immune response renders the synthetic delivery vehicle useless after the initial dose.
How do German cell procurement guidelines impact early-phase developers?
Germany demands highly transparent, localized donor networks for any biological material used in clinical applications. While this increases upfront procurement costs, it yields pristine manufacturing documentation that effortlessly clears subsequent European clinical audits.
What intellectual property creates the strongest competitive moat?
Patents covering active electroporation loading techniques provide an absolute advantage. Companies holding these patents control the fundamental method of getting the therapeutic payload into the vesicle without destroying its integrity.
How does Canada's market dynamic differ from the United States?
Canada functions as a high-value intellectual property incubator rather than an end-to-end commercial hub. Academic clusters in Toronto generate foundational engineering patents, but the lack of domestic late-stage bioprocessing capacity forces clinical directors to out-license manufacturing to US operators.
What is the commercial consequence of failing to achieve high brain-targeting efficiency?
If the engineered surface peptides fail to direct the exosome specifically to the central nervous system, a massive percentage of the dose is lost to peripheral organs. This forces developers to exponentially increase the administered dose, ruining the cost-of-goods economics of the therapy.
How are large pharmaceutical companies hedging their delivery vector bets?
They maintain internal R&D programs focused on next-generation synthetic nanoparticles while simultaneously signing exclusive licensing deals with exosome biotechs. This ensures they have a biological fallback option if their synthetic CNS pipelines hit unforeseen toxicity walls in Phase I.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.