Disease-Modifying MS Therapies Market
Disease-Modifying MS Therapies Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Disease-Modifying MS Therapies Market Forecast and Outlook 2026 to 2036
The disease-modifying MS therapies market is likely to be valued at USD 31,915.8 million in 2026 and reach USD 63,372.5 million by 2036, at a CAGR of 7.1%. Value expansion reflects the structural shift in multiple sclerosis management toward sustained immune modulation over long treatment horizons. Disease-modifying therapies are prescribed for continuous use spanning decades, which concentrates spending in chronic care rather than episodic intervention. Treatment initiation occurs earlier in the disease course, extending cumulative therapy duration per patient. Oral agents, injectables, and monoclonal antibodies coexist within treatment algorithms, with selection guided by disease activity, safety tolerance, and monitoring capacity rather than substitution dynamics. Pricing and utilization remain shaped by long-term outcome evidence, relapse suppression consistency, and the operational ability of healthcare systems to support ongoing surveillance. Hospital and specialty neurology settings capture a large share of value due to therapy initiation, escalation decisions, and safety oversight requirements.
Quick Stats for Disease-Modifying MS Therapies Market
- Disease-Modifying MS Therapies Market Value (2026): USD 31,915.8 million
- Disease-Modifying MS Therapies Market Forecast Value (2036): USD 63,372.5 million
- Disease-Modifying MS Therapies Market Forecast CAGR 2026 to 2036: 7.1%
- Leading Product Category by Demand Share: Oral Therapies
- Fastest-Growing Countries: India, China, Brazil, USA, UK
- Top Players in Global Demand: Biogen, Roche, Novartis, Sanofi, Merck KGaA
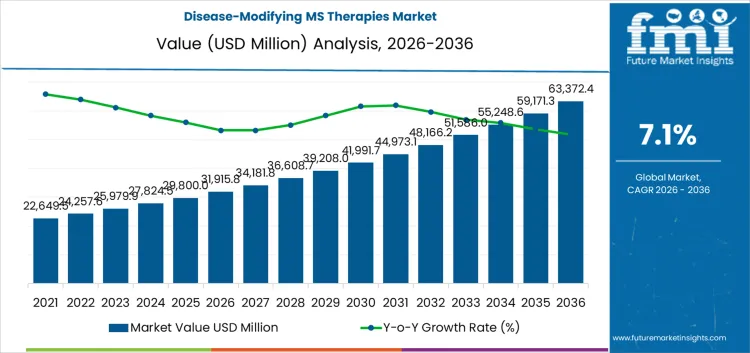
Growth is anchored in patient identification and persistence rather than rapid shifts in therapeutic classes. Improved diagnostic accuracy and broader neurologist access expand the treated population, while therapy switching within the same patient lifecycle sustains volume. Constraints remain tied to safety monitoring intensity, reimbursement thresholds, and biologic manufacturing complexity, which limit uncontrolled uptake of high-efficacy agents. Real-world evidence increasingly influences formulary positioning as payers assess disability progression outcomes over extended timelines. Access varies by healthcare infrastructure maturity, with systems capable of long-term follow-up absorbing higher therapy complexity. Expansion reflects continuity of care, earlier intervention, and prolonged treatment exposure rather than short-term prescribing surges or innovation-driven displacement of established disease-modifying regimens.
Disease-Modifying MS Therapies Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 31,915.8 million |
| Market Forecast Value (2036) | USD 63,372.5 million |
| Forecast CAGR 2026 to 2036 | 7.1% |
Why is the Demand for Disease-Modifying MS Therapies Growing?
Demand for disease-modifying multiple sclerosis (MS) therapies is increasing as neurologists and healthcare systems prioritize early intervention strategies that slow disease progression and reduce relapse frequency in people living with MS. These therapies target immune system pathways to modify the underlying disease course rather than solely addressing symptoms, and clinical guidelines recommend initiation soon after diagnosis to preserve neurological function and delay disability accumulation. Advances in understanding MS pathophysiology have expanded available mechanism-based treatment options, which supports more tailored therapy selection by clinicians based on disease subtype, activity level, and patient tolerance. Procurement teams in hospitals and specialty clinics assess comparative efficacy, safety profiles, and long-term outcome data when adding disease-modifying therapies to formularies, with focus on consistent supply and treatment continuity across outpatient and infusion settings.
Growth in MS prevalence, driven by improved diagnostic capabilities and broader screening in neurology practices, reinforces uptake of disease-modifying agents that align with care standards emphasizing sustained relapse reduction and brain lesion suppression. Payers and health systems increasingly incorporate real-world evidence and long-term disability outcomes into reimbursement decisions, which supports access to therapies demonstrating sustained benefit in diverse patient populations. Neurologists also integrate magnetic resonance imaging results and biomarker data into treatment planning, encouraging use of disease-modifying therapies that align with monitored disease activity. These clinical and operational imperatives are contributing to sustained growth in the disease-modifying MS therapies market.
How Are the Segments Classified in the Disease-Modifying MS Therapies Market?
Demand for disease-modifying multiple sclerosis therapies is shaped by relapse control objectives, disability progression management, and long-term treatment adherence requirements. Clinical practice emphasizes early intervention to reduce inflammatory activity and preserve neurological function. Adoption aligns with evolving treatment algorithms, improved diagnostic rates, and broader access to specialty care. Therapy selection prioritizes efficacy, safety monitoring needs, and administration convenience. Segment classification reflects differentiation by product modality, disease phenotype application, and end-user dispensing setting influencing prescribing behavior, monitoring intensity, and continuity of care.
Which Product Category Holds the Largest Share in Disease-Modifying MS Therapies?
Oral therapies account for 38.0%, representing the largest share among product categories due to administration convenience. Oral dosing reduces injection burden and supports long-term adherence. Simplified administration aligns with outpatient management models and patient preference. Monitoring requirements remain manageable within routine neurology follow-up. Injectables maintain relevance through established safety profiles and long-term data. Monoclonal antibodies provide high efficacy with infusion-based administration and intensive monitoring. Product segmentation reflects preference for therapies balancing efficacy with ease of use and sustained patient compliance.
Key Points
- Oral therapies improve adherence through convenient administration.
- Outpatient suitability supports long-term treatment continuity.
- Other modalities address efficacy or long-established use patterns.
How Does Application Segmentation Influence Therapy Utilization?
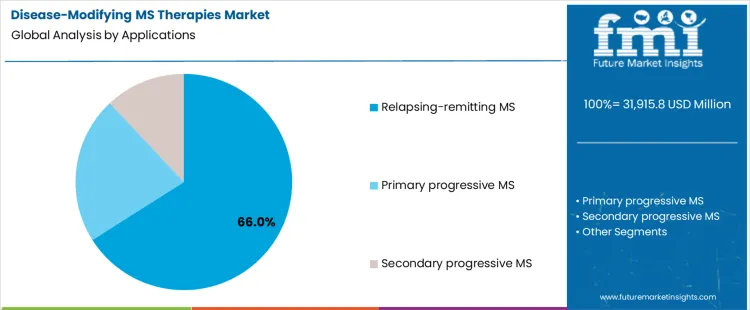
Relapsing-remitting multiple sclerosis accounts for 66.0%, representing the largest share among disease applications. High diagnosis rates and defined relapse patterns drive active disease-modifying treatment. Therapeutic intervention focuses on reducing relapse frequency and lesion accumulation. Clinical guidelines strongly support early and sustained treatment in this population. Primary progressive disease presents limited therapeutic options with slower uptake. Secondary progressive disease requires tailored escalation strategies. Application segmentation reflects concentration where disease prevalence, guideline support, and measurable treatment response sustain the highest therapy utilization.
Key Points
- Relapsing-remitting disease drives primary therapy use.
- Early treatment targets relapse reduction and lesion control.
- Progressive forms involve more selective treatment approaches.
What Role Does End User Distribution Play in Therapy Delivery?
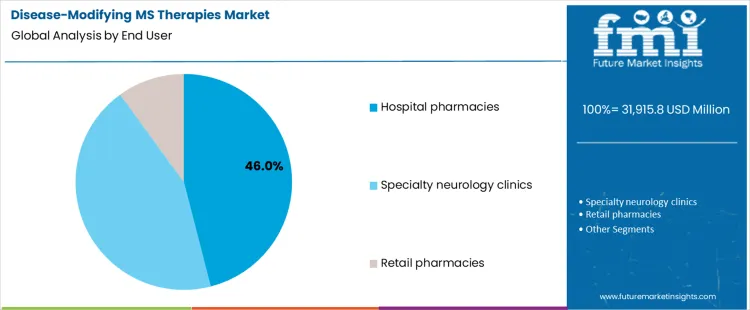
Hospital pharmacies account for 46.0%, representing the largest share among end-user settings. Hospitals manage initiation of advanced therapies and coordinate safety monitoring. Infusion services and multidisciplinary oversight support complex treatment protocols. Specialty neurology clinics follow closely with outpatient-focused care and ongoing management. Retail pharmacies dispense select oral therapies with limited monitoring scope. End-user segmentation reflects concentration where infrastructure, specialist involvement, and monitoring capabilities support safe and effective long-term management of multiple sclerosis.
Key Points
- Hospitals support initiation and monitoring of advanced therapies.
- Specialty clinics manage ongoing outpatient care.
- Retail channels serve limited dispensing roles.
What are the Key Dynamics in the Disease-Modifying MS Therapies Market?
Demand for disease-modifying multiple sclerosis therapies reflects need to reduce relapse frequency, delay disability progression, and control inflammatory activity in relapsing and progressive MS. Adoption concentrates in neurology clinics, specialty hospitals, and infusion centers. Global scope aligns with guideline-driven long-term disease management. Usage centers on injectable, oral, and infused agents targeting immune modulation, lymphocyte trafficking, or cell depletion across defined MS phenotypes.
How do disease heterogeneity and long-term treatment objectives shape adoption?
Multiple sclerosis presents with variable relapse patterns, progression rates, and treatment response. Demand increases where early intervention aims to suppress inflammatory activity before irreversible neurological damage occurs. Therapy selection depends on disease activity, imaging findings, and patient risk tolerance. High-efficacy agents gain use in patients with aggressive disease to limit lesion accumulation. Oral therapies support adherence in patients seeking reduced treatment burden. Infused monoclonal antibodies address high disease activity through sustained immune suppression. Treatment sequencing reflects balance between efficacy and safety over multi-year horizons. Regular monitoring requirements influence therapy choice in routine practice. Adoption reflects clinical focus on individualized, long-duration disease control rather than short-term symptom management.
How do safety monitoring burden and access constraints influence market scalability?
Disease-modifying therapies carry risks including infection, malignancy, and immune reconstitution complications. Demand sensitivity rises where monitoring infrastructure limits safe administration. Laboratory surveillance and imaging increase care complexity and cost. Risk mitigation programs restrict prescribing to trained centers for certain agents. Treatment discontinuation due to adverse events affects long-term utilization. High therapy cost influences reimbursement decisions and patient access across regions. Switching constraints emerge due to washout requirements and rebound risk. Manufacturing complexity for biologics affects supply stability. Scalability remains constrained by safety oversight demands, payer controls, and need for sustained specialist follow-up.
How Is Demand for Disease-Modifying MS Therapies Evolving Globally?
Demand for disease-modifying MS therapies is expanding globally due to improved diagnosis, longer treatment duration, and broader access to neurologic care. Earlier initiation and sustained use across relapsing forms of multiple sclerosis support prescription volumes. Public reimbursement frameworks and inclusion of oral and injectable options influence uptake. Treatment switching within therapy classes sustains baseline demand. Growth rates in India at 8.8%, China at 8.6%, Brazil at 8.3%, USA at 7.7%, and the UK at 6.6% indicate sustained expansion driven by patient identification, therapy continuity, and healthcare access scaling rather than breakthrough therapy substitution.
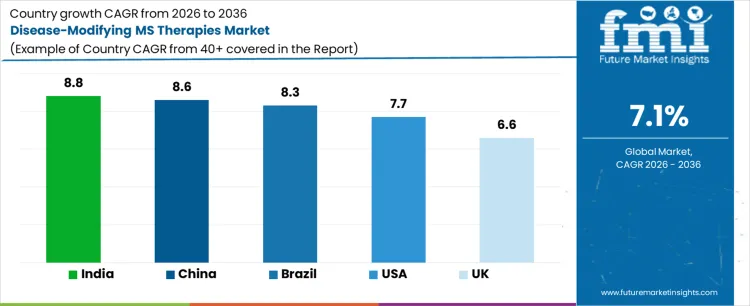
| Country | CAGR (%) |
|---|---|
| India | 8.8% |
| China | 8.6% |
| Brazil | 8.3% |
| USA | 7.7% |
| UK | 6.6% |
What Is Driving Strong Growth of MS Disease-Modifying Therapy Demand in India?
Demand for disease-modifying MS therapies in India is growing at a CAGR of 8.8%, supported by improved neurologist access and rising diagnostic awareness. Expansion of tertiary hospitals and neurology clinics increases treatment initiation. Public and private insurance coverage for essential MS therapies improves affordability. Younger patient demographics sustain longer treatment durations. Increased availability of oral formulations supports adherence. Growth reflects access expansion and continuity of care rather than rapid shifts toward high-cost biologic intensification.
- Improved MS diagnosis and neurologist availability
- Insurance coverage for essential MS therapies
- Younger treated population extending therapy duration
- Growing adoption of oral treatment options
Why Is China Experiencing Rapid Uptake of Disease-Modifying MS Therapies?
Disease-modifying MS therapy demand in China is expanding at a CAGR of 8.6%, driven by improved neurological care infrastructure and diagnostic capacity. Large hospital networks expand access to standardized MS treatment protocols. Reimbursement inclusion of selected therapies supports sustained utilization. Growing awareness among clinicians increases early treatment initiation. Domestic manufacturing and licensing arrangements improve supply consistency. Growth reflects system-level access improvements and protocol adherence rather than aggressive uptake of premium therapies.
- Expansion of neurology services in hospital networks
- Reimbursement inclusion of selected MS therapies
- Increased clinician awareness and early diagnosis
- Improved domestic supply availability
How Is Brazil Supporting Growth in MS Disease-Modifying Therapy Usage?
Demand for disease-modifying MS therapies in Brazil is growing at a CAGR of 8.3%, supported by public healthcare delivery and rising MS recognition. National treatment guidelines prioritize long-term disease modification. Public hospitals rely on established injectable and oral therapies for cost control. Expansion of specialized neurology centers improves patient reach. Treatment adherence programs support continuity. Growth reflects public system scale and guideline-driven care rather than rapid transition toward high-cost infusion therapies.
- National guideline support for long-term MS treatment
- Public hospital reliance on established therapies
- Expansion of specialized neurology centers
- Adherence programs sustaining therapy continuity
What Factors Are Driving MS Therapy Demand Growth in United States?
Disease-modifying MS therapy demand in the United States is expanding at a CAGR of 7.7%, supported by early diagnosis and long treatment persistence. Broad insurance coverage enables access across therapy classes. High neurologist density supports individualized regimen selection and switching. Oral and injectable therapies maintain large treated populations. Aging MS cohorts extend duration of care. Growth reflects therapy continuity and patient volume stability rather than increased reliance on newly launched agents.
- Early diagnosis and long-term treatment persistence
- Broad insurance coverage across therapy classes
- High neurologist access enabling regimen optimization
- Aging MS population extending care duration
Why Is the UK Showing Moderate Growth in Disease-Modifying MS Therapies?
Demand for disease-modifying MS therapies in United Kingdom is growing at a CAGR of 6.6%, driven by NHS treatment pathways and rising prevalence recognition. National guidelines emphasize early initiation and sustained therapy. Health technology assessments guide therapy selection within cost-effectiveness thresholds. Access to higher-cost options remains structured. Stable neurology service capacity supports ongoing treatment. Growth remains moderate, reflecting controlled adoption and budget-managed expansion rather than rapid escalation in therapy intensity.
- NHS guideline emphasis on early sustained treatment
- Cost-effectiveness-driven therapy selection
- Structured access to higher-cost options
- Stable neurology service capacity
What is the competitive landscape of demand for the disease-modifying MS therapies market globally?
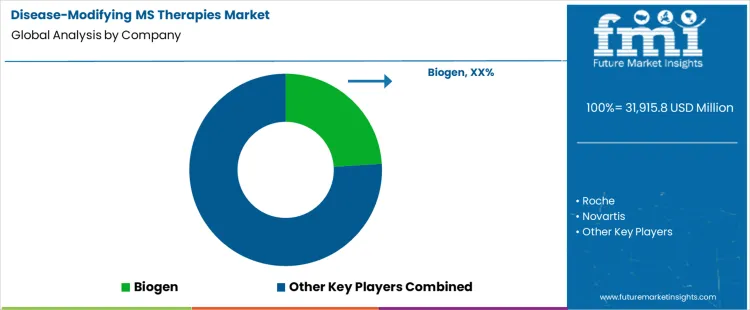
Demand for disease-modifying multiple sclerosis therapies is driven by early diagnosis rates, relapse prevention goals, and long-term disability management needs. Neurologists and payers evaluate relapse reduction efficacy, progression delay evidence, safety monitoring burden, administration route, and dosing frequency. Procurement behavior centers on formulary positioning, reimbursement eligibility, real-world effectiveness data, and long-term safety confidence. Purchasing decisions emphasize treatment adherence potential, patient stratification suitability, and budget impact management across chronic therapy lifecycles. Trend in the disease-modifying MS therapies market reflects continued shift toward high-efficacy agents, expanded use of monoclonal antibodies, and preference for therapies demonstrating durable disease control.
Biogen leads competitive positioning through a long-established MS portfolio spanning injectables, oral therapies, and infusion-based treatments. Roche competes through high-efficacy monoclonal antibody therapies with strong adoption in relapsing and progressive MS segments. Novartis supports demand with oral and infusion therapies emphasizing convenience and sustained disease control outcomes. Sanofi maintains relevance via immunomodulatory treatments positioned for specific patient subgroups and long-term management strategies. Merck KGaA participates with oral disease-modifying therapies supported by established clinical use and global market access. Competitive differentiation centers on efficacy durability, safety management requirements, access negotiations, and alignment with evolving MS treatment algorithms.
Key Players in the Disease-Modifying MS Therapies Market
- Biogen
- Roche
- Novartis
- Sanofi
- Merck KGaA
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Products | Injectables; Oral therapies; Monoclonal antibodies |
| Applications | Relapsing-remitting MS; Primary progressive MS; Secondary progressive MS |
| End User | Hospital pharmacies; Specialty neurology clinics; Retail pharmacies |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, UK, and 40+ countries |
| Key Companies Profiled | Biogen; Roche; Novartis; Sanofi; Merck KGaA; Others |
| Additional Attributes | Dollar sales by product type, application, end user, and sales channel; treatment efficacy and relapse reduction profiles across injectables, oral therapies, and monoclonal antibodies; safety monitoring requirements and administration frequency influencing therapy choice; pricing and reimbursement dynamics across public and private healthcare systems; prescribing patterns shaped by disease progression stage and long-term patient adherence considerations. |
Disease-Modifying MS Therapies Market by Segment
Products:
- Injectables
- Oral therapies
- Monoclonal antibodies
Applications
- Relapsing-remitting MS
- Primary progressive MS
- Secondary progressive MS
End User:
- Hospital pharmacies
- Specialty neurology clinics
- Retail pharmacies
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2023). WHO adds cladribine, glatiramer acetate, and rituximab to Essential Medicines List for multiple sclerosis treatment (2023 update). World Health Organization.
- Multiple Sclerosis International Federation. (2023). Atlas of MS 2023: Mapping multiple sclerosis around the world. MSIF.
- National Institute for Health and Care Excellence. (2023). Multiple sclerosis in adults: Management (NG220, updated guidance). NICE.
- European Medicines Agency. (2023). Guideline on clinical investigation of medicinal products for the treatment of multiple sclerosis (revised). European Medicines Agency.
- USA Food and Drug Administration. (2024). Multiple Endpoints in Clinical Trials – Guidance for Industry (Revised edition). FDA Center for Drug Evaluation and Research.
Frequently Asked Questions
How big is the disease-modifying ms therapies market in 2026?
The global disease-modifying ms therapies market is estimated to be valued at USD 31,915.8 million in 2026.
What will be the size of disease-modifying ms therapies market in 2036?
The market size for the disease-modifying ms therapies market is projected to reach USD 63,372.4 million by 2036.
How much will be the disease-modifying ms therapies market growth between 2026 and 2036?
The disease-modifying ms therapies market is expected to grow at a 7.1% CAGR between 2026 and 2036.
What are the key product types in the disease-modifying ms therapies market?
The key product types in disease-modifying ms therapies market are injectables, oral therapies and monoclonal antibodies.
Which applications segment to contribute significant share in the disease-modifying ms therapies market in 2026?
In terms of applications, relapsing-remitting ms segment to command 66.0% share in the disease-modifying ms therapies market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Injectables
- Oral therapies
- Monoclonal antibodies
- Injectables
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Relapsing-remitting MS
- Primary progressive MS
- Secondary progressive MS
- Relapsing-remitting MS
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital pharmacies
- Specialty neurology clinics
- Retail pharmacies
- Hospital pharmacies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Biogen
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche
- Novartis
- Sanofi
- Merck KGaA
- Others
- Biogen
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Products
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Products
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Products
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Products
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Products
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

