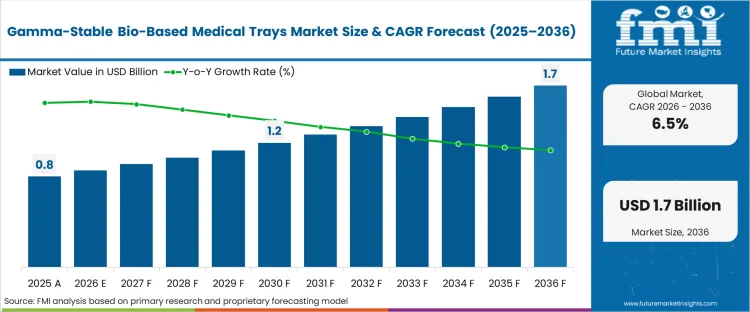
The gamma-stable bio-based medical trays market was valued at USD 0.8 billion in 2025. The sector is set to reach USD 0.9 billion in 2026 at a CAGR of 6.57% during the forecast period. Revenue expansion lifts the total opportunity to USD 1.7 billion through 2036 as medical device manufacturers integrate renewably sourced polymers that maintain polymer chain integrity during high-dose ionizing radiation cycles.
Quality assurance directors in the MedTech sector are moving beyond simple material substitution toward a fundamental recalculation of the sterile barrier system lifecycle. The decision is no longer about whether a tray is bio-based, but whether it can withstand 25 to 50 kGy of gamma radiation without losing the impact resistance required for heavy orthopedic kits. Delaying this transition creates a significant audit risk as global healthcare systems implement carbon-accounting protocols for their primary suppliers. Practitioners often find that the perceived fragility of bioplastic packaging is a myth; when formulated with specific stabilizing agents, these resins can actually exhibit superior dimensional stability compared to legacy fossil-based alternatives post-irradiation.
The primary inflection point for widespread adoption depends on the validation of five-year shelf-life stability in bio-based formats. Once a critical mass of orthopedic OEMs completes the rigorous ISO 11607 testing for bio-resins, the subsequent adoption becomes a procurement standard rather than an R&D experiment. Regulatory bodies and hospital purchasing groups trigger this shift by prioritizing vendors who provide verifiable lifecycle assessment data for their sterile packaging systems.
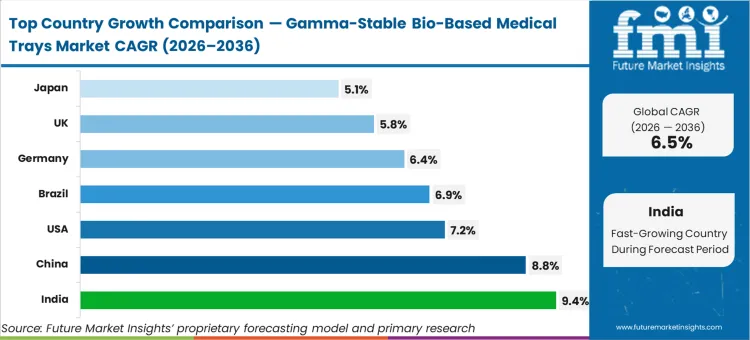
Demand for medical trays in India is set to grow at 9.4%, followed by China at 8.8% as both nations expand their domestic medical device manufacturing capacity. The United States market is anticipated to record a 7.2% CAGR, while Brazil follows at 6.9%. Germany’s sector is likely to post a CAGR of 6.4%, with the United Kingdom and Japan recording 5.8% and 5.1% respectively. This structural divergence reflects the variance between export-driven manufacturing hubs in Asia and the policy-heavy procurement environments in North America and Western Europe.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.9 Billion |
| Industry Value (2036) | USD 1.7 Billion |
| CAGR (2026-2036) | 6.57% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
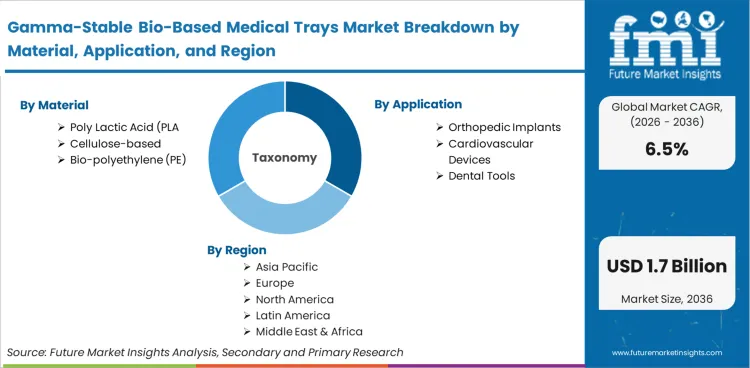
The industry comprises the production and sale of thermoformed or injection-molded primary packaging trays derived from renewable biological feedstocks specifically engineered for gamma sterilization. These trays must maintain mechanical properties, microbial barrier integrity, and visual clarity after exposure to ionizing radiation. This market is analytically distinct from general bioplastics due to the high-performance requirements of the sterile medical supply chain.
This market includes rigid trays made from PLA, bio-PE, and bio-PP designed for terminally sterilized medical devices. It covers specialized secondary components such as bio-based inserts or lids that form part of the sterile barrier system. Within scope are thermoformed medical packaging solutions that utilize plant-derived resins to support orthopedic, cardiovascular, and dental surgical kits.
Excluded from this scope are fossil-based trays like standard PETG, high-impact polystyrene (HIPS), or PVC, even if used for medical applications. Flexible medical pouches, non-sterile primary packaging, and trays intended only for steam or ethylene oxide (EtO) sterilization are also excluded. These exclusions are based on the unique material science challenges associated with gamma-induced polymer degradation which do not apply to non-irradiated or fossil-plastic categories.
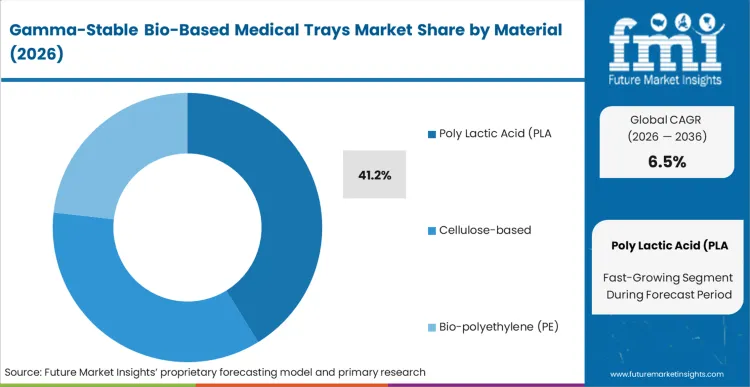
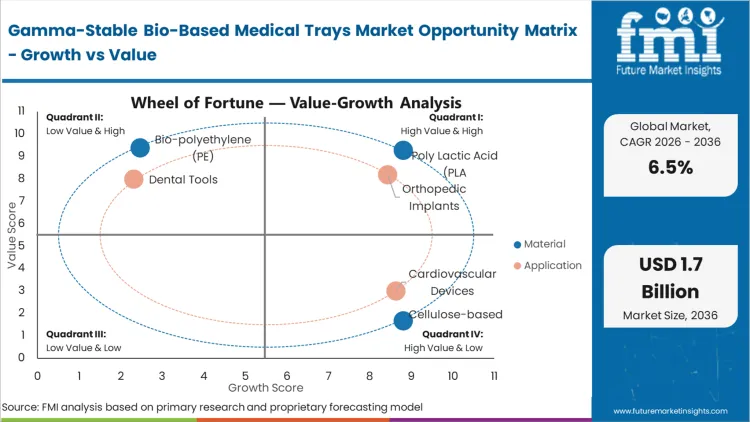
The displacement of legacy PETG and HIPS is accelerating as bio-based resins overcome the historic hurdle of radiation-induced embrittlement. Poly Lactic Acid (PLA) holds 41.2% of this market because it offers the most established pathway to USP Class VI compliance among renewable alternatives. Procurement directors at orthopedic device firms are no longer viewing PLA as an experimental material; it is the only commercially scalable option that maintains the high-stiffness profile needed for heavy surgical kits. According to FMI’s assessment, the adoption of polylactic acid is driven by its ability to undergo thermoforming on existing high-speed equipment without requiring massive capital expenditure. As hospital systems tighten their carbon accounting, the operational consequence of sticking with fossil-based trays is a loss of preferred vendor status in major procurement tenders.
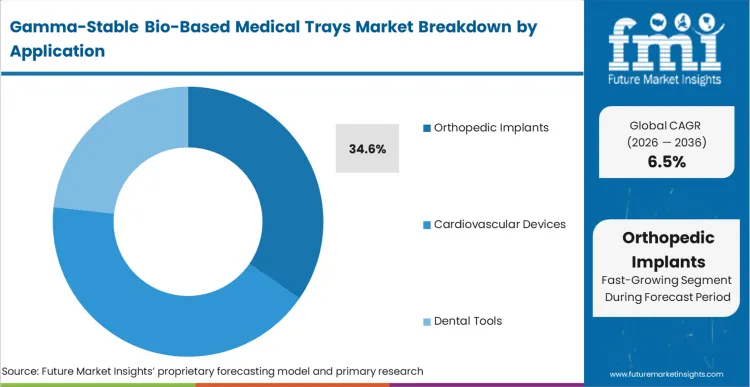
The consequence of orthopedic implant manufacturers failing to modernize their packaging is an immediate friction in high-throughput surgical environments. Orthopedic Implants represent the leading application with 34.6% share because these devices often feature complex geometries and sharp edges that stress the tray's structural integrity. FMI analysts opine that the transition to orthopedic implants packaging in bio-based formats is moving fastest because these high-value items can better absorb the initial unit-cost premium of specialized resins. Buyers are finding that the dimensional stability of gamma-stable bio-trays allows for tighter tolerances in kit design, reducing the overall footprint of the surgical setup. For surgeons, the clarity of the tray is a functional requirement for verifying the implant size before opening the sterile barrier, making high-clarity bio-polymers essential.
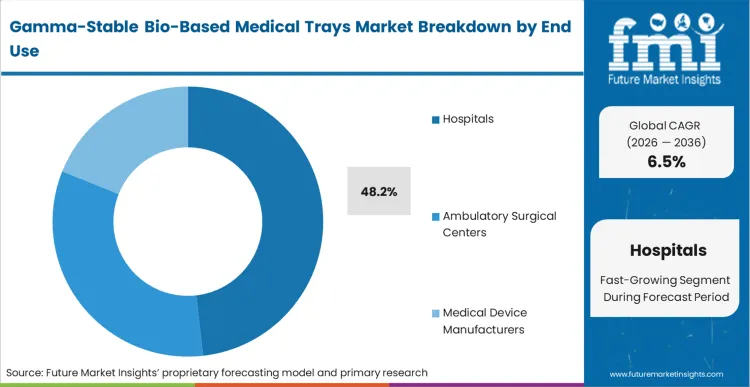
Hospital procurement groups are the primary actors forcing the shift toward sustainable sterile barrier systems. Hospitals hold a dominant 48.2% share because they act as the final decision-maker in the medical supply chain, increasingly weighting environmental impact in their RFP scoring. Based on FMI's view, the decision to mandate healthcare packaging that is both gamma-stable and bio-based is an operational response to the massive volume of plastic waste generated in surgical suites. Clinical staff are being retrained to identify bio-based trays to ensure they enter the correct waste stream for industrial composting or recycling, rather than being incinerated with hazardous waste. The commercial consequence for device manufacturers who ignore this hospital-level pressure is a slow but steady exclusion from the most lucrative integrated delivery networks.
The structural forcing condition for this market is the integration of extended producer responsibility into healthcare procurement. Large brand owners in the medical device space are being compelled by their institutional investors to decouple revenue growth from fossil-plastic consumption. This creates a decision for sourcing directors: invest in the qualification of sustainable packaging now or face future carbon taxes that will erode margins on high-volume single-use kits. The driver is not a temporary trend but a permanent shift in how MedTech firms evaluate their total cost of ownership, where the end-of-life disposal cost is finally being factored into the initial purchase price.
The primary restraint remains the lack of standardized validation protocols for bio-resins post-gamma exposure. Most current ISO standards were written with fossil-plastics in mind, leaving a gap where manufacturers must perform expensive, custom testing to prove that a bio-tray won't lose its microbial barrier over a five-year shelf life. This friction is organizational; the risk-averse nature of regulatory affairs departments often blocks adoption even when the engineering team has proven performance parity. While new testing consortia are emerging to share the burden of this validation, the current landscape requires each OEM to bear the high cost of individual material qualification.
Based on the regional analysis, the Gamma-Stable Bio-Based Medical Trays market is segmented into North America, Europe, Asia Pacific, Latin America, Middle East, and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.4% |
| China | 8.8% |
| USA | 7.2% |
| Brazil | 6.9% |
| Germany | 6.4% |
| UK | 5.8% |
| Japan | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The North American market is shaped by the rapid consolidation of hospital procurement into massive group purchasing organizations (GPOs). These GPOs have begun to include sustainability scores as a tie-breaking factor in competitive bids for orthopedic and cardiovascular kits. As per FMI's projection, the move toward medical device packaging that uses renewable feedstock is becoming a defensive necessity for manufacturers protecting their US market share.
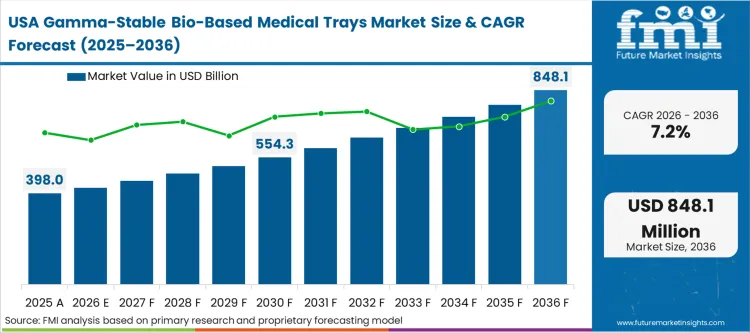
FMI’s report includes a detailed assessment of Canada and Mexico, where market dynamics are increasingly synchronized with U.S. hospital procurement standards. The structural pattern across these North American markets reveals that the move toward medical device packaging using renewable feedstock is becoming a defensive necessity for manufacturers protecting their regional market share. As large Group Purchasing Organizations (GPOs) consolidate their influence, the availability of "packaging transparency" files is shifting from a premium feature to a baseline requirement for any vendor seeking long-term contract eligibility in the region.
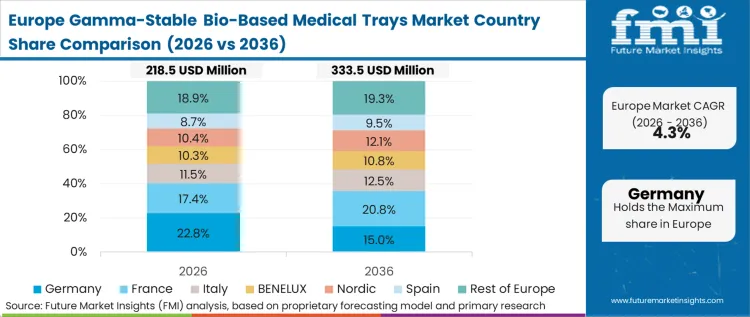
Europe's adoption is strictly policy-led, with the updated Packaging and Packaging Waste Regulation (PPWR) creating a direct financial penalty for non-recyclable or high-carbon medical plastics. European manufacturers are forced to treat the tray as a structural component of their environmental compliance strategy.
FMI's analysis covers additional key markets including France, Italy, and the Benelux region, where adoption is strictly anchored to the European Union's updated Packaging and Packaging Waste Regulation (PPWR). The structural pattern across these additional European countries shows that legislative measures, such as national bans on specific single-use plastic formats, are forcing manufacturers to treat the tray as a structural component of their broader environmental compliance strategy. This transition is accelerating as domestic chemical industries pivot toward bio-monomer production, effectively lowering the administrative and financial burdens for firms reporting on plastic usage under national transparency laws.
Infrastructure development in Asia Pacific is creating a unique opportunity for "leapfrogging," where new medical device plants are built from the ground up to use bio-based materials. This avoids the high cost of re-validating existing legacy lines that slows down adoption in Western markets.
FMI's report includes detailed analysis for South Korea, Japan, and Southeast Asian markets like Thailand and Indonesia, where infrastructure development is creating a unique opportunity for "leapfrogging". The structural pattern across these additional countries reveals that those with existing bio-polymer infrastructure, often supported by government mandates for "green manufacturing", are adopting bio-based trays roughly 18 months faster than those reliant on imported resins. This avoids the high cost of re-validating legacy lines, allowing new medical device plants to be built from the ground up with sustainable materials to maintain global export competitiveness.
The competitive structure of this market is moderately concentrated, as the capital requirements for medical-grade thermoforming and cleanroom operations act as a significant barrier to entry. Leading players like Nelipak Healthcare Packaging and DuPont rely on their deep regulatory expertise to distinguish themselves from generalist packaging firms. Buyers in this market do not choose vendors based on price alone; they select based on the vendor’s ability to provide a "validation package" that includes data on material performance after 50 kGy of gamma exposure
Incumbents such as NatureWorks LLC and Mitsubishi Chemical hold a structural advantage because they control the proprietary additive packages that prevent bio-resins from yellowing. A challenger must build more than just a thermoforming line; they must develop a specialized compounding capability to ensure their bio-based trays can survive the ionizing radiation environment. This requires an investment in rigid medical packaging R&D that typically takes three to five years to yield a commercially viable product that passes ISO 11607.
The trajectory through 2036 shows a tension between large buyers wanting to avoid vendor lock-in and the reality that only a few suppliers can meet the rigorous purity standards for sterile trays. Large medical device OEMs are resisting lock-in by qualifying at least two different bio-resin sources for each tray design, even if it doubles their initial validation cost. This trend ensures that the market remains competitive, though the advantage persists for those who can offer a verifiable lifecycle assessment as part of their standard product data sheet.
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.9 Billion to USD 1.7 Billion, at a CAGR of 6.57% |
| Market Definition | Rigid primary packaging trays derived from biological feedstocks, specifically stabilized to maintain sterile barrier integrity after gamma radiation exposure. |
| Material Segmentation | Poly Lactic Acid (PLA), Cellulose-based, Bio-polyethylene (PE), Bio-polypropylene (PP) |
| Application Segmentation | Orthopedic Implants, Cardiovascular Devices, Surgical Instruments, Dental Tools |
| End Use Segmentation | Hospitals, Ambulatory Surgical Centers, Medical Device Manufacturers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, MEA |
| Countries Covered | USA, Germany, China, India, Japan, UK, Brazil, and 40 plus countries |
| Key Companies Profiled | Nelipak Healthcare Packaging, TEQ, Novolex, Placon, DuPont, Mitsubishi Chemical, NatureWorks, Arkema |
| Forecast Period | 2026 to 2036 |
| Approach | FMI uses a supply-side analysis of medical-grade bio-resins combined with demand-side modeling of irradiated surgical units. Forecasts are validated via interviews with sterilization service providers. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Gamma-Stable Bio-Based Medical Trays in 2026?
The industry is expected to be valued at USD 0.9 billion in 2026. This figure signals that the market has moved beyond the "pilot" phase, where bio-based resins are now being integrated into standard production lines for high-volume orthopedic surgical kits.
What will it be valued at by 2036?
By 2036, the total opportunity is projected to reach USD 1.7 billion. This doubling of value reflects the structural transition where bio-based materials become the default standard for single-use medical trays in North America and Europe, displacing fossil-based PETG.
What CAGR is projected for the forecast period?
The market is set to grow at a CAGR of 6.57% through 2036. This rate is constrained by the multi-year validation cycles required for sterile medical products, meaning the growth reflects actual verified adoption rather than speculative interest.
Which material segment leads the market?
Poly Lactic Acid (PLA) leads with a 41.2% share because it offers the inherent visual clarity required for pre-surgical inspection. The mechanism here is functional; surgeons must see the device through the tray to ensure it is the correct size before breaking the sterile barrier, and PLA currently provides the best transparency among bio-resins.
Which application segment leads the market?
Orthopedic Implants lead because the sharp edges and heavy weight of these devices require high-modulus materials that bio-resins like PLA can provide. The adoption sequence here is driven by the fact that high-value orthopedic kits can absorb the initial cost premium of specialized bio-based materials more easily than low-cost dental tools.
Which end use segment leads the market?
Hospitals lead with 48.2% share as they are the final decision-makers implementing green procurement mandates. The mechanism is a top-down forcing condition where hospital GPOs penalize medical device manufacturers who cannot provide carbon-neutral primary packaging.
What is the primary driver for rapid growth?
The shift toward value-based procurement acts as the primary driver, where the "environmental cost" of a tray is finally being calculated alongside its purchase price. This is a structural insight because it changes the buyer's focus from immediate unit cost to total lifecycle compliance.
What is the primary restraint on market adoption?
The main restraint is the lack of universal data on long-term shelf-life stability for irradiated bio-polymers. This creates a structural friction where risk-averse regulatory departments delay adoption until they can see multi-year validation data, regardless of the material's immediate performance.
Which country is growing the fastest?
India is the fastest-growing country at 9.4% CAGR. Compared to the United States (7.2%), India’s growth is driven by the creation of new, green-field medical device manufacturing hubs that adopt bio-based standards from day one, avoiding the "re-validation lag" that slows down Western markets.
How does gamma radiation specifically affect bio-based trays compared to PETG?
While PETG is highly stable, standard bio-resins can embrittle; however, specialized medical-grade bio-blends utilize antioxidants to maintain polymer chain length. The mechanism involves tuning the resin's molecular weight distribution to ensure that the cross-linking induced by gamma radiation does not lead to a loss of impact resistance.
Can these trays be recycled in a standard hospital environment?
Structurally, these trays are often compatible with bio-based recycling streams, but the gap exists in hospital waste segregation infrastructure. To capture the full sustainability benefit, manufacturers are increasingly partnering with hospitals to create closed-loop collection programs specifically for rigid medical bioplastics.
Why are hospitals willing to pay a premium for bio-based trays?
The premium is justified by the reduction in Scope 3 emissions that hospitals must report to regulatory and financial stakeholders. This structural insight reveals that the "buyer" is not just the surgeon using the kit, but the sustainability officer who uses the packaging data to meet institutional ESG goals.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.