Hypopituitarism Diagnostics Market Size, Market Forecast and Outlook By FMI
Summary of the Hypopituitarism Diagnostics Market
- Demand and Growth Drivers
- Improving diagnostic imaging and endocrine assessment capabilities are expanding identification of hypopituitarism, increasing the patient population requiring hormone stimulation testing and ongoing monitoring.
- MRI or CT scan accounts for 41.2% of the test type segment, reflecting the critical role of pituitary imaging in structural assessment and differential diagnosis.
- Hospitals hold 47.5% of end-user share, confirming specialized endocrine centers as the primary diagnostic and treatment delivery settings.
- Product and Segment View
- MRI or CT scan holds 41.2% of test type share in 2026, confirming imaging as the primary diagnostic modality.
- Clinical diagnosis accounts for 44.5% of application share, reflecting diagnostic workup as the primary demand driver.
- Immunoassay technology represents 40.3% of technology share, confirming its role in hormone level quantification.
- Geography and Competitive Outlook
- Japan (6.9%) and the UK (6.8%) are expected to lead growth, driven by advanced endocrine diagnostic infrastructure and established pituitary care programs.
- The USA (6.6%) reflects strong diagnostic imaging investment and endocrine specialty care.
- Siemens Healthineers holds an estimated 15.0% market share, reflecting the fragmented diagnostic market with multiple imaging and assay platform manufacturers.
- Analyst Opinion
- The hypopituitarism diagnostics market is expanding as advanced imaging, hormone assay technology, and genetic testing improve identification of pituitary hormone deficiencies.
- Growth reflects increasing awareness of hypopituitarism as an underdiagnosed condition, with expanding screening programs in post-traumatic brain injury and post-radiation patient populations.
- Diagnostic companies with integrated imaging and immunoassay platforms are positioned to capture demand from hospital endocrine departments.
- Point-of-care hormone testing and rapid immunoassay platforms are improving diagnostic accessibility outside specialized tertiary centers.
- Companies investing in AI-assisted pituitary imaging interpretation and multiplex hormone panel development are expected to reshape competitive dynamics.
Hypopituitarism Diagnostics Market Definition
The Hypopituitarism Diagnostics Market encompasses imaging systems, hormone assay platforms, and diagnostic testing services used in identifying and monitoring pituitary hormone deficiencies.
Hypopituitarism Diagnostics Market Inclusions
Market scope includes all commercially traded diagnostics categorized by test type (MRI/CT scan, hormone stimulation tests, visual field check, insulin tolerance test), application (clinical diagnosis, screening, monitoring), technology (immunoassay, molecular diagnostics, imaging technology), and end user (hospitals, specialty clinics, academic research institutes, others). Revenue spans 2026 to 2036.
Hypopituitarism Diagnostics Market Exclusions
The scope does not include general brain imaging not specific to pituitary assessment, general blood chemistry analyzers, or pituitary tumor surgical equipment.
Hypopituitarism Diagnostics Market Research Methodology
- Primary Research: FMI analysts conducted interviews with industry participants, product specialists, and distribution channel experts across key markets.
- Desk Research: Combined industry publications, regulatory filings, trade association data, and manufacturer disclosures.
- Market Sizing and Forecasting: Bottom-up aggregation across product segments and regional adoption curves, validated through top-down cross-referencing.
- Data Validation: Cross-checked quarterly against industry production data and manufacturer-reported figures.
Why is the Hypopituitarism Diagnostics Market Growing?
- Advanced MRI imaging with dedicated pituitary protocols is improving diagnostic precision for hypopituitarism, enabling earlier identification and treatment initiation.
- Expanding screening in at-risk populations including post-traumatic brain injury and post-radiation patients is broadening the diagnostic addressable market.
- Japan (6.9%) leads growth, reflecting advanced endocrine diagnostic infrastructure and established pituitary care expertise.
Demand for hypopituitarism diagnostics reflects improving imaging and assay technology that enables more accurate identification of pituitary hormone deficiencies. Dedicated pituitary MRI protocols with dynamic contrast enhancement provide high-resolution structural assessment, while hormone stimulation testing quantifies specific deficiency patterns. This combined diagnostic approach supports targeted hormone replacement therapy decisions.
Clinical diagnosis (44.5% of application share) represents the largest demand category, driven by endocrinologists and neurologists evaluating patients with suspected pituitary dysfunction. Screening applications are expanding as clinical awareness grows regarding hypopituitarism prevalence in traumatic brain injury survivors and patients who have received cranial radiation therapy.
Immunoassay technology (40.3% share) underpins hormone quantification for pituitary function assessment. Advances in multiplex immunoassay platforms enable simultaneous measurement of multiple pituitary hormones from a single sample, improving diagnostic efficiency and reducing patient burden.
Market Segmentation Analysis
- MRI/CT scan holds 41.2% of test type share, confirming imaging as the primary diagnostic modality.
- Clinical diagnosis accounts for 44.5% of application share, reflecting diagnostic workup demand.
- Immunoassay represents 40.3% of technology share, confirming its role in hormone quantification.
The market is segmented by test type (MRI/CT, hormone stimulation tests, visual field check, insulin tolerance test), application (clinical diagnosis, screening, monitoring), technology (immunoassay, molecular diagnostics, imaging), and end user (hospitals, specialty clinics, academic research institutes, others).
Insights into the MRI or CT Scan Test Type
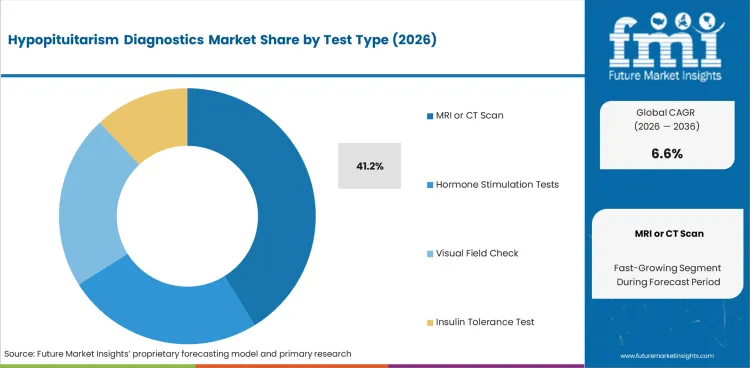
In 2026, MRI or CT scan is expected to represent 41.2% of the test type segment. Pituitary MRI with dynamic contrast enhancement is the standard imaging modality for evaluating pituitary gland structure, identifying tumors, and detecting stalk abnormalities.
Demand is supported by expanding imaging access and improving MRI protocols that enhance pituitary visualization.
Insights into the Hospital End User
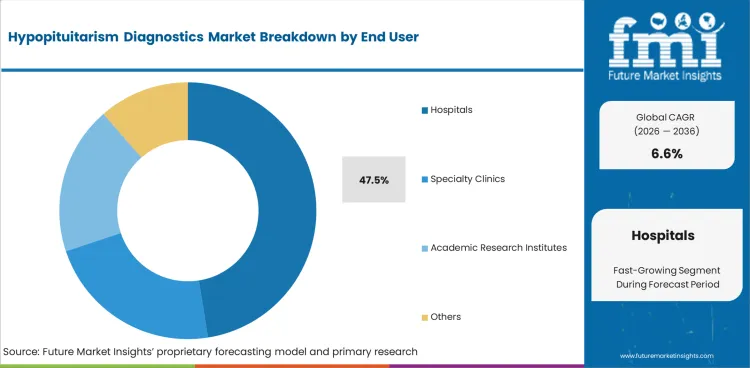
Hospitals account for 47.5% of end-user demand. Endocrine diagnostic workups for suspected hypopituitarism require specialized imaging, hormone stimulation testing, and multi-disciplinary clinical interpretation typically available in hospital settings.
Specialty clinics serve outpatient follow-up monitoring and screening applications.
Hypopituitarism Diagnostics Market Drivers, Restraints, and Opportunities
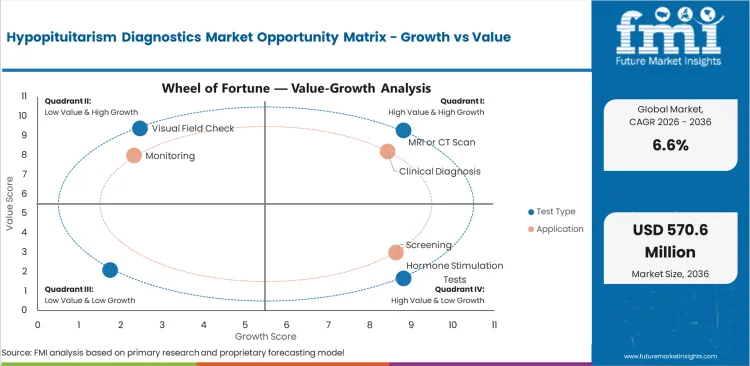
- Advancing diagnostic imaging and assay technology are improving hypopituitarism identification rates.
- Diagnostic complexity and limited clinical awareness outside specialist centers constrain timely patient identification.
- AI-assisted imaging and multiplex hormone panels offer efficiency and accuracy improvement opportunities.
The market benefits from diagnostic technology advancement and expanding screening programs. Awareness gaps and diagnostic complexity present constraints, while AI-assisted diagnostics and point-of-care testing offer growth opportunities.
Diagnostic Imaging and Assay Advancement
Demand is shaped by improving MRI imaging protocols and immunoassay platforms that increase diagnostic sensitivity and specificity for pituitary hormone deficiencies.
Clinical Awareness and Screening Gaps
Growth is constrained by limited awareness of hypopituitarism outside specialized endocrine centers. Many patients with post-traumatic or post-radiation pituitary dysfunction remain undiagnosed.
AI-Assisted Diagnostic Interpretation
Adoption is supported by emerging AI tools that assist in pituitary MRI interpretation, identifying subtle structural abnormalities that may be missed in standard radiological review.
Point-of-Care Hormone Testing
Growth reflects development of rapid hormone testing platforms that enable preliminary pituitary function assessment outside specialized laboratory settings.
Analysis of Hypopituitarism Diagnostics Market By Key Countries
.webp)
| Country |
CAGR |
| Japan |
6.9% |
| UK |
6.8% |
| USA |
6.6% |
| South Korea |
6.3% |
| EU |
6.2% |
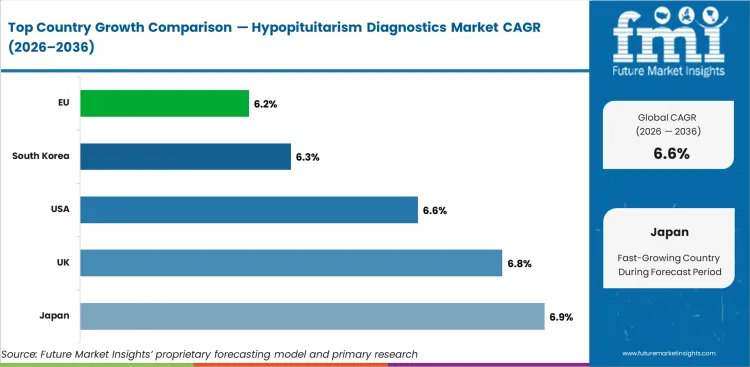
- Japan leads growth at 6.9% CAGR, driven by advanced endocrine diagnostic infrastructure and established pituitary care programs.
- The UK (6.8%) and USA (6.6%) sustain growth through expanding diagnostic capabilities and screening programs.
- South Korea (6.3%) and the EU (6.2%) support demand through healthcare infrastructure investment.
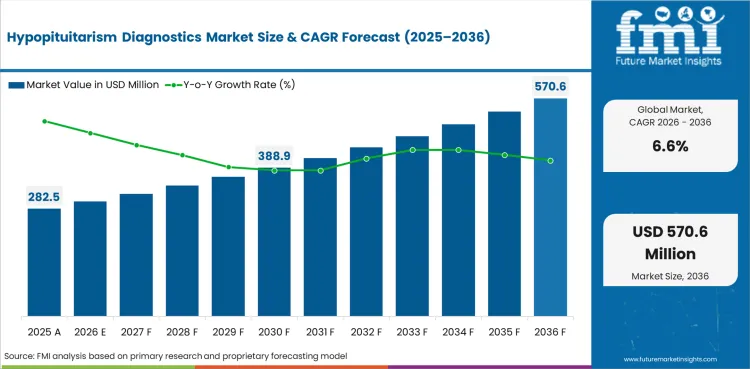
The global Hypopituitarism Diagnostics Market is expected to expand at a CAGR of 6.6% from 2026 to 2036.
Demand Outlook for Hypopituitarism Diagnostics Market in Japan
Japan is expected to lead growth at 6.9% through 2036, driven by advanced endocrine diagnostic infrastructure, established pituitary care programs, and high clinical awareness.
- Advanced diagnostic infrastructure supports comprehensive evaluation.
- Established pituitary care programs ensure specialist access.
- High clinical awareness facilitates timely diagnosis.
Future Outlook for Hypopituitarism Diagnostics Market in UK
The UK is expected to grow at 6.8% through 2036, reflecting NHS endocrine programs, expanding screening, and advanced imaging investment.
- NHS endocrine programs support structured diagnostic pathways.
- Screening programs improve at-risk population identification.
- Imaging investment enhances diagnostic capability.
Opportunity Analysis of Hypopituitarism Diagnostics Market in USA
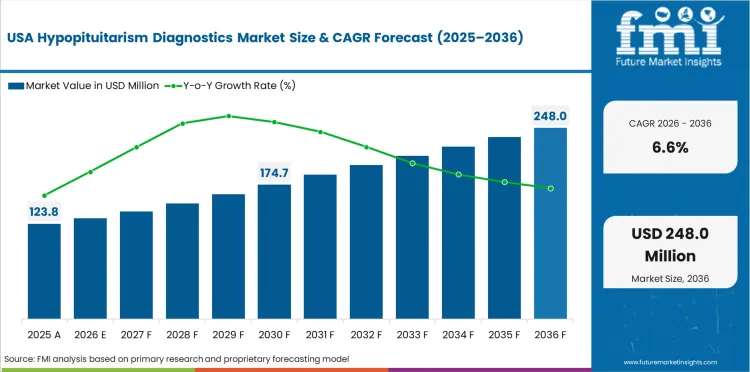
The USA is expected to grow at 6.6% through 2036, supported by advanced imaging infrastructure, endocrine specialty care, and expanding screening awareness.
- Advanced imaging infrastructure supports pituitary assessment.
- Endocrine specialty care provides comprehensive evaluation.
- Screening awareness growth improves patient identification.
In-depth Analysis of Hypopituitarism Diagnostics Market in South Korea
South Korea is expected to grow at 6.3% through 2036, driven by healthcare investment and expanding diagnostic capabilities.
- Healthcare investment improves diagnostic infrastructure.
- Advanced imaging adoption supports pituitary assessment.
- Clinical awareness expansion improves identification rates.
Sales Analysis of Hypopituitarism Diagnostics Market in EU
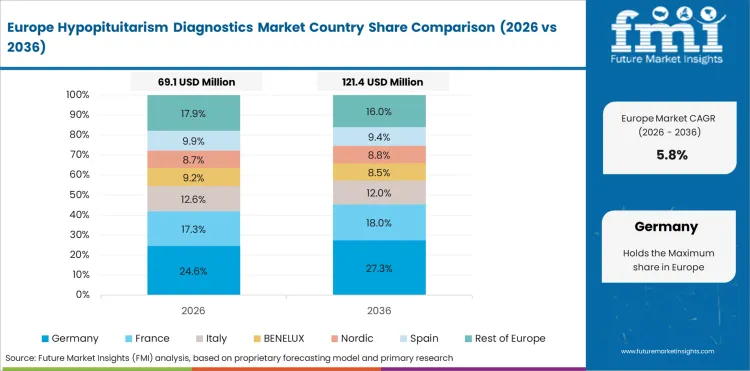
The EU is expected to grow at 6.2% through 2036, supported by cross-border diagnostic standardization and expanding endocrine care.
- Cross-border standardization improves diagnostic consistency.
- Endocrine care expansion supports patient identification.
- Imaging technology adoption improves across member states.
Competitive Landscape and Strategic Positioning
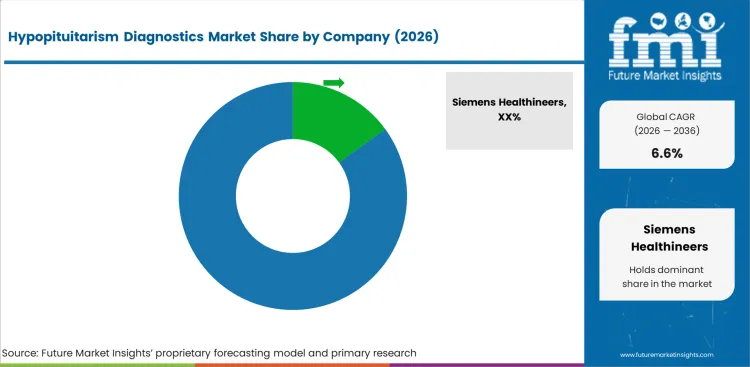
- Siemens Healthineers holds an estimated 15.0% market share, reflecting the fragmented diagnostic market.
- Abbott Laboratories and Thermo Fisher Scientific hold positions through broad immunoassay and diagnostic platforms.
- Specialized diagnostic companies serve pituitary-specific testing needs.
Siemens Healthineers participates through its MRI imaging systems and immunoassay platforms used in pituitary hormone assessment. The company's integrated diagnostic portfolio supports hospital endocrine departments.
Abbott Laboratories and Thermo Fisher Scientific compete through broad immunoassay platforms that include pituitary hormone panels for endocrine diagnostic workups.
F. Hoffmann-La Roche and Beckman Coulter (Danaher) serve the market through automated immunoassay analyzers used in hospital and reference laboratory settings.
Bio-Rad Laboratories and QIAGEN provide specialized diagnostic reagents and molecular testing platforms for endocrine assessment applications.
Key Companies in the Hypopituitarism Diagnostics Market
Key global companies leading the hypopituitarism diagnostics market include:
- Siemens Healthineers (Germany), Abbott Laboratories (USA), Thermo Fisher Scientific (USA), and F. Hoffmann-La Roche (Switzerland) maintain positions through broad diagnostic portfolios.
- Danaher/Beckman Coulter (USA), Bio-Rad Laboratories (USA), and bioMerieux (France) hold positions through specialized assay and diagnostic platforms.
- QIAGEN (Netherlands), PerkinElmer (USA), and emerging diagnostic companies represent additional competition through molecular and specialized testing.
Competitive Benchmarking: Hypopituitarism Diagnostics Market
| Company |
Imaging Capability |
Assay Platform |
Distribution Reach |
Endocrine Focus |
| Siemens Healthineers |
High |
High |
Global |
Moderate |
| Abbott Laboratories |
Low |
High |
Global |
Moderate |
| Thermo Fisher Scientific |
Low |
High |
Global |
Moderate |
| F. Hoffmann-La Roche |
Low |
High |
Global |
Moderate |
| Danaher/Beckman Coulter |
Low |
High |
Global |
Low |
| Bio-Rad Laboratories |
Low |
Moderate |
Global |
Low |
| bioMerieux |
Low |
Moderate |
Global |
Low |
| QIAGEN |
Low |
Moderate |
Global |
Low |
| PerkinElmer |
Low |
Moderate |
Global |
Low |
| GE HealthCare |
High |
Low |
Global |
Low |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Hypopituitarism Diagnostics Market
- In 2025, Siemens Healthineers introduced an AI-assisted pituitary MRI interpretation module for automated structural abnormality detection.
- In 2025, Abbott Laboratories expanded its pituitary hormone multiplex immunoassay panel for comprehensive endocrine evaluation on its core laboratory platform.
Key Players in the Hypopituitarism Diagnostics Market
Major Global Players
- Siemens Healthineers
- Abbott Laboratories
- Thermo Fisher Scientific
- Bio-Rad Laboratories, Inc.
- bioMerieux SA
- Danaher Corporation
- F. Hoffmann-La Roche AG
Emerging Players/Startups
- QIAGEN
- PerkinElmer
- Beckman Coulter
- GE HealthCare
- Hologic Inc.
- DiaSorin
- Tosoh Corporation
- Ortho Clinical Diagnostics
- Fujirebio
Report Scope and Coverage
| Parameter |
Details |
| Quantitative Units |
USD 301.15 million to USD 570.62 million, at a CAGR of 6.6% |
| Market Definition |
The hypopituitarism diagnostics market includes imaging systems, hormone assay platforms, and diagnostic testing for pituitary hormone deficiencies. |
| Regions Covered |
North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered |
Japan, UK, USA, South Korea, EU, 30 plus countries |
| Key Companies Profiled |
Siemens Healthineers, Abbott, Thermo Fisher, Bio-Rad, bioMerieux, Danaher, Roche, QIAGEN, PerkinElmer |
| Forecast Period |
2026 to 2036 |
| Approach |
Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Market Segmentation Analysis
Hypopituitarism Diagnostics Market Segmented by Test Type:
- MRI or CT Scan
- Hormone Stimulation Tests
- Visual Field Check
- Insulin Tolerance Test
Hypopituitarism Diagnostics Market Segmented by Application:
- Clinical Diagnosis
- Screening
- Monitoring
Hypopituitarism Diagnostics Market Segmented by Technology:
- Immunoassay
- Molecular Diagnostics
- Imaging Technology
Hypopituitarism Diagnostics Market Segmented by End User:
- Hospitals
- Specialty Clinics
- Academic Research Institutes
- Others
Hypopituitarism Diagnostics Market by Region:
- North America
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- 1. Endocrine Society. (2025). Endocrine Society Clinical Practice Guidelines: Hypopituitarism. ES.
- 2. European Society of Endocrinology. (2024). ESE Clinical Guidance on Pituitary Assessment. ESE.
- 3. World Health Organization. (2025). WHO Diagnostic Technology Assessment Database. WHO.
- 4. USA Food and Drug Administration. (2025). FDA Diagnostic Device Clearances. FDA.
- 5. National Institutes of Health. (2024). NIH Endocrine Research Portfolio. NIH.
This bibliography is provided for reader reference.
This Report Answers
- Estimating the size of the market and revenue projections from 2026 to 2036.
- Segmentation by test type, application, technology, and end user.
- Regional and country-level demand analysis across key markets.
- Competitive landscape assessment.
- Diagnostic imaging and assay technology trends.
- Screening program expansion impact analysis.
- Growth opportunities in AI-assisted diagnostics and point-of-care testing.
Frequently Asked Questions
What is the global market size in 2026?
In 2026, the global Hypopituitarism Diagnostics Market is expected to be valued at USD 301.15 million.
How large will the market be in 2036?
By 2036, the market is expected to reach USD 570.62 million.
What is the projected CAGR?
The market is projected to expand at a CAGR of 6.6% from 2026 to 2036.
Which test type is expected to lead?
MRI or CT scan is expected to account for 41.2% in 2026, reflecting its essential role in pituitary structural assessment.
What is driving demand in Japan?
Japan is expected to grow at 6.9% through 2036, driven by advanced endocrine diagnostics and established pituitary care.
What is driving demand in the UK?
The UK is expected to grow at 6.8% through 2036, supported by NHS endocrine programs and expanding screening.
What does this report cover?
The market includes imaging, hormone assay platforms, and diagnostic testing for identifying and monitoring pituitary hormone deficiencies.
How does FMI validate the forecast?
Forecasting uses a hybrid bottom-up and top-down approach, starting with verified diagnostic equipment and assay data.




.webp)





