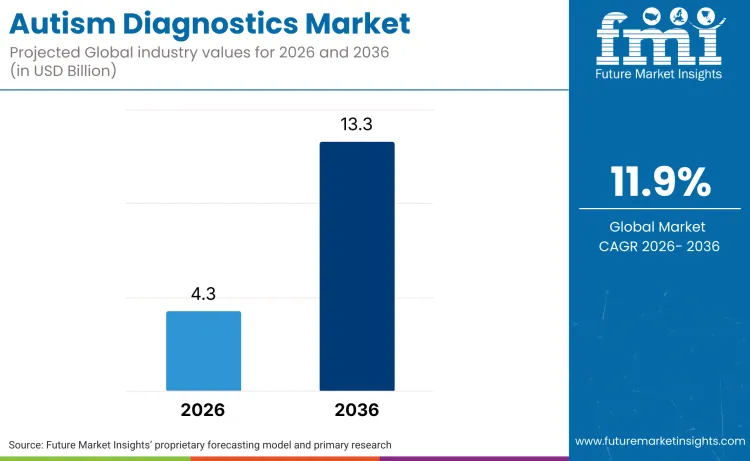
The global autism diagnostics market is projected to reach a valuation of USD 4.3 billion in 2026 and accelerate to USD 13.3 billion by 2036, expanding at a robust 11.9% CAGR. Demand for autism diagnostics, as per Future Market Insights, is being driven by a critical widening of the gap between rising global prevalence and existing clinical capacity, a reality confirmed by the World Health Organization in September 2025 when it reported that nearly 62 million people globally are living with the condition.
Major genomic players are aggressively pivoting their corporate strategies to address this health crisis through precision medicine and expanded access. "Jacob Thaysen", CEO of Illumina, emphasized this commitment by stating: "At Illumina, we work relentlessly in our mission to improve human health by unlocking the power of the genome, and our CSR strategy is an essential part of that." This executive focus highlights how the industry is moving beyond simple behavioral observation toward scalable, biologically-validated autism spectrum disorder management solutions that can meet the needs of millions.
Manufacturers are responding to increased identification by moving beyond FDA clearance to secure essential payer coverage, which is now the primary gatekeeper for commercial success.
What Is Growth Outlook for Autism Diagnostics Market as per Future Market Insights Projection?
Future Market Insights projects the autism diagnostics market to grow at a robust CAGR of 11.9% from 2026 to 2036, expanding from USD 4.3 billion in 2026 to USD 13.3 billion by 2036.
FMI Research Approach: Based on FMI’s proprietary bottom‑up forecasting model that incorporates global prevalence trends, diagnostic rate normalization, primary interviews with manufacturers and clinicians, and reimbursement + regulatory adoption scenario modeling.
How Do FMI Analysts Perceive Autism Diagnostics Market to Evolve?
FMI analysts perceive the market shifting from traditional subjective behavioral observation toward objective, scalable, reimbursable diagnostic pathways, combining AI‑driven screening, standardized assessments, and biological validation.
FMI Research Approach: Supported by evidence of increased AI‑based eye‑tracking adoption, broader insurance coverage for diagnostic software and genomic tests, deeper integration into pediatric and primary care settings, and the industry’s pivot to precision‑medicine‑aligned biomarkers.
Which Country Holds Largest Share in the Global Autism Diagnostics Market?
The United States holds the largest share of the global autism diagnostics market by value.
FMI Research Approach: Based on country‑level revenue modeling across hospitals and specialty clinics, high diagnostic penetration driven by CDC surveillance systems, early payer adoption of reimbursable digital diagnostic tools, and strong presence of leading diagnostic and genomic companies.
How Large Will the Autism Diagnostics Market Be by 2036?
The global autism diagnostics market is expected to reach USD 13.3 billion by 2036.
FMI Research Approach: Long‑term revenue forecasts accounting for offering and application mix, scaling assumptions for screening tools and standardized behavioral assessments, exclusion of therapy and non‑clinical tests, and cross‑validation with historical diagnostic and genomic market growth patterns.
What Is the Definition of Autism Diagnostics Market?
The autism diagnostics market includes commercial screening tools, diagnostic instruments, genetic testing panels, and assessment software used to identify ASD in both pediatric and adult populations.
FMI Research Approach: Structured using FMI’s taxonomy defining inclusion/exclusion criteria, validation across behavioral, genetic, and AI‑based tools, mapping across patient segments, and exclusion of therapy services, advocacy funding, and non‑validated consumer tests.
What Are Globally Unique Trends Shaping the Autism Diagnostics Market?
Globally unique trends include AI‑driven rapid screening, commercialization of physiological biomarkers, and regulatory maturation that is enabling laboratory innovation and reimbursement alignment.
FMI Research Approach: Evidenced by adoption of eye‑tracking and AI behavioral platforms, commercialization partnerships in hair, stool, and genomic biomarker testing, regulatory clarification around laboratory‑developed tests, and deployment of mass‑screening infrastructure in high‑population emerging markets.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4.3 Billion |
| Industry Value (2036) | USD 13.3 Billion |
| CAGR (2026 to 2036) | 11.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary researc
Specialty clinics are currently overwhelmed by unprecedented demand, creating an urgent operational need for advanced cognitive diagnostics that can drastically reduce appointment duration without sacrificing accuracy. Clinical piloting of the EarliPoint device at the University of Nebraska Medical Center in March 2024 proved that technology can reduce appointment times from approximately 4 hours to just 1 hour and 15 minutes, effectively tripling clinic capacity.
Sharief Taraman, CEO of Cognoa, captured this urgency with the industry maxim: "For Pediatric Developmental Health, Early Is Everything." This drive for efficiency is forcing a fundamental transition from subjective, time-consuming observation to objective measurement and rapid triage.
LinusBio validated this new direction in April 2025 by partnering with Autism Speaks to utilize ClearStrand-ASD, a biochemical test using hair strands to identify molecular signatures. The integration of such physiological biomarkers allows clinicians to triage cases faster, ensuring that scarce specialist time is allocated to complex diagnoses rather than initial screening.
The autism diagnostics market is segmented by offering, application, end user, and region to reflect the diverse pathways of care. By offering, the market includes screening tools, diagnostic instruments, and standardized behavioral assessment kits that serve as the primary clinical interface. By application, demand spans pediatrics and adult diagnosis, reflecting the growing need for cognitive assessment and training across the entire human lifespan. By end user, adoption is led by hospitals, specialty clinics, and diagnostic laboratories, though schools and primary care centers are emerging as key intake points.
FMI analysis suggests that while technological tools are growing fast, the market structure remains heavily reliant on trusted clinical frameworks. This segmentation highlights a dual-track market where traditional psychological tools provide depth while new digital solutions provide breadth and scale.
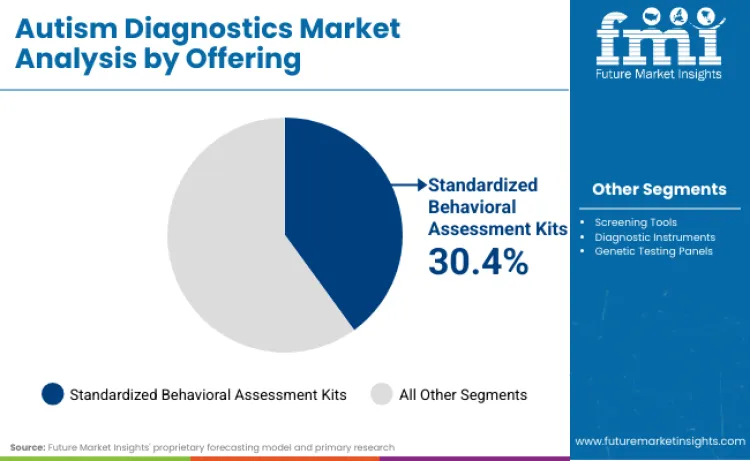
Standardized behavioral assessment kits command a dominant 30.4% share because they remain the clinically accepted "gold standard" required for insurance reimbursement and educational placement. Pearson Clinical, a leader in this space, continues to defend this position by maintaining deep trust and emotional connection with the psychologist community.
"Clay Richey", VP of Portfolio Management at Pearson Clinical Assessment, explained this personal stake: " For years, I've led the Clinical Assessment product development team, collaborating with brilliant psychologists and child development researchers to create assessment tools to help identify and support neurodivergent individuals. I'd review research data, approve design changes, and celebrate product launches,all with quiet satisfaction that our work was making a difference out there." This deep-seated clinician loyalty protects the segment from rapid displacement by unproven tech, as practitioners prefer tools they know. Traditional tools are evolving, with many now integrating digital scoring components to streamline the workflow for behavioral health professionals.
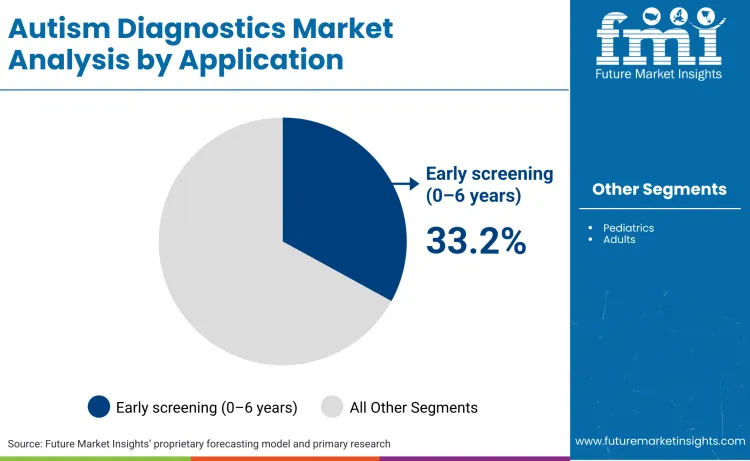
Early screening segment for children aged 0 to 6 years leads the autism diagnostics market at 33.2%. Factors supporting the dominance to surging parental awareness, routine pediatric screenings, and proven benefits of early intervention for better developmental outcomes. Rising prevalence and policy emphasis further amplify demand for reliable early diagnostics tools in this segment. Such dynamics position early screening as the cornerstone of sustained market expansion.
The genetic testing segment is accelerating rapidly as clinical evidence increasingly links specific chromosomal variances to autism phenotypes, making it a medical necessity. LabCorp supported commercialization in May 2024 by publishing comprehensive clinical guidance on navigating ASD complexity using SNP Microarrays, further solidifying the link between genotype and phenotype. This validation transforms neuro-immunoassay and genetic panels from elective research add-ons into standard components of a modern diagnostic workup. As coverage expands, clinicians are empowered to order these tests earlier in the patient journey, driving revenue growth for diagnostic labs that can offer integrated reporting.
Artificial intelligence is fundamentally reshaping assessment by replacing subjective human observation with quantifiable data collected through advanced eye tracking systems. The EarliPoint Evaluation for Autism device demonstrated an accuracy rating of about 80% in identifying children on the spectrum during pivotal 2024 trials, proving that machines can detect subtle behavioral cues. This technology allows non-specialists to gather high-fidelity data, effectively relieving the bottlenecks that currently paralyze specialty centers. It enables earlier intervention, which is critical for long-term outcomes, effectively functioning as a form of AI-powered behavioral therapy guidance by pinpointing specific deficits years earlier than standard methods.
The autism diagnostics sector is pivoting aggressively from relying solely on behavioral criteria to incorporating objective biological evidence that can be measured in a lab. LinusBio and Autism Speaks formed a strategic partnership in April 2025 to deploy physiological testing that analyzes hair strands for metabolic history, a breakthrough in non-invasive sampling.
Manish Arora, founder and CEO at LinusBio described the joint mission: " Together, we aim not only to raise awareness among families, healthcare providers, and clinicians about the benefits of biomarker testing for autism, and how it can support earlier diagnosis and intervention,but also to make a tangible, immediate impact for families currently seeking answers." This shift moves diagnostics closer to the precision of magnetoencephalography and other advanced neurological tools by providing a "biological positive." It reduces the agonizing "wait and see" period for toddlers, allowing families to access support services months or years sooner than traditional behavioral observation methods allow.
Increased regulatory clarity is finally stabilizing the investment landscape for diagnostic labs, allowing for more confident R&D spending. On September 19, 2025, the FDA issued a final rule reverting the text of the regulation regarding Laboratory Developed Tests (LDTs) to its status prior to May 2024, removing a major compliance hurdle.
This regulatory reversal lowers the barrier to entry for specialized labs to introduce new assays without facing the immediate, crushing burden of full device clearance. It fosters an environment where niche diagnostic tests can enter the market faster, directly supporting research into complex areas like 3D neuroscience and personalized medicine. Smaller labs are now freer to innovate on specific biomarker panels, knowing that the regulatory framework will not arbitrarily stifle their commercialization pathways.
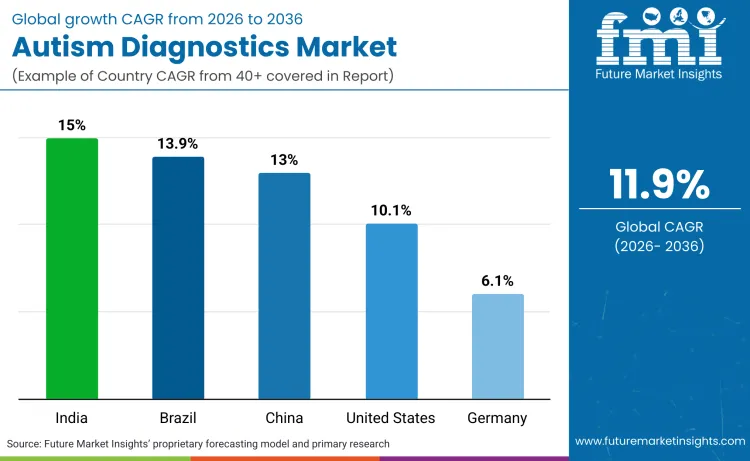
Sales in emerging markets are significantly outpacing the global average, driven by aggressive government mandates for early screening and disability certification. While the global market expands at 11.9%, developing nations are leapfrogging traditional clinical models by deploying mass-screening infrastructure directly in communities. India leads this growth with a 15.0% CAGR, followed closely by Brazil at 13.9% and China at 13.0%, creating a new axis of demand in the Global South. In contrast, mature markets like the United States (10.1%) and Germany (6.1%) are focused on efficiency and reimbursement rather than raw volume expansion. FMI analysis indicates that future growth will come from the democratization of access in these high-population regions.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 15.0% |
| Brazil | 13.9% |
| China | 13.0% |
| United States | 10.1% |
| Germany | 6.1% |
Source: FMI historical analysis and forecast data.
India is growing at a rapid 15.0% CAGR, driven primarily by regulatory standardization that is forcing the formalization of the disability sector. The Department of Empowerment of Persons with Disabilities notified new guidelines for assessing disabilities in March 2024, creating a uniform, state-sanctioned pathway for diagnosis and certification. This policy change streamlines access to government benefits, effectively incentivizing parents to seek formal diagnosis rather than relying on informal family support.
FMI predicts that the volume of certified assessments will triple by 2030 as rural adoption increases and stigma decreases. The integration of therapeutic robots in urban centers is also beginning to assist with therapy, but the core driver remains the government's push for a documented, measurable disabled population.
China is expanding at a 13.0% CAGR, fueled by massive state-funded pediatric initiatives that aim to screen every child. As of March 2025, the Beijing Municipal Health Commission established 80 pediatric expert clinics to provide comprehensive free screenings for children aged 0-6, setting a precedent for other provinces.
Celeste Saulo, Secretary-General of the WMO, commented on China's tech sharing in a broader context: "This technology not only benefits China, but is also open source. [It is] a good example to bridge the technology gap among different countries through AI."
This infrastructure build-out includes advanced systems showcased by China Mobile in July 2025, which support the country's "Internet Hospital" vision. The integration of healthcare companion robots into these clinics suggests a future where automated, AI-driven screening becomes the primary filter for the country's massive pediatric population.
Brazil's market is rising at a 13.9% CAGR due to significantly improved data visibility and government tracking. The 2024 PeNSE national health survey and Census data now actively track autism diagnoses by health professionals, removing the "invisible" nature of the demographic. Brazil is positioned as an attractive destination for international research funding. This influx of research can lead to the development of more tailored interventions and therapies that resonate with the specific cultural and social contexts of the Brazilian population. As local researchers collaborate with international partners, they can share invaluable insights that enhance the understanding of autism and its variations in different demographics. FMI analysis indicates that this scientific integration will make Brazil the primary hub for Latin American clinical trials and digital therapeutics validation.
The USA is growing at a steady 10.1% CAGR, characterized by a fundamental shift from identification to insurance coverage. The CDC's 2025 Community Report confirms that identification strategies are working, but the bottleneck has moved to payer access and waiting lists. "John Reviere", VP of Clinical Affairs at EarliTec Diagnostics, noted the collaborative solution: "We are thrilled to partner with MMI on clinical implementation of EarliPoint™️ to accelerate early and efficient diagnosis of autism in young toddlers."
With the FDA's September 2025 ruling on LDTs, the regulatory environment has finally stabilized for labs. The industry is focused on optimizing models where diagnosis can initiate immediate, reimbursable home-based interventions, reducing the time between detection and care.
Established leaders like Pearson Clinical protect their market share through deep community integration and an extensive library of 300,000+ assessment tools. They leverage major events like the 2024 "Virtual Autism Summit" to maintain loyalty among psychologists who act as the primary gatekeepers for diagnosis. Startups are challenging this dominance by securing direct payer reimbursement, creating a new competitive front. "Alice Shillingsburg", Director of the Integrated Center for ASD at UNMC, validated the clinical utility of these new entrants: "For many children, we will be able to use this device to confirm the presence of autism, when combined with clinical judgment and developmental history."
Strategic partnerships are further defining the landscape, such as LinusBio's collaboration with advocacy giant Autism Speaks, which moves competition into the realm of patient advocacy. Even manufacturers of peripheral tools like decompression toys are finding ways to align with diagnostic brands to offer holistic support packages to families.
Recent Development:
The autism diagnostics market represents revenue generated from the sale of screening tools, diagnostic instruments, genetic testing kits, and assessment software used to identify Autism Spectrum Disorder (ASD). As operationally defined in the article, the market measures the commercial value of consumables, capital equipment, and software licenses sold to healthcare providers, labs, and research institutions.
The market includes standardized behavioral assessment kits (e.g., ADOS-2, ADI-R), molecular and genetic testing panels (e.g., chromosomal microarray, Whole Exome Sequencing), and emerging AI-based diagnostic aids (e.g., eye-tracking devices). It covers adoption by hospitals, specialized pediatric clinics, diagnostic laboratories, and increasing use in primary care settings.
Revenue from research grants, non-profit advocacy donations, and therapy services (post-diagnosis) is excluded. General mental health screening tools not specific to autism and consumer-grade "tests" without clinical validation are also not counted as market revenue.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 4.3 Billion |
| Offering Segments | Screening Tools, Diagnostic Instruments, Standardized Behavioral Assessment Kits, Genetic Testing Panels |
| Application Segments | Pediatrics, Adults |
| End User Segments | Hospitals, Specialty Clinics, Diagnostic Laboratories, Research Institutes, Schools |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Middle East & Africa |
| Countries Covered | United States, China, India, Brazil, Germany, United Kingdom, Japan + 40+ countries |
| Key Companies Profiled | Pearson Clinical, Cognoa, Illumina, EarliTec Diagnostics, LabCorp, LinusBio, Quest Diagnostics, Roche Diagnostics, Thermo Fisher Scientific, Akili Interactive, Click Therapeutics |
| Additional Attributes | Regulatory landscape analysis (FDA LDT Rule), reimbursement trends (Medicaid coverage), AI integration impact, physiological biomarker validation, mass-screening infrastructure in emerging markets, supply chain resilience analysis |
Source: FMI historical analysis and forecast data.
What is the current global market size for Autism Diagnostics?
The global market is valued at USD 4.3 billion in 2026, driven by rising prevalence and the introduction of AI-based screening tools.
What is the projected CAGR for the Autism Diagnostics market?
Sales are projected to increase at a 11.9% CAGR from 2026 to 2036, significantly outpacing general medical device growth.
Which segment currently dominates the industry?
Screening tools hold the leading share of 33.2%, while standardized behavioral assessment kits account for 30.4% of the offering share.
What are the primary drivers for adoption in emerging markets?
Government mandates, such as India's revised disability guidelines and China's free clinic initiative, are the primary engines for growth in developing regions.
Who are the leading players in the Autism Diagnostics space?
Pearson Clinical leads in behavioral assessments, while Cognoa and Illumina are dominating the AI and genomic testing segments respectively.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.