Demand for dengue vaccines in Japan is valued at USD 4.9 million in 2025 and is projected to reach USD 9.0 million by 2035, reflecting a CAGR of 6.2%. Valuation from USD 3.6 million in 2020 to USD 4.9 million in 2025 reflects a gradual uptake centered on travel-related prevention and pediatric immunization planning. Qdenga holds a marginal lead in market share due to its broader age-group suitability. Pediatric use accounts for the larger share of administered doses, as the preventive focus remains aligned with school-age travelers and households with overseas relocation. Direct procurement by government and public health bodies forms the largest distribution route. Hospitals and travel medicine clinics account for steady secondary administration volumes. Early phase demand remains limited by low domestic disease incidence and narrowly defined risk groups.
From 2026, demand rises from USD 5.2 million to USD 7.0 million by 2031, then advances to USD 9.0 million by 2035 through consistent annual additions. Growth reflects expanded immunization coverage among outbound travelers, defense personnel, and long-stay expatriate populations. Hospitals maintain a stable role for pre-travel consultation and administration. Travel medicine clinics gain incremental share as private vaccination services widen across urban centers. NGO supported programs remain limited in scale and focused on high-risk overseas assignments. Adult vaccination volumes increase steadily as awareness improves among older travelers and business groups. After 2030, growth reflects repeat vaccination cycles rather than first-time uptake alone. Annual value additions remain narrow, indicating controlled expansion shaped by travel patterns, public health guidance, and procurement planning practices in Japan.
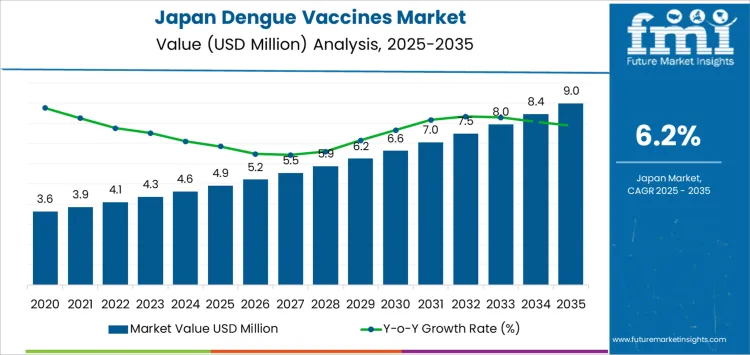
Between 2025 and 2030, demand for dengue vaccines in Japan is projected to rise from USD 4.9 Between 2025 and 2030, demand for dengue vaccines in Japan increases from USD 4.9 million to USD 6.6 million, representing an absolute expansion of USD 1.7 million over five years. Average annual value addition during this phase remains close to USD 0.3 million, indicating steady but controlled uptake. Growth is guided by travel medicine prescriptions, occupational vaccination for overseas assignments, and gradual inclusion within private preventive care programs. Earlier demand levels were constrained by limited domestic risk perception. Near term expansion reflects higher outbound travel volumes, sharper clinical screening for febrile illnesses, and availability of vaccine options via hospital programs.
From 2030 to 2035, demand rises from USD 6.6 million to USD 9.0 million, representing an additional USD 2.4 million over the final five years of the forecast. Yearly value additions in this phase average close to USD 0.5 million, signaling a faster expansion profile than the earlier period. Uptake strengthens through structured adult immunization programs, outbound tourism growth among older populations, and institutional procurement by travel clinics. Market progression becomes less episodic and more program-driven. Earlier growth relied on reactive demand, while later growth reflects planned prevention strategies embedded within routine healthcare delivery systems. Clinical adoption improves across regional care
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 4.9 million |
| Forecast Value (2035) | USD 9.0 million |
| Forecast CAGR (2025-2035) | 6.2% |
Demand for dengue vaccines in Japan remained limited for most of the modern vaccination era because domestic transmission stayed rare and case counts relied on imported infections among travelers and short term visitors. Public health priorities focused on influenza, childhood immunization, and adult boosters for established pathogens. Surveillance studies monitored vector presence and sporadic outbreaks without sustained circulation. Historical procurement occurred through research programs, small clinical stockpiles, and travel medicine providers serving outbound tourism. Risk perception stayed low across the general population as hospitalization burden remained minimal. The absence of routine recommendations kept unit volumes constrained. Public confidence depended on localized risk assessment rather than broad prevention policy. Manufacturing presence existed through regional pharmaceutical capability rather than mass immunization supply chains. Demand patterns followed travel advisories and regional outbreaks globally.
Future demand for dengue vaccines in Japan is expected to track shifts in travel patterns, vector surveillance, and regulatory positioning of next generation vaccines. Public sector demand may remain targeted toward travelers, expatriates, and occupational exposure groups. Climate trends support longer mosquito activity seasons in parts of the country, raising monitoring intensity. Health authorities evaluate cost effectiveness, post marketing safety, and cross protection against multiple serotypes. Historical demand relied on episodic import risk. Future demand depends on contingency planning, traveler vaccination guidance, and stockpile policy. Supply strategies emphasize cold chain control, shelf life management, and distribution through hospital networks rather than mass campaigns. Uptake will reflect public trust, clinical guidance, and perceived exposure risk across mobile populations. Domestic production capacity and international licensing arrangements influence availability and pricing stability nationwide.
Demand for dengue vaccines in Japan is shaped by exposure to outbound travel, rising awareness of mosquito-borne disease risk, and targeted immunization in pediatric populations. Qdenga leads by product based on broader age eligibility and growing clinical uptake. Pediatric use leads by age group, as childhood vaccination programs and travel-related prevention drive early adoption. Hospital pharmacies, travel medicine clinics, and public health procurement channels drive procurement. Import reliance remains high because dengue vaccines are fully sourced from overseas manufacturers. Substitution pressure from vector control and personal protection measures exists within low-risk populations. Demand stability is supported by sustained international travel and preventive immunization planning.
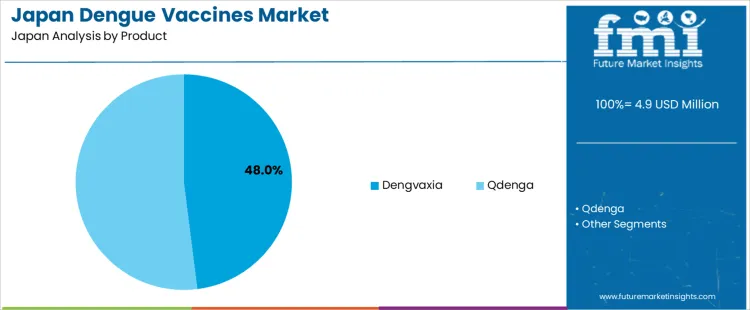
Qdenga accounts for 52% of the demand for dengue vaccines in Japan by product, reflecting its broader indication coverage and suitability across serostatus groups. Consumption intensity is driven by pre-travel vaccination, pediatric immunization scheduling, and physicians' preference for single-product coverage. Usage remains stable because dosing is structured under defined immunization intervals with predictable follow-up. Hospital outpatient pharmacies and travel health clinics lead procurement. Price sensitivity remains moderate because vaccination cost is positioned within preventive care spending. Specification control emphasizes strain coverage consistency, cold chain integrity, and post market safety tracking under national vaccine monitoring frameworks.
Qdenga also generates steady repeat demand through seasonal travel cycles and routine pediatric vaccination scheduling. Repeat utilization remains predictable as clinics restock based on appointment bookings and travel season peaks. Buyers favor products with simplified storage and handling protocols to reduce wastage risk. Margin structure remains controlled under regulated vaccine pricing and distribution contracts. Regulatory exposure remains elevated due to pharmacovigilance reporting and batch release oversight. Import reliance persists for finished dose supply. Substitution pressure from Dengvaxia remains limited due to narrower use criteria.
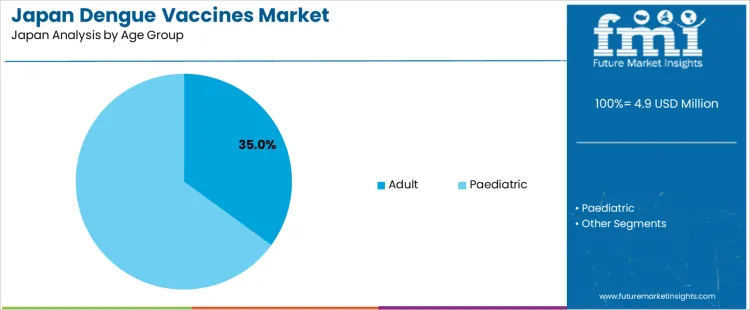
The pediatric group represents 65.0% of the demand for dengue vaccines in Japan by age group, reflecting preventive focus on early life immunization and family travel health planning. Consumption intensity is driven by school age children traveling to endemic regions and clinical guidance favoring early immune priming. Usage remains stable because vaccination timing aligns with routine pediatric visit schedules. Procurement is dominated by pediatric hospitals, community clinics, and travel medicine centers serving family travelers. Price sensitivity remains moderate because vaccines are viewed as essential preventive interventions. Specification control emphasizes age appropriate dosing accuracy, adverse event monitoring, and storage compliance under pediatric care standards.
Pediatric vaccination also generates consistent repeat demand through sibling vaccinations and follow on age cohort coverage each year. Repeat utilization remains predictable as new birth cohorts enter the eligible age range. Buyers favor vaccines supported by clear clinical guidance and post immunization monitoring protocols. Margin structure remains controlled under public health procurement and clinic administered pricing. Regulatory exposure remains elevated due to pediatric safety surveillance and consent standards. Import reliance persists across all pediatric supply. Substitution pressure from non-vaccine prevention methods remains present in low travel frequency households.
Demand for dengue vaccines in Japan reflects rising concern over imported infections, expanding outbound travel to Southeast Asia, and climate patterns that support mosquito survival. Japan records periodic locally acquired dengue cases linked to international movement and urban vector exposure. Public health authorities treat dengue as a preparedness priority due to absence of natural population immunity. Travel medicine clinics, airport health centers, and infectious disease hospitals support vaccination within defined risk groups. Physicians focus on pre travel counseling for business travelers, students, and long stay tourists. Demand aligns with infection prevention strategy and surveillance readiness rather than domestic outbreak volume alone.
Japan corporate assignments, student exchange programs, and tourism contribute to steady outbound movement toward dengue endemic regions. Travelers to Thailand, Indonesia, Vietnam, and the Philippines face year round exposure risk. Occupational health units in trading companies and manufacturing groups coordinate vaccination for employees deployed abroad. Universities guide students toward immunization before long academic stays. Medical tourism and volunteer programs also contribute to pre travel vaccination demand. This structured travel driven uptake anchors demand in predictable scheduling cycles tied to corporate transfer periods, academic semesters, and long holiday travel seasons rather than unexpected domestic transmission events nationwide.
Rising summer temperatures and high urban humidity support wider mosquito breeding activity across Japanese cities. Public memory of past local dengue transmission remains present within health departments and municipal disease control units. Surveillance programs track mosquito density near parks, rivers, and festival areas with heavy foot traffic. Vaccination demand gains support through local preparedness planning for workers in outdoor maintenance, emergency services, and large event operations. This prevention focused framing treats dengue as an intermittent domestic risk requiring advance control readiness. Demand grows through risk management logic tied to seasonal vector presence and urban exposure patterns.
Dengue vaccination in Japan remains confined by medical eligibility rules, cost exposure, and limited public familiarity with the disease. Many travelers avoid vaccination due to perceived low probability of infection during short trips. Out of pocket payment constrains uptake among students and budget travelers. Contraindications narrow the eligible population further under existing clinical guidance. Primary care physicians outside travel clinics often give lower priority to dengue education compared with influenza or hepatitis. These behavioral and structural limits keep demand focused within travel medicine channels and high awareness groups rather than spread evenly across the general adult population nationwide.
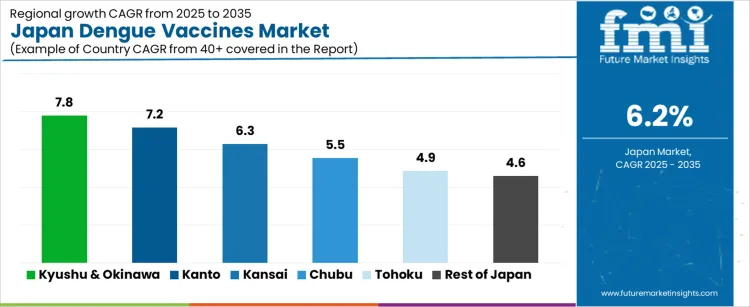
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 7.8% |
| Kanto | 7.2% |
| Kansai | 6.3% |
| Chubu | 5.5% |
| Tohoku | 4.9% |
| Rest of Japan | 4.6% |
The demand for dengue vaccines in Japan is rising steadily across all regions, led by Kyushu and Okinawa at a 7.8% CAGR. Growth in this region is supported by warmer climatic conditions, higher mosquito exposure risk, and stronger preventive vaccination programs linked to international travel and public health preparedness. Kanto follows at 7.2%, driven by dense urban populations, high outbound travel volumes, and early vaccine adoption through large hospital networks. Kansai records 6.3% growth, reflecting steady demand from travel clinics and infectious disease departments. Chubu at 5.5% shows moderate uptake linked to regional healthcare outreach and occupational travel screening. Tohoku and the Rest of Japan, at 4.9% and 4.6%, reflect slower growth shaped by lower exposure risk, fewer travel related cases, and more conservative preventive vaccination adoption patterns.
Demand for dengue vaccines in Kyushu and Okinawa is advancing at a CAGR of 7.8% through 2035, supported by subtropical climate exposure, rising mosquito activity, and growing public health surveillance programs. Local health authorities promote vaccination for travel related protection and outbreak preparedness. High tourist inflows also influence preventative immunization strategies among hospitality workers and port staff. Growth reflects steady public awareness campaigns, improved access through clinics and pharmacies, and broader integration of dengue prevention within regional infectious disease control planning.
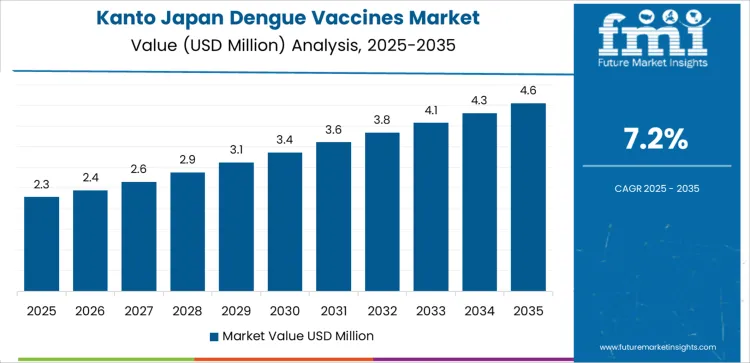
Demand for dengue vaccines in Kanto is rising at a CAGR of 7.2% through 2035, driven by high outbound travel activity, dense population mobility, and strong presence of international transport hubs. Vaccination demand remains tied to overseas travel advisories, business travel, and student mobility. Hospitals and travel clinics report steady pre travel immunization appointments. Growth reflects continuous global travel recovery, employer supported vaccination programs, and greater integration of tropical disease prevention within routine travel health services.
Demand for dengue vaccines in Kansai is progressing at a CAGR of 6.3% through 2035, supported by steady regional travel, rising awareness of mosquito borne infections, and routine adoption within infectious disease prevention programs. Kansai hospitals apply targeted vaccination for immunocompromised patients and overseas workers. Public education campaigns support seasonal uptake patterns. Growth reflects balanced healthcare access, stable travel activity, and gradual expansion of preventative vaccine offerings within regional medical institutions.
Demand for dengue vaccines in Chubu is advancing at a CAGR of 5.5% through 2035, supported by outbound leisure travel, growing medical tourism awareness, and structured disease prevention planning by regional health offices. Chubu residents show rising compliance with vaccination advisories linked to Southeast Asia travel routes. Clinics report steady annual appointment inflow for travel vaccines. Growth reflects moderate travel volume expansion, structured healthcare coordination, and steady public sector funding for infectious disease preparedness.
Demand for dengue vaccines in Tohoku is advancing at a CAGR of 4.9% through 2035, supported by outbound travel protection needs and controlled public health monitoring programs. Local infection risk remains low due to cooler climate conditions. Vaccination demand remains concentrated among international travelers, aid workers, and students studying abroad. Growth reflects gradual increases in global travel participation, steady compliance with travel medicine guidance, and controlled expansion of preventative vaccine stock across regional healthcare providers.
Demand for dengue vaccines in Rest of Japan is advancing at a CAGR of 4.6% through 2035, supported by selective use among outbound travelers, gradual inclusion in regional healthcare services, and steady education on global infectious disease risks. These areas show limited local transmission risk. Vaccination demand remains focused on individuals traveling to endemic regions. Growth remains steady and guided by travel patterns, public awareness programs, and gradual expansion of travel health services beyond major urban centers.
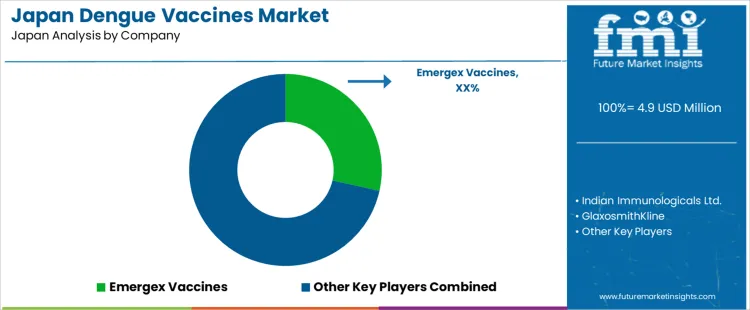
The demand for dengue vaccines in Japan is shaped by imported dengue cases linked to overseas travel, rising awareness among clinicians, and national infectious disease preparedness planning. GlaxoSmithKline and Merck influence the preventive vaccine landscape through global clinical development programs and Japanese regulatory engagement for mosquito borne diseases. Japanese health authorities focus demand on travel medicine clinics, port city hospitals, and public health research centers that monitor vector borne infection risk. Indian Immunologicals Ltd. participates through international supply programs referenced in global dengue control rather than routine domestic use. Emergex Vaccines and Codagenix remain positioned within research and early development pathways influencing long term vaccine preparedness strategy rather than near term clinical uptake in Japan.
Vaccine policy in Japan is governed by Ministry of Health surveillance data, disease notification trends, and risk assessment tied to inbound travel and climate driven mosquito expansion. Routine vaccination demand remains limited due to low domestic transmission. Strategic interest centers on outbreak readiness, stockpile access, and rapid deployment capability during regional surges in Asia Pacific. Buyer preference at the public health level favors vaccines with strong safety records, stable cold chain performance, and robust post vaccination monitoring systems. Demand visibility tracks outbound travel recovery, regional dengue activity across Southeast Asia, and national budget allocation for emerging infectious disease preparedness across prefectural public health institutes and quarantine stations.
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Type | Dengvaxia, Qdenga |
| Application | Paediatric Vaccination, Adult Travel Vaccination |
| End User | Direct Sales to Governments and International Organizations, Travel Medicine Clinics, Hospitals, NGOs |
| Regions Covered | Kyushu and Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Emergex Vaccines, Indian Immunologicals Ltd., GlaxoSmithKline, Merck, Codagenix |
| Additional Attributes | Dollar sales by product and age group, travel driven vaccination demand cycles, government and public health procurement patterns, repeat vaccination linked to overseas assignments and tourism flows, cold chain storage and distribution requirements, regulatory oversight for vaccine safety monitoring, outbreak preparedness stockpile planning tied to vector surveillance |
How big is the demand for dengue vaccines in Japan in 2025?
The demand for dengue vaccines in Japan is estimated to be valued at USD 4.9 million in 2025.
What will be the size of dengue vaccines in Japan in 2035?
The market size for the dengue vaccines in Japan is projected to reach USD 9.0 million by 2035.
How much will be the demand for dengue vaccines in Japan growth between 2025 and 2035?
The demand for dengue vaccines in Japan is expected to grow at a 6.2% CAGR between 2025 and 2035.
What are the key product types in the dengue vaccines in Japan?
The key product types in dengue vaccines in Japan are dengvaxia and qdenga.
Which age group segment is expected to contribute significant share in the dengue vaccines in Japan in 2025?
In terms of age group, adult segment is expected to command 35.0% share in the dengue vaccines in Japan in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.