Demand for Dengue Vaccines in USA
Demand for Dengue Vaccines in USA Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Demand for Dengue Vaccines in USA 2025 to 2035
Demand for dengue vaccines in the USA is valued at USD 31.0 million in 2025 and is projected to reach USD 66.2 million by 2035, reflecting a CAGR of 7.9%. Growth is driven by the expanding risk of dengue transmission linked to increased travel to endemic regions and climate-related spread of Aedes mosquitoes. Public health preparedness, vaccination for high-risk populations, and heightened awareness of severe dengue complications support stronger uptake.
Qdenga leads in product adoption due to its broader eligibility profile for both seropositive and seronegative individuals. Clinicians emphasize protection for travelers, military personnel, and residents in areas experiencing mosquito activity. Continued clinical research and post-authorization surveillance reinforce vaccination confidence and guide optimized immunization strategies.
The West, South, and Northeast regions show notable demand due to travel hubs, warmer seasonal conditions, and targeted vaccination initiatives for at-risk segments. Key suppliers include Sanofi Pasteur, GSK Vaccines, Merck & Co., Emergent BioSolutions, and Codagenix Inc. Their focus includes advancing next-generation formulations, scaling strategic distribution partnerships, and aligning vaccination access with evolving vector-borne disease management priorities in the USA.
Quick Stats for USA Dengue Vaccines Demand
- USA Dengue Vaccines Sales Value (2025): USD 31.0 million
- USA Dengue Vaccines Forecast Value (2035): USD 66.2 million
- USA Dengue Vaccines Forecast CAGR (2025-2035): 7.9%
- Leading Product in USA Dengue Vaccines Demand: Qdenga
- Key Growth Regions in USA Dengue Vaccines Demand: West USA, South USA, Northeast USA
- Top Players in USA Dengue Vaccines Demand: Sanofi Pasteur, GSK Vaccines, Merck & Co., Emergent BioSolutions, Codagenix Inc.
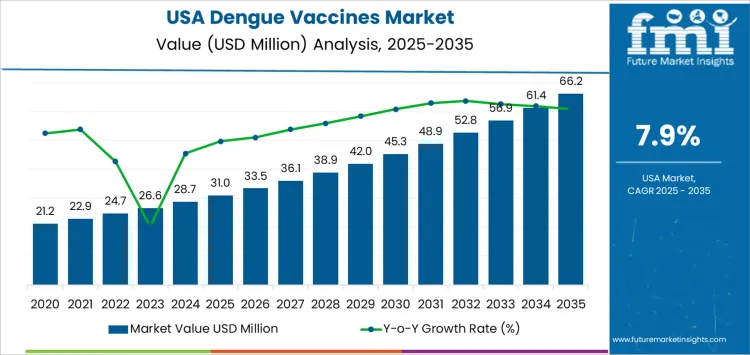
What is the Growth Forecast for Dengue Vaccines Industry in USA through 2035?
Demand for dengue vaccines in the United States contributes unevenly to growth across healthcare segments. The largest contribution comes from preventive public health programs that aim to reduce imported and climate-driven dengue cases in southern states. Rising traveler immunization needs support steady expansion in outpatient vaccination services. Pharmaceutical manufacturers contribute through new vaccine introductions supported by regulatory approvals and scaled production. Hospitals and infectious disease clinics generate incremental demand through risk-based immunization for vulnerable groups. Consumer awareness campaigns add moderate contribution by improving acceptance in regions with periodic mosquito-borne illness alerts.
The contribution from insurers remains limited because many vaccination decisions occur outside routine reimbursement pathways. Climate-sensitive regions in Florida, Texas, Arizona, and Hawaii create disproportionate contributions because vector presence elevates regional urgency. Research institutes also participate through procurement for surveillance and field studies. Growth is driven by multiple smaller contributors rather than a single dominant sector.
USA Dengue Vaccines Key Takeaways
| Metric | Value |
|---|---|
| USA Dengue Vaccines Sales Value (2025) | USD 31.0 million |
| USA Dengue Vaccines Forecast Value (2035) | USD 66.2 million |
| USA Dengue Vaccines Forecast CAGR (2025-2035) | 7.9% |
Why is the Demand for Dengue Vaccines in the USA Changing?
Demand for dengue vaccines in the USA is limited but potentially rising due to increased travel-associated cases and occasional local mosquito-borne transmission in territories such as Puerto Rico and regions with established Aedes mosquito populations. Public health concern over sporadic outbreaks drives interest among at-risk individuals and travelers seeking protection before visiting endemic zones or returning from abroad. Only one vaccine is approved for a narrow group of children with confirmed prior dengue infection living in endemic areas. Approval restrictions and the need for diagnostic confirmation before vaccination so far restrict broad uptake. That limits demand among the general population.
Development of newer vaccines in international markets draws attention from USA public health authorities and travel medicine providers, especially as global dengue incidence rises. If regulatory approval expands to include broader age groups or travelers, demand could grow substantially. Challenges include low incidence of dengue in most of the continental USA, which reduces perceived benefit and dampens large-scale demand. Regulatory and safety requirements related to prior infection screening add complexity. Many people rely on preventative measures such as mosquito control and bite prevention instead of vaccination.
Which Products, Age Groups, and End Users Lead Demand for Dengue Vaccines in the United States?
Demand for dengue vaccines in the United States reflects rising preventive focus for travelers and individuals in territories with mosquito-borne disease exposure. Distribution expands through medical facilities that serve outbound tourism, military personnel, and communities requiring international travel immunization. Vaccine selection depends on eligibility guidelines, safety profiles, and physician recommendations. Growth reflects education efforts around disease risks associated with global travel and climate-linked vector expansion.
By Product, Qdenga Holds the Larger Share
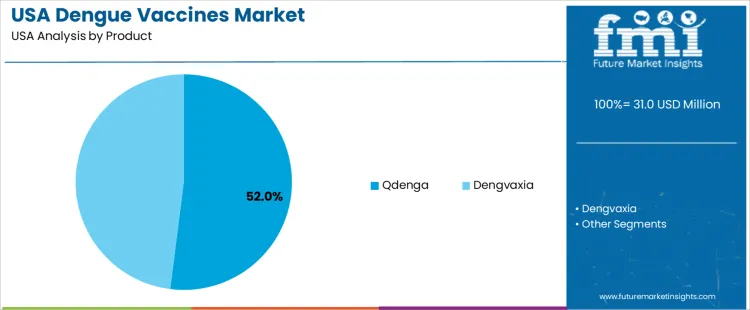
Qdenga represents 52.0%. Eligibility for broader population groups strengthens its vaccination footprint across USA travel medicine providers. Dengvaxia accounts for 48.0% and remains directed toward individuals with confirmed prior dengue exposure. Vaccine preferences reflect regulatory safety standards and screening requirements before administration. Procurement agreements with health systems and availability through specialized clinics support operational readiness for outbreak response. Inventory strategies emphasize preventive access for travel seasons and international deployments.
Key Points:
- Qdenga supports expanded coverage across eligible demographics
- Dengvaxia serves patients with documented infection history
- Availability influences rapid deployment for high-risk travelers
By Age Group, Paediatric Immunization Holds the Larger Share
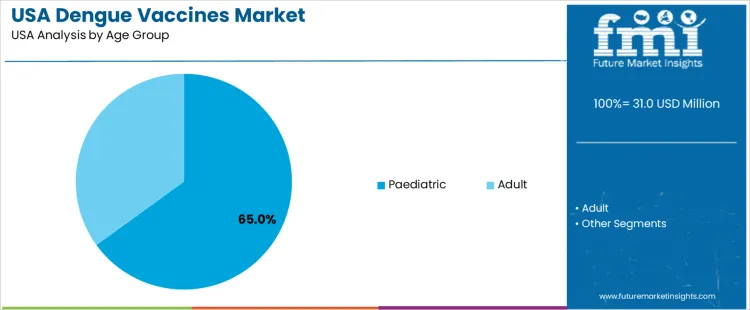
Paediatric vaccination accounts for 65.0%. Protection for adolescents engaging in travel to dengue-endemic regions drives this share. Adults represent 35.0%. Adult demand concentrates in frequent travelers, humanitarian workers, and government personnel. Usage patterns relate to physician guidance on exposure risks and adherence to immunization schedules. Engagement strategies target families planning overseas visits through specialized vaccination centers.
Key Points:
- Paediatric groups show highest uptake due to preventive healthcare focus
- Adult adoption aligns with frequent international travel
- Uptake depends on verified exposure history for specific vaccine products
By End User, Government-Linked Procurement Leads Demand
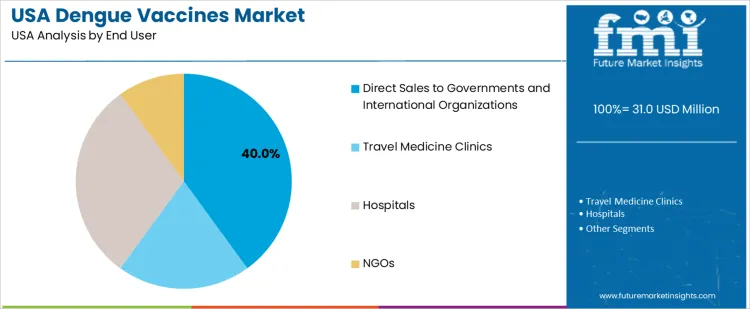
Direct sales to governments and international organizations hold 40.0%, supporting structured vaccination programs covering eligible populations. Hospitals account for 30.0%, integrating vaccines into travel health services. Travel medicine clinics represent 20.0%, serving outbound tourists requiring disease-specific protection. NGOs hold 10.0%, focusing on humanitarian deployments. Each channel operates with defined eligibility verification to ensure safe and compliant delivery.
Key Points:
- Government procurement supports stock availability and broader preparedness
- Hospitals maintain consistent access for clinical travel guidance
- NGOs and travel clinics support operational readiness for international movement
What are the Key Dynamics in the Dengue Vaccines Industry?
Rise in dengue incidence in USA territories, growing mosquito-borne disease awareness and increasing travel-linked importation support constrained vaccine demand.
In the United States, dengue remains endemic in territories such as Puerto Rico, the USA Virgin Islands and certain Pacific-affiliated states. Local outbreaks and occasional cases in parts of Florida, Texas, California and Hawaii underline ongoing regional risk. Public health authorities and clinicians in those regions, along with travelers returning from endemic areas, seek protection for children and adolescents to reduce severe dengue incidence. Vaccine availability supports targeted prevention efforts among previously infected individuals living in or visiting high-risk zones. These risk-related drivers generate a continuing though limited baseline demand for approved vaccines and interest in next-generation candidates.
Limited approval criteria, narrow indication scope and manufacturer withdrawal plans restrain overall market growth.
Currently only one vaccine is approved in the USA and its use is restricted to individuals aged 9–16 who have laboratory-confirmed prior dengue infection and live in endemic territories. That narrow indication excludes most of the continental USA population and many travelers, which limits uptake. The manufacturer has announced plans to discontinue production due to low global demand, creating supply uncertainty for future immunization campaigns. Additional vaccine candidates remain unapproved under USA regulatory oversight, which delays broader prophylactic coverage. These factors significantly constrain expansion toward a general-use dengue immunization market.
Renewed vaccine development efforts, increasing public health surveillance and growing interest in next-generation vaccines define key trends.
Global approval of newer vaccines and WHO prequalification generate pressure on USA regulators and public health agencies to revisit dengue prevention strategies. Institutions investing in expanded surveillance and diagnostic confirmation may increase identification of eligible individuals, which could revive demand if a broader-use vaccine becomes available. Research into improved vaccine candidates with safer profiles for seronegative individuals and more inclusive age coverage raises the possibility of expanded demand in future years. Heightened public awareness of vector-borne disease risks, climate-linked mosquito spread and travel-related cases may further support demand for safer, more comprehensive dengue vaccines across the USA.
Analysis of the Demand for Dengue Vaccines in the USA by Region
Demand for dengue vaccines in the United States continues to rise as public health agencies strengthen preparedness strategies for mosquito-borne diseases. Imported infection cases from international travel create continuous monitoring expectations in coastal and metropolitan regions. The market includes preventive immunization for high-risk travelers, residents in warm-climate zones, and individuals requiring protection during seasonal mosquito activity. Regulatory evaluation prioritizes safety data across different age groups. Digital vaccination-record systems support dose-completion tracking and inventory planning. West USA leads growth at 9.1% CAGR, followed by South USA at 8.1%, Northeast USA at 7.3%, and Midwest USA at 6.3%. Procurement strategies evaluate cold-chain logistics, risk-area mapping, and physician-training coverage.
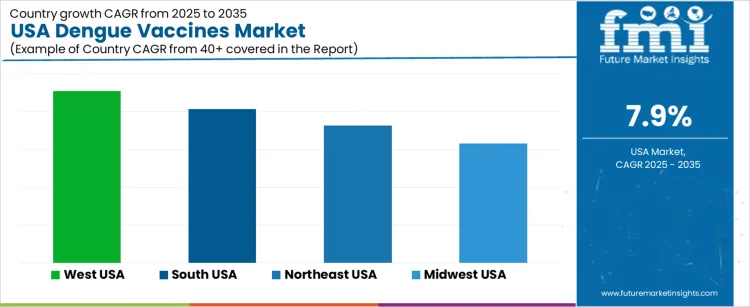
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 9.1% |
| South USA | 8.1% |
| Northeast USA | 7.3% |
| Midwest USA | 6.3% |
How is the West USA driving demand for dengue vaccines?
West USA records a 9.1% CAGR, supported by elevated travel volumes, outdoor recreation patterns, and expanding awareness of mosquito-borne disease risk. Travel clinics in California and Washington advise pre-travel immunization for individuals visiting endemic locations. Public health officials monitor warming temperatures that impact local mosquito populations and infection exposure. Insurance networks consider vaccination reimbursement based on preventive-care frameworks within primary care. Providers focus on accurate risk identification and scheduling efficiency for dose management. Logistics teams maintain tight handling standards to preserve vaccine-potency performance in regional supply routes.
- Travel-linked protection shaping vaccine consultations
- Climate-adjusted exposure monitoring guiding immunization plans
- Insurance-aligned reimbursement policies affecting access
- Cold-chain continuity supporting safety assurance
How is the South USA shaping demand?
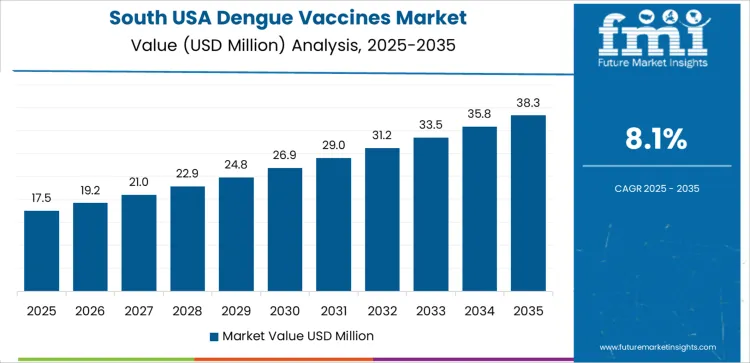
South USA grows at 8.1% CAGR, driven by tropical and subtropical environmental conditions in Texas, Florida, and Gulf Coast states. Persistent mosquito presence increases vaccination attention among residents with seasonal exposure concerns. Health departments communicate disease-prevention messaging through community networks. Travel and tourism segments support ongoing advisory programs for outbound visitors. Clinical administrators evaluate vaccine scheduling windows aligned with weather-driven risk patterns. Inventory availability must remain dependable across urban and rural healthcare access points.
- Seasonal mosquito presence guiding preventive adoption
- Public-health outreach improving vaccine awareness
- Travel activity influencing clinical recommendations
- Multi-location access improving dose-completion consistency
How is the Northeast USA influencing vaccination needs?
Northeast USA posts 7.3% CAGR, linked to densely populated hubs in New York, New Jersey, and Massachusetts with sizable international-travel flows. Providers emphasize pre-travel immunization for individuals visiting endemic zones. Professional organizations issue periodic guidance to maintain confidence in vaccine-protocol adherence. Regional demand strengthens around university health centers and employer-supported care programs. Procurement teams check data on temperature-control compliance in pharmacies and clinics.
- Travel-medicine services supporting vaccination uptake
- Academic and employer health programs enabling coverage
- Guidance publications assisting clinical decision support
- Cold-storage compliance shaping operational execution
How is the Midwest USA contributing to demand growth?
Midwest USA advances at 6.3% CAGR, reflecting measured but continuous adoption across states such as Illinois, Michigan, and Ohio. Demand originates from travel-risk consultations in primary-care facilities serving globally mobile populations. Distribution networks align stocking models with pre-travel seasonality. Healthcare providers educate patients about dose timing to ensure complete protection during international trips. Buyers focus on supply reliability and validated handling protocols consistent with national immunization standards.
- Travel-linked consultations maintaining baseline demand
- Seasonal forecasting improving inventory stability
- Patient-education efforts supporting dose compliance
- Standardized logistics reinforcing vaccination reliability
What is the competitive landscape of demand for dengue vaccines in the United States?
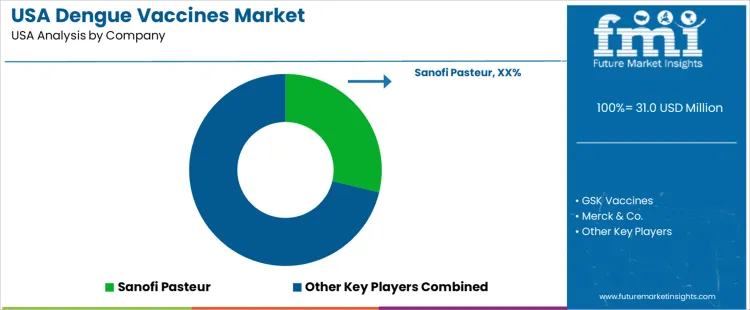
Demand for dengue vaccines in the United States reflects interest from healthcare systems located in regions with travel-related exposure risk. Public health agencies track dengue cases entering the country from tropical locations. Risk mitigation strategies include targeted immunization in specialized clinics serving travelers and military personnel deployed to dengue-endemic zones. Buyers assess immunogenic response data, dosing complexity, cold-chain suitability, and transparency in population-specific safety results. Procurement groups align decisions with CDC guidance and large-scale readiness planning led by federal vaccination programs.
Sanofi Pasteur holds an estimated 28.8% share. Its live attenuated vaccine supports structured delivery in pre-screened individuals with documented prior dengue infection. Specialized prescribing requirements create a narrow but stable deployment channel. GSK Vaccines conducts work on next-generation constructs under clinical evaluation. Merck & Co. invests in viral vector science used in global dengue studies with knowledge transfer applicable to USA readiness planning. Emergent BioSolutions supplies biological defense infrastructure capable of advanced vaccine scale-up and contract development. Codagenix Inc. progresses synthetic biology candidates designed to fit streamlined dosing and rapid production objectives.
Competitive positioning depends on validated clinical data across diverse age groups, inventory stability for travel immunization centers, and preparedness alignment with national biodefense planning. Engagement with regulatory bodies supports ongoing requirements for USA deployment of dengue vaccination solutions.
Key Players in USA Dengue Vaccines Demand
- Sanofi Pasteur
- GSK Vaccines
- Merck & Co.
- Emergent BioSolutions
- Codagenix Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product | Qdenga, Dengvaxia |
| Age Group | Paediatric, Adult |
| End User | Direct Sales to Governments and International Organizations, Travel Medicine Clinics, Hospitals, NGOs |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Sanofi Pasteur, GSK Vaccines, Merck & Co., Emergent BioSolutions, Codagenix Inc. |
| Additional Attributes | Dollar sales by vaccine product type, target patient age groups, and end-use channels; regional adoption influenced by travel-related vaccination demand and mosquito-borne disease risk in southern states; competitive landscape including live-attenuated and next-generation vaccine developers; regulatory environment and CDC guidance on dengue prevention; purchasing patterns through government programs, hospitals, and travel medicine providers. |
USA Dengue Vaccines Demand by Segments
Product:
- Qdenga
- Dengvaxia
Age Group:
- Paediatric
- Adult
End User:
- Direct Sales to Governments and International Organizations
- Travel Medicine Clinics
- Hospitals
- NGOs
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Frequently Asked Questions
How big is the demand for dengue vaccines in USA in 2025?
The demand for dengue vaccines in USA is estimated to be valued at USD 31.0 million in 2025.
What will be the size of dengue vaccines in USA in 2035?
The market size for the dengue vaccines in USA is projected to reach USD 66.2 million by 2035.
How much will be the demand for dengue vaccines in USA growth between 2025 and 2035?
The demand for dengue vaccines in USA is expected to grow at a 7.9% CAGR between 2025 and 2035.
What are the key product types in the dengue vaccines in USA?
The key product types in dengue vaccines in USA are qdenga and dengvaxia.
Which age group segment is expected to contribute significant share in the dengue vaccines in USA in 2025?
In terms of age group, paediatric segment is expected to command 65.0% share in the dengue vaccines in USA in 2025.
Table of Content
- Executive Summary
- USA Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- USA Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- USA Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2025 to 2035
- Qdenga
- Dengvaxia
- Y to o to Y Growth Trend Analysis By Product , 2020 to 2024
- Absolute $ Opportunity Analysis By Product , 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Age Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Age Group, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Age Group, 2025 to 2035
- Paediatric
- Adult
- Y to o to Y Growth Trend Analysis By Age Group, 2020 to 2024
- Absolute $ Opportunity Analysis By Age Group, 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Direct Sales to Governments and International Organizations
- Travel Medicine Clinics
- Hospitals
- NGOs
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Age Group
- By End User
- Competition Analysis
- Competition Deep Dive
- Sanofi Pasteur
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- GSK Vaccines
- Merck & Co.
- Emergent BioSolutions
- Codagenix Inc.
- Sanofi Pasteur
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: USA Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: USA Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 3: USA Market Value (USD Million) Forecast by Age Group, 2020 to 2035
- Table 4: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 5: USA Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: USA Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 7: USA Market Value (USD Million) Forecast by Age Group, 2020 to 2035
- Table 8: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: USA Market Pricing Analysis
- Figure 2: USA Market Value (USD Million) Forecast 2020-2035
- Figure 3: USA Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 4: USA Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 5: USA Market Attractiveness Analysis by Product
- Figure 6: USA Market Value Share and BPS Analysis by Age Group, 2025 and 2035
- Figure 7: USA Market Y-o-Y Growth Comparison by Age Group, 2025-2035
- Figure 8: USA Market Attractiveness Analysis by Age Group
- Figure 9: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 11: USA Market Attractiveness Analysis by End User
- Figure 12: USA Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: USA Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 14: USA Market Attractiveness Analysis by Region
- Figure 15: USA Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: USA Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: USA Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 18: USA Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 19: USA Market Attractiveness Analysis by Product
- Figure 20: USA Market Value Share and BPS Analysis by Age Group, 2025 and 2035
- Figure 21: USA Market Y-o-Y Growth Comparison by Age Group, 2025-2035
- Figure 22: USA Market Attractiveness Analysis by Age Group
- Figure 23: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 24: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 25: USA Market Attractiveness Analysis by End User
- Figure 26: USA Market - Tier Structure Analysis
- Figure 27: USA Market - Company Share Analysis

