Jellyfish-Derived Peptide Ingredient Concepts Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The jellyfish-derived peptide ingredient concepts market is segmented by Ingredient Type (Collagen peptides, Hydrolysates, Peptide chelates, Peptide complexes), Application (Nutricosmetics, Supplements, Topical care, Biomaterials), Form (Powder, Liquid, Gel, Encapsulated), Source Species (Cannonball, Rhopilema, Nomura, Aurelia), End Use (Dietary supplements, Skincare brands, Ingredient houses, Research labs), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Jellyfish-Derived Peptide Ingredient Concepts Market Size, Market Forecast, and Outlook By FMI
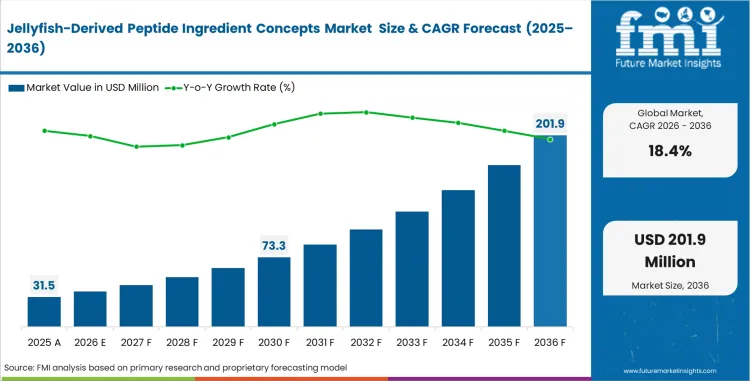
The jellyfish-derived peptide ingredient concepts market was valued at USD 26.6 million in 2025. Demand is projected to reach USD 31.5 million in 2026 and USD 170.5 million by 2036, expanding at a CAGR of 18.4% during the forecast period. Growth is being supported by early interest in marine-derived peptide ingredients for beauty, wellness, and biomaterial applications.
Summary of Jellyfish-Derived Peptide Ingredient Concepts Market
- The market is forecast to reach USD 170.5 million by 2036.
- The market is expected to grow at a CAGR of 18.4% from 2026 to 2036.
- The market was estimated at USD 26.6 million in 2025.
- The forecast period represents an incremental opportunity of USD 139.0 million.
- The market remains a concept-stage marine bioactive ingredient segment focused on collagen peptides, hydrolysates, and related peptide formats rather than large-scale commodity trade.
- Demand is rising due to abundant but underutilized jellyfish biomass and increasing research on collagen and peptide applications across beauty, supplements, and biomaterials.
- Skincare and nutricosmetic applications are gaining the strongest traction, supported by emerging anti-aging and skin-health research.
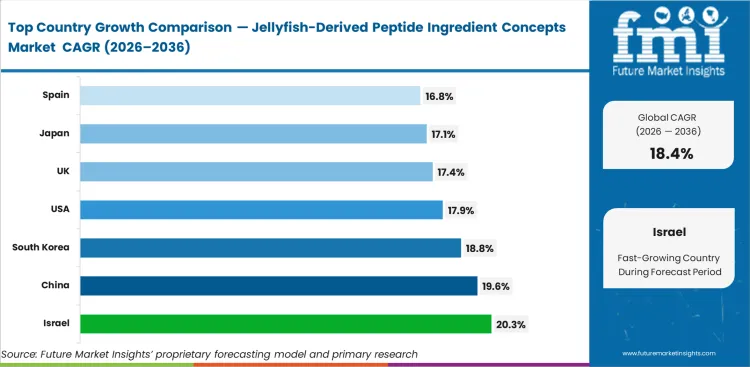
- Israel leads projected growth at 20.3% CAGR, followed by China, South Korea, the United States, the United Kingdom, Japan, and Spain.
- Collagen peptides dominate the ingredient type segment with a 46.0% share, reflecting current commercial focus on hydrolyzed peptide fractions.
- Nutricosmetics lead the application segment with a 34.0% share, driven by ingestible beauty trends.
- Powder form holds a 52.0% share due to advantages in stability, transport, and formulation flexibility.
- Cannonball jellyfish accounts for a 38.0% share of the source species segment, supported by commercialization and research activity.
- Dietary supplements represent the leading end-use segment with a 31.0% share.
- Key companies include Certified Nutraceuticals, JellyCoUSA, Jellagen, Qortein, Creative Enzymes, Baoding Faithful Industry, and Chemipan Corporation.
- The market remains fragmented due to limited commercialization, regulatory uncertainty, and reliance on specialized extraction and formulation capabilities.
Commercial interest in marine collagen alternatives is rising, but supplier depth remains limited across early-stage jellyfish-derived inputs. Jellyfish biomass is increasingly being evaluated as a distinct input rather than being grouped within broader marine ingredient categories. Brands are looking at species-specific mesoglea extracts to build more differentiated peptide concepts for nutricosmetics and related uses. At the same time, raw biomass variability remains a real constraint, which keeps species access, harvest consistency, and processing control central to scale-up.
Broader functional-food adoption is likely to remain limited until clearer safety and approval pathways emerge for specific mesoglea-derived ingredients. Progress depends on more reliable sourcing models and tighter consistency across extraction and characterization. Until then, most activity is likely to remain concentrated in pilot-scale, specialty, or research-led applications rather than broad commercial runs.
Israel is expected to record a 20.3% CAGR from 2026 to 2036 as startup activity supports early commercial development. China is anticipated to register a 19.6% CAGR, supported by established edible jellyfish processing capacity that can shorten ingredient conversion cycles. South Korea is set to post an 18.8% CAGR, backed by beauty-sector demand for new marine peptide formats. The United States is projected to witness a 17.9% CAGR as preventive health formulations widen ingredient adoption. The United Kingdom is likely to rise at a 17.4% CAGR, supported by biomaterial development programs. Japan is estimated to expand at a 17.1% CAGR as functional beverage use supports commercial uptake. Spain is poised to register a 16.8% CAGR, with Mediterranean raw material access supporting supply despite novel-food approval constraints.
Segmental Analysis
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by Ingredient Type
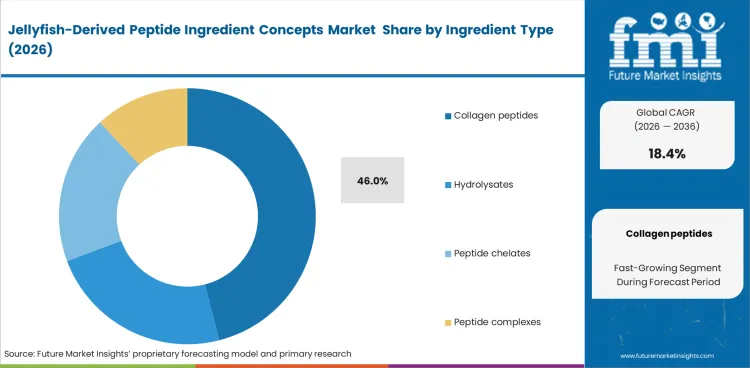
Structural similarity to human collagen keeps commercial interest concentrated in peptide formats rather than less refined jellyfish protein fractions. Collagen peptides are expected to hold 46.0% share, as manufacturers continue to favor formats that fit more easily into supplement, skincare, and biomaterial development pipelines. Demand remains strongest where bioactive peptide positioning supports higher-value formulation claims and clearer differentiation from conventional marine proteins. Processing compatibility also supports this position, since hydrolyzed peptide formats are easier to standardize for solubility and formulation consistency than more complex structural extracts. Low extraction yields still restrict the availability of bulk jellyfish collagen peptides, keeping supply tight and preserving premium pricing across early-stage commercial use. This combination of formulation fit and constrained scalable output is likely to keep collagen peptides ahead of other ingredient types in 2026.
- Initial screening: Jellyfish peptide molecular weight distribution remains a key checkpoint because bioavailability, solubility, and formulation fit all depend on it. Wide yield variation can weaken batch consistency and reduce early project viability.
- Pilot validation: Standard enzymatic cleavage methods often require adjustment to suit mesoglea composition and processing behavior. Poor adaptation at this stage can raise material loss and weaken pilot economics.
- Commercial expansion: Multi-year extraction agreements help manufacturers reduce input volatility and improve supply visibility. Limited access to stable processing capacity can still slow scale-up for later entrants.
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by Application
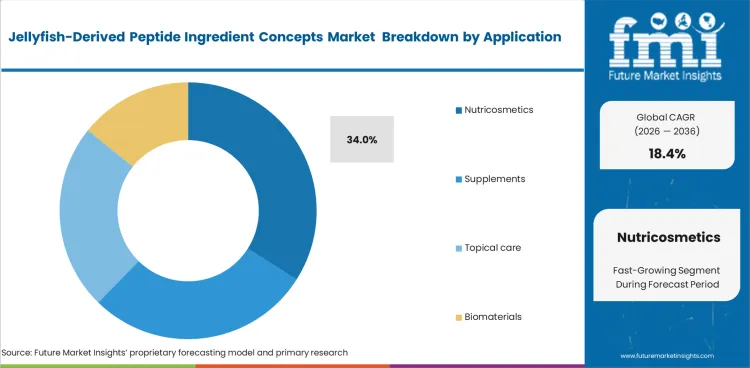
Commercial traction is strongest in ingestible beauty because marine novelty supports premium positioning more effectively in supplements than in topical formats. Consumer acceptance is also easier to build where nutraceutical ingredients can be linked to routine oral use rather than uncertain transdermal performance. Nutricosmetics are anticipated to capture 34.0% share of the application segment in 2026, as manufacturers use nutricosmetic peptide ingredients to separate new launches from crowded fish-based collagen lines. Pressure on cosmeceutical collagen actives remains higher because efficacy claims often move ahead of strong delivery validation. Premium pricing potential stays attractive, but only where bioactivity, stability, and formulation credibility hold together in a commercially usable format.
- Procurement savings: Bulk mesoglea purchases during peak bloom periods can reduce input cost at the sourcing stage. Storage, preservation, and spoilage risk can narrow that advantage quickly.
- Hidden processing: Specialized filtration remains important where contaminant control and heavy metal reduction affect ingredient acceptance. Added equipment needs can weaken early margin assumptions.
- Lifecycle comparison: Total formulation cost must stay aligned with premium retail positioning for the concept to remain viable. Products that fail to justify higher shelf pricing often face early discontinuation.
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by Form
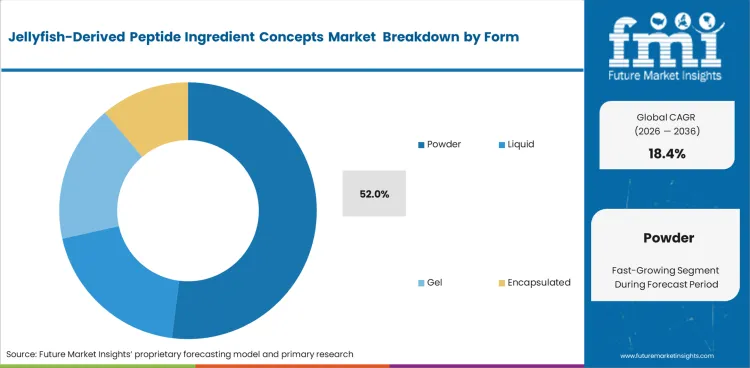
Dry formats remain commercially favored because shelf stability, transport efficiency, and handling control matter more than presentation variety at this stage of market development. International distribution is also easier when dry marine collagen-based products move through supply chains with lower microbial exposure and fewer storage constraints than liquid alternatives. Format preference is further supported by jellyfish peptide ingredient formulation needs, since dry inputs are easier to standardize across blends, premixes, and controlled-dose systems. Consistency in moisture control also matters for preserving jellyfish collagen peptide bioavailability, particularly where stability loss can weaken product performance and shorten usable shelf life. Powder is projected to secure 52.0% share, as manufacturers continue to favor formats that balance storage life, formulation flexibility, and distribution practicality.
- Agglomeration prevention: Customized dehumidification helps maintain powder flowability during filling and blending. Weak atmospheric control can interrupt production and reduce batch consistency.
- Residual moisture: Batch testing remains critical because small moisture shifts can accelerate degradation and shorten usable shelf life. Tight control is needed to avoid rejection risk.
- Full utilization: Blender configuration affects residual powder loss, line efficiency, and unit cost control. Poor equipment fit can reduce usable output from each batch.
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by Source Species
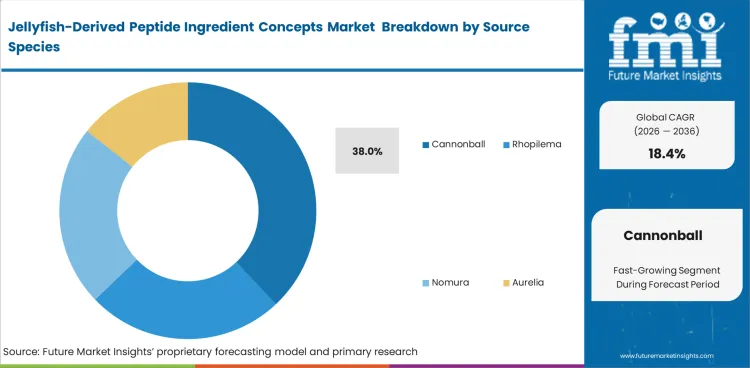
Commercial supply remains concentrated in species with established harvest access and workable processing familiarity. Predictable landing patterns and existing fishery infrastructure still matter because peptide supplements production depends on steady biomass input and a dependable jellyfish peptide raw material partner rather than irregular bloom abundance alone. Cannonball is projected to secure 38.0% share of the source species segment in 2026, supported by established harvesting networks across the Americas and stronger near-term processing readiness. Seasonal variability still limits supply reliability, making species diversification important where manufacturers need better input continuity. This keeps Cannonball in a leading position, even though long-term scale remains exposed to harvest volatility.
- Primary extractors: Regional fishery groups often control early access to raw biomass through local harvesting rights and established landing networks. Buyers usually need direct relationships with local operators to secure material.
- Volume constraints: Short harvest windows can flood processing sites with more biomass than facilities can handle efficiently. Weak handling capacity can raise spoilage and reduce usable extraction yield.
- Long-term landscape: Controlled aquaculture is likely to gain attention as manufacturers look for better supply stability. Wild harvesting alone may not support a larger commercial volume over time.
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by End Use
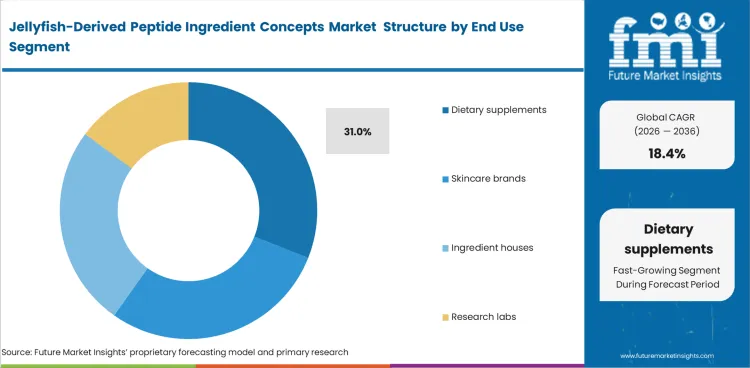
Commercial adoption is moving first through dietary supplements because oral formats offer a clearer route to dosage control and repeat-use positioning. Demand also fits well with marine nutraceutical ingredients that still need stronger standardization before broader scale-up. Dietary supplements are anticipated to account for 31.0% share of the end-use segment in 2026, as manufacturers continue to position these inputs within beauty, joint health, and wellness-focused collagen supplements. Wider expansion still depends on tighter control over peptide profile and dosing consistency at commercial scale.
- Early formulators: Niche nutricosmetic brands are likely to adopt first because premium positioning can absorb higher early-stage R&D costs.
- Scale operators: Mainstream supplement manufacturers are likely to follow only after extraction yields improve, since lower price points require stronger cost efficiency.
- Late converts: Traditional pharmaceutical companies are likely to wait for broader clinical standardization, with regulatory certainty remaining the main requirement.
Jellyfish-Derived Peptide Ingredient Concepts Market Drivers, Restraints, and Opportunities
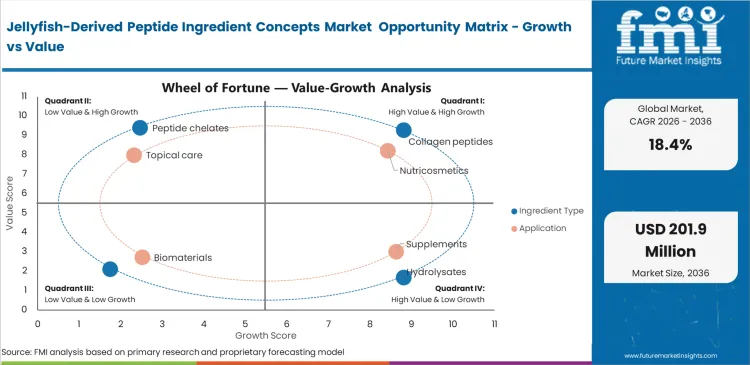
Interest in non-mammalian structural proteins is supporting evaluation of jellyfish-derived peptide concepts, especially in biomaterial and wound-care research. For developers, the main attraction is differentiation: marine-derived inputs may offer an alternative route where bovine or porcine sourcing creates safety, positioning, or regulatory questions. Even so, this remains an early-stage opportunity rather than a scaled commercial market.
Fragmented harvesting infrastructure remains a real restraint because raw biomass availability is still tied to bloom patterns, handling conditions, and site-level processing readiness. That makes annual forecasting difficult and leaves brands exposed to yield variation. In practical terms, supply inconsistency continues to slow commercial adoption even where buyer interest is rising.
Opportunities in the Jellyfish-Derived Peptide Ingredient Concepts Market
- Clinical trial partnerships: Co-funding efficacy studies with specialized ingredient developers could help brands secure earlier access to more standardized extract formats.
- Decentralized processing: Small extraction facilities located closer to harvest zones could help reduce degradation risk and improve raw material handling.
- Aquaculture development: Controlled cultivation could improve supply stability over the long term, although this remains a forward-looking opportunity rather than a near-term commercial norm.
Regional Analysis
Based on regional analysis, the jellyfish-derived peptide ingredient concepts market is segmented into North America, Europe, East Asia, and Middle East and Africa, with country-level discussion focused on the key markets covered in this report.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Israel | 20.3% |
| China | 19.6% |
| South Korea | 18.8% |
| United States | 17.9% |
| United Kingdom | 17.4% |
| Japan | 17.1% |
| Spain | 16.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Middle East and Africa Jellyfish-Derived Peptide Ingredient Concepts Market Analysis
Israel defines the region’s commercial profile because development is still concentrated in high-value peptide and biomaterial applications rather than in bulk ingredient volume. Limited large-scale marine processing capacity keeps the regional market narrow, while stronger technical emphasis on purity, extraction control, and specialty use supports early adoption. Commercial progress across Middle East and Africa remains more innovation-led than supply-led. Regional growth is likely to depend on whether pilot-scale work can move into repeatable manufacturing without losing quality consistency.
- Israel: Technical development remains concentrated in specialized peptide and biomaterial pathways where higher-value use cases can absorb early processing cost. Demand for jellyfish-derived peptide ingredient concepts in Israel is expected to rise at a CAGR of 20.3% from 2026 to 2036, supported by stronger innovation activity and a clearer focus on specialty applications. This keeps Israel ahead within the region, especially where commercial viability depends more on formulation precision than on bulk supply scale.
FMI's report includes South Africa and GCC countries. Early laboratory interest in validating local species points to longer-term commercialization potential, even though scalable infrastructure remains limited today. South Africa adds relevance through marine biodiversity and research potential, while GCC markets could influence future demand through premium wellness and nutraceutical consumption.
East Asia Jellyfish-Derived Peptide Ingredient Concepts Market Analysis
East Asia remains the most commercially prepared regional base for jellyfish-derived peptide ingredient concepts because edible-biomass processing familiarity already exists across parts of the marine value chain. Existing handling infrastructure, stronger ingredient acceptance, and established fish collagen and functional nutrition markets reduce some of the early conversion friction seen elsewhere. Regional momentum is also supported by faster adaptation of marine processing assets into higher-value extraction work. Commercial progress in East Asia is likely to stay ahead where processing readiness and application fit move together.
- China: Existing edible-jellyfish processing capacity gives China a practical advantage in moving biomass toward higher-value peptide extraction. Demand for jellyfish-derived peptide ingredient concepts is projected to expand at a 19.6% CAGR from 2026 to 2036, supported by processing scale, lower conversion cost, and stronger manufacturing readiness. This keeps China central to regional supply development as commercial volumes begin to build.
- South Korea: Premium beauty and ingestible wellness positioning give South Korea a stronger route for early product uptake than many other markets. Sales of jellyfish-derived peptide ingredient concepts in South Korea are likely to advance at a CAGR of 18.8%. The faster formulation cycles and cleaner-label demand support new marine ingredient adoption. Commercial relevance remains highest where differentiated beauty concepts can justify higher retail pricing.
- Japan: Functional beverage and healthy aging applications continue to shape market logic more than rapid mass-market rollout. Stronger validation discipline and tighter product acceptance standards also keep commercialization more selective than in faster-moving regional markets. Demand for jellyfish-derived peptide ingredient concepts in Japan is anticipated to rise at a 17.1% CAGR from 2026 to 2036. This supports a more controlled adoption path where product credibility and formulation acceptance matter as much as launch speed.
FMI's report includes Taiwan. Local contract manufacturers already show workable capability in handling specialized marine powders, supporting future regional brand partnerships. Taiwan also remains relevant through its strength in contract manufacturing and export-oriented nutraceutical production.
North America Jellyfish-Derived Peptide Ingredient Concepts Market Analysis
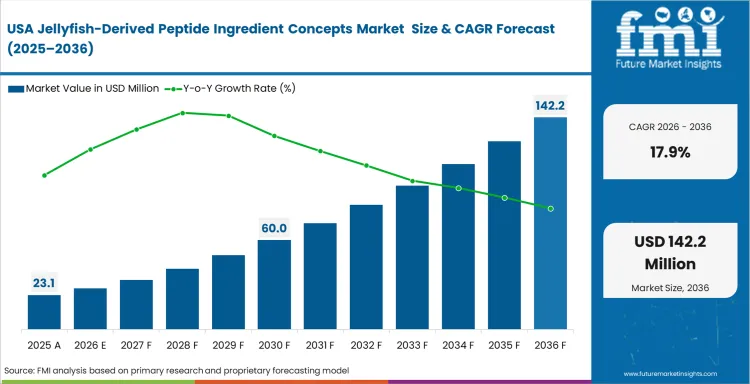
Preventive health and active-aging demand define the regional opportunity more clearly than broad marine ingredient substitution. Commercial interest remains concentrated in supplement and wellness applications where dosage control, origin differentiation, and formulation standardization carry more weight. Regulatory scrutiny also keeps product development disciplined, especially where manufacturers need cleaner documentation around processing, safety, and consistency. North America remains relevant as a premium-positioned market rather than a volume-led supply base.
- United States: Domestic sourcing matters because localized biomass access can reduce dependence on longer international supply chains and improve raw material visibility. Demand for jellyfish-derived peptide ingredient concepts in the United States is expected to rise at a CAGR of 17.9% from 2026 to 2036, supported by premium supplement positioning and stronger interest in differentiated marine bioactives. Commercial progress is likely to depend on how far domestic extraction and standardization capabilities can improve over the forecast period.
FMI's report includes Canada. Regional regulatory bodies continue to assess novel marine ingredient safety profiles, shaping the early framework for future functional food approvals. Canada also adds relevance through its strong supplement market and consumer acceptance of wellness-led marine ingredients.
Europe Jellyfish-Derived Peptide Ingredient Concepts Market Analysis
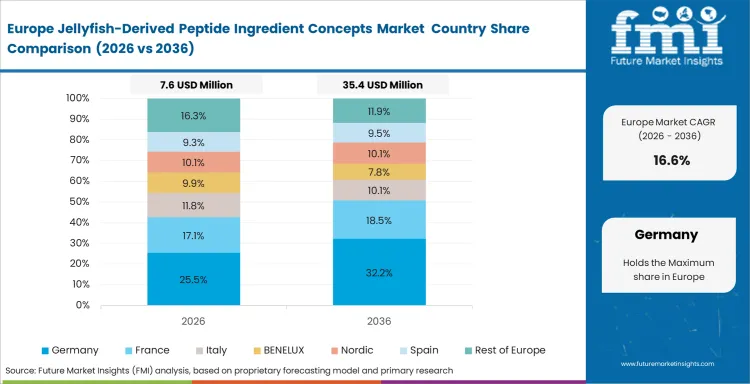
Regulatory discipline shapes the regional market more than raw material access or early consumer interest. Novel-food approval requirements keep ingestible expansion slower across Europe, while biomedical and topical pathways remain more workable for early commercialization. Mediterranean biomass availability supports technical development, but adoption still depends on species-specific safety characterization and tighter documentation standards. Europe mains commercially relevant where validation quality and regulatory navigation matter more than rapid volume buildout.
- United Kingdom: Biomedical development continues to shape demand more strongly than mainstream nutrition use. Demand for jellyfish-derived peptide ingredient concepts in the United Kingdom is likely to advance at a 17.4% CAGR from 2026 to 2036, supported by a stronger emphasis on medical-grade extracts and application-led validation. This keeps the country important, where commercialization depends on technical credibility and controlled use environments.
- Spain: Mediterranean biomass access gives Spain a practical supply advantage, but ingestible adoption still moves more slowly under tighter approval conditions. Near-term activity remains more aligned with topical and adjacent non-ingestible uses, while demand for jellyfish-derived peptide ingredient concepts in Spain is anticipated to rise at a 16.8% CAGR from 2026 to 2036. Regulatory progress will remain central to whether Spain expands beyond a supply-linked opportunity into a broader commercial market.
FMI's report includes France, Germany, and Italy. Formulators in these countries actively monitor authorization progress, preparing rapid product launches once legal barriers fall. The Netherlands is likely to influence the sector as well, supported by its strong nutraceutical distribution base and established role in specialty ingredient commercialization across Europe.
Competitive Aligners for Market Players
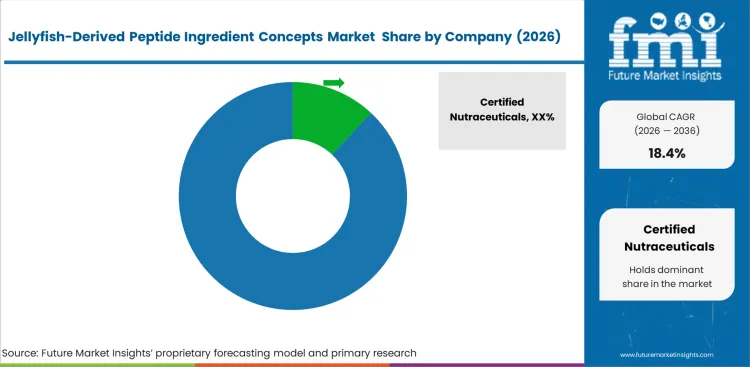
Competitive position depends more on extraction quality than on raw biomass access alone. Jellagen and Certified Nutraceuticals stand apart through tighter control over enzymatic processing methods that preserve structural integrity during refinement. Supplier selection in this market is shaped less by headline pricing and more by stability, solubility, and repeatable bioactivity across finished peptide output, which raises the bar for any jellyfish peptide ingredient supplier. A jellyfish collagen peptide manufacturer without this level of processing control often struggles to produce hydrolysates with the consistency needed for premium supplement, skincare, or biomaterial use. Keeping technical refinement capability central to value realization reduces the role of undifferentiated supply in higher-margin applications.
Early participants also benefit from a stronger documentation base built around safety, characterization, and application testing. Existing dossiers and validation data can shorten qualification cycles materially, especially where product claims and regulatory acceptance remain closely linked. Newer entrants face a slower path because safety substantiation still takes time, cost, and usable technical evidence before premium programs for bioactive marine peptides move forward. This raises the entry barrier for any peptide complexes provider and can delay progress from initial inquiry to a serious jellyfish collagen ingredient quote.
Large cosmetic and supplement manufacturers also try to avoid overdependence on a single processor, particularly where harvest variability and site-level disruption can affect supply continuity. Dual-sourcing interest is rising for that reason, even though specialized ingredient houses still need volume visibility to justify capacity expansion. Competitive advantage increasingly rests with processors that can maintain comparable bioactive profiles across more than one sourcing or processing location. That capability improves supply confidence and makes those suppliers more relevant in premium skincare and supplement programs.
Key Players in Jellyfish-Derived Peptide Ingredient Concepts Market
- Certified Nutraceuticals
- JellyCoUSA
- Jellagen
- Qortein
- Creative Enzymes
- Baoding Faithful Industry
- Chemipan Corporation
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 31.5 Million to USD 170.5 Million, at a CAGR of 18.4% |
| Market Definition | Jellyfish-derived peptide ingredient concepts cover enzymatically isolated protein fractions sourced from medusozoan biomass. Commercial applications require purified components demonstrating specific functional, cosmetic, or biomedical utility. |
| Segmentation | Ingredient Type, Application, Form, Source Species, End Use, Region |
| Regions Covered | North America, Europe, East Asia, Middle East and Africa |
| Countries Covered | United States, United Kingdom, Spain, China, Japan, South Korea, Israel |
| Key Companies Profiled | Certified Nutraceuticals, JellyCoUSA, Jellagen, Qortein, Creative Enzymes, Baoding Faithful Industry, Chemipan Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Biomass harvest volumes converted through known enzymatic yield ratios. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Jellyfish-Derived Peptide Ingredient Concepts Market Analysis by Segments
Ingredient Type
- Collagen peptides
- Hydrolysates
- Peptide chelates
- Peptide complexes
Application
- Nutricosmetics
- Supplements
- Topical care
- Biomaterials
Form
- Powder
- Liquid
- Gel
- Encapsulated
Source Species
- Cannonball
- Rhopilema
- Nomura
- Aurelia
End Use
- Dietary supplements
- Skincare brands
- Ingredient houses
- Research labs
Region
- North America
- United States
- Europe
- United Kingdom
- Spain
- East Asia
- China
- Japan
- South Korea
- Middle East and Africa
- Israel
Bibliography
- Food and Agriculture Organization of the United Nations. (2024). The State of World Fisheries and Aquaculture 2024: Blue Transformation in action. FAO.
- European Commission. (2024). Summary of applications and notifications. Directorate-General for Health and Food Safety.
- European Commission Maritime Forum. (2024, July 18). Discover food4future - An innovative project unlocking the potential of macroalgae as a sustainable food source. European Commission.
- Gao, J., Ning, C., Wang, M., Wei, M., Ren, Y., & Li, W. (2024). Structural, antioxidant activity, and stability studies of jellyfish collagen peptide-calcium chelates. Food Chemistry X, 23, 101706.
- Hu, B., Zong, Z., Han, L., Cao, J., Yang, J., Zheng, Q., Zhang, X., Liu, Y., & Yao, Z. (2025). Jellyfish collagen: A promising and sustainable marine biomaterial with emerging applications in food, cosmetics, and biomedical-A review. Applied Food Research, 5, 101165.
- Yu, J., Xing, S., Sun, R., Wang, L., Yang, X., & Zeng, M. (2025). Jellyfish collagen peptides: Anti-aging mechanism and potential application in skincare. Journal of Functional Foods, 134, 107070.
- Villalba-Urquidy, B. del S., Torres-Arreola, W., Medina, I., Hernández-Aguirre, L. E., Chan-Higuera, J. E., & Ezquerra-Brauer, J. M. (2025). Bioactivity and in silico insights of collagen-derived peptides from jellyfish (Stomolophus sp. 2) mesoglea. Marine Drugs, 23(11), 427.
- Pesterau, A.-M., Popescu, G., Lupu, D., Turcanu, A.-A., Radu, G.-L., Dobre, T., & Oancea, F. (2025). Marine jellyfish collagen and other bioactive natural compounds from the sea, with significant potential for wound healing and repair materials. Marine Drugs, 23(6), 252.
- Karunarathne, A. D., & Cumaranatunga, P. R. T. (2025). Jellyfish fisheries in southern Asia. Aquatic Living Resources, 38, 5.
- European Food Safety Authority. (2024). Novel Foods Authorisation Database. EFSA.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Supply chain friction surrounding unpredictable seasonal medusozoan biomass blooms.
- Formulation shifts replacing bovine materials with marine Type 0 structures.
- Commercialization gaps between pilot extraction facilities and full-scale manufacturing.
- Extreme hygroscopic behavior inherent in advanced marine peptide powders.
- Stringent novel-food authorization hurdles delaying European market entry.
- Concentration of venture capital funding within Israeli biomedical startups.
- Decentralization requirements for securing viable long-term extract sourcing.
- Stability testing protocols needed for precise clinical batch standardization.
Frequently Asked Questions
How big could the jellyfish-derived peptide ingredient concepts market become by 2036?
Revenue is projected to reach USD 170.5 million by 2036, supported by early commercial interest in marine-derived peptide ingredients for beauty, wellness, and related applications.
What are jellyfish-derived peptides used for?
These extracts are being explored mainly in nutricosmetics, supplements, skincare, and selected biomaterial applications. The commercial appeal comes from novelty, marine sourcing, and possible functional differentiation rather than from broad mainstream use at this stage.
Are jellyfish collagen peptides safe for supplements?
Safety depends on species-specific characterization, processing quality, and regulatory clearance. For ingestible use, suppliers still need stronger safety documentation, including contaminant control and heavy-metal testing.
How are jellyfish peptides extracted?
They are typically extracted through cleaning, hydrolysis, filtration, and fractionation steps designed to produce usable peptide profiles from jellyfish biomass.
Do jellyfish peptides work in skincare?
Early research points to possible skincare relevance, but commercial use still depends on stronger validation around efficacy, stability, and formulation performance.
Which companies are actively commercializing jellyfish collagen peptides?
Companies such as Certified Nutraceuticals, Jellagen, and JellyCoUSA are among the more visible participants in this space.
Is jellyfish collagen a real alternative to fish collagen?
It may offer a differentiated alternative in selected formulations, but the commercial comparison with fish collagen still depends on safety, efficacy, supply reliability, and regulatory acceptance.
What regulations could slow jellyfish peptide ingredient adoption in Europe?
Novel-food authorization remains one of the main factors slowing ingestible adoption in Europe, since suppliers need stronger species-specific safety documentation before broader commercialization can occur.
How does jellyfish biomass turn into high-value peptide ingredients?
Harvested biomass undergoes immediate local desalination and enzymatic hydrolysis. Facility operators must process the raw material quickly to prevent degradation. Precise filtration then isolates the target protein fractions from the remaining biological material.
What species are used most often for jellyfish collagen peptides?
Cannonball and Rhopilema species dominate commercial sourcing. Resource managers prioritize these specific medusozoan types because they display predictable seasonal aggregation patterns that support consistent processing facility operations.
How do operators evaluate jellyfish peptides vs bovine collagen peptides?
Procurement directors assess pathogen transmission risk and structural homology. Medusozoan extracts eliminate the mammalian disease vectors associated with bovine sourcing while maintaining the highly desirable Type 0 structural integrity.
What defines jellyfish collagen vs porcine collagen formulations?
Cultural and religious dietary restrictions frequently disqualify porcine sourcing. Medusozoan alternatives provide a clean, universally acceptable protein scaffold that formulators can deploy globally without reformulating for distinct regional restrictions.
Why choose jellyfish collagen or fish collagen for nutricosmetics?
Product managers seeking absolute premium positioning favor medusozoan extracts to differentiate their lines. Fish collagen faces heavy commoditization, while jellyfish sourcing currently commands distinct narrative value and specific hypoallergenic claims.
Why do cosmetic chemists prefer jellyfish peptides for skincare?
Marine hydrogels provide superior moisture retention compared to synthetic alternatives. The unique triple-helix configurations integrate seamlessly into human dermal matrices to support elasticity and cellular repair.
What drives demand for jellyfish collagen peptides for supplements?
Active aging demographics increasingly demand sustainable, clean-label joint support. Early clinical data supporting enhanced mobility positions these specialized marine extracts as premium alternatives to saturated glucosamine product lines.
How do operators limit immediate global adoption?
Extreme harvest unpredictability destroys standard manufacturing schedules. Procurement directors cannot rely on centralized oceanic harvesting, forcing them to build decentralized extraction networks that dramatically increase initial capital requirements.
How does powder format retain dominance?
Moisture control remains vital for international shipping. Facility operators prefer dry material to mitigate microbial growth risks, though this requires highly specialized, dehumidified packing environments to prevent catastrophic agglomeration.
Why does Israel lead regional compound growth?
Venture capital overwhelmingly targets specialized biomedical applications over bulk foods. Local operators prioritize high-margin regenerative medicine trials, establishing critical intellectual property moats before international competitors can deploy pilot facilities.
How do East Asian supply chains compress timelines?
Decades of handling edible biomass provide established infrastructure. Process engineers adapt existing extraction equipment, bypassing the massive capital expenditures required to build processing facilities from scratch.
What creates tension between suppliers and buyers?
Ingredient houses demand long-term volume commitments to fund necessary facility expansions. Conversely, brand sourcing directors demand geographic harvest diversity to prevent catastrophic stockouts during localized bloom failures.
Why do topical applications face regulatory exposure?
Marketing claims frequently outpace actual clinical transdermal delivery data. Compliance officers must ensure substantiation matches formulation reality to avoid severe regulatory penalties and brand damage.
How does the United States market structure differ?
Preventive aging and sports nutrition dictate demand rather than pure cosmetic applications. This positions raw material qualification around strict dietary supplement guidelines rather than simple topical safety profiles.
What dictates ultimate commercial survival for startups?
Controlling proprietary enzymatic cleavage protocols determines success. Manufacturers lacking technology to maintain triple-helix integrity during refinement produce inferior hydrolysates relegated to low-margin animal feed applications.
Why do large brands require dual-sourcing?
Single-facility reliance creates unacceptable risk profiles for flagship products. Operations managers actively fund secondary processors to ensure production continuity during inevitable regional harvest shortages.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Collagen peptides
- Hydrolysates
- Peptide chelates
- Peptide complexes
- Collagen peptides
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Nutricosmetics
- Supplements
- Topical care
- Biomaterials
- Nutricosmetics
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder
- Liquid
- Gel
- Encapsulated
- Powder
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source Species
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Source Species, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Source Species, 2026 to 2036
- Cannonball
- Rhopilema
- Nomura
- Aurelia
- Cannonball
- Y to o to Y Growth Trend Analysis By Source Species, 2021 to 2025
- Absolute $ Opportunity Analysis By Source Species, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Dietary supplements
- Skincare brands
- Ingredient houses
- Research labs
- Dietary supplements
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Application
- By Form
- By Source Species
- By End Use
- Competition Analysis
- Competition Deep Dive
- Certified Nutraceuticals
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- JellyCoUSA
- Jellagen
- Qortein
- Creative Enzymes
- Baoding Faithful Industry
- Chemipan Corporation
- Certified Nutraceuticals
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Source Species, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Form
- Figure 12: Global Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Source Species
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Form
- Figure 38: North America Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Source Species
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Application
- Figure 51: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Form
- Figure 54: Latin America Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Source Species
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Application
- Figure 67: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Form
- Figure 70: Western Europe Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Source Species
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Application
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Form
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Source Species
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Application
- Figure 99: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Form
- Figure 102: East Asia Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Source Species
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Source Species
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Source Species, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Source Species, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Source Species
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

