Mediterranean Pre-Diabetes Ingredient Concepts Market
The Mediterranean Pre-Diabetes Ingredient Concepts Market is segmented by Ingredient type (Olive polyphenols, Grape seed extracts, Citrus flavonoids, Soluble fibers, Mineral blends), Format (Powder blends, Capsule fills, Sachet mixes, Liquid shots, Softgels), Function (Glucose support, Insulin sensitivity, Weight control, Oxidative stress, Microbiome support), Channel (Dietary supplements, Functional foods, Medical nutrition, Practitioner brands, Direct-to-consumer), End user (Adults 35–54, Adults 55+, Overweight adults, Women’s health users, Preventive care users), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mediterranean Pre-Diabetes Ingredient Concepts Market Size, Market Forecast and Outlook By FMI
The Mediterranean pre-diabetes ingredient concepts market crossed a valuation of USD 0.7 billion in 2025. The industry is expected to reach USD 0.8 billion in 2026 at a CAGR of 8.5% during the forecast period. Demand outlook carries the market valuation to USD 1.8 billion by 2036 as preventive healthcare increasingly focuses on targeted, clinically substantiated metabolic interventions.
Formulators at mid-tier supplement brands face intense pressure from major retail buyers to provide clinical data for blood sugar claims. Purchasing departments reject traditional botanical blends lacking standardized active compound profiles. Companies must transition toward characterized nutraceutical ingredients to secure shelf space in premium metabolic health aisles. Delaying this qualification process forces brands into lower-margin generic categories where price competition destroys profitability. What procurement directors often misunderstand is how sourcing consistency dictates regulatory compliance across multiple jurisdictions.
Summary of Mediterranean Pre-Diabetes Ingredient Concepts Market
- Mediterranean Pre-Diabetes Ingredient Concepts Market Definition
- Standardized bioactives derived from Mediterranean agricultural sources target physiological pathways related to insulin sensitivity and glucose regulation. These ingredients function as preventative interventions for populations exhibiting borderline metabolic dysfunction prior to clinical diagnosis.
- Demand Drivers in the Market
- Rising diagnostic rates of pre-diabetes compel consumers to seek non-pharmaceutical interventions.
- Retail pharmacy category managers require brands to reformulate with ingredients possessing specific health claim authorizations.
- Contract manufacturers push brands toward standardized extracts to ensure batch consistency and regulatory compliance.
- Key Segments Analyzed in the FMI Report
- Olive polyphenols: anticipated to hold 31.0% share in 2026 based on robust clinical dossiers supporting insulin sensitivity mechanisms.
- Powder blends: projected to hold 43.0% share in 2026 driven by versatile formulation capabilities across diverse delivery formats.
- Glucose support: poised to hold 34.0% share in 2026 owing to direct consumer recognition of blood sugar management benefits.
- Dietary supplements: set to hold 46.0% share in 2026 as the primary channel for high-dose therapeutic applications.
- Adults 35–54: expected to hold 38.0% share in 2026 due to this demographic experiencing initial clinical warnings regarding metabolic health.
- Spain: 10.4% compound growth supported by integrated supply chains connecting domestic agriculture directly to advanced extraction facilities.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Analyst, Food & Beverage, at FMI, observes that "Measurement frameworks in metabolic supplements typically prioritize isolated active compound concentration. Procurement directors optimize for the lowest cost-per-kilogram of raw material based on these specifications. They miss how poor bioavailability within specific delivery formats destroys actual clinical efficacy. Standardizing the active compound means very little when the chosen extract fails to survive digestive transit. Brands prioritizing raw material cost over validated delivery mechanisms experience terrible consumer retention rates because end users see absolutely no change in their fasting glucose numbers."
- Strategic Implications / Executive Takeaways
- Formulation scientists need clinically validated delivery technologies to ensure bioactives reach intended absorption sites.
- Procurement managers must secure multi-year supplier agreements to buffer against agricultural yield volatility in Mediterranean regions.
- Marketing directors require rigorous clinical substantiation to navigate strict regulatory environments governing metabolic health claims.
- Methodology
- Procurement patterns analyzed among top-tier dietary supplement manufacturers.
- Clinical trial registries evaluated to map emerging applications for specific botanical bioactives.
- Market models anchored by verified extraction capacities of primary Mediterranean ingredient suppliers.
- Forecasting cross-validated against demographic shifts in regional pre-diabetes prevalence.
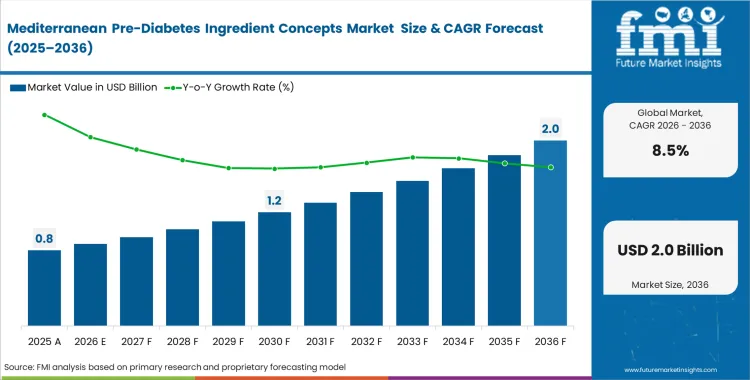
Mediterranean Pre-Diabetes Ingredient Concepts Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.8 billion |
| Industry Value (2036) | USD 1.8 billion |
| CAGR (2026-2036) | 8.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Crossing the threshold of specific health claim approvals changes the adoption math entirely. Once regulatory bodies authorize explicit glucose-regulating language for a defined compound, large-scale consumer packaged goods manufacturers commit to multi-year supply contracts. Pre-approval behavior involves small pilot runs in specialty channels. Securing these authorizations requires massive upfront clinical investment.
Spain expands at 10.4% supported by localized extraction infrastructure near primary olive cultivation zones. Italy tracks closely at 9.8% due to high domestic consumption of clinically backed metabolic formulations. Greece advances at 9.5% based on established agricultural cooperatives pivoting to high-value bioactives. Turkey grows at 9.2% by leveraging competitive processing costs for regional export. Morocco records 9.0% through government-backed agricultural modernization initiatives. Egypt registers 8.7% driven by rising domestic health awareness and expanding pharmacy distribution. France follows at 8.1% with strict regulatory frameworks favoring heavily researched premium ingredients.
Mediterranean Pre-Diabetes Ingredient Concepts Market Definition
Functional compounds derived from Mediterranean diets specifically isolated and standardized address impaired glucose tolerance before formal diabetes onset. This category focuses strictly on active bioactives modulating insulin sensitivity and carbohydrate metabolism. Formulations center around clinically validated phytochemicals and fibers naturally occurring in traditional Mediterranean agricultural products. Focus remains on physiological mechanisms regulating postprandial glucose spikes.
Mediterranean Pre-Diabetes Ingredient Concepts Market Inclusions
Standardized botanical extracts from olives, grapes, and citrus fruits validated for metabolic applications fall directly into this scope. Formulations targeting borderline dysglycemia or insulin resistance mechanisms qualify. Naturally derived soluble fibers and specific mineral complexes synergistic with these plant compounds are covered. Products intended for integration into both supplements and functional food formats require consideration.
Mediterranean Pre-Diabetes Ingredient Concepts Market Exclusions
Synthetically derived pharmaceuticals intended for diagnosed type 2 diabetes treatment remain strictly outside this analysis. Generic vitamins without specific clinical backing for glycemic control are deliberately omitted. Whole foods like unprocessed olives or raw citrus fruits fall completely outside scope. Ingredients targeting general weight loss without verified primary mechanisms acting on glucose metabolism pathways receive zero inclusion.
Mediterranean Pre-Diabetes Ingredient Concepts Market Research Methodology
- Primary Research: Procurement directors at functional food brands and R&D formulation scientists evaluating botanical bioactives.
- Desk Research: Clinical trial registries, EFSA novel food application archives, and supplier ingredient monographs.
- Market-Sizing and Forecasting: Baseline established through total regional agricultural output allocated specifically to high-grade extraction facilities.
- Data Validation and Update Cycle: Import volumes of standardized botanical extracts cross-referenced against retail point-of-sale data for metabolic health products.
Segmental Analysis
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by Ingredient type
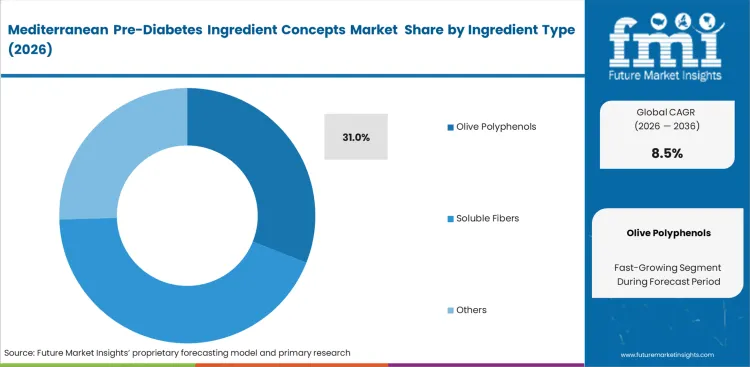
Mechanisms determining market dominance center entirely on clinical trial volume and resulting regulatory authorizations. Olive polyphenols hold 31.0% share in 2026 driven by extensive research demonstrating efficacy in modulating insulin receptors and lipid profiles. R&D directors at major dietary supplements companies specify these exact extracts because EFSA claims protect them from regulatory action. Procurement teams face significant volatility tied directly to European olive harvest yields. Generalist reporting focuses entirely on the antioxidant properties of these compounds. The actual practitioner reality is that supply chain instability forces brands to constantly reformulate or accept massive margin compression during poor harvest years. Brands failing to secure resilient supply networks risk complete out-of-stock situations during peak demand seasons.
- Dossier validation: R&D directors select olive extracts based on complete clinical dossiers rather than raw material cost. Purchasing unverified ingredients leads directly to rejected health claims and forced market withdrawal.
- Harvest dependency: Procurement managers must navigate extreme price fluctuations caused by regional climate events affecting olive yields. Securing long-term contracts provides the only reliable defense against raw material cost spikes.
- Synergistic combinations: Formulation scientists increasingly pair olive polyphenols with specific fiber supplements to enhance overall metabolic impact. Creating these combinations requires specialized processing to maintain stability over the required shelf life.
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by Format
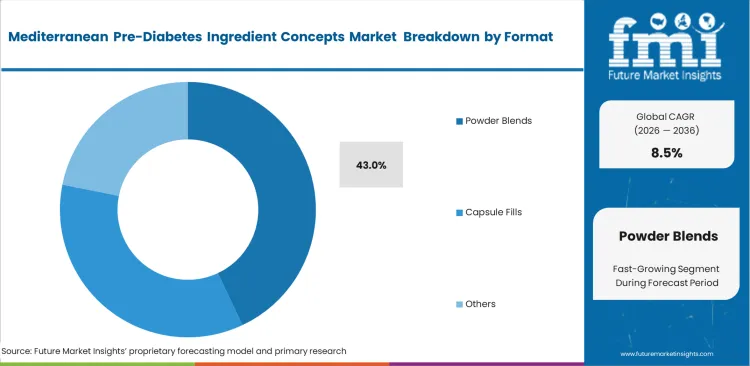
Versatility across manufacturing lines dictates format adoption among contract manufacturers. Powder blends command 43.0% share in 2026 based on their ability to integrate seamlessly into diverse end products. Facility operations managers favor powders because they eliminate the need for specialized encapsulation or liquid-fill equipment. Formulation scientists encounter persistent challenges with bulk density variations between different botanical extract batches. Product brochures highlight the convenience of powder formats for consumer dosing. The non-obvious reality is that unpredictable powder flowability forces manufacturers to overcompensate fill weights, quietly eroding unit economics on large production runs. Overlooking these flow characteristics results in costly line stoppages and significant material waste during high-speed packaging operations.
- Manufacturing efficiency: Operations managers prioritize powder formats to maximize throughput on existing production lines. Selecting complex liquid formats requires massive capital investment in specialized filling equipment.
- Formulation masking: Flavor chemists must actively mask the inherent bitterness of high-concentration botanical ingredients. Failing to achieve acceptable taste profiles guarantees product failure in the functional beverage category.
- Density management: Quality control technicians constantly monitor bulk density to ensure accurate dosing in final retail packaging. Ignoring these variations causes serious regulatory compliance issues regarding stated label claims.
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by Function
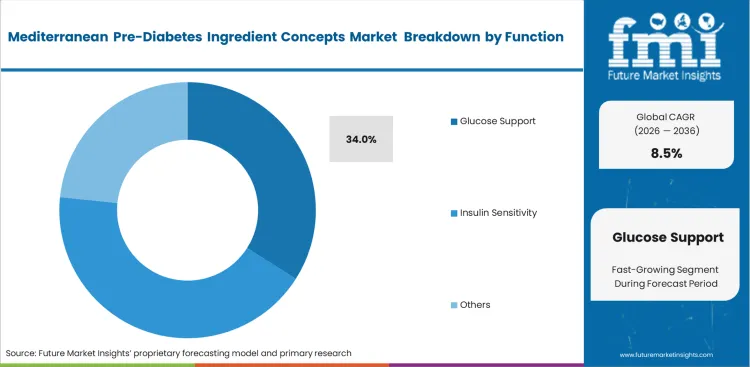
Consumer recognition directly shapes formulation strategies across the metabolic health category. Glucose support captures 34.0% share in 2026 as consumers explicitly seek preventative measures following elevated HbA1c test results. Brand managers design front-of-pack messaging around this specific function to capture immediate intent. Navigating the regulatory requirements for these claims requires precise alignment between the selected extract and available clinical data. Marketing teams emphasize long-term metabolic health in their campaigns. Practitioners understand that consumers actually judge product efficacy based on short-term energy stability and the absence of post-meal fatigue. Brands neglecting to formulate for these immediate experiential benefits suffer from poor long-term subscription retention.
- Claim alignment: Regulatory affairs officers meticulously match functional claims to the specific clinical evidence backing the chosen ingredient. Approving unsupported language invites immediate regulatory penalties and forced product recalls.
- Experiential feedback: Formulation scientists include complementary bioactives to provide noticeable energy stability alongside long-term glucose management. Relying solely on invisible physiological changes makes it very difficult to secure repeat consumer purchases.
- Diagnostic triggers: Marketing directors target consumer acquisition immediately following routine medical appointments. Failing to intercept consumers during this high-anxiety window results in lost market share to established botanical bioactives brands.
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by Channel
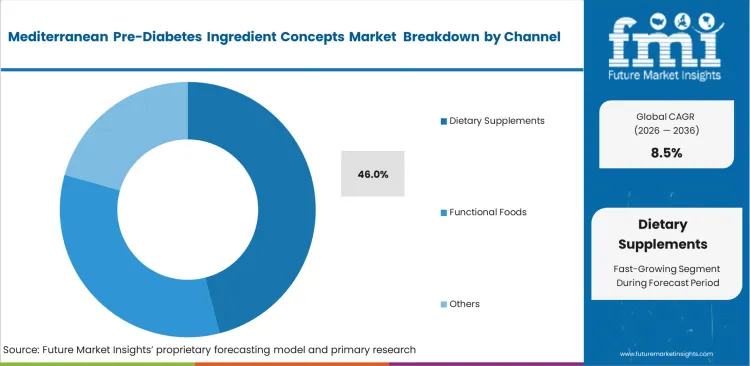
Therapeutic dosage requirements dictate distribution strategies for specialized metabolic ingredients. Dietary supplements account for 46.0% share in 2026 due to their capacity to deliver clinically relevant concentrations of active compounds. Category buyers at major pharmacy chains allocate prime shelf space to established brands possessing verified clinical backing. Achieving distribution in these channels demands robust stability data and comprehensive quality control documentation. Industry narrative highlights the rapid growth of direct-to-consumer online channels. The hidden dynamic involves medical nutrition channels acting as the primary stealth growth engine, driven by specific practitioner recommendations that bypass traditional retail entirely. Brands unable to penetrate these practitioner networks miss the most loyal and price-insensitive consumer segment available.
- Dosage delivery: Formulation scientists utilize supplement formats to achieve specific therapeutic thresholds difficult to reach through fortified foods. Attempting to deliver active doses through food formats often destroys the product's taste and texture.
- Retail gatekeeping: Category managers at major pharmacy chains require comprehensive clinical dossiers before authorizing new product listings. Presenting products without this documentation guarantees immediate rejection from premium retail placements.
- Practitioner advocacy: Sales directors targeting medical channels must provide detailed mechanism-of-action data to healthcare professionals. Brands succeeding in this channel gain highly credible recommendations that drive long-term herbal supplements sales.
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by End user
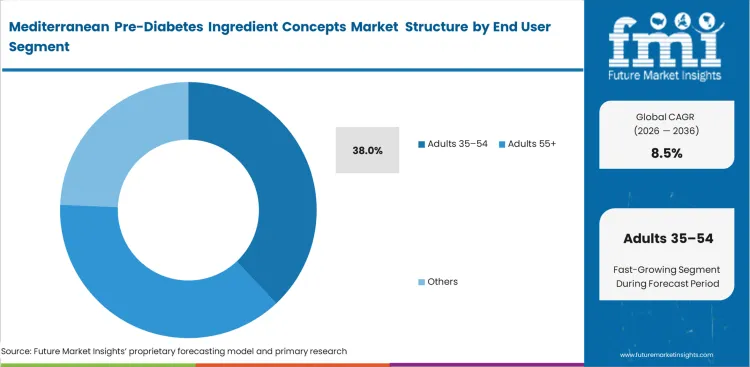
Preventative healthcare behaviors accelerate sharply following initial medical warnings. Adults 35–54 represent 38.0% share in 2026 as this demographic increasingly receives borderline metabolic diagnoses during routine screenings. Product developers target this cohort because they possess high disposable income and intense motivation to avoid pharmaceutical interventions. Engaging this demographic requires formulations that fit seamlessly into busy, active lifestyles. Standard analysis suggests older demographics represent the primary target for metabolic products. The practitioner reality reveals that the 35–54 group demands immediate experiential effects to justify continued use, unlike older cohorts who dutifully consume traditional pill formats. Failing to provide these immediate lifestyle benefits ensures this valuable demographic quickly abandons the regimen.
- Lifestyle integration: Product developers must create convenient formats that busy professionals can easily incorporate into daily routines. Designing complex dosing schedules leads directly to poor compliance and eventual product abandonment.
- Alternative seeking: Marketing directors position these products as verified natural alternatives for consumers terrified of progressing to prescription medications. Messaging must balance clinical authority with accessible lifestyle aspirations to drive initial trial.
- Retention metrics: Brand managers track repeat purchase rates rigorously within this demographic to gauge true formulation success. Poor experiential feedback causes immediate subscription cancellations regardless of the underlying clinical validity.
Mediterranean Pre-Diabetes Ingredient Concepts Market Drivers, Restraints, and Opportunities
Major retail pharmacy category managers mandate explicit clinical substantiation for any product entering the metabolic health aisle. Purchasing departments refuse to list generic botanical blends, effectively forcing supplement brands to qualify standardized extracts with proven glucose-modulating mechanisms. Transitioning to these characterized functional food ingredients requires substantial formulation overhauls and complete label redesigns. Brands failing to secure these verified bioactives face immediate delisting from premium retail environments. Securing this retail real estate dictates commercial survival in an increasingly crowded preventative health landscape.
Agricultural yield volatility creates massive structural friction for procurement departments attempting to stabilize long-term pricing. Sourcing standardized olive or grape extracts remains completely dependent on unpredictable regional climate conditions across the Mediterranean basin. Securing consistent active compound concentrations requires sophisticated blending strategies when individual harvests fail to meet strict specifications. Emerging precision fermentation technologies offer theoretical alternatives, but current regulatory frameworks prevent these synthesized compounds from utilizing natural botanical health claims.
Opportunities in the Mediterranean Pre-Diabetes Ingredient Concepts Market
- Combinatorial formulations: R&D directors secure premium pricing by validating synergistic effects between olive polyphenols and specific prebiotic ingredient profiles.
- Beverage integration: Formulation scientists unlock new demographics by successfully masking bitter botanical bioactives within ready-to-drink metabolic health formats.
- Practitioner channels: Sales directors capture high-margin recurring revenue by providing dedicated educational platforms for dietitians recommending preventative metabolic protocols.
Regional Analysis
Based on regional analysis, Mediterranean Pre-Diabetes Ingredient Concepts Market is segmented into Western Europe, Southern Europe, North Africa, and Middle East across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Spain | 10.4% |
| Italy | 9.8% |
| Greece | 9.5% |
| Turkey | 9.2% |
| Morocco | 9.0% |
| Egypt | 8.7% |
| France | 8.1% |
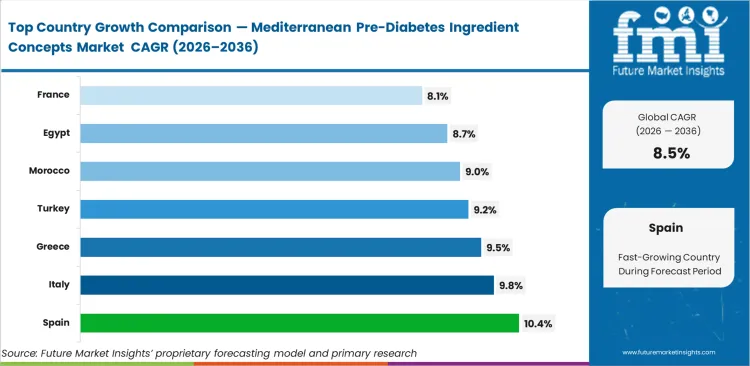
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Southern Europe Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis
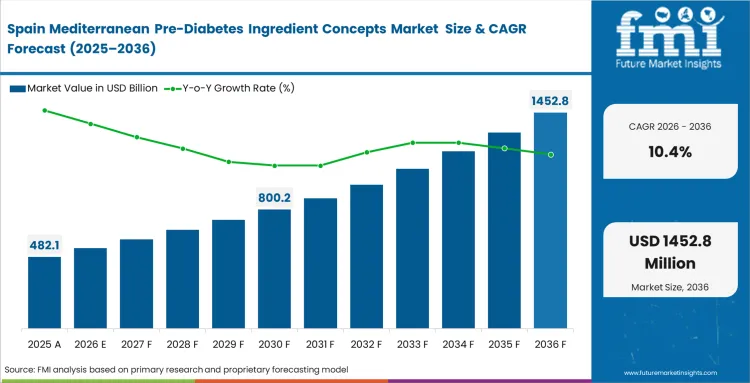
Integrated agricultural supply chains directly connect domestic cultivation with advanced extraction infrastructure. Processing facilities locate operations precisely near major olive and grape growing regions to minimize raw material degradation during transport. Procurement directors at regional supplement manufacturers leverage these local networks to secure preferred pricing on premium standardized bioactives. Establishing these localized partnerships provides a critical buffer against global supply chain disruptions. FMI observes that companies attempting to import competing botanicals face severe consumer pushback from populations highly educated on the benefits of local Mediterranean diets.
- Spain: A strong domestic olive base gives Spanish extract producers a clear supply advantage. Raw material is available at scale, which helps processors secure longer-term contracts and manage costs with more stability. Spain is projected to record a CAGR of 10.4%. That supply strength also supports the country’s position in the wider European export market.
- Italy: In Italy, consumer familiarity with Mediterranean diet-based health products supports demand for metabolic supplements built around local ingredients. Italy is set to grow a 9.8% CAGR. Products with domestic botanical sourcing often carry stronger appeal in pharmacy and health retail channels.
- Greece: Greek cooperatives are moving beyond conventional crop supply and placing greater focus on standardized bioactive production. This is opening room for ingredient development based on regional plant varieties with distinct functional profiles. Growth in Greece is forecast at 9.5% CAGR.
FMI's report includes Portugal and Cyprus. Subsidized agricultural modernization programs rapidly expand the capacity for pharmaceutical-grade botanical extraction in these adjacent markets.
North Africa Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis
Government-backed agricultural modernization initiatives rapidly transform raw material export economies into sophisticated ingredient processing hubs. Extraction companies upgrade facilities to meet strict European pharmaceutical standards, allowing direct competition in premium botanical markets. Quality control managers implement rigorous testing protocols to verify active compound concentrations required by international buyers. Navigating complex export certifications remains the primary operational hurdle for these emerging suppliers. According to FMI's estimates, companies successfully establishing verified analytical testing capabilities immediately secure lucrative contracts with major European supplement brands.
- Morocco: State-sponsored investments in agricultural processing elevate local facilities to international pharmaceutical standards. Quality control managers leverage these upgrades to secure premium European export contracts, advancing at 9.0%. Achieving these certifications transforms local agriculture into a high-value bioactives industry.
- Egypt: Expanding pharmacy networks drive domestic demand for preventative metabolic health products. Sales directors navigate complex regional distribution models to achieve 8.7% growth. Successfully integrating products into these localized pharmacy chains dictates total market penetration.
FMI's report includes Tunisia and Algeria. Emerging regulatory frameworks slowly align with European standards, gradually opening new avenues for standardized botanical exports.
Western Europe Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis
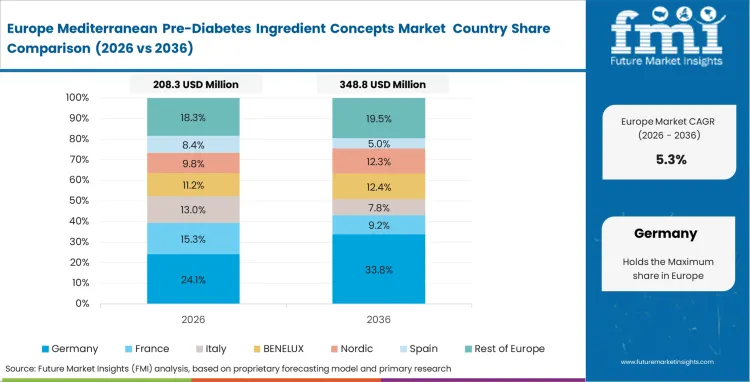
Strict regulatory frameworks actively shape formulation strategies and marketing capabilities across the region. Regulatory affairs officers navigate complex EFSA requirements to secure legal health claims for specific botanical bioactives. Companies possessing authorized claims dominate the premium retail spaces within major pharmacy chains. Formulating without these verified claims restricts products to generic wellness categories with significantly lower margins. Based on FMI's assessment, brands investing heavily in specific clinical trials effectively weaponize these regulatory barriers against smaller competitors lacking R&D capital.
- France: Rigorous national health directives force supplement manufacturers to utilize extensively researched botanical ingredients. Regulatory affairs officers rely on comprehensive clinical dossiers to navigate these requirements, resulting in 8.1% growth. Brands lacking this documentation find it functionally impossible to secure placement in major retail pharmacies.
- Turkey: Competitive processing costs allow domestic facilities to supply standardized ingredients across the broader European region. Operations managers optimize high-volume extraction techniques to maintain 9.2% growth. Mastering this low-cost, high-quality processing model establishes local facilities as indispensable nodes in the global supply chain.
FMI's report includes Germany and the United Kingdom. Consumer demand for heavily researched functional foods forces major grocery chains to rethink their approach to the metabolic health category.
Competitive Aligners for Market Players
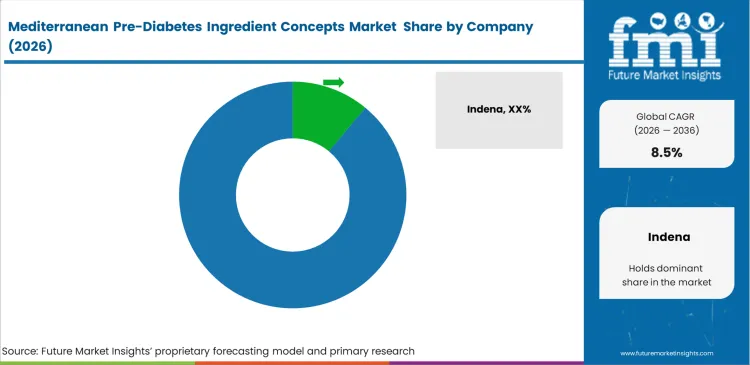
Extracting standardized bioactives from variable agricultural inputs requires specialized processing infrastructure and deep sourcing networks. Indena and Euromed control significant market share because they possess decades of proprietary clinical data specific to their exact extraction methods. Purchasing directors at major supplement brands specify these exact branded ingredients because substituting a cheaper generic alternative voids the existing health claims. Competing in this top tier demands massive upfront investment in both pharmaceutical-grade facilities and human clinical trials.
Incumbents maintain dominance by locking down long-term agricultural supply contracts across the Mediterranean basin. Challengers struggle to secure consistent raw material volumes during poor harvest years, forcing them to blend lower-quality inputs that fail to meet strict active compound specifications. Formulators demand consistency above all else to ensure accurate retail labeling. Established players leverage their vast processing scale to absorb natural agricultural variations, delivering completely uniform ingredient profiles batch after batch.
Procurement teams at large consumer packaged goods companies actively qualify secondary suppliers to mitigate supply chain risks tied to regional climate events. Brands resist total dependence on single extraction facilities, forcing ingredient suppliers to differentiate through advanced delivery technologies rather than just raw material pricing. Moving forward, competitive advantage hinges entirely on providing highly bioavailable formats that function seamlessly within complex food and beverage matrices without compromising taste or stability.
Key Players in Mediterranean Pre-Diabetes Ingredient Concepts Market
- Indena
- Euromed
- ADM
- Kerry
- dsm-firmenich
- Givaudan
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.8 billion to USD 1.8 billion, at a CAGR of 8.5% |
| Market Definition | Standardized Mediterranean botanical bioactives function as preventative interventions targeting physiological pathways related to insulin sensitivity and glucose regulation. |
| Segmentation | Ingredient type, Format, Function, Channel, End user, Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East and Africa |
| Countries Covered | Spain, Italy, Greece, Turkey, Morocco, Egypt, France |
| Key Companies Profiled | Indena, Euromed, ADM, Kerry, dsm-firmenich, Givaudan |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline established through agricultural extraction capacity cross-validated against regional demographic data regarding pre-diabetes prevalence. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Mediterranean Pre-Diabetes Ingredient Concepts Market Analysis by Segments
Ingredient type:
- Olive polyphenols
- Grape seed extracts
- Citrus flavonoids
- Soluble fibers
- Mineral blends
Format:
- Powder blends
- Capsule fills
- Sachet mixes
- Liquid shots
- Softgels
Function:
- Glucose support
- Insulin sensitivity
- Weight control
- Oxidative stress
- Microbiome support
Channel:
- Dietary supplements
- Functional foods
- Medical nutrition
- Practitioner brands
- Direct-to-consumer
End user:
- Adults 35-54
- Adults 55+
- Overweight adults
- Women’s health users
- Preventive care users
Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- South Asia and Pacific
- East Asia
- Middle East and Africa
Bibliography
- International Diabetes Federation. (2025). IDF Diabetes Atlas 2025.
- American Diabetes Association Professional Practice Committee. (2025). Diagnosis and classification of diabetes: Standards of care in diabetes-2025. Diabetes Care, 48(Supplement_1).
- Zalewska, M., Kretowski, A., Adamska-Patruno, E., Citko, A., Bauer, W., Mlyszko, A., Mlyszko, J., Jelski, W., Kretowski, A. J., & Leoniuk, A. (2025). Beneficial effect of the Mediterranean diet on the reduction of prediabetes-Results of the Bialystok PLUS study. Nutrients, 17(12), 2034.
- Veronese, N., Bonaccio, M., Dinu, M., Sofi, F., Persichillo, M., Loffredo, L., Costanzo, S., Iacoviello, L., & Gensini, G. F. (2026). 2025 national guidelines on the Mediterranean diet: Executive summary of a joint report by Italian scientific societies and the National Institute of Health task force on clinical practice guidelines. Nutrition Reviews.
- Li, R., Shu, L., Zhu, Q., & Lu, D. (2025). Dietary total antioxidant capacity and risk of prediabetes and diabetes mellitus: A systematic review and dose-response meta-analysis of 170,919 participants. Frontiers in Nutrition, 12, 1541734.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Procurement strategies for mitigating olive harvest volatility affecting standardized polyphenol pricing.
- Formulation requirements for maintaining active compound stability within complex powder blends.
- Regulatory pathways for securing specific glucose support health claims across European jurisdictions.
- Distribution dynamics shaping the expansion of medical nutrition practitioner channels.
- Lifestyle integration techniques required to capture the high-value 35-54 demographic.
- Capital investment requirements for upgrading North African extraction facilities to pharmaceutical standards.
- Supply chain logistics defining raw material acquisition across Southern European agricultural cooperatives.
- Clinical substantiation requirements mandated by major retail pharmacy category managers.
Frequently Asked Questions
What is the baseline valuation for these preventative ingredients?
FMI calculated total revenue at USD 0.8 billion in 2026. This figure reflects the immediate commercial pivot by mid-tier brands away from generic botanical blends toward clinically verified standardized extracts.
What structural condition drives the 8.5% growth rate?
Retail pharmacy category managers refuse to list metabolic products lacking specific clinical substantiation. This strict gatekeeping forces rapid formulation overhauls across the entire dietary supplement sector.
Why do olive polyphenols dominate the ingredient category?
Robust clinical dossiers supporting their exact physiological impact on insulin receptors provide brands with defensible health claims. Formulation scientists rely on this verified data to navigate strict EFSA regulations.
How does agricultural volatility impact procurement strategies?
Regional climate events across the Mediterranean severely disrupt consistent raw material supplies. Procurement directors must secure multi-year contracts or face massive margin compression during poor harvest seasons.
What operational consequence drives the adoption of powder formats?
Versatility across existing manufacturing lines allows contract manufacturers to maximize throughput without purchasing specialized equipment. Operations managers avoid expensive liquid-fill machinery by standardizing around these flowable blends.
How do bulk density variations affect manufacturing efficiency?
Unpredictable powder flowability forces quality control technicians to constantly recalibrate filling equipment. Failing to monitor these variations leads to inaccurate dosing and subsequent regulatory compliance issues.
Why is glucose support the dominant functional claim?
Consumers actively seek preventative interventions immediately following concerning HbA1c test results. Marketing directors target this high-anxiety window to capture intent before consumers transition to pharmaceutical options.
What hidden factor influences consumer retention for metabolic products?
Practitioners know consumers evaluate efficacy based on short-term energy stability rather than invisible physiological changes. Products failing to prevent post-meal fatigue suffer massive subscription cancellation rates.
Why are dietary supplements the primary distribution channel?
Delivering clinically relevant concentrations of active bioactives remains exceptionally difficult in standard food matrices. Formulation scientists utilize capsules and powders to achieve required therapeutic thresholds without destroying taste profiles.
How does the medical nutrition channel operate differently?
Sales directors bypass traditional retail gatekeepers by providing detailed mechanism-of-action data directly to healthcare professionals. This approach secures highly credible practitioner recommendations that drive price-insensitive recurring revenue.
What demographic shift accelerates adoption across the sector?
Adults aged 35–54 increasingly receive borderline metabolic diagnoses during routine medical screenings. Product developers target this cohort because they possess high disposable income and intense motivation to avoid prescription medications.
How do usage patterns differ between demographic cohorts?
Younger professionals demand immediate experiential benefits that integrate easily into active lifestyles. Older demographics generally demonstrate higher compliance with traditional, rigid supplement dosing schedules regardless of immediate lifestyle feedback.
What structural difference explains Spain's rapid expansion?
Massive domestic olive cultivation provides local extraction facilities with unparalleled access to raw materials. Establishing processing infrastructure directly adjacent to these agricultural zones minimizes degradation and significantly lowers transport costs.
How do French regulations shape the regional landscape?
Rigorous national health directives force manufacturers to utilize exclusively researched botanical ingredients. Brands lacking comprehensive clinical dossiers find it functionally impossible to secure placement within major French retail pharmacies.
What defines the trajectory of North African processing capabilities?
State-sponsored agricultural modernization initiatives elevate local extraction facilities to international pharmaceutical standards. Quality control managers leverage these verified testing protocols to secure premium European export contracts.
Why do incumbent suppliers maintain significant market share?
Established companies possess decades of proprietary clinical data tied directly to their specific extraction methods. Substituting cheaper generic alternatives instantly voids the health claims brands rely upon for retail placement.
How do competitors navigate strict agricultural supply constraints?
Large consumer packaged goods companies actively qualify multiple secondary suppliers to mitigate climate-related harvest failures. Procurement teams resist total dependence on single extraction facilities to maintain pricing leverage.
What defines the integration challenges for these botanical extracts?
Flavor chemists must actively mask the inherent bitterness of high-concentration bioactives. Failing to achieve acceptable taste profiles guarantees product failure when brands attempt to transition from supplements to functional beverages.
How do synergistic combinations enhance product positioning?
R&D directors validate the combined physiological effects of specific polyphenols paired with targeted soluble fibers. Creating these complex blends allows brands to command premium pricing through highly differentiated functional claims.
What role do agricultural cooperatives play in regional sourcing?
Historical cooperatives actively pivot from bulk commodity export toward producing high-value standardized bioactives. R&D directors utilize these unique regional varietals to create specialized products with distinct phytochemical profiles.
How does clinical trial volume impact formulation decisions?
Regulatory affairs officers meticulously match functional claims to the specific evidence backing the chosen ingredient. Approving unsupported language invites immediate regulatory penalties and forced product recalls across multiple jurisdictions.
What defines the eventual structural end state for this sector?
Competitive advantage hinges entirely on providing highly bioavailable formats functioning seamlessly within complex food matrices. Future growth relies on moving these verified clinical interventions out of the supplement aisle and into daily dietary habits.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Olive Polyphenols
- Soluble Fibers
- Others
- Olive Polyphenols
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Format, 2026 to 2036
- Powder Blends
- Capsule Fills
- Others
- Powder Blends
- Y to o to Y Growth Trend Analysis By Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Function, 2026 to 2036
- Glucose Support
- Insulin Sensitivity
- Others
- Glucose Support
- Y to o to Y Growth Trend Analysis By Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Channel, 2026 to 2036
- Dietary Supplements
- Functional Foods
- Others
- Dietary Supplements
- Y to o to Y Growth Trend Analysis By Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Adults 35–54
- Adults 55+
- Others
- Adults 35–54
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Format
- By Function
- By Channel
- By End User
- Competition Analysis
- Competition Deep Dive
- Indena
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Euromed
- ADM
- Kerry
- dsm-firmenich
- Givaudan
- Indena
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Format
- Figure 9: Global Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Function
- Figure 12: Global Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Channel
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Format
- Figure 35: North America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Function
- Figure 38: North America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Channel
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Function
- Figure 54: Latin America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Channel
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Function
- Figure 70: Western Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Channel
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Function
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Channel
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Function
- Figure 102: East Asia Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Channel
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Function
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Channel
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Function
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Channel
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

