Mediterranean Mood and Stress Support Complexes Market
The Mediterranean Mood And Stress Support Complexes Market Is Segmented By Ingredient Base (Saffron Extracts, Lemon Balm, Lavender, Chamomile, Olive Polyphenols), Formulation Type (Multi-Botanical Blends, Single Extracts, Mineral Blends, Probiotic Blends, Drink Powders), Delivery Format (Capsules, Tablets, Gummies, Sachets, Liquids), End Use (Dietary Supplements, Functional Foods, Functional Beverages, Clinical Nutrition), Sales Channel (B2B Ingredients, Branded Retail, Practitioner Sales, Online Direct), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
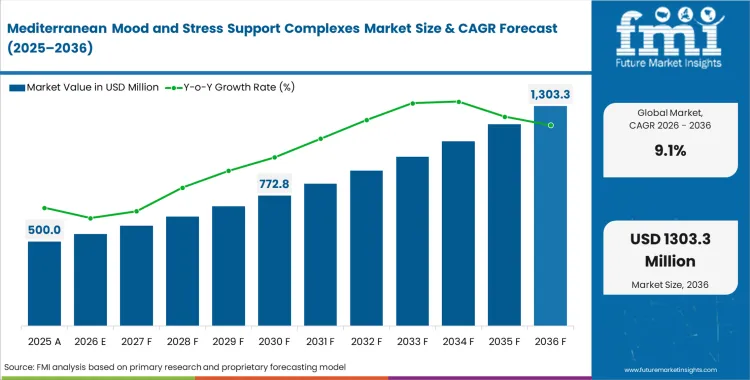
Mediterranean Mood and Stress Support Complexes Market Size, Market Forecast and Outlook By FMI
The mediterranean mood and stress support complexes market secured a solid growth of USD 400 million in 2025. Steady increase in revenue is further expected to cross USD 500 million in 2026 at a CAGR of 9.1% during the forecast period with the industry demand propelling total valuation to USD 1,100 million through 2036 as supply-chain localization of European botanical extracts aligns directly with tightening clinical validation requirements.
Formulation directors at mid-tier nutraceutical companies face immediate regulatory pressure to replace synthetic anxiety compounds with clinically backed, region-specific botanicals. Delaying this transition risks product delisting in major European retail chains demanding transparent sourcing for mediterranean mood support ingredients. Finding a reliable mood enhancing supplement source proves difficult when crop yield variability dictates pricing. Procurement managers realize that securing a stable stress relief supplement supply chain matters more than identifying novel ingredients.
Summary of Mediterranean Mood and Stress Support Complexes Market
- Mediterranean Mood and Stress Support Complexes Market Definition:
- Standardized botanical formulations derived from Mediterranean flora targeting neurological pathways comprise this sector. Efficacy requires specific phytochemical concentrations modulating the hypothalamic-pituitary-adrenal axis rather than generic raw plant material.
- Demand Drivers in the Market:
- Supply chain transparency requirements force procurement managers to secure direct extraction facility contracts.
- Clinical substantiation demands push R&D directors toward proprietary branded ingredients over generic plant powders.
- Consumer skepticism regarding synthetic compounds drives marketing teams to mandate whole-plant traceable sourcing.
- Key Segments Analyzed in the FMI Report:
- Saffron Extracts: 31.0% share in 2026, benefiting from unparalleled clinical data regarding serotonin reuptake modulation.
- Multi-Botanical Blends: 46.0% share in 2026, addressing complex consumer demands for synergistic botanical supplements profiles.
- Capsules: 37.0% share in 2026, effectively masking the bitter astringent profiles of concentrated polyphenols.
- Dietary Supplements: 58.0% share in 2026, driven by favorable regulatory pathways compared to clinical nutrition channels.
- B2B Ingredients: 52.0% share in 2026, as specialized extraction requires massive capital distinct from retail branding.
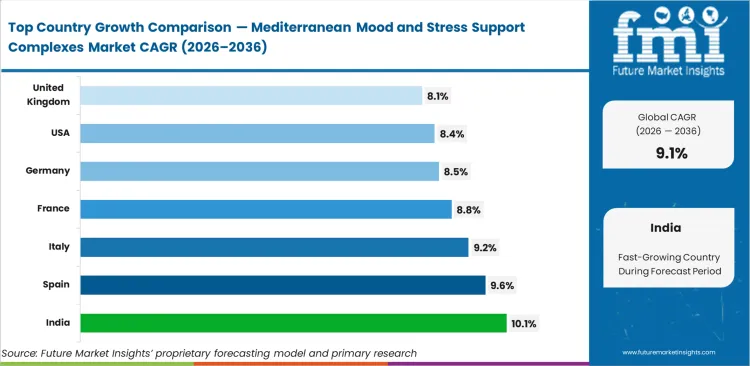
- India: 10.1% CAGR, reflecting a massive shift in contract manufacturing capabilities toward premium export components.
- Analyst Opinion at FMI:
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, observes that, "R&D directors spend months navigating dietary supplement formulation mediterranean herbs to design the perfect synergistic ratio based on clinical literature. Procurement teams then spend the next year fighting a losing battle against the reality of European crop yield variability. The fluctuating phytochemical densities across different harvest seasons create an operational nightmare. Buyers who prioritize unit cost over multi-year agricultural contracts quickly find their 'clinically backed' products failing basic potency tests upon delivery."
- Strategic Implications / Executive Takeaways:
- Formulation chemists must prioritize supply chain stability over novel synergistic claims when selecting specific botanical sources.
- Procurement teams face severe supply bottlenecks unless they secure multi-year off-take agreements with vertically integrated extractors.
- Marketing departments risk significant regulatory action if they base product claims on generic literature rather than batch-specific trial data.
- Methodology:
- Primary Research: Direct interviews with chief formulation officers determine immediate procurement priorities.
- Desk Research: Clinical trial databases reveal shifting substantiation standards for neurological claims.
- Market-Sizing and Forecasting: Wholesale extract volume shipments anchor the baseline valuation.
- Data Validation and Update Cycle: Regional extraction yield rates cross-validate long-term pricing projections.
Once top-tier extraction facilities standardize crocin levels across multiple harvests, formulation of mediterranean stress support botanicals becomes highly predictable. Reliable phytochemistry removes the primary hesitation for large-scale contract manufacturers evaluating these ingredients. Guaranteed potency allows brands to confidently list specific physiological claims without fear of batch failure, directly impacting sleep supplement applications.
India leads at 10.1% as contract manufacturers pivot rapidly toward premium European-sourced complexes for export channels. Spain tracks at 9.6% due to its domestic agricultural dominance in saffron and olive cultivation, anchoring the mediterranean stress support ingredients forecast. Italy follows closely at 9.2% based on aggressive vertical integration by regional processors. France expands at 8.8% while the United States grows at 8.4% as domestic brands seek imported actives. Germany registers 8.5% and the United Kingdom advances at 8.1% with differing regulatory approaches shaping final product formats.
Mediterranean Mood and Stress Support Complexes Market Definition
The mediterranean mood and stress support complexes comprise standardized botanical formulations derived primarily from flora native to the Mediterranean basin, intended specifically for neurological and psychological applications. Active ingredients must possess verified mechanisms for modulating the hypothalamic-pituitary-adrenal axis or altering neurotransmitter activity. Efficacy relies on specific phytochemical concentrations rather than raw plant mass.
Mediterranean Mood and Stress Support Complexes Market Inclusions
Formulations integrating saffron, lemon balm, lavender, chamomile, and specific olive polyphenols fall strictly within scope as mediterranean anti-stress complexes. Standardized extracts intended for herbal supplements targeting cognitive function, anxiety reduction, and sleep architecture improvement qualify for analysis. Single-ingredient standardized botanical preparations and multi-botanical synergistic blends both receive full coverage.
Mediterranean Mood and Stress Support Complexes Market Exclusions
Synthetic nootropics and non-botanical amino acid preparations do not meet inclusion criteria due to their differing regulatory pathways and manufacturing processes. Formulations relying entirely on adaptogens outside the Mediterranean geographic classification, such as Ashwagandha or Rhodiola rosea, sit outside this specific dataset. Essential oils intended exclusively for topical or aromatherapeutic use without internal ingestion pathways face exclusion.
Mediterranean Mood and Stress Support Complexes Market Research Methodology
- Primary Research: Procurement directors and chief formulation officers at leading contract manufacturing organizations.
- Desk Research: European Food Safety Authority health claim registries and proprietary supplier clinical trial databases.
- Market-Sizing and Forecasting: Wholesale botanical extract volume shipments cross-referenced with regional extraction yield rates.
- Data Validation and Update Cycle: Import-export customs logs for high-value botanical commodities calibrate quarterly projections.
Segmental Analysis
Mediterranean Mood and Stress Support Complexes Market Analysis by Ingredient Base
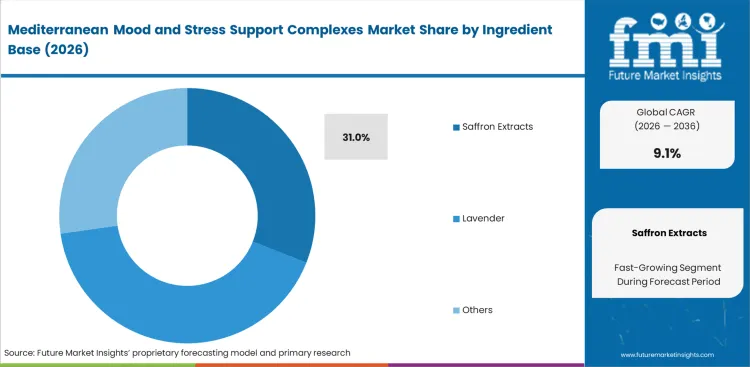
Generic plant powders fail modern clinical substantiation requirements entirely, making saffron extracts to dominate by a 31.0% share, as FMI analysts note that procurement directors actively prioritize saffron extract for stress support. High crocin concentrations deliver verifiable results for botanical ingredients applications. Securing lemon balm extract for mood support requires deep relationships with Spanish and Iranian agricultural cooperatives. Widespread origin fraud renders generic literature useless if the supply chain lacks strict vertical integration. Buyers accepting unverified botanical extracts face immediate recall risks when independent testing reveals synthetic adulteration.
- Targeted Serotonin Modulation: Standardized crocin profiles directly influence neurotransmitter reuptake mechanisms within neural pathways. Formulation officers achieve consistent therapeutic outcomes.
- Agricultural Supply Constraints: Genuine Crocus sativus cultivation relies on highly variable seasonal weather patterns across limited geographical zones. Procurement managers face intense pricing volatility.
- Clinical Validation Standards: European regulators demand exact phytochemical matching between commercial batches and original trial materials. Quality control directors must reject non-compliant shipments.
Mediterranean Mood and Stress Support Complexes Market Analysis by Formulation Type
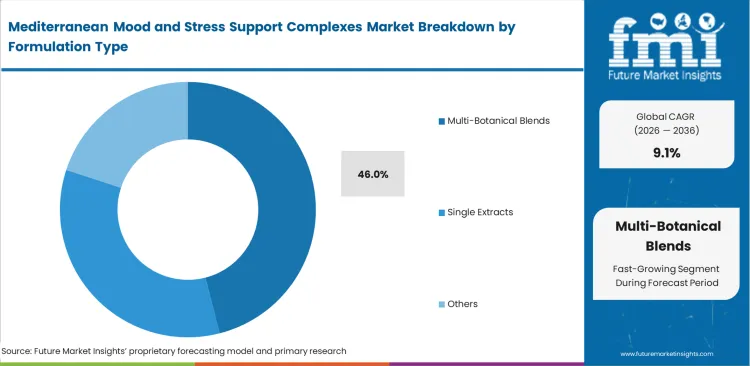
Brands balance consumer desire for comprehensive solutions against the high cost of therapeutic dosing, and, by combining multiple botanical bioactives creates stability issues requiring advanced encapsulation technologies. This dilemma leads research and development heads struggle to maintain efficacy across complex saffron lemon balm mood blend architectures, thereby, directing the multi-botanical blends to secure a 46.0% share, representing a significant formulation challenge. Complex synergy claims frequently mask sub-therapeutic dosing of the most expensive raw materials within the blend while companies failing to validate final product stability experience rapid potency degradation on retail shelves, even when sourcing the best Mediterranean botanicals for stress and mood support.
- Synergistic Interaction Testing: Combining lavender and lemon balm alters individual pharmacokinetic absorption rates. Chief scientific officers require extensive in-vitro validation protocols.
- Excipient Compatibility Challenges: Highly concentrated olive polyphenols interact unpredictably with standard binding agents. Manufacturing engineers must redesign traditional compression workflows.
- Cost Optimization Pressures: Blending expensive saffron with more abundant chamomile lowers overall unit production expenses. Category managers balance margin requirements against efficacy.
Mediterranean Mood and Stress Support Complexes Market Analysis by Delivery Format
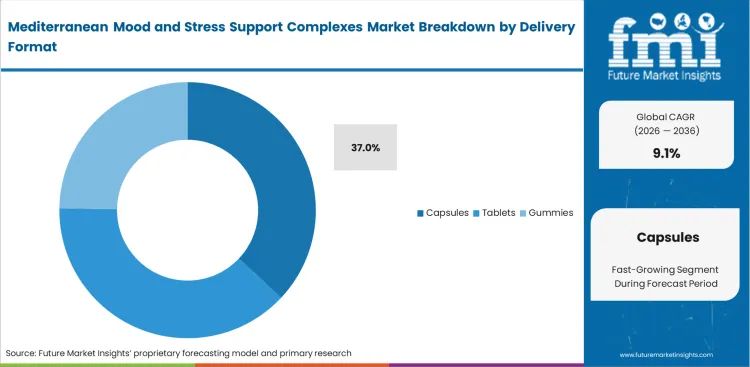
Avoiding severe taste aversion dictates final product engineering for high-concentration botanicals. Capsules command 37.0% share based on this specific mechanical advantage with consumers expecting dietary supplements to remain flavor-neutral during ingestion. Protecting sensitive melatonin sleep supplements compounds from oxidation requires specialized shell technologies. This format perfectly hides the highly astringent and bitter taste profiles of concentrated Mediterranean extracts that routinely destroy gummy formulations.
- Moisture Barrier Properties: Hydroxypropyl methylcellulose shells prevent hygroscopic botanical powders from absorbing ambient humidity. Packaging engineers ensure long-term shelf stability.
- Disintegration Timing Control: Acid-resistant banding allows sensitive actives to bypass gastric degradation entirely. Clinical researchers confirm higher intestinal absorption rates.
- Astringency Masking Capability: Enclosing raw polyphenol powders eliminates immediate tongue receptor activation. Product development managers avoid complex flavor-masking formulations.
Mediterranean Mood and Stress Support Complexes Market Analysis by End Use
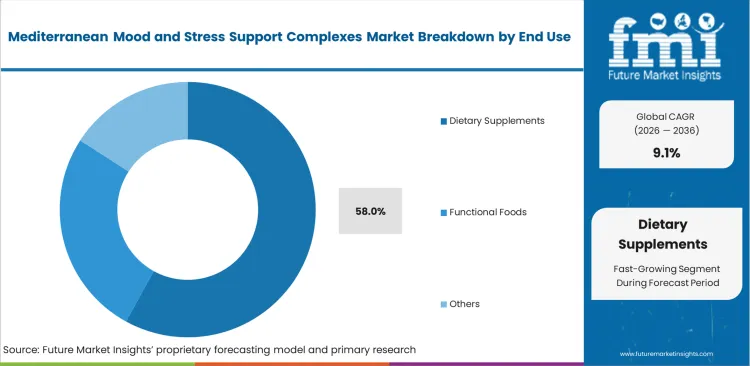
Consumers seeking accessible anxiety management prioritize immediate retail availability over prescription pathways. Creating effective herbal nutraceuticals requires less stringent pre-market approval than medical foods. Lower barriers to entry encourage rapid innovation cycles for novel adaptogenic beverages concepts, including functional beverages for relaxation support. At the same time, heavy over-the-counter dominance actually highlights a critical limitation being clinical nutrition channels demanding significantly more rigorous human trials than most current suppliers possess, propelling the dietary supplements to secure a 58.0% share as formulation teams navigate favorable regulatory environments. Brands ignoring medical-grade substantiation remain permanently locked out of high-margin practitioner distribution networks.
- Retail Distribution Speed: Standard dietary classifications allow rapid shelf placement across major pharmacy chains. Category buyers prioritize products with clear structural-function claims.
- Regulatory Compliance Thresholds: Existing monographs provide safe-harbor formulations for traditional Mediterranean herbs. Legal officers avoid costly novel food application processes.
- Consumer Self-Selection Behavior: Shoppers independently evaluate symptom relief without requiring specialized medical consultations. Marketing directors focus entirely on direct educational campaigns.
Mediterranean Mood and Stress Support Complexes Market Analysis by Sales Channel
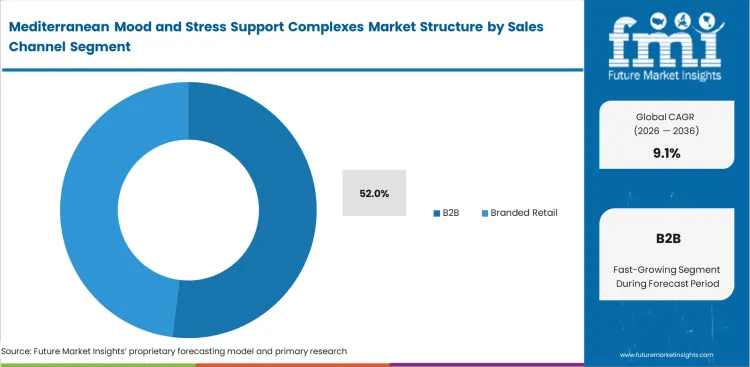
Specialized extraction capabilities require massive capital investment completely distinct from consumer branding operations. As sophisticated processors dominate the value chain, B2B ingredients accomplishes a 52.0% share, while supplying validated cognitive health supplement components requires millions in analytical testing infrastructure. Based on FMI's assessment, mediterranean mood support ingredient suppliers recognize that few retail brands possess this level of technical competency. White-labeling proprietary extracts to multiple consumer brands proves significantly more profitable for saffron extract manufacturers for mood supplements than competing directly in the saturated retail space. Retailers attempting backward integration routinely fail to achieve the necessary economies of scale for a viable nootropic supplement product.
- Extraction Facility Capital: Supercritical fluid extraction units demand extreme initial financial commitments. Operations directors must secure long-term client contracts.
- Analytical Certification Requirements: Providing continuous third-party potency verification creates immense overhead costs. Quality assurance managers maintain massive compliance databases.
- Custom Formulation Services: Supplying pre-mixed botanical blends simplifies manufacturing for downstream consumer brands. Sales executives command higher premiums for complete solutions.
Mediterranean Mood and Stress Support Complexes Market Drivers, Restraints, and Opportunities
Tighter regulation around synthetic additives is pushing formulation teams to look for clinically supported botanical alternatives. Brands that continue to rely on older synthetic stress-support ingredients may face greater retail and compliance pressure, especially in Europe. This is making early sourcing more important, particularly for premium natural ingredients used in cortisol support products. Many procurement teams now witness advance supply planning as a practical way to avoid disruption and protect product continuity.
Extreme variability in seasonal crop yields creates massive long-term pricing instability for specific Mediterranean botanicals. Unpredictable rainfall directly alters the final phytochemical density of harvested materials intended for a brain health supplement. Contracting for precise active molecule volumes becomes nearly impossible when underlying plant mass fluctuates wildly. Vertical integration offers partial protection, but regional climate realities strictly cap total agricultural output.
- Precision Fermentation Scaling: Engineering specific yeast strains to produce targeted saffron molecules bypasses agricultural limitations entirely. Biotech startup founders secure massive venture capital funding.
- Biomarker Tracking Integration: Pairing sleep and emotional balance supplement blends with consumer wearable data proves real-time efficacy. Digital product managers create highly sticky subscription models.
- Microbiome Pathway Validation: Mapping exactly how specific olive polyphenols influence gut flora opens novel serotonin supplement applications. Clinical researchers unlock entirely new patentable mechanisms.
Regional Analysis
Based on regional analysis, the market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa across 40 plus countries. The global landscape for these botanical complexes reveals a stark divide between regions cultivating the raw materials and those aggressively consuming the finished premium products. Extraction technology, localized agricultural dominance, and shifting regulatory frameworks, in the end, dictate the flow of these highly specialized ingredients across international borders.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.1% |
| Spain | 9.6% |
| Italy | 9.2% |
| France | 8.8% |
| Germany | 8.5% |
| United States | 8.4% |
| United Kingdom | 8.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Mediterranean Mood and Stress Support Complexes Market Analysis
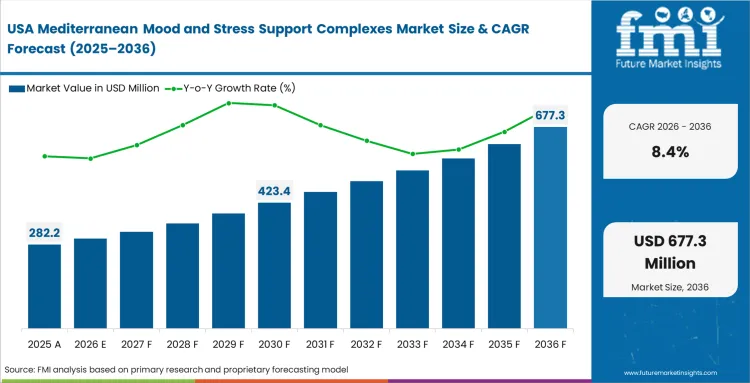
Instead of waiting for extensive pre-market clinical validation, formulation officers in North America prioritize speed to commercialization. To elevate their premium product tiers, these brands actively hunt for imported Mediterranean materials. Local extraction facilities simply lack the specific soil conditions needed to replicate optimal phytochemical densities, shaping the competitive dynamics across the continent. The procurement teams as a result, cultivate deep relationships with European suppliers to circumvent domestic agricultural shortfalls.
- United States: In the United States, demand for natural stress-support ingredients is rising as consumers show more interest in botanical alternatives to older synthetic formulations. The U.S. market for Mediterranean mood and stress support complexes is projected to grow at a CAGR of 8.4% from 2026 to 2036. This shift is leading brands to increase their use of imported ingredients such as saffron and lemon balm extracts, especially in products positioned around calm, mood balance, and daily stress support. For sourcing teams, that means managing longer supply chains and more complex import requirements. Companies that secure dependable access to clinically supported European extracts may gain an advantage over brands using less differentiated ingredient inputs.
FMI assesses, North America remains heavily dependent on cross-Atlantic supply chains to fuel its emotional wellness category. Until domestic biotechnology can reliably synthesize these exact botanical signatures, the region will operate primarily as an aggressive buyer rather than a primary producer of these specialized compounds.
Europe Mediterranean Mood and Stress Support Complexes Market Analysis
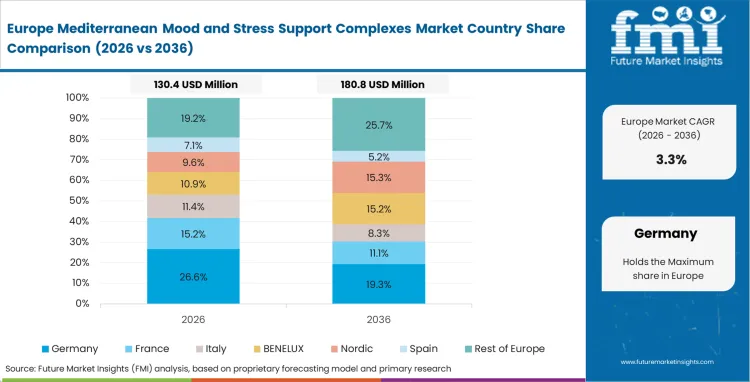
The supply chain reality across this continent is entirely defined by the aggressive vertical integration efforts of regional agricultural cooperatives. Proximity to traditional Mediterranean cultivation zones gives local processors the unique ability to control material quality straight from the seed to the final therapeutic extract. Meanwhile, strict European Food Safety Authority regulations mandate rigorous substantiation for any functional health claim. Successful enterprises distance themselves from generic literature, choosing instead to build massive proprietary clinical dossiers that easily justify their premium ingredient pricing.
- Spain: Spain has a strong position in this market because of its agricultural base and steady access to saffron and olive-derived raw materials. Local processors benefit from tighter control over supply, which helps them manage costs and improve extraction efficiency. Spain is projected to grow at 9.6% CAGR. That supply advantage also supports the country’s standing in premium botanical exports.
- Italy: Italy is building its position through extraction quality rather than raw material scale alone. Regional processors are investing in methods that better preserve delicate active compounds, especially polyphenols that can degrade during processing. Italy is forecast to expand at 9.2% CAGR. This technical focus is helping Italian suppliers hold a premium place in the market.
- France: In France, capabilities developed in fragrance and cosmetic extraction are supporting growth in nutraceutical ingredients as well. Producers are bringing that same attention to purity, profile, and consistency into botanical processing. France is expected to register 8.8% CAGR. That background gives local firms an advantage in premium ingestible formulations.
- Germany: Germany remains a quality-focused market where buyers place strong importance on testing, documentation, and controlled manufacturing standards. Suppliers that meet these expectations are better placed to serve more demanding channels. Germany is set to record 8.5% CAGR. The market rewards consistency and technical credibility more than price-led positioning.
- United Kingdom: In the United Kingdom, clean-label expectations are influencing how supplements are formulated and presented at retail. Brands are under more pressure to reduce synthetic excipients, which makes stability and handling of botanical ingredients more important. The United Kingdom is likely to grow at 8.1% CAGR. Suppliers that solve these formulation issues are in a better position to gain retail acceptance.
Per FMI’s analysis, Europe functions as both the agricultural heartland and the regulatory gatekeeper for the global category. The continent's processors will continue to command premium margins as long as they maintain their monopoly on clinically validated, geographically authentic raw materials.
Asia Pacific Mediterranean Mood and Stress Support Complexes Market Analysis
Across the Asian manufacturing landscape, massive contract infrastructure is rapidly pivoting away from generic Ayurvedic formulations in favor of premium European-sourced ingredients. Facilities focused entirely on exports now recognize the substantially higher margin potential attached to clinically validated Mediterranean botanicals. However, procurement teams face intense logistical hurdles as they scramble to build entirely new sourcing relationships with distant agricultural cooperatives. Facility profitability in the end hinges on how well these managers can balance steep import costs against lucrative final export margins.
- India: In India, contract manufacturers are strengthening their testing capabilities to work with more complex imported phytochemical ingredients. Many facilities are investing in advanced chromatography systems so they can meet tighter quality and documentation requirements from overseas clients. India is projected to record the fastest growth in this market, with revenue rising at a CAGR of 10.1%. As analytical standards improve, Indian manufacturers are becoming better placed to secure production mandates from international psychobiotic and botanical supplement brands.
FMI's report includes extensive analysis of emerging extraction hubs across Northern Africa and the Middle East. Shifting climate patterns force major agricultural cooperatives to explore novel cultivation zones for critical mediterranean herb nootropic complexes ingredients.
Competitive Aligners for Market Players
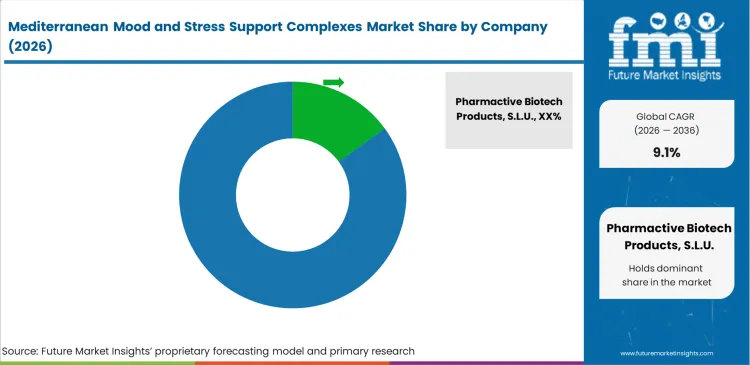
Proprietary clinical evidence remains one of the clearest competitive advantages in this market. It creates a gap between established Mediterranean mood-support ingredient suppliers and companies that buy botanicals on the spot market. For formulation teams, switching suppliers is not just a sourcing decision, because many product claims are tied to a specific branded ingredient and the studies behind it. That is why companies such as Pharmactive Biotech Products and Activ’Inside hold a strong position. Their clinically studied saffron and lemon balm extracts give brands a level of substantiation that is difficult and expensive to recreate with a lower-cost alternative. Euromed also strengthens its position by giving mid-sized brands access to supporting validation material, which helps reduce compliance risk and speeds up product development.
Supply consistency is another area where leading processors stand apart. Long-term agricultural mapping, raw material profiling, and tighter analytical control help them manage variation across harvests, especially when drought or shifting growing conditions affect plant chemistry. This matters because mood-support botanicals need more than volume; brands also want stable active compound levels from batch to batch. Indena has built this advantage through botanical fingerprinting and standardization capabilities that help maintain more reliable phytochemical ratios even when crop conditions are less predictable. Nektium addresses the same issue through continuous analytical verification, giving buyers more confidence in the consistency of ingredients used in stress- and anxiety-support products. Suppliers that cannot offer this level of control are more likely to stay in lower-value bulk powder segments.
At the same time, buyers are trying to reduce dependence on any one extract supplier. Some are doing that by shifting toward multi-ingredient systems, while others prefer partners that can provide ready-to-launch formats instead of raw materials alone. PharmaLinea supports this shift by offering private label Mediterranean mood-support solutions that help brands move faster without handling the full sourcing and qualification process themselves. The market is also changing as these ingredients move into more difficult delivery formats such as powders, stick packs, and beverages. In those applications, stability and taste become harder to manage. Monteloeder is addressing that challenge through delivery technologies and microencapsulation systems that improve ingredient performance in liquid and functional beverage products, opening the door to formats that standard extract suppliers often struggle to support.
Key Players in Mediterranean Mood and Stress Support Complexes Market
- Pharmactive Biotech Products, S.L.U.
- Activ’Inside
- Indena S.p.A.
- Euromed S.A.
- Monteloeder S.L.
- Nektium Pharma S.L.
- PharmaLinea d.o.o.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 500 million to USD 1,100 million, at a CAGR of 9.1% |
| Market Definition | Standardized botanical formulations derived from Mediterranean flora targeting neurological pathways comprise this sector. Efficacy requires specific phytochemical concentrations modulating the hypothalamic-pituitary-adrenal axis rather than generic raw plant material. |
| Segmentation | By Ingredient Base, By Formulation Type, By Delivery Format, By End Use, By Sales Channel, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa |
| Countries Covered | United States, Spain, Italy, France, Germany, United Kingdom, India, and others |
| Key Companies Profiled | Pharmactive Biotech Products, S.L.U., Activ’Inside, Indena S.p.A., Euromed S.A., Monteloeder S.L., Nektium Pharma S.L., PharmaLinea d.o.o. |
| Forecast Period | 2026 to 2036 |
| Approach | Wholesale botanical extract volume shipments cross-referenced with regional extraction yield rates. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
By Ingredient Base:
- Saffron extracts
- Lemon balm
- Lavender
- Chamomile
- Olive polyphenols
By Formulation Type:
- Multi-botanical blends
- Single extracts
- Mineral blends
- Probiotic blends
- Drink powders
By Delivery Format:
- Capsules
- Tablets
- Gummies
- Sachets
- Liquids
By End Use:
- Dietary supplements
- Functional foods
- Functional beverages
- Clinical nutrition
By Sales Channel:
- B2B ingredients
- Branded retail
- Practitioner sales
- Online direct
Regions:
- North America & Latin America
- United States
- Canada
- Mexico
- Brazil
- Argentina
- Chile
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia-Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- ANZ
- Middle East & Africa
- UAE
- Saudi Arabia
- South Africa
Bibliography
- Smith, T., Lang, C., & Craft, E. (2025). US sales of herbal supplements increase 5.4% in 2024. HerbalGram, 144, 46-69.
- Allcock, L., Mantzioris, E., & Villani, A. (2024). Adherence to a Mediterranean diet is inversely associated with anxiety and stress but not depression: A cross-sectional analysis of community-dwelling older Australians. Nutrients, 16(3), 366.
- Council for Responsible Nutrition. (2024). CRN survey shows consistent supplement usage with increase of specialty product use over time.
- Amadieu, C., et al. (2025). Effect of saffron extract supplementation on mood in healthy adults with subclinical symptoms of depression: A randomized, double-blind, placebo-controlled study. The American Journal of Clinical Nutrition.
- Lopresti, A. L., et al. (2025). An examination into the effects of a saffron extract (Affron) on depression, anxiety, stress, and sleep in adults with low mood: A randomized, double-blind, placebo-controlled trial. The Journal of Nutrition.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Regulatory substitution pathways forcing procurement directors to secure specific Mediterranean botanical supplies.
- Crocin standardization mechanisms allowing saffron extracts to command 31.0% category dominance.
- Vertical integration strategies protecting Spanish extraction facilities against crop yield fluctuations.
- Taste-masking requirements making capsule formats the preferred delivery system for astringent polyphenols.
- Clinical validation hurdles keeping dietary supplements highly separated from practitioner-only medical foods.
- Analytical verification investments driving Indian contract manufacturers toward 10.1% compound expansion.
- B2B extractor dominance restricting retail brands from achieving backward integration profitability.
- Clean label compliance pushing formulation chemists toward novel microencapsulation technologies over synthetic binders.
Frequently Asked Questions
What are Mediterranean mood and stress support complexes?
Standardized botanical formulations derived from Mediterranean flora targeting neurological pathways comprise this sector. Efficacy requires specific phytochemical concentrations modulating the hypothalamic-pituitary-adrenal axis rather than generic raw plant material.
Which ingredients dominate Mediterranean stress support formulas today?
Saffron extracts command a 31.0% position because specific crocin concentrations deliver verifiable serotonin reuptake modulation required by modern clinical substantiation standards.
Why is saffron used for mood support supplements over generic alternatives?
Extractors build massive clinical dossiers to justify premium pricing because targeted crocin profiles consistently alter neurotransmitter activity in ways generic plant mass cannot replicate.
How do formulators compare saffron lemon balm and lavender for mood supplements?
Chief scientific officers test these components vigorously, recognizing that combining lavender and lemon balm radically alters individual pharmacokinetic absorption rates and demands advanced microencapsulation technologies.
How does saffron vs ashwagandha for stress support differ operationally?
Ashwagandha falls entirely outside the Mediterranean geographic classification, pushing buyers seeking strictly European clean-label positioning to prioritize saffron for its highly localized, transparent supply chain.
What is the verdict on lemon balm vs melatonin for sleep and mood?
Brands aggressively substitute hormonal components with botanical extracts like lemon balm because consumers increasingly demand non-habit-forming botanical bioactives for mental wellness that bypass complex prescription regulations.
What is the current valuation of this specific category?
Valuation crosses USD 500 million in 2026, signaling a major procurement shift toward clinically verified Mediterranean botanical extraction networks.
How large will this sector become by the end of the forecast?
Total revenue reaches USD 1,100 million by 2036, driven primarily by impending European regulatory crackdowns on synthetic anxiety ingredients.
What specific growth rate characterizes this trajectory?
The sector advances at a 9.1% CAGR, reflecting intense formulation pressure to secure reliable, crop-dependent phytochemical yields.
What drives multi-botanical blends to dominate formulation types?
Securing a 46.0% share, multi-botanical blends satisfy consumer demands for comprehensive neurological support despite the intense stability challenges they present to manufacturing engineers.
How do capsules maintain their formatting advantage?
Capturing 37.0% of demand, capsules successfully hide the highly astringent and bitter taste profiles of concentrated Mediterranean polyphenols that routinely destroy gummy formulations.
Why do dietary supplements lead the end-use applications?
Commanding 58.0% share, standard dietary supplements offer rapid retail placement and require less stringent pre-market approval compared to strictly regulated clinical nutrition channels.
What structurally protects B2B ingredient suppliers?
Maintaining a 52.0% position, B2B extraction requires massive capital investment in supercritical fluid units and continuous third-party potency verification that retail brands cannot replicate.
How does India outpace global average growth?
Expanding at 10.1%, Indian contract manufacturers secure highly lucrative export contracts by heavily upgrading analytical testing capabilities for complex imported European phytochemicals.
Why does Spain maintain such a strong geographic presence?
Tracking at 9.6%, Spanish extraction facilities leverage their immediate proximity to massive domestic saffron and olive cultivation zones to dictate global raw material pricing.
What structural friction slows adoption for mid-tier brands?
Extreme variability in seasonal crop yields alters final phytochemical density, making precise active molecule volumes nearly impossible to guarantee without multi-year agricultural contracts.
How do Italian processors differentiate their output?
Growing at 9.2%, Italian chief scientific officers develop advanced solvent-free extraction methodologies specifically engineered to preserve delicate polyphenol structures during processing.
What role do European Food Safety Authority regulations play?
Strict regulatory frameworks force formulation directors to base functional product claims exclusively on batch-specific proprietary trials rather than generic historical plant literature.
How do synthetic substitutions impact purchasing behavior?
Widespread origin fraud and synthetic adulteration in spot markets push quality control directors to demand complete vertical integration from seed to final standardized extract.
What limits functional beverage applications for these botanicals?
High concentrations of active Mediterranean compounds cause severe excipient compatibility challenges and unpredictable precipitation in liquid environments over extended shelf lives.
How do large consumer brands resist extractor lock-in?
Formulation officers actively design complex multi-botanical formulas where no single agricultural supplier controls the entire synergistic efficacy profile of the final product.
What specific capability do clean-label mandates require?
Packaging engineers must stabilize extremely hygroscopic botanical powders using only natural shell technologies because major pharmacy chains strictly ban traditional synthetic moisture barriers.
How does agricultural climate change affect future pricing?
Shifting rainfall patterns directly cap regional agricultural output, forcing major cooperatives to explore entirely novel cultivation zones to maintain critical botanical ingredient supplies.
What potential does precision fermentation offer?
Engineering specific yeast strains to produce targeted saffron molecules bypasses traditional agricultural limitations entirely and provides procurement managers with completely predictable unit costs.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Base
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Base , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Base , 2026 to 2036
- Saffron Extracts
- Lavender
- Others
- Saffron Extracts
- Y to o to Y Growth Trend Analysis By Ingredient Base , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Base , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Formulation Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Formulation Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Formulation Type, 2026 to 2036
- Multi-Botanical Blends
- Single Extracts
- Others
- Multi-Botanical Blends
- Y to o to Y Growth Trend Analysis By Formulation Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Formulation Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Delivery Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Delivery Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Delivery Format, 2026 to 2036
- Capsules
- Tablets
- Gummies
- Capsules
- Y to o to Y Growth Trend Analysis By Delivery Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Delivery Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Dietary Supplements
- Functional Foods
- Others
- Dietary Supplements
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- B2B
- Branded Retail
- B2B
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Base
- By Formulation Type
- By Delivery Format
- By End Use
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Pharmactive Biotech Products, S.L.U.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Activ’Inside
- Indena S.p.A.
- Euromed S.A.
- Monteloeder S.L.
- Nektium Pharma S.L.
- Pharmactive Biotech Products, S.L.U.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Base , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Formulation Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Base
- Figure 6: Global Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Formulation Type
- Figure 9: Global Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Delivery Format
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Sales Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Base
- Figure 32: North America Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Formulation Type
- Figure 35: North America Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Delivery Format
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Sales Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Base
- Figure 48: Latin America Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Formulation Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Delivery Format
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Base
- Figure 64: Western Europe Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Formulation Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Delivery Format
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Base
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Formulation Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Delivery Format
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Base
- Figure 96: East Asia Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Formulation Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Delivery Format
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Base
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Formulation Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Delivery Format
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Base , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Base , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Base
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Formulation Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Formulation Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Formulation Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Delivery Format
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

