Mediterranean Women’s Cycle Support Ingredient Systems Market
The Mediterranean Women’s Cycle Support Ingredient Systems Market Is Segmented By Ingredient System (Magnesium-B6 Blends, Omega-3 Blends, Probiotic Blends, Botanical Blends, Multi-Ingredient Blends), Format (Capsules, Gummies, Powders, Liquids, Sachets), Application (PMS Support, Cramp Relief, Mood Support, Bloating Support, Cycle Balance), Channel (Online Retail, Pharmacies, Health Stores, Clinics, Subscriptions), Ingredient Origin (Mediterranean Botanicals, Marine Lipids, Mineral Systems, Fermented Cultures, Vitamin Premixes), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mediterranean Women’s Cycle Support Ingredient Systems Market Size, Market Forecast and Outlook By FMI
The mediterranean women’s cycle support ingredient systems market scaled a USD 211.7 million in 2025 with a consistent progress assessing it to surpass the predicted value of USD 228.0 million in 2026 at a CAGR of 7.7% during this forecast period. The market is expected to reach USD 478.7 million by 2036, as retailers increasingly require clinical substantiation before accepting cycle support formulations, pushing formulators toward standardized regional botanical extracts.
Formulators face intense pressure from major retail category managers who now demand double-blind clinical validation for PMS claims before allocating shelf space. Delaying this qualification cycle costs mid-tier brands their distribution agreements within eighteen months. These clinical requirements also lock procurement directors into specific Mediterranean cultivation supply chains, because changing ingredient origins would invalidate existing efficacy data. FMI analysis indicates women's health supplement formulations utilizing precise regional saffron or chasteberry cannot easily substitute geographic origins without invalidating existing efficacy data.
Summary of Mediterranean Women’s Cycle Support Ingredient Systems Market
- Mediterranean Women’s Cycle Support Ingredient Systems Market Definition:
- Clinically substantiated botanical and nutritional complexes target specific physiological phases across menstrual cycles. These inputs require precise bioactive standardization and robust stability data for functional formulation.
- Demand Drivers in Market:
- Shelf-space qualification requirements force retail category managers to mandate double-blind clinical data for any cycle-related product claims.
- High formulation failure rates push contract manufacturing operations directors to source pre-validated synergistic complexes over individual raw materials.
- Complex regulatory monographs compel compliance officers to source ingredients from documented Mediterranean agricultural supply chains.
- Key Segments Analyzed in FMI Report:
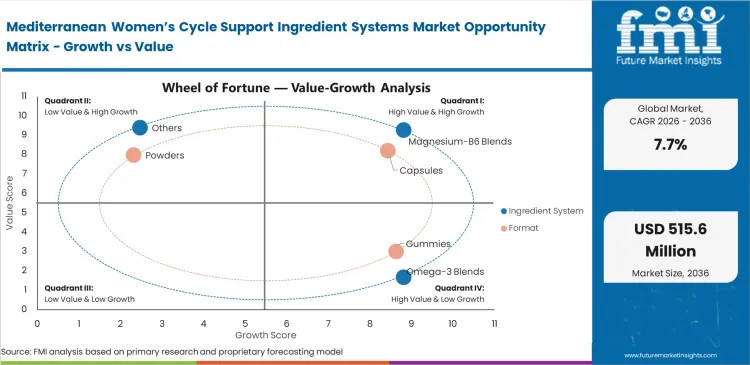
- Magnesium-B6 Blends: 31.0% share in 2026, driven by superior solubility metrics in automated encapsulation lines.
- Capsules: 38.0% share in 2026, satisfying stringent stability requirements for volatile botanical extracts.
- PMS Support: 36.0% share in 2026, mirroring consumer demand for clinically validated premenstrual interventions.
- Online Retail: 44.0% share in 2026, enabled by direct-to-consumer brands controlling extensive educational narratives.
- Mediterranean Botanicals: 34.0% share in 2026, anchored by proprietary regional extraction technologies.
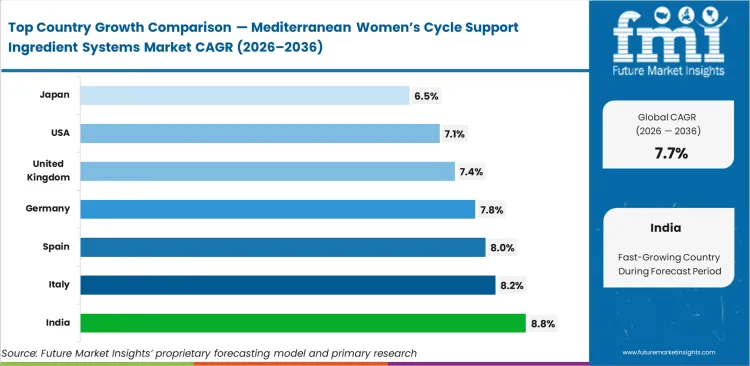
- India: 8.8% CAGR, reflecting massive domestic pivot toward high-margin export-grade active ingredient processing.
- Analyst Opinion at FMI:
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, points out: "Purchasing managers often assume that buying active ingredients separately will lower unit costs. In practice, the outcome is not always that simple. FMI notes that combining individual Mediterranean botanicals with standard vitamins can create moisture-related stability issues, especially during warmer production months. That can lead to batch failures, slower processing, and shorter shelf life. Because of this, formulators often prefer pre-integrated ingredient systems that run more smoothly in production and help maintain product quality. Teams that focus only on raw material cost may end up losing those savings through manufacturing delays and product instability."
- Strategic Implications / Executive Takeaways:
- Formulators must secure long-term extraction contracts to guarantee bioactive consistency across multi-year retail distribution agreements.
- Contract manufacturers face urgent requirements to upgrade blending equipment for handling highly volatile regional botanical inputs.
- Quality control directors risk entire product lines by substituting native Mediterranean extracts with cheaper generic equivalents.
- Methodology:
- Interviews with formulation chemists reveal actual factory-floor blending challenges.
- Regulatory registry analysis exposes accelerating shifts toward clinically substantiated active components.
- Import volume tracking provides grounded baseline valuation for specialized regional agricultural outputs.
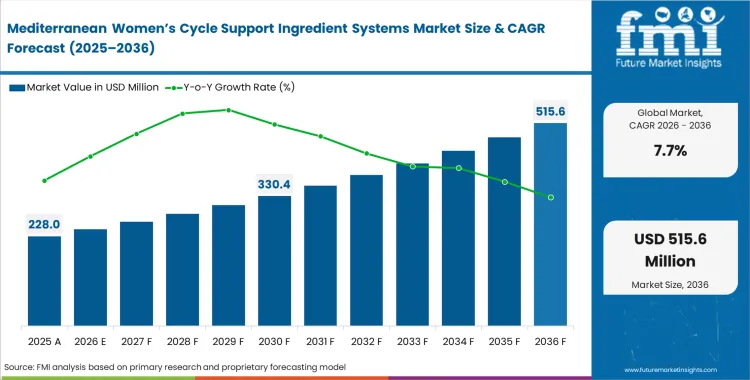
Contract manufacturers need to match the dissolution performance of complex botanical blends to that of standard vitamin premixes before brands can scale into mass retail. Once operations managers solve this manufacturing problem, higher production volumes bring unit costs down. Consistent batch quality allows brands to move from health food stores into national pharmacy and grocery chains.
India leads regional growth at 8.8%, as domestic contract manufacturers are upgrading their facilities to produce export-grade nutraceuticals. Italy follows at 8.2% due to proximity to native botanical supply chains. Spain grows at 8.0%, as manufacturers use existing marine lipid processing equipment to handle botanical encapsulation. Germany registers 7.8%, shaped by strict herbal medicine monographs that set ingredient quality standards. The United Kingdom grows at 7.4% as pharmacy chains standardize their wellness product ranges. The United States shows 7.1%, driven by direct-to-consumer brand launches relying on pre-validated formulations. Japan grows at 6.5%, as an aging population increases demand for perimenopausal support products. Quality standards differ across these regions, and facilities that meet export-grade requirements are better placed to win international contracts.
Mediterranean Women’s Cycle Support Ingredient Systems Market Definition
The Mediterranean Women's Cycle Support Ingredient Systems market covers clinically substantiated raw material complexes formulated to address physiological fluctuations across menstrual phases. This category uses regionally sourced agricultural inputs that meet precise bioactive standards, rather than generic nutritional isolates. Generic multivitamins without cycle-specific dosing protocols are outside this category. Procurement officers review stability data and clinical trial endpoints when selecting these ingredient systems, not basic nutritional content.
Mediterranean Women’s Cycle Support Ingredient Systems Market Inclusions
Scope covers standardized botanical extracts natively cultivated around mediterranean basin geographies. Marine lipid complexes formulated for specific prostaglandin regulation fall inside these parameters. FMI assesses menstrual care formulations containing synergistic magnesium-B6 ratios optimized for neurotransmitter synthesis as core inclusions. Fermented probiotic cultures targeting estrogen metabolization within gut microbiomes represent critical components. Delivery matrices enabling controlled release of specific bioactives qualify for inclusion.
Mediterranean Women’s Cycle Support Ingredient Systems Market Exclusions
Synthetic hormone replacement therapies require entirely different regulatory pathways and sit outside this framework. Generic calcium carbonate inputs lack necessary cycle-specific clinical data. General dietary supplements focused on overarching wellness without targeted menstrual phase clinical endpoints fail our inclusion criteria. Single-ingredient commodity vitamins lacking synergistic formulation data remain excluded.
Mediterranean Women’s Cycle Support Ingredient Systems Market Research Methodology
- Primary Research: Chief formulation officers and procurement directors at specialized nutraceutical contract manufacturers.
- Desk Research: European Food Safety Authority novel food registries and proprietary clinical trial databases focusing on dysmenorrhea interventions.
- Market-Sizing and Forecasting: Verified botanical extract import volumes across major European and North American processing hubs.
- Data Validation and Update Cycle: Independent extraction yield data cross-referenced against major contract manufacturer purchasing records.
Segmental Analysis
Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis by Ingredient System
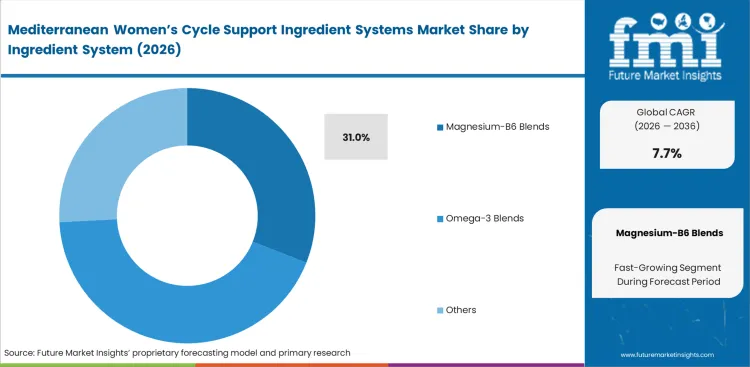
Magnesium-B6 blends account for 31.0% of total revenue share in the hormonal health supplements during the forecast period. This high share reflects their operational reliability in large-scale production environments. This mineral-vitamin combination helps control moisture when used with volatile botanical extracts sourced from different regions. Stable moisture levels reduce formulation inconsistencies during processing. Production teams define exact blend ratios to avoid equipment blockages, especially during high humidity conditions in summer months. Even minor deviations in composition can disrupt continuous manufacturing lines. These hormonal health supplements formulations also function as anti-caking systems, though this role is rarely highlighted by formulation chemists. The flow stability they provide supports smooth encapsulation and uniform fill weight. Manufacturers that replace these blends with lower-cost individual ingredients often face frequent line stoppages. These substitutions also weaken capsule structure and reduce overall batch quality.
- Machine Flow Optimization: Pre-granulated matrices prevent powder bridging inside automated hopper systems. Operations managers achieve continuous production runs without requiring costly humidity-controlled cleanroom upgrades.
- Stability Protocol Preservation: Controlled particle sizing ensures uniform active ingredient distribution throughout every batch. Quality directors avoid costly product recalls stemming from inconsistent dosing across individual capsules.
- Formula Consolidation: Blended mineral complexes eliminate three separate weighing steps during initial staging. Floor supervisors reduce labor costs while simultaneously eliminating multiple points of potential human error.
Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis by Format
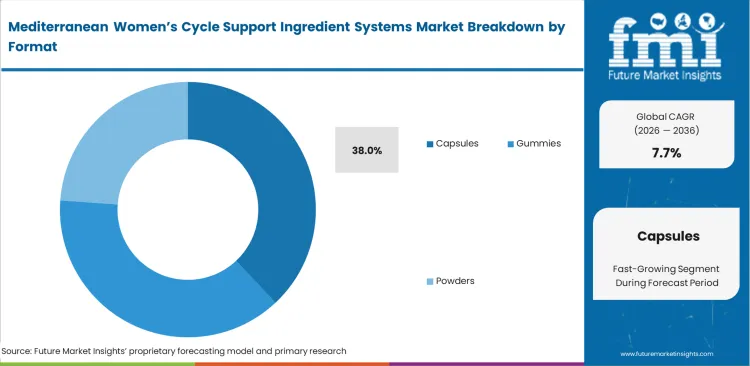
Avoiding taste-masking steps shortens product development timelines for formulations that include highly bitter regional extracts. This approach supports faster iteration cycles for formulation teams working on stable delivery formats. FMI analysis found that sealing active botanicals within capsule shells removes up to 12 weeks of flavor development work in the stress relief supplement segment. This reduction lowers formulation costs and speeds up product launch timelines. Capsules account for 38.0% of revenue share in the forecast period. This share reflects their suitability for complex botanical formulations. Marketing teams often promote chewable formats, but procurement teams prioritize two-piece capsule shells for their moisture resistance. These shells protect sensitive probiotic strains from degradation during storage and transport.
- Flavor Profile Bypass: Hermetic sealing isolates intensely bitter compounds from consumer taste receptors. Formulation scientists incorporate maximum clinical dosages without utilizing expensive artificial masking agents.
- Moisture Barrier Integrity: Standardized shell compositions protect highly hygroscopic extracts from ambient humidity degradation. Quality control officers guarantee longer shelf life across diverse retail environments.
- Rapid Iteration Capacity: Standard shell sizing requires zero custom tooling changes on manufacturing floors. Product development teams execute rapid formula updates without incurring extensive equipment changeover delays.
Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis by Application
Formulators face intense pressure balancing broad physiological claims against specific regulatory evidence requirements, reflecting stringent buyer demands for clearly validated efficacy markers. FMI's analysis indicates period cramp supplement retail category managers refuse shelf placement without double-blind trials demonstrating specific symptom reduction. Clinical directors utilize this application category precisely because standard measurement scales exist for premenstrual parameters, pushing the PMS support by a 36.0% share in command. Brands wanting to market general cycle balance discover that vague wellness claims fail completely during major pharmacy procurement reviews.
- Clinical Endpoint Validation: Standardized symptom tracking allows clear statistical proof of formula efficacy. Regulatory affairs directors easily secure necessary compliance approvals for specific physiological claims.
- Retail Buyer Acceptance: Quantifiable data satisfies strict corporate pharmacy procurement guidelines. Distribution managers unlock premium shelf placement across major national drug store chains.
- Consumer Expectation Alignment: Specific symptom targeting matches exact consumer search queries. Brand managers achieve significantly lower customer acquisition costs compared to broad wellness positioning.
Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis by Channel
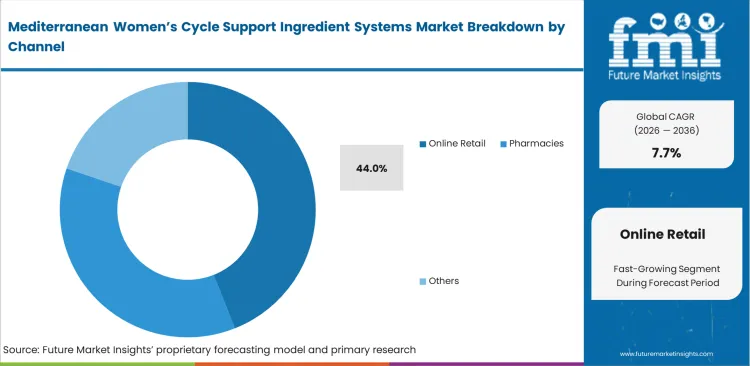
Complex formulation narratives require expansive educational real estate to justify premium pricing structures. Online retail holds 44.0% share, providing vital digital space for extensive ingredient substantiation. FMI observes that women probiotic brand managers utilize expansive web properties to detail specific Mediterranean agricultural sourcing practices. Brick-and-mortar pharmacy cartons lack sufficient physical space for explaining nuanced synergistic mechanisms. Digital marketing directors leverage this channel to connect direct clinical trial data with immediate purchasing actions. Companies relying strictly on physical retail presence struggle continuously to communicate why their standardized botanical systems cost double the price of generic vitamins.
- Educational Real Estate Expansion: Unlimited digital space permits deep dives into proprietary extraction methods. Brand managers successfully justify premium pricing through extensive clinical data presentation.
- Subscription Model Integration: Direct platforms enable automated monthly replenishment tied precisely to user cycles. Financial officers secure predictable recurring revenue streams while lowering overall customer churn.
- Feedback Loop Acceleration: Direct consumer purchasing data provides immediate insights regarding formula efficacy. Product development teams iterate new synergistic blends based on real-time symptom relief reports.
Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis by Ingredient Origin
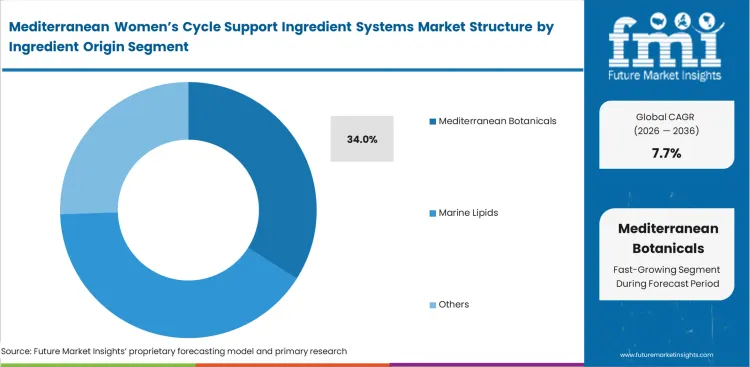
Mediterranean botanicals accomplish a 34.0% share as generic agricultural inputs consistently fail to replicate precise bioactive profiles required by regulatory monographs, acting as foundational pillars for clinically validated formulations. FMI analysts note that procurement directors cannot substitute cheaper Asian-sourced equivalents without completely invalidating their existing efficacy data. Original benchmark trials utilized herbal supplements cultivated specifically within unique Mediterranean microclimates. Formulation officers understand that substituting precise geographic inputs alters crucial phytochemical ratios, triggering immediate regulatory non-compliance. Brands attempting geographic substitution to reduce costs immediately lose their ability to print specific health claims on packaging.
- Efficacy Data Preservation: Sourcing from original clinical trial microclimates guarantees consistent phytochemical ratios. Regulatory directors maintain active compliance without requiring expensive new validation studies.
- Supply Chain Monopolization: Limited geographic growing regions create natural barriers against mass-market generic encroachment. Procurement officers secure exclusive regional contracts to lock competitors out of high-quality material.
- Extraction Yield Optimization: Native growing conditions produce superior concentrations of targeted bioactive compounds. Manufacturing engineers extract maximum value per kilo during downstream processing stages.
Mediterranean Women’s Cycle Support Ingredient Systems Market Drivers, Restraints, and Opportunities
Retail pharmacy category managers now mandate absolute clinical substantiation before assigning premium shelf space to new cycle support formulations. Brands face immediate commercial exile if they cannot provide specific vitamin and mineral supplement double-blind data proving symptom reduction. This strict gatekeeping forces procurement officers to abandon cheap, isolated commodity ingredients. Formulators must invest in specialized, pre-validated systems because retailers refuse to accept extrapolated data from disconnected raw materials.
Severe hygroscopic instability during commercial-scale blending represents a massive operational friction point. Manufacturing directors discover that mixing highly concentrated regional extracts with standard vitamins creates immediate powder caking inside automated encapsulation hoppers. Facility managers must install million-dollar humidity control infrastructure or suffer crippling batch failure rates during summer months. This physical reality forces many food supplement ingredients formulators to dilute their active concentrations simply to keep production lines running.
- Extraction Infrastructure Upgrades: Installing advanced low-temperature processing technology preserves volatile phytochemicals during initial harvesting. Facility operators capture premium pricing tiers by offering highly stable active compounds.
- Biomarker Tracking Integration: Aligning specific daily ingredient dosages with digital cycle-tracking applications creates powerful consumer stickiness. Digital product directors establish impenetrable recurring revenue loops by syncing deliveries with anticipated physiological phases.
- Clinical Dossier Licensing: Developing proprietary, pre-validated EPA DHA omega 3 ingredients matrices allows ingredient suppliers to lease their efficacy data. Procurement directors eagerly pay premium licensing fees to bypass multi-year independent trial requirements entirely.
Regional Analysis
The regional landscape for Mediterranean women’s cycle support ingredient market reveals a sharp divide between jurisdictions focused on premium extraction capabilities and those prioritizing rapid direct-to-consumer commercialization. Distinct compliance monographs and shifting domestic manufacturing strategies heavily dictate exactly how fast specific geographies adopt these clinically validated ingredient complexes. The market is segmented into North America, Europe, Asia Pacific, and Rest of World across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.8% |
| Italy | 8.2% |
| Spain | 8.0% |
| Germany | 7.8% |
| United Kingdom | 7.4% |
| United States | 7.1% |
| Japan | 6.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis
Digital marketing directors across North America bypass traditional retail gatekeepers completely, aiming their physiological symptom solutions directly at consumers via social platforms. This aggressive direct-to-consumer approach necessitates incredibly stable digestive health supplements matrices that will not degrade during unregulated warehouse storage or erratic shipping conditions. Operations managers heavily favor robust capsule structures over complex liquid formats to ensure product efficacy remains intact upon delivery. To build a strong defensive moat against fast-moving domestic copycats, local procurement teams zero in on exclusive supply agreements for clinically validated regional extracts.
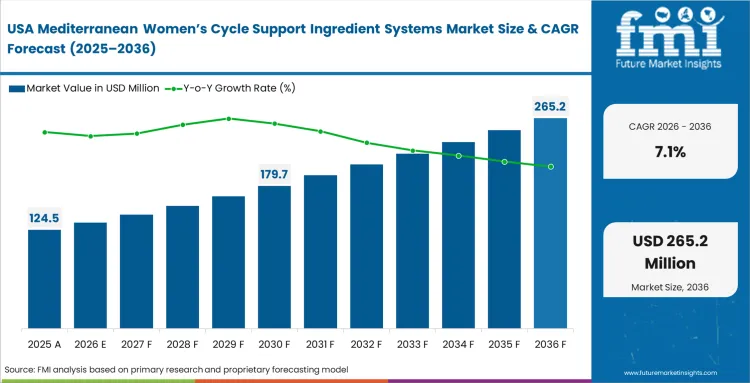
- United States: Strict venture capital expectations force independent brands to execute rapid, high-margin product launches across the country. Financial directors want to skip long research and development cycles entirely, prioritizing ingredient systems that already possess clinical validation. The United States demand for these standardized botanical complexes is expected to scale at a 7.1% CAGR as this heavy reliance on turnkey botanical formulations opens a massive commercial gateway for specialized contract manufacturers who own robust clinical dossiers.
FMI's report includes extensive analysis covering Canada and Mexico within the North American framework. Expanding distribution networks across these unlisted territories accelerates the structural transition from basic multivitamin consumption toward targeted physiological interventions. The regulatory harmonization efforts between cross-border health agencies further streamline the future importation of standardized Mediterranean extracts into these expanding commercial zones.
Europe Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis
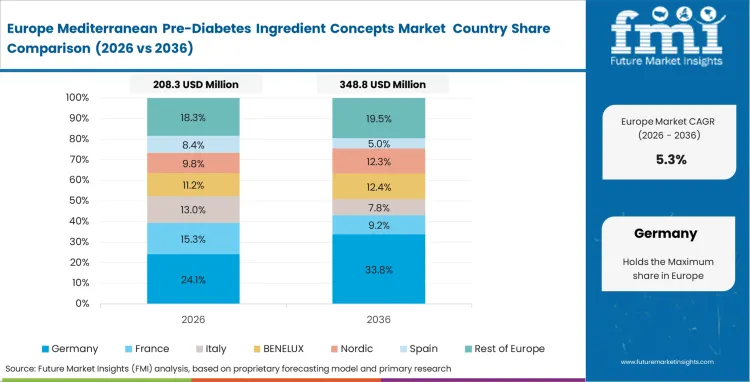
Exact sourcing requirements are strictly governed by complex regulatory monographs throughout established pharmacy networks across Europe. Compliance officers operate under intense scrutiny regarding both the specific origins of botanicals and their precise extraction methodologies. This unyielding regulatory climate essentially forces herbal nutraceuticals purchasing directors to cultivate deep, lasting relationships with agricultural suppliers located directly around native Mediterranean basins. Attempting to substitute these inputs with Asian-sourced alternatives is out of the question, as it would instantly trigger massive and costly relicensing procedures. Local formulation teams, as a result, concentrate entirely on optimizing synergistic matrices to extract maximum bioavailability while staying within these rigid geographic limits.
- Italy: Italy sits close to native botanical cultivation zones, and cycle support ingredient sales record an 8.2% CAGR over the forecast period. Raw materials reach processing units faster, which limits degradation during transit. This proximity helps maintain extract potency at the initial stages. Supply chain teams secure better-quality inputs before chemical breakdown begins. Strong control over sourcing supports Italy’s role in the European supply chain.
- Spain: Spain relies on adapted marine lipid infrastructure to process complex botanical actives. The country records an 8.0% CAGR in cycle support ingredient demand over the forecast period. Facilities handle multi-compound formulations without major process changes. This capability supports contract manufacturing demand from across Europe. Encapsulation systems already in place ensure consistent batch output.
- Germany: Consumer familiarity with standardized herbal medicines remains high across Germany. Cycle support ingredient sales grow at a 7.8% CAGR over the forecast period. Medical teams expect detailed pharmacological backing before approvals. Products must meet strict validation benchmarks to enter regulated channels. This requirement shapes broader acceptance standards across Europe.
- United Kingdom: Retail pharmacies in the United Kingdom apply strict compliance checks across wellness products. Cycle support ingredient sales expand at a 7.4% CAGR over the forecast period. Procurement teams review stability data before listing decisions. Only formulations meeting defined benchmarks reach store shelves. Approved brands gain stronger positioning against online-led competition.
FMI's report includes extensive analysis covering France and the broader Rest of Europe category. Scaling localized extraction infrastructure across these unlisted territories accelerates the structural shift away from generic vitamins toward highly targeted physiological interventions. In addition, unified European Food Safety Authority guidelines continue to shape exactly how new regional botanical blends achieve commercial clearance across borders.
Asia Pacific Mediterranean Women’s Cycle Support Ingredient Systems Market Analysis
Operators of massive domestic formulation facilities across this region are executing a strategic pivot toward processing premium, export-grade active ingredients. Regional plant managers fully realize that churning out basic gut health supplement vitamins offers minimal margins when compared to the lucrative potential of highly specialized synergistic complexes. These facilities are rapidly upgrading their extraction technologies to safely process sensitive botanical inputs that comply with strict Western regulatory monographs. To feed these upgraded manufacturing lines, local procurement teams aggressively import raw Mediterranean agricultural products, aiming to refine them domestically into high-value finished matrices.
India: India’s manufacturing base is shifting from generic drug output toward complex nutraceutical blending. Cycle support ingredient processing and consumption in the country is set to expand at an 8.8% CAGR from 2026 to 2036, supported by export-driven demand. Seasonal humidity often disrupts batch stability, which pushes facility heads to invest in moisture-control systems. These upgrades lower rejection rates and stabilize output quality. This shift positions Indian manufacturers as key partners for global wellness brands handling large-scale, formulation-intensive production.
Japan: Japan’s aging population is redirecting product focus toward perimenopausal support. Cycle support ingredient sales in Japan are projected to rise at a 6.5% CAGR over the forecast period as demand concentrates among older consumers. Formulation teams adjust delivery systems to suit slower digestion and absorption changes. This leads to a stronger focus on bioavailability rather than higher dosage. Domestic brands are tightening formulation standards, which changes how they compete with imported supplements.
FMI's report includes extensive analysis covering China and Australia within the Asia Pacific landscape. Rising middle-class purchasing power across these unlisted territories accelerates the consumer transition from basic nutritional supplements toward targeted physiological interventions. The advancements in domestic supply chain logistics further ensure that imported volatile extracts maintain their clinical integrity upon arrival at regional processing centers.
Competitive Aligners for Market Players
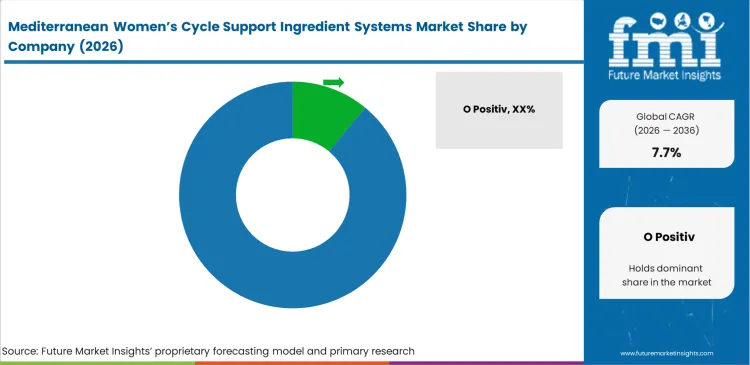
Competition in this market is driven by buyers who care more about consistency, stability, and whether a formulation can hold up across retail, pharmacy, and e-commerce channels, rather than who can buy ingredients at the lowest cost. That gives established brands such as Thorne, MegaFood, O Positiv, and HUM Nutrition a key advantage, since they already work with tested ingredient systems and dependable supply partners.
Another barrier is validation, as it is easier to launch a new blend, but much harder to support it with stability data, efficacy evidence, and repeatable manufacturing. Resulting with brands such as Elix Healing and Boiron benefit from years of formulation work and product credibility, which makes it harder for newer entrants to compete in premium channels.
Many mediterranean botanicals are not easily interchangeable, so changing source regions or extract profiles can affect both product quality and claim support, thereby making supply an imperative aspect. As a result, companies with stronger supplier relationships and better control over blending and sourcing are in a better position to manage crop variation, maintain consistency, and protect retail contracts.
Key Players in Mediterranean Women’s Cycle Support Ingredient Systems Market
- O Positiv
- HUM Nutrition
- OLLY
- Elix Healing
- Thorne
- MegaFood
- Boiron
Scope of the Report
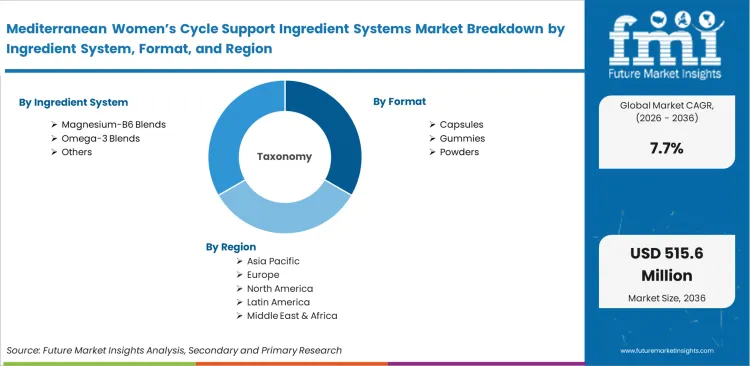
| Metric | Value |
|---|---|
| Quantitative Units | USD 228.0 million to USD 478.7 million, at a CAGR of 7.7% |
| Market Definition | Clinically substantiated raw material complexes formulated specifically to address physiological fluctuations across menstrual phases. |
| Segmentation | Ingredient system, Format, Application, Channel, Ingredient origin |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, Brazil, Mexico |
| Key Companies Profiled | O Positiv, HUM Nutrition, OLLY, Elix Healing, Thorne, MegaFood, Boiron |
| Forecast Period | 2026 to 2036 |
| Approach | Verified botanical extract import volumes across major processing hubs. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Ingredient System
- Magnesium-B6 blends
- Omega-3 blends
- Probiotic blends
- Botanical blends
- Multi-ingredient blends
Format
- Capsules
- Gummies
- Powders
- Liquids
- Sachets
Application
- PMS support
- Cramp relief
- Mood support
- Bloating support
- Cycle balance
Channel
- Online retail
- Pharmacies
- Health stores
- Clinics
- Subscriptions
Ingredient Origin
- Mediterranean botanicals
- Marine lipids
- Mineral systems
- Fermented cultures
- Vitamin premixes
Regions:
- North America
- United States
- Asia Pacific
- Japan
- China
- India
- Europe
- Germany
- United Kingdom
- France
- Italy
- Greece
- Greece
- Spain
- Middle-East
- United Arab Emirates
- Turkey
- Morocco
- Egypt
Bibliography
- Robinson, J., Brown, N., Beck, K. L., O’Sullivan, J. M., von Hurst, P. R., & Kruger, R. (2025). Effect of nutritional interventions on the psychological symptoms of premenstrual syndrome in women of reproductive age: A systematic review of randomized controlled trials. Nutrition Reviews, 83(2), 280–306.
- Oboza, P., Bień, A., & Iwanowicz-Palus, G. (2024). Relationships between premenstrual syndrome (PMS) and diet composition, dietary patterns and eating behaviors. Nutrients, 16(12), 1911.
- Lin, K. C., Hsu, T. J., Yeh, M. L., Su, Y. T., & Tsai, I. J. (2024). Vitamin D supplementation for patients with dysmenorrhoea: A meta-analysis with trial sequential analysis of randomised controlled trials. Nutrients, 16(7), 1089.
- Zakaria, I. A., Mohammed Zain, N. A., Teik, C. K., Abu, M. A., Zainuddin, A. A., Abdul Aziz, N. H., Safian, N., Mohd Mokhtar, N., Raja Ali, R. A., & Ngu, B. K. (2024). The role of probiotics in improving menstrual health in women with primary dysmenorrhoea: A randomized, double-blind, placebo-controlled trial (the PERIOD study). Women’s Health, 20, 17455057241234524.
- Güzeldere, H. K. B., Özcan, M., & Yıldız, A. (2024). The relationship between dietary habits and menstruation problems in women: A cross-sectional study. BMC Women’s Health, 24, 397.
- Ajorlouie, Z., Moshkian, P., Baghdadi, G., Amiri, R., Biglari, F., & Rahimlou, M. (2025). The association between the Mediterranean diet and the prime diet quality score and polycystic ovary syndrome: A case control study. BMC Nutrition, 11(1), 80.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Clinical validation bottlenecks preventing commodity extract suppliers from securing mass retail distribution.
- Formulation instability resulting from substituting precise magnesium threonate supplement matrices with disparate raw material inputs.
- Rigid compliance monographs determining exact geographic sourcing parameters for functional botanical imports.
- Strategic tension between maintaining proprietary efficacy dossiers and achieving mass-market unit economics.
- Specialized manufacturing upgrades required to handle volatile hygroscopic regional crop yields.
- Digital channel dominance enabled by extensive menopause supplement educational requirements regarding cycle biochemistry.
- Rapid domestic pivot by Indian contract manufacturers targeting high-margin active ingredient processing.
- Direct-to-consumer brand vulnerability stemming from reliance on unvalidated, generic extraction methodologies.
Frequently Asked Questions
What value will Mediterranean Women’s Cycle Support Ingredient Systems record in 2026?
Total revenue is poised to cross USD 228.0 million in 2026 based on surging clinical requirements.
What total valuation will this sector reach by 2036?
Cumulative buildup takes total valuation to USD 478.7 million through 2036.
What compound growth rate applies to this category?
Sustained investment drives a 7.7% CAGR across the forecast period.
Why do Magnesium-B6 blends command the largest ingredient share?
Formulation chemists utilize these specific omega 3 matrices primarily for superior moisture control during automated encapsulation rather than pure nutritional efficacy.
Why do capsules maintain dominance over gummies?
Standard shell sizing completely bypasses expensive flavor-profiling development needed to mask intensely bitter regional extracts.
What structural condition drives PMS support applications?
Retail buyers demand double-blind symptom reduction data which currently exists primarily within standardized premenstrual clinical endpoints.
Why does online retail capture maximum sales volume?
Complex botanical narratives require expansive digital real estate to justify premium pricing structures over standard multivitamin offerings.
What prevents formulators from substituting Asian botanicals for Mediterranean inputs?
Altering geographic origin invalidates existing efficacy dossiers and immediately triggers massive regulatory relicensing procedures.
Why does India lead regional growth rates?
Domestic facility operators are aggressively upgrading extraction technology to secure premium export-grade contract manufacturing agreements.
How does Italian proximity shape supply chain dynamics?
Direct access to native cultivation sites eliminates transit degradation and allows local processors to monopolize potent primary outputs.
What structural difference explains Spanish growth?
Manufacturing engineers repurpose established marine lipid processing infrastructure to encapsulate complex botanical compounds seamlessly.
Why do German consumers drive strict pharmaceutical standards?
Deep cultural integration of herbal medicine monographs forces medical directors to demand rigorous pharmacological validation.
What limits generic competitors from disrupting pricing?
Securing multi-year double-blind efficacy data requires immense time and creates impenetrable barriers against unvalidated entrants.
How do volatile botanical extracts impact manufacturing operations?
Hygroscopic instability causes massive powder caking that forces plant managers to install million-dollar humidity control infrastructure.
What hidden costs emerge when procurement buys cheaper raw materials?
Inconsistent particle sizing causes capsule dosing variations that inevitably trigger expensive product recalls.
Who primarily initiates the transition toward validated systems?
Major retail category managers mandate absolute clinical substantiation before assigning premium shelf space.
What operational consequence follows format changes?
Switching from hard capsules to gummies sacrifices active ingredient concentration to accommodate necessary sugar matrices.
Why do direct-to-consumer brands rely on turnkey formulations?
Venture capital expectations force financial directors to utilize pre-validated systems to minimize lengthy product development cycles.
How do subscription models alter revenue stability?
Direct platforms enabling automated monthly replenishment tied to user cycles lower customer churn dramatically.
What residual risk remains even when buying premium extracts?
Seasonal crop variations demand continuous formulation adjustments to maintain consistent phytochemical ratios across batches.
How does perimenopausal demand impact Japanese formulations?
Aging demographics shift R&D focus toward highly bioavailable delivery formats tailored specifically for maturing digestive tracts.
What finally converts skeptical pharmacy buyers?
Quantifiable clinical endpoint validation satisfies strict corporate procurement guidelines and unlocks premium shelf placement.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient System
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient System , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient System , 2026 to 2036
- Magnesium-B6 Blends
- Omega-3 Blends
- Others
- Magnesium-B6 Blends
- Y to o to Y Growth Trend Analysis By Ingredient System , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient System , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Format, 2026 to 2036
- Capsules
- Gummies
- Powders
- Capsules
- Y to o to Y Growth Trend Analysis By Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- PMS Support
- Cramp Relief
- Others
- PMS Support
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Channel, 2026 to 2036
- Online Retail
- Pharmacies
- Others
- Online Retail
- Y to o to Y Growth Trend Analysis By Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Origin
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Origin, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Origin, 2026 to 2036
- Mediterranean Botanicals
- Marine Lipids
- Others
- Mediterranean Botanicals
- Y to o to Y Growth Trend Analysis By Ingredient Origin, 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Origin, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient System
- By Format
- By Application
- By Channel
- By Ingredient Origin
- Competition Analysis
- Competition Deep Dive
- O Positiv
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- HUM Nutrition
- OLLY
- Elix Healing
- Thorne
- MegaFood
- O Positiv
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient System , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Origin, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient System
- Figure 6: Global Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Format
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Channel
- Figure 15: Global Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Ingredient Origin
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient System
- Figure 32: North America Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Format
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Channel
- Figure 41: North America Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Ingredient Origin
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient System
- Figure 48: Latin America Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Channel
- Figure 57: Latin America Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Ingredient Origin
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient System
- Figure 64: Western Europe Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Channel
- Figure 73: Western Europe Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Ingredient Origin
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient System
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Channel
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Ingredient Origin
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient System
- Figure 96: East Asia Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Channel
- Figure 105: East Asia Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Ingredient Origin
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient System
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Channel
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Ingredient Origin
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient System , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient System , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient System
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Channel
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Origin, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Origin, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Ingredient Origin
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

