Bergamot Polyphenol Ingredient Systems Market
The bergamot polyphenol ingredient systems market is segmented by Ingredient Type (Polyphenol fractions, Phytosome systems, Blend systems, Juice extracts, Peel extracts), Form (Powders, Capsules, Tablets, Granules, Liquids), Application (Cardiometabolic health, Lipid management, Weight management, Liver support, Sports recovery), Channel (Branded ingredients, Private label, Contract blends, Distributor packs, Clinical supply), End Use (Dietary supplements, Functional foods, Functional beverages, Medical nutrition, Personal care), aand Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Bergamot Polyphenol Ingredient Systems Market Size, Market Forecast and Outlook By FMI
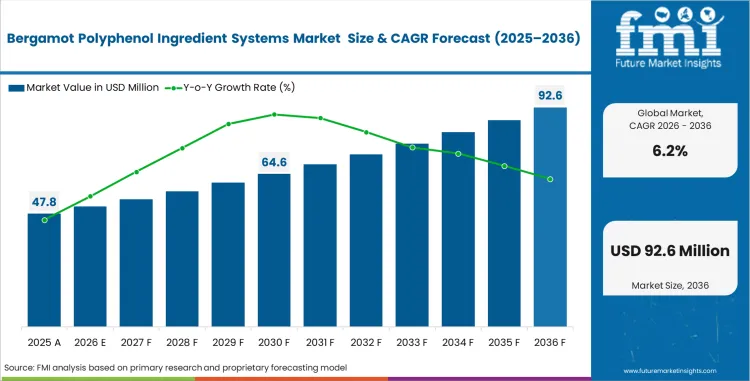
The bergamot polyphenol ingredient systems market was valued at USD 45.0 million in 2025 and is expected to reach USD 47.8 million in 2026. Market value is projected to rise to USD 87.2 million by 2036, reflecting a CAGR of 6.2% during 2026 to 2036.
Bergamot Polyphenol Ingredient Systems Market Report Summary
- The market is forecast to reach USD 87.2 million by 2036.
- The market is expected to grow at a CAGR of 6.2% from 2026 to 2036.
- The market was estimated at USD 45.0 million in 2025.
- The forecast period represents an incremental opportunity of USD 39.4 million.
- The market is projected to reach USD 47.8 million in 2026.
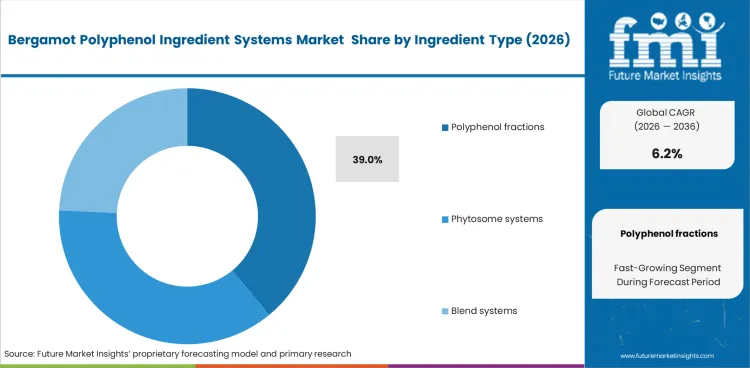
- Polyphenol fractions lead the ingredient type segment with a 39.0% share, driven by demand for standardized flavonoid-rich systems.
- Powder form dominates with a 61.0% share, supported by ease of use in capsules, tablets, and premixes.
- Cardiometabolic health applications account for 48.0% share, reflecting strong clinical and commercial focus.
- Dietary supplements lead end use with a 58.0% share, as most commercialization remains supplement-driven.
- Branded ingredients hold a 44.0% share, supported by differentiation through clinical validation and standardization.
- Key companies in the market include Herbal & Antioxidant Derivatives (HP Ingredients), Bionap, Indena, PRG Pharma, Safic-Alcan, Nutraceuticals Group Europe, and Greenyn Biotechnology.
Bergamot Polyphenol Ingredient Systems Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 47.8 million |
| Industry Value (2036) | USD 87.2 million |
| CAGR (2026-2036) | 6.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Demand for dose-defined formulations continues to support adoption, while commercial expansion remains tied to the narrow Calabrian raw material base that supports standardization.
Nutraceutical and functional food developers continue to use bergamot polyphenol systems in cardiometabolic and healthy aging products. Interest remains strongest where finished products need a Mediterranean botanical with clearer technical positioning than generic citrus extracts. Published literature continues to support attention on bergamot polyphenol fractions in lipid management and related metabolic applications. That keeps powders, capsules, and combination formulas at the center of commercialization.
Buyer preference is moving toward standardized botanical ingredients with defined polyphenol content. Clear specifications make finished product positioning easier and reduce qualification issues during sourcing and formulation work. Suppliers that maintain extraction quality and batch consistency are better placed in this market. Supply still remains tight because commercial bergamot sourcing is concentrated in Southern Italy, and material grown outside that corridor does not always align with the target profile used in premium systems.
Expansion is expected to stay measured through the forecast period rather than accelerate sharply. Cultivar choice, extraction control, and batch consistency remain central to ingredient performance. Companies with secure Calabrian sourcing and validated ingredient systems are likely to retain an advantage through 2036.
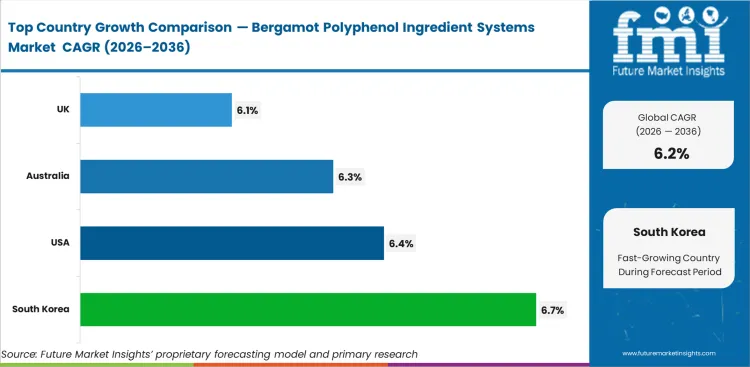
South Korea is projected to expand at a CAGR of 6.7% during 2026 to 2036, supported by demand for metabolic supplements that is moving ahead of domestic supply. Demand in the United States is expected to rise at a CAGR of 6.4% over the same period, as practitioner-led adoption continues to support category consumption. Australia is estimated to register a CAGR of 6.3% through 2036, while the United Kingdom is likely to record 6.1% during the forecast period. Germany is anticipated to grow at a CAGR of 6.0% from 2026 to 2036, followed by Japan at 5.6% and Italy at 5.1%. This variation across countries reflects differences in import reliance, local production depth, and the balance between domestic supply and market demand.
Segmental Analysis
Bergamot Polyphenol Ingredient Systems Market Analysis by Ingredient Type
Polyphenol fractions lead ingredient type because buyers want bergamot systems with tighter standardization and cleaner specification control. Clinical positioning is easier to support with defined fractions than with broad extracts. That preference keeps the polyphenol fractions segment at 39.0% share in 2026. Wider expansion into adjacent ingredients, including rosemary citrus synergy antioxidant systems and other citrus ingredients, does not change the fact that buyers in this niche still prioritize standardization first.
- Dossier anchoring: Buyers favor fractions that support a more consistent clinical and marketing position, especially in lipid-management applications where specification detail matters.
- Compression behavior: Standardized dry fractions are easier to qualify for tableting and filling than less-consistent raw extracts, which supports repeatable manufacturing.
- Practitioner channel access: Products positioned through practitioner-led channels benefit from ingredients that offer clearer standardization and more disciplined documentation.
Bergamot Polyphenol Ingredient Systems Market Analysis by Form
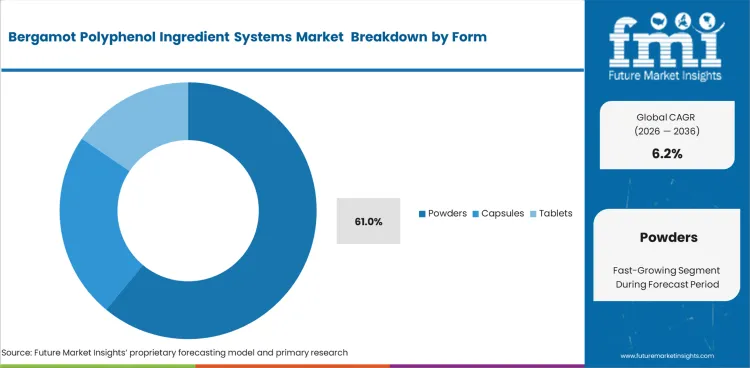
Powdered formats remain ahead in this segment because dry bergamot ingredients align more easily with established supplement production systems than liquid alternatives. Powders are estimated to account for 61.0% of market share in 2026. They also align better with routine blending, filling, and premix handling. Competition from adjacent functional food ingredients does not change the current preference for dry-format bergamot actives.
- Blending uniformity: Dry formats are easier to dose and blend consistently in standard supplement manufacturing lines.
- Excipient compatibility: Powders fit more naturally with common tableting and encapsulation systems used across nutraceutical production.
- Throughput optimization: Dry, flowable materials help manufacturers keep filling and compression processes more stable at scale.
Bergamot Polyphenol Ingredient Systems Market Analysis by Application
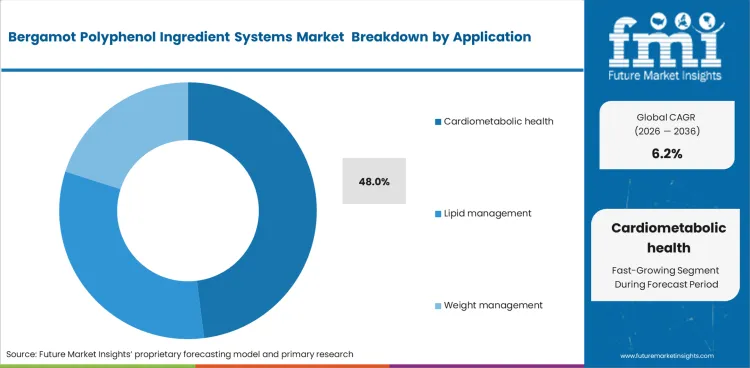
Cardiometabolic health remains the leading application because most commercial interest and published discussion still center on lipid management and related metabolic support. The segment is likely to secure 48.0% share in 2026. That concentration also reinforces demand for clinically positioned botanical extracts, while interest in hesperidin and related citrus actives points to broader overlap across metabolic-health formulations.
- Endpoint targeting: Suppliers and brands continue to focus on measurable metabolic and lipid-related use cases because those applications carry the clearest commercial fit.
- Claims defense: Better-documented ingredients are easier to position where marketers need tighter support for metabolic-health claims.
- Formulation focus: Cardiometabolic applications remain the clearest anchor for bergamot polyphenol systems, which keeps broader wellness use secondary.
Bergamot Polyphenol Ingredient Systems Market Analysis by Channel
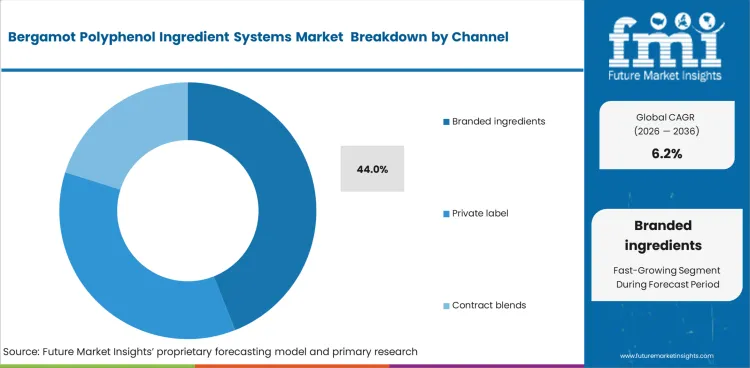
Branded ingredients are likely to lead the channel segment because they reduce formulation risk for product makers. The segment is expected to account for 44.0% of market share in 2026. Price remains important, but buyers usually place more value on clinical support and standardization.
- Dossier inheritance: Finished brands gain a stronger commercial platform when ingredient suppliers already support the product with evidence and formulation documentation.
- Bioavailability guarantees: Delivery technologies help suppliers justify higher pricing when they offer a clearer point of differentiation.
- Adulteration defense: Buyers place value on traceability and identity control, especially where premium botanical positioning depends on sourcing confidence.
Bergamot Polyphenol Ingredient Systems Market Analysis by End Use
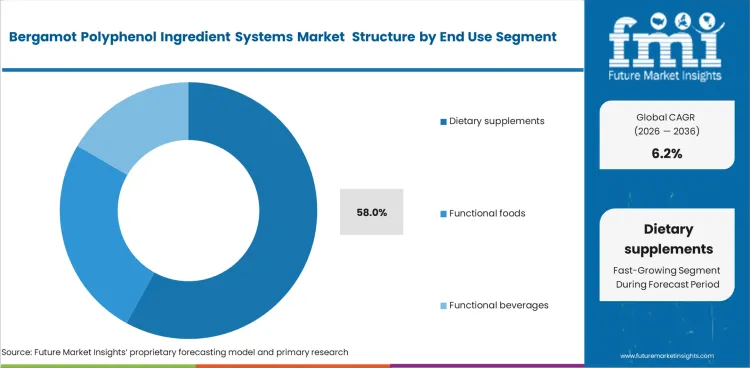
Dietary supplements lead end use because bergamot polyphenols are easier to commercialize in capsules and tablets than in foods or beverages. The dietary supplements segment is expected to account for 58.0% of market share in 2026. Bitterness, dose loading, and formulation limits still hold back wider beverage use, even where adjacent citrus flavors supports interest in flavor-led innovation.
- Flavor masking: High-polyphenol bergamot systems are harder to carry in mainstream food and beverage formats because bitterness remains difficult to manage.
- Payload capacity: Supplement formats handle clinically positioned actives more easily when dose loading is higher.
- Format expectation: Capsules and tablets remain the most natural route for commercialization in this category.
Bergamot Polyphenol Ingredient Systems Market Drivers, Restraints, and Opportunities
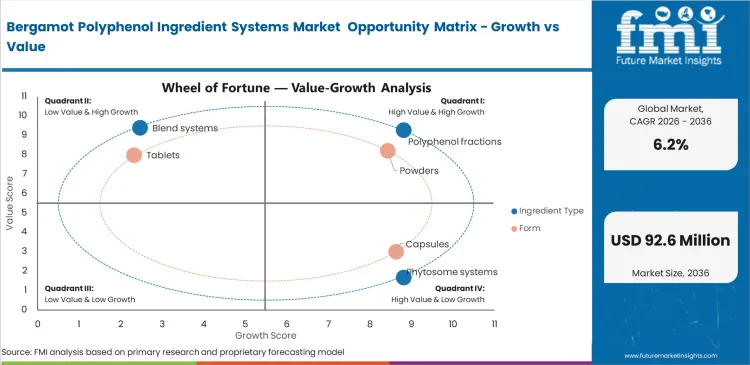
Cardiovascular screening thresholds are pushing more preventive health consumers toward dietary lipid-management options before prescription statins. The bergamot polyphenol ingredient systems market is likely to benefit from this shift as brands develop formulations for earlier use. Reliable access to citrus oils and standardized polyphenol fractions helps brands move into more premium categories. Standardized fractions are likely to attract stronger interest from formulators that want clearer claims and lower pill burden. Some brands also prefer standardized citrus derivatives because they are often easier to support than synthetic alternatives.
Agricultural concentration is still likely to limit market expansion. Supply depends on a small number of production zones in Calabria that meet flavonoid standards. This leaves the ingredient base exposed to weather shifts and harvest disruption. Other sourcing regions are harder to qualify because their active composition often differs from accepted profiles. As a result, companies rely more on longer-term supply agreements and tighter sourcing control. Smaller brands often face more difficulty expanding because they cannot secure harvest volumes as easily.
Opportunities in the Bergamot Polyphenol Ingredient Systems Market
- Phytosome integration: Advanced delivery systems can help reduce dosage volume and improve format convenience. This creates room for smaller capsule formats in premium supplement lines.
- Beverage microencapsulation: Better taste control can improve bergamot’s fit in functional drink development. This may help brands reach consumers who prefer beverages over tablets or capsules.
- Synergistic stacking: Bergamot fractions combined with artichoke extracts can support more differentiated metabolic health formulations. This gives brands more room to build premium multi-ingredient products.
Regional Analysis
Based on regional analysis, the bergamot polyphenol ingredient systems market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| South Korea | 6.7% |
| United States | 6.4% |
| Australia | 6.3% |
| United Kingdom | 6.1% |
| Germany | 6.0% |
| Japan | 5.6% |
| Italy | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Bergamot Polyphenol Ingredient Systems Market Analysis
Regulatory support for metabolic health products is increasing demand for imported bergamot ingredients across key Asia Pacific markets. Buyers in the region often prefer Calabrian systems with established clinical documentation over local alternatives when health-claim support is important. Imported systems raise unit costs, but they also help premium brands address concerns about consistency and ingredient authenticity. Beverage and supplement formulators are also paying more attention to flavor management because concentrated bergamot extracts can affect product acceptance.
- South Korea: Faster approvals for metabolic health supplements are expected to support South Korea’s bergamot polyphenol ingredient systems market at a 6.7% CAGR through 2036. Product selection leans toward ingredients with stronger clinical documentation. Imported bergamot systems remain important because premium products in this market depend more on proven evidence than local botanical replacement.
- Australia: Practitioner-led supplement demand is expected to support 6.3% CAGR in Australia during 2026 to 2036. Market uptake remains closely tied to preventive cardiovascular use, where consumers show greater willingness to pay for clinically supported products. Better-documented bergamot ingredients are likely to retain their place in premium formulations sold through practitioner-focused channels.
- Japan: Preventive health demand and an aging consumer base are projected to support a 5.6% CAGR in Japan from 2026 to 2036. Formulators in Japan place strong importance on ingredients that fit established function-based health claim frameworks. Bergamot fractions with clearer technical support are likely to remain well placed, especially where product acceptance depends on evidence-backed positioning.
North America Bergamot Polyphenol Ingredient Systems Market Analysis
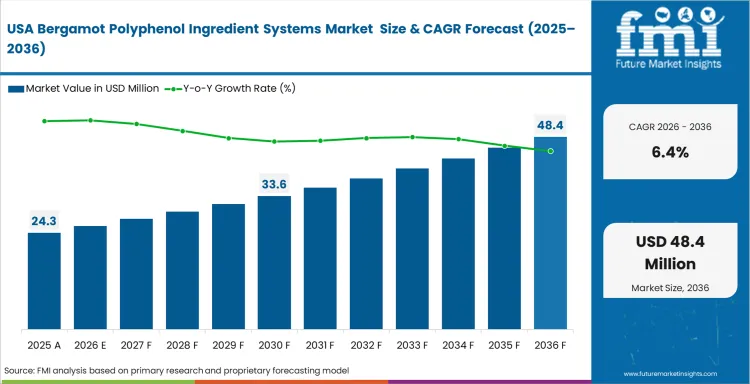
North America has strong demand across practitioner-led and direct-to-consumer cardiovascular supplement channels. Concentrated bergamot fractions remain important because many products are positioned around clinically supported lipid management. Raw material validation matters more in this market, where labeling scrutiny is stricter and formulation claims need stronger support. Proprietary delivery formats also help suppliers stand apart as lower-priced alternatives enter the category. Manufacturing needs are also becoming more specialized because concentrated botanical distillates and powders require tighter handling to avoid cross-contamination.
- United States: Demand in the United States is projected to grow at a CAGR of 6.4% during 2026 to 2036. Growth is supported by competition among branded ingredients and by consumer willingness to pay for clinically positioned cardiovascular support products. Practitioner-dispensed channels continue to strengthen this market, especially where evidence-backed formulations help maintain premium pricing. Supply arrangements linked to differentiated delivery formats can also help products stand out in crowded retail and practitioner-facing categories. This keeps validation, formulation support, and ingredient differentiation central to market performance in the United States.
Europe Bergamot Polyphenol Ingredient Systems Market Analysis
Europe has a different market structure because it sits closer to bergamot origin and extraction networks than most import-led markets. In bergamot and similar coastal botanical extracts, raw material access depends on localized farm networks, harvest timing, and uninterrupted regional processing, making long-term supply relationships more valuable than opportunistic spot buying. This matters because raw material quality and seasonal consistency directly affect standardization and formulation reliability. As a result, European brands sourcing from Mediterranean corridors benefit from closer origin access and stronger provenance, but they are also more vulnerable to weather-related crop disruption, processing interruptions, and batch variability across the supply base.
- United Kingdom: Premium pharmacy channels continue to support demand for more targeted botanical formulations in the United Kingdom. Bergamot systems are gaining acceptance as brands shift away from broad vitamin products and focus more on metabolic and cardiovascular support. Products that combine bergamot fractions with plant sterols are also seeing better uptake, especially in premium formats. The United Kingdom is projected to grow at a 6.1% CAGR from 2026 to 2036.
- Germany: Germany is projected to expand at a 6.0% CAGR from 2026 to 2036, as traceability and regulatory fit remain important in the market. This makes ingredient selection more selective than in several neighboring countries. Products without clear safety backing and careful claim positioning face a narrower path to acceptance, especially in specialized health channels. Suppliers with stronger documentation and technical support are likely to perform better.
- Italy: Italy benefits from close access to raw material sources, giving it a more stable demand structure than most import-led markets. Access to harvest zones and existing extraction infrastructure helps maintain input quality and processing continuity. Local familiarity with bergamot-derived ingredients also supports more stable demand. Italy is expected to grow at a 5.1% CAGR from 2026 to 2036.
Competitive Aligners for Market Players
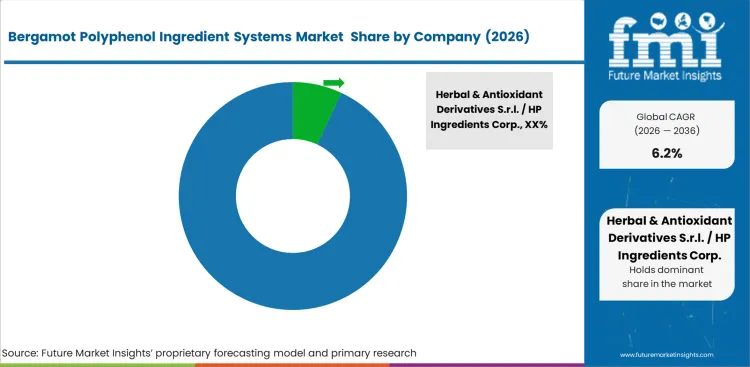
Clinical support remains a clear differentiator between premium ingredient suppliers and bulk intermediaries. Competitive strength in this market depends on more than orchard access. Buyers also look for human study support, strong standardization data, and proven formulation quality. Suppliers with stronger clinical backing often hold an advantage because independent efficacy work adds cost and slows product development. Delivery technology also shapes competition, especially where absorption and formulation performance influence ingredient choice.
Longstanding links with Calabrian farming networks also remain important. Raw material consistency cannot be secured through capital investment alone. Processors with experience across seasonal variation, extraction behavior, and standardization tend to maintain more consistent profiles from one harvest cycle to the next. New entrants often face greater difficulty because process control improves with operating history. Batch variation becomes more visible during qualification when composition moves outside accepted limits, making extraction discipline central to supplier credibility.
Large supplement brands also try to avoid reliance on a single source. Growth in this category depends on both clinical support and reliable supply. Stability work on alternative materials gives buyers more flexibility in supplier negotiations and lowers dependence on incumbent suppliers. Supplier selection in the bergamot polyphenol ingredient systems market is shaped by clinical depth, cost position, standardization consistency, and formulation performance. Suppliers that can support more advanced co-formulations without reducing active strength or product stability are expected to remain better placed.
Key Players in Bergamot Polyphenol Ingredient Systems Market
- Herbal & Antioxidant Derivatives S.r.l. / HP Ingredients Corp.
- Bionap S.r.l.
- Indena S.p.A.
- PRG Pharma Pvt. Ltd.
- Safic-Alcan
- Nutraceuticals Group Europe
- Greenyn Biotechnology Co., Ltd.
Scope of the Report
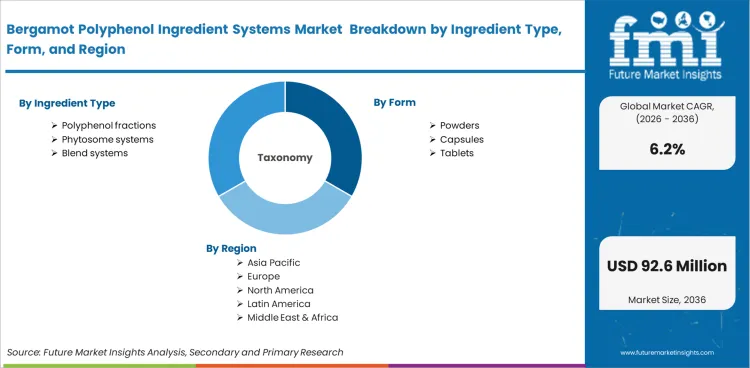
| Metric | Value |
|---|---|
| Quantitative Units | USD 47.8 million to USD 87.2 million, at a CAGR of 6.2% |
| Market Definition | Standardized polyphenolic fractions and phytosome-enhanced systems derived from Calabrian citrus define this analytical boundary. This space isolates commercial activity around formulation-ready nutraceutical ingredients engineered specifically for clinical metabolic applications. |
| Segmentation | By Ingredient Type, By Form, By Application, By Channel, By End Use, and By Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East & Africa |
| Countries Covered | United States, Italy, Germany, United Kingdom, Japan, South Korea, Australia |
| Key Companies Profiled | Herbal & Antioxidant Derivatives S.r.l. / HP Ingredients Corp., Bionap S.r.l., Indena S.p.A., PRG Pharma Pvt. Ltd., Safic-Alcan, Nutraceuticals Group Europe, Greenyn Biotechnology Co., Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Clinical-grade extraction yield volumes compared against premium heart-health supplement retail data anchor baseline sizing. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Bergamot Polyphenol Ingredient Systems Market Analysis by Segments
By Ingredient Type:
- Polyphenol fractions
- Phytosome systems
- Blend systems
- Juice extracts
- Peel extracts
By Form:
- Powders
- Capsules
- Tablets
- Granules
- Liquids
By Application:
- Cardiometabolic health
- Lipid management
- Weight management
- Liver support
- Sports recovery
By Channel:
- Branded ingredients
- Private label
- Contract blends
- Distributor packs
- Clinical supply
By End Use:
- Dietary supplements
- Functional foods
- Functional beverages
- Medical nutrition
- Personal care
Region:
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Rest of Middle East & Africa
Bibliography
- Mazzola, G., Rondanelli, M., Baron, G., Zupo, R., Castellana, F., Clodoveo, M. L., Gasparri, C., Barrile, G. C., Seniga, M., Schiavi, L. M., Moroni, A., Gulec, S., Riso, P., & Perna, S. (2024, October). Bergamot (Citrus bergamia), a (Poly)Phenol-Rich Source for Improving Osteosarcopenic Obesity: A Systematic Review. Foods, 13(21), 3422.
- Spina, A., Amone, F., Zaccaria, V., Insolia, V., Perri, A., Lofaro, D., Puoci, F., & Nobile, V. (2024, November). Citrus bergamia Extract, a Natural Approach for Cholesterol and Lipid Metabolism Management: A Randomized, Double-Blind Placebo-Controlled Clinical Trial. Foods, 13(23), 3883.
- Agosto, N. J., Alambatin, P. B., Bacalso, J., Cabisada, J., & Carating, B. D. (2024, September). The Evaluation of Citrus bergamia Phytochemicals as Potential Cholesterol-Lowering Agents against HMG-CoA Reductase: An In Silico Molecular Docking Study. Biology and Life Sciences Forum, 35(1), 7.
- Ilari, S., Nucera, S., Passacatini, L. C., Scarano, F., Macrì, R., Caminiti, R., Ruga, S., Serra, M., Giancotti, L. A., Lauro, F., Dagostino, C., Mazza, V., Ritorto, G., Oppedisano, F., Maiuolo, J., Palma, E., Malafoglia, V., Tomino, C., Mollace, V., & Muscoli, C. (2024, August). Exploring the Role of Bergamot Polyphenols in Alleviating Morphine-Induced Hyperalgesia and Tolerance through Modulation of Mitochondrial SIRT3. Nutrients, 16(16), 2620.
- Carpenito, M., Coletti, F., Muscoli, S., Guarino, L., Di Cristo, A., Cammalleri, V., Mega, S., Emerenziani, S., Cicala, M., Fanali, C., et al. (2025, May). Unveiling the Power of Bergamot: Beyond Lipid-Lowering Effects. Nutrients, 17(11), 1871.
This Report Addresses
- Polyphenol fractions tracking through strict clinical dossier requirements.
- Powder blending constraints dictating high-speed encapsulation parameters.
- Cardiometabolic diagnostic thresholds triggering premium supplement adoptions.
- Patented phytosome complexes shielding brands from raw agricultural volatility.
- Capsule formulations bypassing severe astringency and bitter flavor masking hurdles.
- Calabrian agricultural dominance creating global supply bottlenecks.
- South Korean regulatory fast-tracking protocols favoring imported branded activities.
- Practitioner channel dynamics rewarding explicit 150 mg standardized flavonoid intakes.
Frequently Asked Questions
What is the difference between bergamot oil and bergamot extract?
Fragrance-grade essential oils utilize volatile aromatic compounds derived from citrus rinds, whereas clinical extracts isolate specific flavonoid fractions. Medical personnel utilize precision extracts specifically to target lipid pathways without introducing toxic aromatic compounds into human digestive systems.
How large is the bergamot polyphenol ingredient systems market?
Demand scales from USD 47.8 million in 2026 to USD 87.2 million by 2036. This trajectory confirms how deeply clinical validation anchors long-term purchasing commitments inside premium cardiovascular supplement supply chains.
What drives growth in bergamot nutraceutical ingredients?
Cardiovascular diagnostic thresholds compel preventative health consumers to seek immediate dietary interventions. Medical personnel exploit specific timing windows by deploying clinical-grade formulations featuring profiles optimized for endothelial support within the bergamot polyphenol ingredient systems market.
Which companies supply bergamot polyphenol fractions?
Key producers include Herbal & Antioxidant Derivatives S.r.l., Bionap S.r.l., Indena S.p.A., and PRG Pharma Pvt. Ltd. Companies choose specific partners based widely on possessing bulletproof cardiovascular efficacy studies.
Why do polyphenol fractions dominate ingredient types?
Clinical trial baseline dependencies require explicit 38.0% concentration guarantees. Formulation officers reject raw peel extracts to ensure consumer products perfectly mirror published cardiovascular research outcomes without relying on generic botanical powders.
How do powders maintain form leadership?
Moisture sensitivity dictates precise delivery vehicle selections. Quality control personnel optimize blending suites specifically for dry actives to prevent secondary processing friction during capsule runs.
What drives cardiometabolic health applications?
Diagnostic trends force immediate lipid-management interventions. Medical personnel target endothelial function pathways specifically to position premium formulations directly alongside prescription alternatives at higher retail price points.
Why do branded ingredients control channel volume?
Intellectual property protection completely overrides spot-market pricing. Companies willingly pay premiums to inherit established clinical dossiers, shifting primary validation costs away from finished product manufacturers entirely.
Why are dietary supplements the primary end use?
Consumer pill fatigue collides with dosage volume requirements. Product development chemists rely on capsules to deliver bitter active doses without triggering complex flavor masking redesigns inside functional beverages.
What separates South Korean adoption from Japan?
South Korean regulatory frameworks explicitly favor imported ingredients backed by robust human data, driving 6.7% growth. Japan integrates fractions slowly into complex FFC architectures at 5.6%.
Why does Italy trail in regional growth?
Domestic agricultural dominance provides unmatched access to raw materials, creating highly mature baselines. Local operators define global standardization metrics rather than aggressively expanding domestic consumption.
How do Calabrian orchards dictate global availability?
Sourcing personnel rely almost entirely on single geographic strips to yield correct flavonoid profiles. Minor local weather events instantly cascade into severe global ingredient shortages.
Why do competitors fund alternative clinical trials?
Large supplement labels fiercely resist proprietary ingredient lock-in. Companies actively validate challenger materials specifically to break incumbent pricing power and establish secondary supply redundancies.
What forces brand managers toward phytosome technologies?
Engineering advanced lipid-based delivery systems dramatically lowers necessary dosage volumes. Formulation chemists capture premium margins by offering consumers smaller, easier-to-swallow capsules.
Why do contract manufacturers avoid generic botanicals?
Purified fractions exhibit distinct physical handling characteristics. Filling operations managers avoid secondary granulation steps by maintaining consistent supplier relationships with standardized extract producers.
How do strict cardiovascular claims alter purchasing?
Regulatory officers demand verifiable Italian-sourced flavonoid profiles to support legal labeling. Compliance constraints eliminate cheaper alternative geographic sources from consideration entirely.
What role do practitioner channels play in Australia?
Clinical naturopaths recommend specific patented fractions based strictly on peer-reviewed outcomes. Brands capturing practitioner trust establish virtually unbreakable recurring subscription revenue streams.
Why do brands license clinical data from suppliers?
Ingredient manufacturers fund primary human clinical trials. Brand marketing teams simply license data to support aggressive retail positioning without absorbing massive independent research costs.
How does astringency limit functional beverage expansion?
High-concentration polyphenols introduce severe bitterness. Beverage formulators struggle to mask notes effectively, quietly enforcing supplement dominance over liquid delivery formats.
What prevents new entrants from matching established yields?
Emerging competitors lack historical extraction yield data necessary to calibrate processing equipment. Attempting to match consistency forces blending variations that premium buyers readily detect during qualification.
Why do brands require specific 150 mg dosage claims?
Medical professionals recommend precise dosages based strictly on published literature. Securing practitioner endorsements demands exact adherence to proven clinical intake thresholds.
How do desiccants impact packaging expenses?
Concentrated powders frequently require sophisticated desiccants to prevent caking in humid environments. Sourcing officers must balance advanced ingredient selection against quietly escalating total packaging costs.
What drives synergistic artichoke stacking?
Combining specific fractions enhances overall endothelial efficacy. Product strategists differentiate commodity formulas by building proprietary multi-ingredient metabolic stacks that competitors cannot easily replicate.
How does European compliance shape Germany consumption?
Strict frameworks severely restrict aggressive cardiovascular marketing. Compliance personnel navigate rules by utilizing only ingredients possessing unimpeachable European clinical trial histories.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Polyphenol fractions
- Phytosome systems
- Blend systems
- Juice extracts
- Peel extracts
- Polyphenol fractions
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powders
- Capsules
- Tablets
- Granules
- Liquids
- Powders
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cardiometabolic health
- Lipid management
- Weight management
- Liver support
- Sports recovery
- Cardiometabolic health
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Channel, 2026 to 2036
- Branded ingredients
- Private label
- Contract blends
- Distributor packs
- Clinical supply
- Branded ingredients
- Y to o to Y Growth Trend Analysis By Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Dietary supplements
- Functional foods
- Functional beverages
- Medical nutrition
- Personal care
- Dietary supplements
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Form
- By Application
- By Channel
- By End Use
- Competition Analysis
- Competition Deep Dive
- Herbal & Antioxidant Derivatives S.r.l. / HP Ingredients Corp.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bionap S.r.l.
- Indena S.p.A.
- PRG Pharma Pvt. Ltd.
- Safic-Alcan
- Nutraceuticals Group Europe
- Greenyn Biotechnology Co., Ltd.
- Herbal & Antioxidant Derivatives S.r.l. / HP Ingredients Corp.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Channel
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Form
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Channel
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Form
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Channel
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Form
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Channel
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Form
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Channel
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Form
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Channel
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Channel
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Channel
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

