Mediterranean Cognitive-First Ingredient Systems Market
The Mediterranean Cognitive-First Ingredient Systems Market Is Segmented By Ingredient Type (Omega-3 Lipids, Olive Polyphenols, Saffron Extracts, Rosemary Extracts, Lemon Balm, Nut Concentrates), Delivery Format (Powder Blends, Softgels, Capsules, Gummies, Beverage Premixes), Application (Memory Support, Focus Support, Mood Support, Healthy Aging, Stress Resilience), Buyer Type (Supplement Brands, Beverage Brands, Functional Food Brands, Cdmos, Pharmacies), Source Geography (Spain, Italy, Greece, France, Blended Sourcing), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mediterranean Cognitive-First Ingredient Systems Market Size, Market Forecast and Outlook By FMI
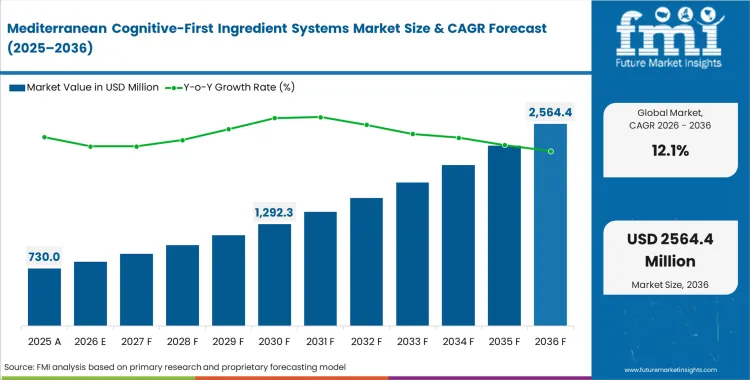
In 2025, the Mediterranean cognitive-first ingredient systems market crossed a valuation of USD 650 million. Revenue is expected to reach USD 730 million in 2026 and USD 2,290 million by 2036, reflecting a CAGR of 12.1% over the forecast period. Growth is being supported by continued investment and by a broader shift in clinical research from single-ingredient studies to more complex botanical combinations, which is expanding the market’s commercial scope.
Summary of Mediterranean Cognitive-First Ingredient Systems Market
- Mediterranean Cognitive-First Ingredient Systems Market Definition
- Standardized botanical and lipid extracts sourced from specific agricultural zones, engineered for measurable neurological support and standardized to precise bioactive markers.
- Demand Drivers in the Market
- Retail shelf-space requirements force supplement directors to replace synthetic options with clean-label mediterranean cognitive health nutraceutical ingredients.
- Consumer demand for experiential efficacy compels formulation chemists to stack fast-acting polyphenols with long-term lipid structures.
- Aging demographic pressures push preventative medicine practitioners to recommend food-based cognitive preservation strategies.
- Key Segments Analyzed in the FMI Report
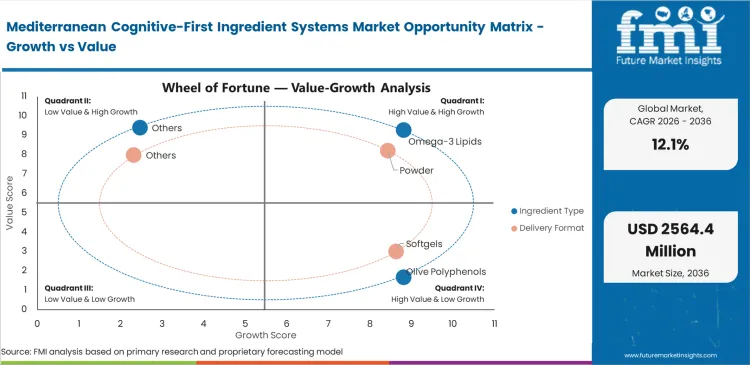
- Omega-3 Lipids: 34.0% share in 2026, driven by foundational requirements for neuronal membrane fluidity.
- Powder Blends: 38.0% share in 2026, powered by high solubility requirements in functional beverages.
- Memory Support: 29.0% share in 2026, linked directly to rising mild cognitive impairment diagnoses.
- Supplement Brands: 46.0% share in 2026, leading early adoption of complex botanical formulations.
- Spain: 31.0% share in 2026, controlling critical extraction infrastructure for regional botanicals.
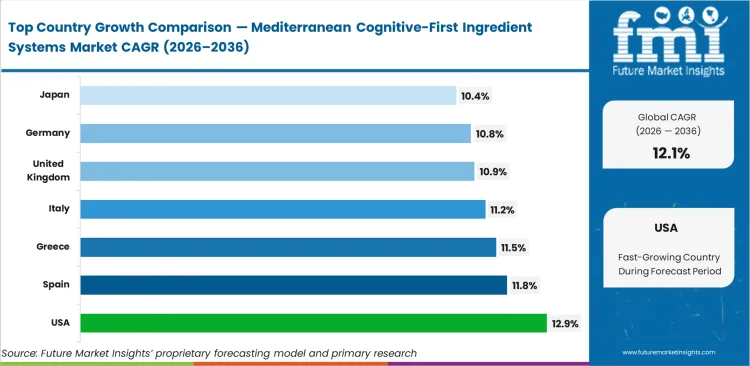
- United States: 12.9% CAGR, powered by rapid expansion in functional beverage retail channels.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, observes that,"Clinical evidence demands a higher standard today and unless extraction methods preserve specific hydroxytyrosol ratios, formulation efficacy drops entirely. Procurement directors focusing strictly on bulk pricing often purchase dead material devoid of cognitive benefit. Clinical validation is tied entirely to extraction methodology as well as with the botanical origin."
- Strategic Implications / Executive Takeaways
- Procurement directors must secure long-term agricultural contracts to guarantee bioactive consistency across harvest seasons.
- Formulation chemists need to prioritize water-soluble delivery systems to penetrate expanding beverage categories.
- CDMO operations should invest in taste-masking technologies for bitter botanical extracts to improve consumer compliance.
- Methodology
- Primary Research: Direct interviews with clinical trial coordinators and extraction facility managers.
- Desk Research: Analysis of patent filings for novel botanical synergistic complexes.
- Market-Sizing and Forecasting: Volume tracking of specialized extracts leaving Mediterranean ports.
- Data Validation and Update Cycle: Triangulation with global nutraceutical retail sales data.
Retail partners now require both immediate experiential effects and long-term cognitive claims from Mediterranean brain health ingredients, pushing formulators to meet both standards. Supplement directors cannot rely solely on isolated synthetic racetams; they must formulate multi-pathway botanical stacks utilizing mediterranean nootropic ingredients. Pharmacy chains that require natural cognitive support options will delist products that do not use validated regional extracts. Identifying reliable standardized polyphenol sources remains difficult for independent cognitive health supplement manufacturers lacking direct supply contracts.
When regional agricultural cooperatives standardize extraction protocols for altitude-stressed botanicals, mass-market uptake becomes more feasible. Contract manufacturers trigger this shift by offering pre-validated blends, establishing themselves as essential mediterranean cognition ingredient suppliers. Brand owners use these turnkey blends to avoid the cost of running their own clinical validation.
United States adoption leads at 12.9%, powered by aggressive functional beverage expansion. Spain follows at 11.8% due to vertically integrated cooperative processing. Greece tracks at 11.5% on back of specialized high-altitude cultivation. Italy grows at 11.2%, leveraging historical extraction expertise. United Kingdom expands at 10.9% as consumer preference shifts toward preventative cognitive care. Germany advances at 10.8% through strict monograph compliance. Japan completes this tier at 10.4%, driven by an aging demographic seeking natural memory preservation. Sourcing constraints dictate regional growth speed.
Mediterranean Cognitive-First Ingredient Systems Market Definition
Mediterranean Cognitive-First Ingredient Systems Market represents standardized botanical and lipid extracts sourced specifically from Mediterranean agricultural zones, designed to support neurological function. Mediterranean cognitive ingredients are defined by strict molecular standardization requirements, bioactives such as hydroxytyrosol must meet specific concentration thresholds, and essential fatty acid ratios must fall within defined ranges. Sourcing geography and clinical focus separate these materials from general nutritional commodities.
Mediterranean Cognitive-First Ingredient Systems Market Inclusions
Standardized extracts of regional herbs, customized mediterranean herb nootropic complexes, specialized marine lipids, and concentrated polyphenol derivatives fall within scope. Functional combinations of these active compounds formulated specifically as mediterranean healthy aging ingredients also qualify.
Mediterranean Cognitive-First Ingredient Systems Market Exclusions
Synthetic nootropics and generic botanical extracts lacking geographical traceability are strictly excluded. Formulations targeting general cardiovascular health without specific cognitive endpoint validation do not qualify. Generic vitamins and minerals remain outside this boundary due to their commodity status.
Mediterranean Cognitive-First Ingredient Systems Market Research Methodology
- Primary Research: Formulation scientists and procurement directors from specialized nutraceutical brands.
- Desk Research: European Food Safety Authority clinical registry filings and regional agricultural cooperative export logs.
- Market-Sizing and Forecasting: Metric tons of standardized botanical extracts cleared for export with cognitive claims.
- Data Validation and Update Cycle: Independent cross-validation against raw material cooperative production yields.
Segmental Analysis
Mediterranean Cognitive-First Ingredient Systems Market Analysis By Ingredient Type
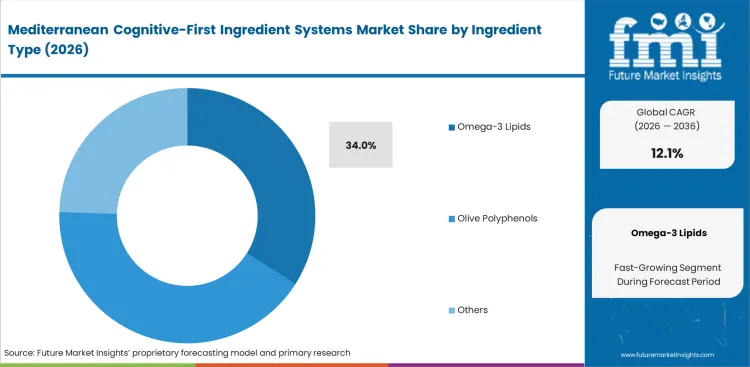
Specific neuronal membrane requirements dictate why Omega-3 lipids capture prominent market position, holding a share of 34.0% in 2026, with the foundational cellular architecture demanding DHA algal oil brain health ingredients before secondary botanical bioactives can function optimally. FMI's analysis indicates formulation chemists prioritize these baseline fats over transient stimulants when buyers compare saffron olive polyphenols and DHA for cognitive supplements. Integrating unstable lipids into dry powder formats remains technically challenging; oxidation ruins entire product batches quickly. Premium omega-3 ingredients now require microencapsulation to maintain stability during standard retail shelf life when stored alongside active botanical extracts. Procurement teams that do not specify oxidative stability metrics risk elevated consumer returns from rancidity.
- Membrane Integration Necessity: Formulators establish baseline efficacy using marine fats. Product development directors avoid launching standalone botanical products without this lipid foundation.
- Oxidation Cascade Risks: Poorly stabilized oils degrade rapidly when mixed with highly active polyphenols. Quality assurance managers must constantly test blend stability across different temperature profiles.
- Microencapsulation Premium: Advanced delivery tech preserves sensitive oils. Brands paying this premium secure significantly longer retail distribution windows without cold-chain requirements.
Mediterranean Cognitive-First Ingredient Systems Market Analysis By Delivery Format
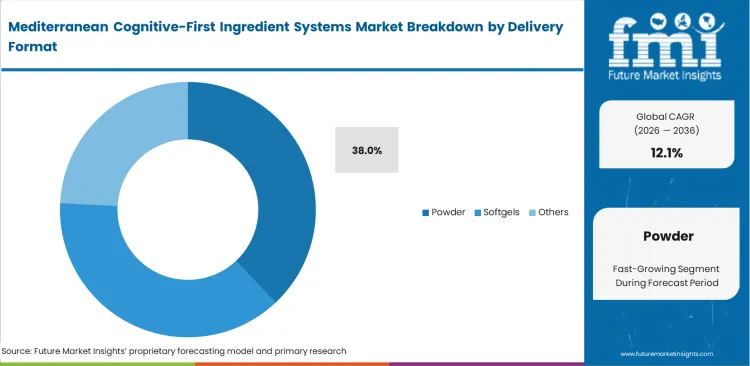
CDMO formulation officers spend significant time and budget solving botanical bitterness and suspension problems for brands that want ready-to-use Mediterranean cognition premixes. Hydrophobic herbal extracts do not disperse evenly in clear liquids and cause cloudiness or separation. The most effective cognitive blends tend to degrade in liquid over time, which makes point-of-use powder mixing more reliable than pre-mixed RTD formats. Brands forcing unstable extracts into liquid functional food ingredients applications experience total efficacy loss within three months of bottling. These formulation constraints push powder blends to a 38.0% share in 2026. Water solubility problems remain the central challenge for beverage formulators working with brain health ingredients. Consumers demand cognitive benefits in convenient drinkable formats, driving adoption of beverage-ready Mediterranean nootropic ingredients.
- Suspension Technology Limits: Active compounds resist uniform liquid distribution. Beverage scientists rely on advanced emulsifiers to prevent unsightly sedimentation in clear packaging.
- Taste Masking Costs: Mediterranean herbs carry aggressive bitter profiles. Flavor chemists allocate significant budget strictly toward covering these intense natural notes.
- Point-Of-Use Superiority: Dry formats protect delicate bioactives until consumption. Supply chain managers prefer shipping powders due to vastly lower weight and eliminating spoilage risks.
Mediterranean Cognitive-First Ingredient Systems Market Analysis By Application
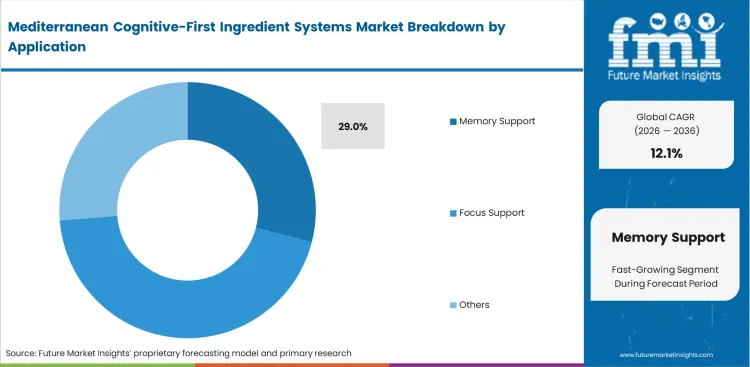
Rising rates of age-related cognitive decline drive specific clinical endpoint targeting. General wellness claims no longer satisfy educated consumers researching which Mediterranean botanicals support memory. Formulating for memory requires multi-ingredient stacks balancing olive polyphenols for cognition against neuroinflammation simultaneously. Many supplement brands overlook the fact that specific polyphenol ratios perform worse at higher doses. The efficacy follows a bell-curve, pushing concentrations beyond a certain point reduces cognitive benefit. Memory support holds a 29.0% share in 2026. Regulatory affairs officers review marketing copy for saffron extract products carefully to ensure claims stay within permitted limits. Companies pushing megadoses waste expensive raw materials and potentially trigger adverse digestive events for users.
- Circulatory Baseline Requirement: Blood flow must improve before neuronal repair begins. Formulators include vasodilating botanicals alongside specific neurogenic compounds.
- Dose Curve Complexity: Higher botanical concentrations do not equal better outcomes. Clinical trial directors struggle to identify exact optimal dosing windows for synergistic extract blends.
- Regulatory Boundary Navigation: Strict limits exist on disease claims. Marketing directors must carefully phrase benefits to avoid pharmaceutical classification and subsequent market removal.
Mediterranean Cognitive-First Ingredient Systems Market Analysis By Buyer Type
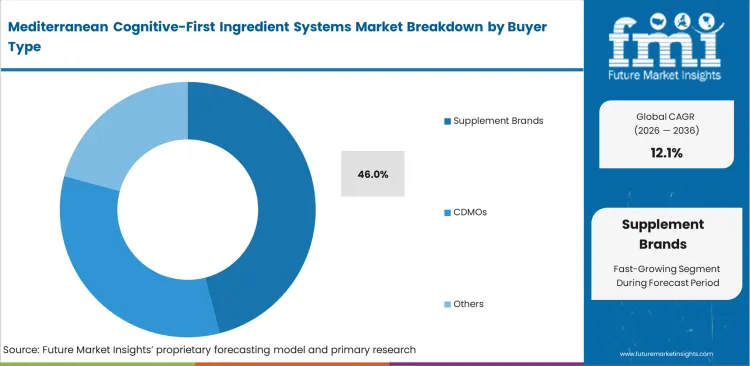
Speed to market separates successful launches from delayed failures in retail nutraceuticals. Independent brands lack capital for decade-long clinical validation pipelines. FMI analysts note that procurement managers increasingly rely on the best suppliers for Mediterranean nootropic blends to provide white-label clinical dossiers, leading the supplement brands to a 46.0% share in 2026, and as sourcing raw materials independently requires internal QA infrastructure most mid-tier brands simply cannot afford. Interestingly, reliance on a single saffron olive omega-3 ingredient blend supplier means hundreds of competing brands essentially sell identical nutraceutical CDMO formulations under different labels. Brands refusing to invest in proprietary custom blends eventually lose retail positioning to cheaper identical competitors.
- Validation Outsourcing: Brands lack internal clinical capabilities. R&D directors depend entirely on ingredient manufacturers to supply safety and efficacy documentation.
- Differentiation Crisis: Shared supplier formulas create identical retail products. Marketing executives struggle to articulate unique value propositions when chemical profiles match competitors exactly.
- Custom Stack Investments: Leading companies commission exclusive botanical combinations. Chief scientific officers use these proprietary blends to block competitor replication entirely.
Mediterranean Cognitive-First Ingredient Systems Market Analysis By Source Geography
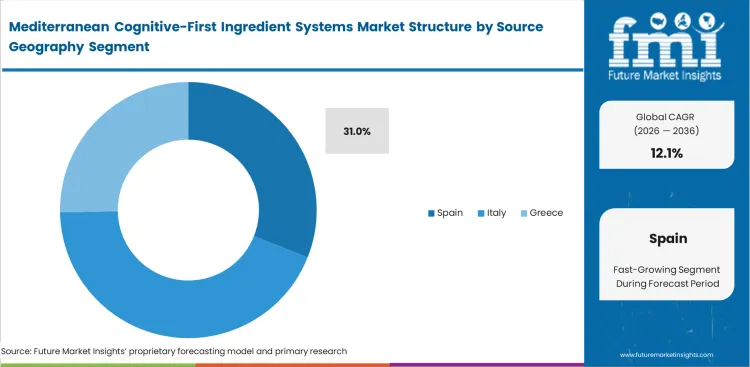
Vertical integration across agricultural and extraction tiers determines material reliability as fragmented supply chains cannot guarantee consistent bioactive yields across unpredictable climate seasons. Buying raw saffron or olive leaf on open markets guarantees inconsistent standardization while only deep industry insiders realize that soil microbiome differences across adjacent Spanish valleys drastically alter final extract efficacy profiles. According to FMI's estimates, regional agricultural cooperatives dominate global supply by linking farms directly to advanced extraction facilities with Spain dominating in this aspect, it manages to secure a 31.0% share in 2026. Supply chain directors who switch between regional suppliers based on spot pricing risk inconsistent bioactive content across batches.
- Climate Stress Necessity: Harsh growing conditions force plants to produce defensive bioactives. Agronomists purposefully restrict water to increase desirable polyphenol yields before harvest.
- Extraction Proximity Limits: Active compounds degrade quickly post-harvest. Facility operations managers must process raw botanicals within hours to capture peak molecular profiles.
- Microclimate Variations: Soil differences change chemical composition. QA directors must constantly recalibrate extraction parameters depending on which specific hillside supplied raw material.
Mediterranean Cognitive-First Ingredient Systems Market Drivers, Restraints, And Opportunities
Supplement buyers now expect to notice a cognitive effect quickly, rather than waiting thirty days of continuous use. This expectation of fast results pushes formulation chemists to move away from traditional single-ingredient products. Supplement directors are now engineering complex botanical stacks that pair the rapid onset of lemon balm extract focus support with the sustained impact of rosemary extract cognitive benefits. If a formulation fails to deliver that day-one experiential validation, retail buyers drop the SKU almost immediately. This results with the brands rushing to adopt highly bioavailable, standardized extract complexes designed specifically to cross the blood-brain barrier within hours.
On the supply side, climate variability is making consistency harder to maintain from one harvest to the next. Even when crop volumes hold up, shifts in weather can change the bioactive profile of the raw material. Dry conditions are especially difficult because they affect not only yield, but also the plant’s chemical composition, which can vary in ways suppliers cannot fully control. Extraction and standardization can help correct some of that variation, but the cost remains a serious constraint. Producers working across both organ support and premium cognitive botanical formulations often need a high level of batch consistency, and that requires advanced processing infrastructure. Smaller agricultural cooperatives may not have the financial capacity to run high-performance chromatography systems throughout the year. As a result, maintaining uniform quality at scale remains a challenge, especially for suppliers operating with limited processing budgets.
- Microencapsulation Technology: Advanced lipid coating protects volatile botanicals. Formulation scientists gain ability to mix previously incompatible Mediterranean antioxidant ingredients for supplements safely.
- Nootropic Beverage Expansion: RTD formats demand soluble ingredients. CDMO executives capture massive contracts by solving botanical bitterness and suspension problems.
- Precision Agriculture Integration: Sensor-driven farming optimizes harvest timing. Cooperative directors maximize exact bioactive yields, reducing waste and increasing premium extract volume.
Regional Analysis
Global adoption of these specialized botanical and lipid formulations fractures distinctly along regional lines. Local regulatory environments and varying levels of supply chain integration completely alter how these cognitive systems penetrate different commercial geographies. Based on regional analysis, Mediterranean Cognitive-First Ingredient Systems Market is segmented into North America, Europe, Asia Pacific, and Rest of World across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 12.9% |
| Spain | 11.8% |
| Greece | 11.5% |
| Italy | 11.2% |
| United Kingdom | 10.9% |
| Germany | 10.8% |
| Japan | 10.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Mediterranean Cognitive-First Ingredient Systems Market Analysis
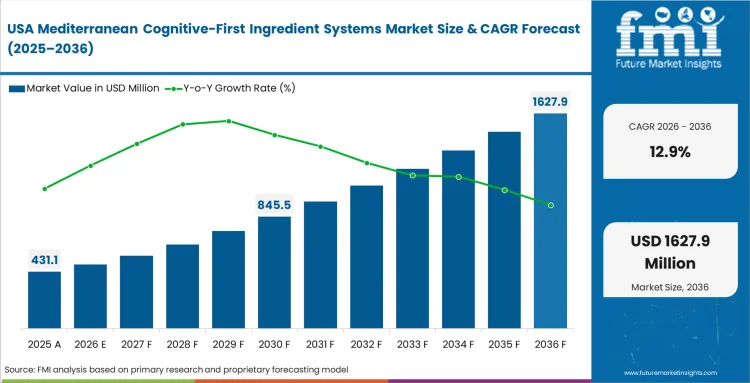
Formulation scientists constantly modify traditional encapsulated herbs to survive highly acidic liquid environments, as aggressive retail expansion of functional beverages dictates ingredient procurement strategies here. Brand managers refuse to sacrifice the clinical efficacy of dry powders while demanding rapid liquid solubility. The CDMO operations as a result offer proprietary suspension technologies to absolutely dominate this commercial landscape, based on FMI's assessment.
- United States: In the United States, rising interest in preventive cognitive health is supporting steady market growth. The market is projected to expand at a CAGR of 12.9%, with competition increasing across specialty retail channels as brands continue to refine product formats and positioning. Beverage formulators are paying close attention to solubility and taste, especially in products where clarity and drinkability matter. Companies that can improve flavor while keeping botanical functionality intact are better placed to secure premium shelf placement.
FMI's report includes Canada and Mexico within this regional scope. Cross-border supply chain integration accelerates rapidly as contract manufacturing capacity expands southward to meet rising North American demand. This operational shift allows brands to scale production efficiently while maintaining strict quality controls over sensitive botanical imports.
Europe Mediterranean Cognitive-First Ingredient Systems Market Analysis
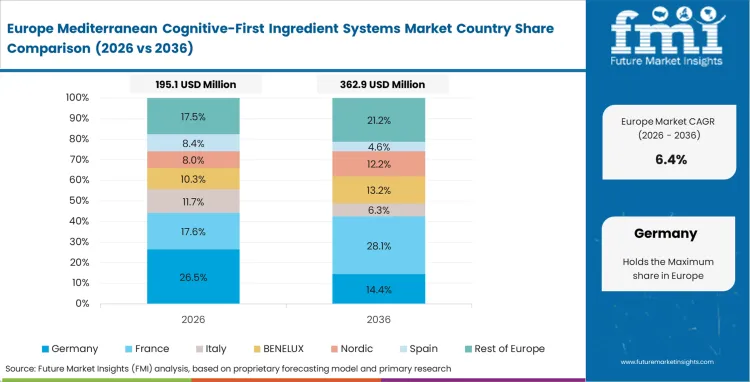
Supply chain proximity allows European brands to source remarkably fresh extracts directly from local Mediterranean cooperatives. However, strict regulatory frameworks governing health claims fundamentally shape how companies formulate and market their products locally. Regulatory affairs officers must carefully navigate complex EFSA monographs, ensuring botanical dosages hit required efficacy thresholds without crossing into restricted pharmaceutical territory.
- Spain: Spain benefits from a well-developed supply base, with close links between cultivation, processing, and extract production. That integrated structure is helping the market grow at a CAGR of 11.8%. Faster post-harvest processing also gives producers a better chance of preserving sensitive active compounds in olive, saffron, and other Mediterranean botanicals. This has helped Spanish suppliers build a strong position in premium extract markets.
- Greece: In Greece, cultivation conditions are a key part of the value proposition. Mountain growing regions and more demanding climate conditions can support higher polyphenol content in some botanicals, which gives local ingredients a distinct profile. The market is projected to expand at a CAGR of 11.5%. Interest from international buyers is rising, especially for concentrated botanical ingredients tied to origin, growing conditions, and extract quality.
- Italy: Italy continues to hold a premium position through its strength in extraction and ingredient refinement. The market is expected to grow at a CAGR of 11.2%, supported by demand for highly purified botanical fractions used in advanced supplement and health-focused formulations. Long-standing processing expertise remains an advantage, but it is now being reinforced by modern analytical methods and tighter quality control systems.
- United Kingdom: In the United Kingdom, demand is being shaped by growing consumer interest in products positioned around cognitive support and healthy aging. The market is forecast to grow at a CAGR of 10.9%. Retailers are giving more attention to formulations that combine natural positioning with stronger product backing, especially in categories where consumers are willing to pay more for ingredients that feel both credible and easy to understand.
- Germany: Germany is developing at a steady pace, with the market projected to grow at a CAGR of 10.8%. Product development in the country tends to follow a more structured path, with strong attention to botanical guidance, documentation, and formulation discipline. That approach can limit flexibility in some cases, but it also supports high standards for product quality and regulatory acceptance across the market.
FMI's detailed report also includes analysis of France and Switzerland. Precision extraction machinery manufacturing concentrates heavily near these specific borders, supporting the broader European ecosystem. This localized technical expertise ensures that extraction facilities maintain the advanced equipment necessary for highly standardized bioactive yields.
Asia Pacific Mediterranean Cognitive-First Ingredient Systems Market Analysis
Regional procurement directors actively seek validated European botanical extracts to blend securely with traditional local medicinal frameworks. Quality assurance managers demand extensive documentation verifying extract origin and purity before authorizing any import. This intense scrutiny occurs as rapidly aging demographics force national health systems to explore preventative nutritional interventions seriously.
- Japan: The strict import quality requirements filter out low-grade commodity botanicals entirely to protect vulnerable buyers, with this central focus on quality leading the industry to experience a rise of 10.4% of CAGR value, in conjunction with the extreme demographic aging that creates massive baseline demand for reliable memory preservation formulations. The clinical research directors as a result run extensive trials verifying imported Mediterranean extract efficacy within local populations, ensuring only premium standardized extracts survive regulatory scrutiny here.
FMI's analytical report includes specific coverage of China and Australia within this geographic segment. Heavy investment in local extraction infrastructure attempts to reduce long-term reliance on expensive European imports. Despite these focused efforts to domesticate production, securing identical botanical stress profiles outside the Mediterranean basin remains a profound agricultural challenge.
Competitive Aligners for Market Players
Technical formulation strength matters more than raw material access alone. Many brands can source botanical inputs, but far fewer can turn them into stable, effective finished ingredients. Leading CDMOs stand out because they can handle problems such as poor solubility, oxidation, and shelf-life instability that smaller brands often cannot solve on their own. Companies such as DSM-Firmenich and Kemin Industries have built strong positions by developing delivery systems that help protect sensitive polyphenols and improve how these ingredients perform in real formulations. Nutraceutical buyers prioritize technical support and formulation depth when choosing partners.
Clinical backing also shapes competition in this space. Established suppliers already have substantial documentation around their proprietary blends, which gives customers more confidence during product development and regulatory review. New entrants do not have that advantage. To compete at the same level, they often need to invest in costly human studies. Large pharmaceutical and health-focused companies are usually cautious about adding new ingredients unless safety and efficacy data are well established. That makes it difficult for low-cost suppliers without strong evidence to win premium manufacturing contracts.
At the same time, buyers do not want to depend too heavily on a single supplier. Large retail and supplement brands often qualify backup vendors for important extracts so they are less exposed to supply disruption. Procurement teams are also spreading contracts across more than one growing region, especially as weather variability disrupts harvest quality and volumes in single-region sourcing. In practice, suppliers with broader cultivation networks are in a stronger position than those tied to one sourcing area.
Key Players in Mediterranean Cognitive-First Ingredient Systems Market
- DSM-Firmenich
- Kemin Industries
- Pharmactive Biotech Products
- Euromed S.A.
- Indena S.p.A.
- Monteloeder
- Givaudan
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 730 million to USD 2,290 million, at a CAGR of 12.1% |
| Market Definition | Standardized botanical and lipid extracts sourced specifically from Mediterranean agricultural zones, engineered for measurable neurological support and standardized to precise bioactive markers. |
| Segmentation | Ingredient type, Delivery format, Application, Buyer type, Source geography, Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Spain, Greece, Italy, United Kingdom, Germany, Japan |
| Key Companies Profiled | dsm-firmenich, Kemin Industries, Pharmactive Biotech Products, Euromed S.A., Indena S.p.A., Monteloeder, Givaudan |
| Forecast Period | 2026 to 2036 |
| Approach | Volume tracking of specialized extracts leaving Mediterranean ports triangulated with retail sales data. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Ingredient Type:
- Omega-3 lipids
- Olive polyphenols
- Saffron extracts
- Rosemary extracts
- Lemon balm
- Nut concentrates
Delivery Format:
- Powder blends
- Softgels
- Capsules
- Gummies
- Beverage premixes
Application:
- Memory support
- Focus support
- Mood support
- Healthy aging
- Stress resilience
Buyer Type:
- Supplement brands
- Beverage brands
- Functional food brands
- CDMOs
- Pharmacies
Source Geography:
- Spain
- Italy
- Greece
- France
- Blended sourcing
Regions:
- North America
- United States
- Asia Pacific
- Japan
- China
- India
- Europe
- Germany
- United Kingdom
- Italy
- Greece
- Spain
- Middle-East
- United Arab Emirates
Bibliography
- Global Organization for EPA and DHA Omega-3s. (2024). 2024 annual report. GOED.
- International Nut and Dried Fruit Council. (2024). Nuts & dried fruits statistical yearbook 2024. INC.
- International Olive Council. (2024, December 17). World market of olive oil and table olives - data from December 2024. IOC.
- Kehtari, T., Farzaei, M. H., & Bahramsoltani, R. (2025). From mood to memory: Unlocking saffron’s potential in brain disorders. Frontiers in Pharmacology.
- McLeod, A., Espeland, M. A., Espinoza, S. E., Hildreth, K. L., Hayden, K. M., Coday, M., Rushing, J., Snyder, D. C., & Bowen, M. E. (2025). Long-term outcomes of a Mediterranean diet with or without weight loss on cognition in older African American, Hispanic, and White adults. Contemporary Clinical Trials Communications, 47, 101522.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Ingredient type specifications governing standardized extract inclusion limits.
- Delivery format challenges facing functional beverage formulation teams.
- Application protocols necessary for substantiating preventative memory claims.
- Buyer type behaviors shifting toward pre-validated clinical dossiers.
- Source geography constraints affecting global botanical supply consistency.
- Clinical testing standards required by major retail pharmacy chains.
- Taste-masking technologies protecting bitter herbal extraction profiles.
- Vertical integration strategies protecting cooperative agricultural yields.
Frequently Asked Questions
What is the forecast for Mediterranean cognition ingredients through 2036?
The valuation builds toward USD 2,290 million by the end of the forecast period as expansion relies fundamentally on mass retail channels requiring clean-label botanical formulations that carry validated clinical backing rather than traditional herbal folklore.
Which Mediterranean-origin ingredients are used in brain health supplements?
Core inputs include olive polyphenols, altitude-stressed saffron extracts, rosemary derivatives, and specialized marine lipids, which formulation chemists combine because their unique bioactives survive harsh digestion while crossing the blood-brain barrier effectively.
What limits functional beverage adoption of these extracts?
Water solubility remains incredibly difficult to achieve without destroying active compounds, as formulation scientists struggle to suspend bitter, hydrophobic botanicals in clear liquids without heavy emulsifiers that ruin clean-label aesthetics.
Why do supplement brands dominate purchasing?
Because independent brands lack internal clinical validation capabilities, procurement directors rely heavily on ingredient suppliers who provide complete, ready-to-use efficacy dossiers, allowing rapid retail launch without conducting expensive independent human trials.
How does climate alter extract quality?
Harsh growing conditions force botanicals to produce defensive polyphenols; thus, agronomists monitor drought stress carefully since too much water actually reduces bioactive concentrations, meaning well-irrigated crops produce mathematically inferior cognitive extracts.
What advantage does Spain hold in sourcing?
Vertically integrated agricultural cooperatives dominate Spanish production, where facility operations managers process botanicals immediately upon harvest, capturing volatile compounds before degradation occurs and granting them absolute control over premium extract pricing.
Why is microencapsulation necessary?
Mixing highly active polyphenols with unstable lipids causes rapid oxidation, prompting quality assurance directors to demand advanced coating technologies to prevent formulation rancidity during standard retail shelf-life periods.
What stops commodity substitution?
Strict regulatory monographs require precise bioactive ratios for health claims, meaning regulatory affairs officers immediately reject cheap, unstandardized botanical powders because they fail laboratory testing and risk immediate product recall.
How does Japan evaluate imported extracts?
Stringent national health system standards filter out low-grade materials, requiring clinical research directors to run extensive local population trials to verify imported European efficacy data before allowing widespread retail distribution.
Why do higher doses sometimes fail?
Specific botanical synergies follow a bell-curve efficacy profile, with clinical trial coordinators noting that pushing concentrations too high actually reduces cognitive benefit while increasing adverse digestive reactions.
What role do contract manufacturers play?
CDMOs act as technology gateways for smaller brands by allowing chief scientific officers at these facilities to develop proprietary taste-masking and suspension matrices, creating a deep competitive moat against generic blenders.
How do buyers resist vendor lock-in?
Procurement directors constantly split supply contracts across different geographic cooperatives, since sourcing from multiple microclimates prevents complete supply chain failure if severe weather destroys a single region's harvest.
Why are Omega-3 lipids considered foundational?
Cellular membrane fluidity requires structural fats before secondary botanicals can function properly, leading product development scientists to refuse launching premium cognitive stacks without securing this baseline lipid architecture first.
What drives United States adoption speed?
Aggressive retail expansion of functional lifestyle beverages normalizes preventative cognitive care, driving brand managers to demand highly soluble, fast-acting extracts to satisfy consumers transitioning away from synthetic energy drinks.
How do strict monographs impact Germany?
Conservative regulatory boundaries ensure high product safety by forcing product development teams to formulate strictly within established botanical guidelines, guaranteeing retail approval while avoiding risky experimental combinations.
Why do synthetic racetams face replacement?
Retail pharmacy chains demand clean-label, natural alternatives, compelling formulators to transition toward validated botanical stacks to maintain shelf space as consumer preference shifts heavily toward food-based preventative medicine.
What happens when formulations lack clinical data?
Major retailers delist unsupported products immediately, meaning marketing executives cannot rely on historical herbal folklore and must instead provide peer-reviewed documentation proving specific cognitive endpoints.
How does altitude affect Greek botanicals?
Mountain cultivation stresses plants naturally, prompting agronomists to harvest these unique variants because they yield exceptionally high concentrations of protective polyphenols compared to sea-level equivalents.
What makes independent QA testing difficult?
Standardizing complex botanical matrices requires expensive chromatography equipment that small brands cannot afford, forcing their total reliance on supplier-provided certificates of analysis.
Why are generic vitamins excluded from this category?
Commodity nutrients lack specific, targeted cognitive endpoints, whereas this market strictly bounds ingredients engineered and standardized precisely for neurological support, separating them from general wellness multivitamins.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Omega-3 Lipids
- Olive Polyphenols
- Others
- Omega-3 Lipids
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Delivery Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Delivery Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Delivery Format, 2026 to 2036
- Powder
- Softgels
- Others
- Powder
- Y to o to Y Growth Trend Analysis By Delivery Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Delivery Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Memory Support
- Focus Support
- Others
- Memory Support
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Buyer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Buyer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Buyer Type, 2026 to 2036
- Supplement Brands
- CDMOs
- Others
- Supplement Brands
- Y to o to Y Growth Trend Analysis By Buyer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Buyer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source Geography
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Source Geography, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Source Geography, 2026 to 2036
- Spain
- Italy
- Greece
- Spain
- Y to o to Y Growth Trend Analysis By Source Geography, 2021 to 2025
- Absolute $ Opportunity Analysis By Source Geography, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Delivery Format
- By Application
- By Buyer Type
- By Source Geography
- Competition Analysis
- Competition Deep Dive
- DSM-Firmenich
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Kemin Industries
- Pharmactive Biotech Products
- Euromed S.A.
- Indena S.p.A.
- Monteloeder
- Givaudan
- DSM-Firmenich
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Delivery Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Buyer Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Source Geography, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Delivery Format
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Buyer Type
- Figure 15: Global Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Source Geography
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Delivery Format
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Buyer Type
- Figure 41: North America Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Source Geography
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Delivery Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Buyer Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Source Geography
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Delivery Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Buyer Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Source Geography
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Delivery Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Buyer Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Source Geography
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Delivery Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Buyer Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Source Geography
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Delivery Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Buyer Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Source Geography
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Delivery Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Delivery Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Delivery Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Buyer Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Buyer Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Buyer Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Source Geography, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Source Geography, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Source Geography
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

