Mediterranean Metabolic Syndrome Support Ingredient Packs Market
The Mediterranean Metabolic Syndrome Support Ingredient Packs Market Is Segmented By Ingredient Type (Polyphenols, Omega-3s, Probiotics, Prebiotics, Berberine), Pack Format (Capsule Packs, Sachet Packs, Powder Blends, Stick Packs, Softgels), Health Focus (Glucose Support, Lipid Support, Weight Support, Gut Support, Inflammation Support), End Use (Dietary Supplements, Functional Foods, Medical Nutrition, Personalized Packs, Practitioner Packs), Distribution Channel (B2B Formulation, Brand Owners, Pharmacies, E-Commerce, Clinics), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Size, Market Forecast and Outlook By FMI
The Mediterranean metabolic syndrome support ingredient packs market surpassed a valuation of USD 270 million in 2025 and is expected to reach USD 310 million in 2026. From 2026 to 2036, market revenue is projected to grow at a 12.8% CAGR, taking total sales to about USD 1,030 million by 2036. Growth is being supported by rising demand for more targeted Mediterranean ingredient systems designed to address multiple metabolic health markers, as buyers move beyond broad botanical positioning toward clinically supported formulations.
Formulation directors at major nutraceutical brands face an immediate requirement to validate physiological mechanisms rather than simply marketing traditional diets. Regulatory bodies increasingly demand precise clinical endpoints for cardiovascular and glycemic claims. Delaying the transition to standardized, bio-assayed olive phenolic complexes risks immediate delisting from clinical nutrition formularies. Practitioners now require multi-ingredient combinations that address lipid oxidation and insulin resistance simultaneously, heavily relying on mediterranean diet bioactives for metabolic syndrome. Isolated extracts fail to provide the combined effects documented in regional clinical trials. Suppliers must deliver pre-blended, validated nutraceutical ingredients that guarantee exact active ratios per serving. Brand owners that do not secure validated combinations face stronger generic competition and closer regulatory scrutiny.
Summary of Mediterranean Metabolic Syndrome Support Ingredient Packs Market
- Mediterranean Metabolic Syndrome Support Ingredient Packs Market Definition
- Pre-formulated, standardized combinations of bioactive compounds specifically calibrated to target insulin resistance and dyslipidemia, providing contract manufacturers with clinically validated, ready-to-encapsulate metabolic interventions.
- Demand Drivers in the Market
- Rigorous clinical endpoint requirements compel formulation directors to source pre-validated, multi-pathway bioactive combinations.
- Escalating metabolic syndrome rates force public health administrators to integrate standardized nutritional interventions into chronic care protocols.
- Complex extraction protocols drive contract manufacturers to rely on specialized B2B formulation partners for consistent active ratios.
- Key Segments Analyzed in the FMI Report
- Polyphenols: 29.0% share in 2026, driven by extensive clinical data supporting endothelial function and lipid oxidation prevention.
- Capsule Packs: 38.0% share in 2026 due to superior active compound protection and precise dosage delivery mechanics.
- Glucose Support: 31.0% share in 2026 as practitioner prescribing shifts toward early-stage insulin resistance management.
- Dietary Supplements: 46.0% share in 2026, sustained by rapid consumer adoption of clinically validated metabolic therapies.
- B2B Formulation: 41.0% share in 2026, reflecting the high technical barrier of stabilizing multi-ingredient bioactive combinations.
- Egypt: 13.6% CAGR, fueled by state-level integration of standardized metabolic interventions into subsidized public health frameworks.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage analysts observe that, "The isolated bioactives frequently degrade when combined without proper matrix stabilization with the true constraint lying in achieving batch-to-batch pharmacokinetic consistency across multiple active compounds simultaneously. Contract manufacturers who master this complex co-processing capture immediate, sticky institutional supply agreements, while those attempting to blend raw extracts internally face chronic stability failures and subsequent regulatory rejection."
- Strategic Implications / Executive Takeaways
- B2B formulators must secure proprietary stabilization technologies to prevent active compound degradation in multi-ingredient matrices.
- Procurement heads face immediate pressure to lock in long-term supply agreements with primary extractors to avoid raw material volatility.
- Clinical nutrition brands risk severe market share erosion if they fail to upgrade from generic botanical claims to specific, validated pharmacokinetic profiles.
- Methodology
- Primary interviews with formulation scientists map the technical transition from single-ingredient sourcing to integrated pack procurement.
- Analysis of clinical trial registries quantifies the pipeline of validated multi-pathway metabolic interventions.
- Aggregation of B2B supply contracts anchors baseline demand for standardized omega 3 concentrates.
- Cross-referencing extraction yields against final product formulations validates total volume projections.
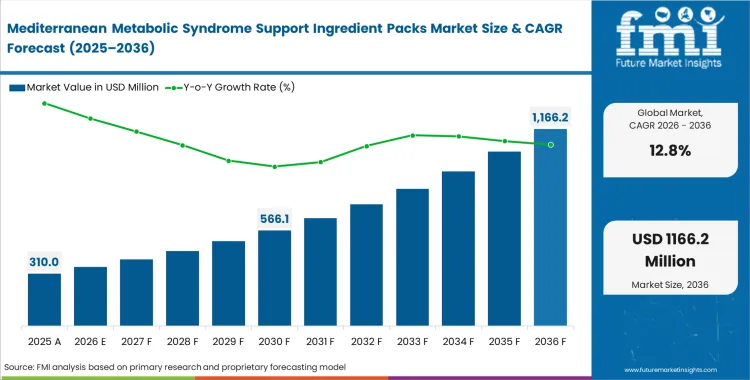
Once pharmaceutical-grade extraction facilities achieve standardized batch-to-batch active ingredient ratios, food supplement ingredients transition from alternative health options to front-line medical nutrition prescriptions. Dietary supplement manufacturers dictate this crossing point by requiring specific pharmacokinetic profiles, particularly for emerging GLP-1 companion nutrition ingredients, before signing supply contracts. Validated multi-pathway combinations improve the likelihood of inclusion in clinical guidelines for metabolic management. Formulators who lock in these validated supply chains early capture institutional contracts that smaller brands cannot service.
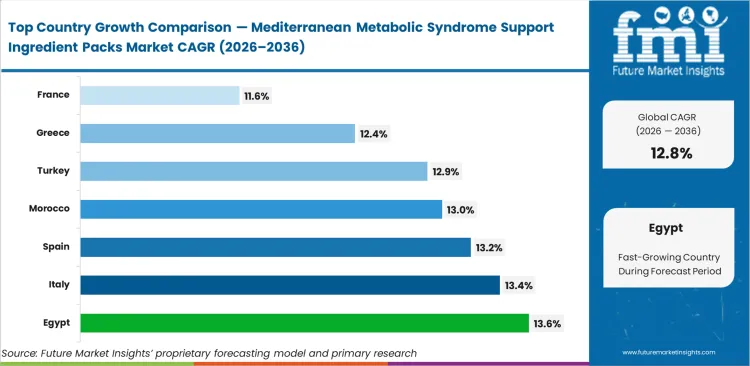
Egypt leads expansion at 13.6% as state-sponsored metabolic health initiatives subsidize clinically validated combinations. Italy tracks at 13.4%, with regional agricultural cooperatives forming direct supply agreements with medical nutrition ingredients for metabolic syndrome brands. Spain follows at 13.2% on the back of aggressive practitioner-led bioactive ingredient prescribing frameworks. Morocco advances at 13.0% as domestic extraction infrastructure scales to meet European pharmaceutical standards. Turkey expands at 12.9%, prioritizing nutraceutical actives within chronic disease management protocols. Greece registers 12.4% as heritage olive producers pivot to high-value extract processing. France climbs at 11.6%, focusing strictly on registered medical nutrition pathways. Divergence across these regions stems entirely from the speed at which local regulatory bodies integrate standardized combinations into reimbursable clinical guidelines.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Definition
Mediterranean Metabolic Syndrome Support Ingredient Packs represent pre-formulated, standardized blends of bioactive compounds specifically calibrated to address insulin resistance, dyslipidemia, and localized inflammation. These combinations require precise molecular ratios mimicking regional dietary benefits, delivered in clinically validated dosages. Contract manufacturers rely on these integrated systems to bypass complex internal formulation challenges and accelerate time-to-market for targeted metabolic therapies.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Inclusions
Standardized polyphenol extracts, concentrated omega-3 fractions, targeted prebiotics, and specific multistrain bacterial cultures fall directly within scope. Raw material combinations must feature clinical validation for metabolic syndrome endpoints, utilizing targeted gut microbiome metabolic health pack ingredients. Supply formats cover bulk powder blends, specialized beadlets, and stabilized liquid emulsions intended for secondary manufacturing, often featuring an olive leaf bergamot berberine blend. Co-processed EPA DHA omega 3 ingredients designed for immediate encapsulation are also captured.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Exclusions
Single-ingredient botanical powders lacking metabolic clinical validation sit outside this classification. Generic olive oils, unstandardized fish oils, and bulk commodities intended for general food manufacturing are excluded. These baseline materials lack the required batch-to-batch active compound standardization necessary for medical nutrition claims. General wellness multivitamins and standalone mineral supplements also fail to meet the specific multi-pathway intervention criteria defining this category.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Research Methodology
- Primary Research: Procurement directors, formulation scientists, and clinical nutrition executives
- Desk Research: European Food Safety Authority novel food registries, clinical trial databases, and botanical extract patent filings
- Market-Sizing and Forecasting: Validated active compound extraction volumes converted to standardized pack equivalents
- Data Validation and Update Cycle: Independent contract manufacturing output logs and B2B nutraceutical supply contracts
Segmental Analysis
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis By Ingredient Type
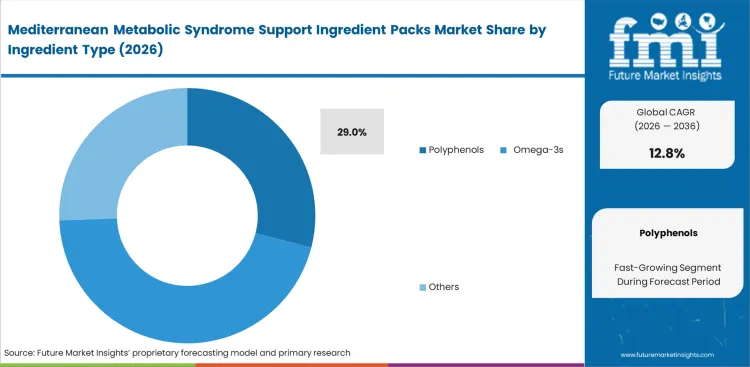
Clinical validation pipelines dictate formulation priorities across this category with polyphenols holding a 29.0% share because specific phenolic compounds demonstrate measurable impact on endothelial function and lipid oxidation. According to FMI's estimates, sourcing teams at clinical nutrition brands are giving more attention to prebiotic ingredient networks and high-grade olive polyphenol ingredients as they work to meet stricter practitioner expectations in metabolic health formulations. Securing consistent hydroxytyrosol and oleuropein ratios requires sophisticated extraction infrastructure that few suppliers possess. Formulation scientists attempting to blend generic extracts frequently encounter severe degradation profiles during shelf-life testing, a common failure point for standard olive polyphenol supplements.
- Clinical Qualification: Standardized phenolic profiles satisfy rigorous European Food Safety Authority guidelines for cardiovascular health claims. Formulation scientists gain immediate access to reimbursable medical nutrition pathways.
- Extraction Infrastructure: Sophisticated concentration techniques ensure exact active ratios per batch. Procurement directors avoid costly raw material rejections and streamline final product manufacturing.
- Stability Management: Specialized matrices prevent rapid oxidation of sensitive phenolic compounds. If active levels drop below label claims before expiration, quality control managers must address potential product recalls.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis By Pack Format
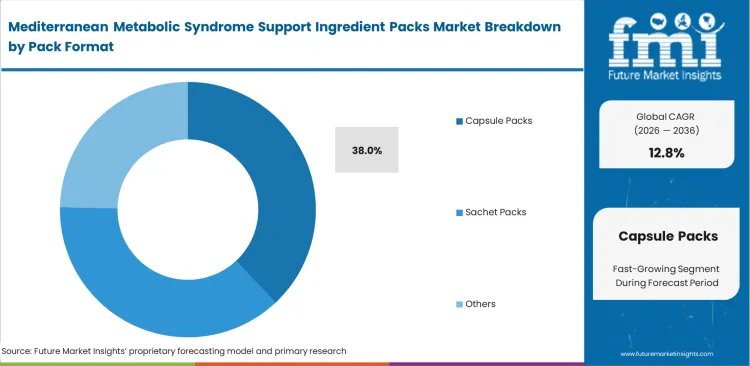
Precise dosage delivery mechanics determine adoption trajectories for final product delivery as they offer superior protection against environmental degradation while maintaining exact active combinations. FMI analysts note that production managers at multistrain probiotics facilities favor this format to bypass complex liquid stabilization challenges. Hermetically sealed environments prevent cross-reactivity between aggressive phenolic compounds and sensitive lipid fractions. Brand owners transitioning from loose powders to precision capsules dramatically reduce consumer compliance failures, especially when launching private label metabolic syndrome supplement packs, sealing the capsule packs sale to a 38.0% share in 2026. Polymer composition determines whether active compounds survive the upper gastrointestinal tract. Incompatible encapsulation materials cause active degradation before the ingredients reach their target absorption sites.
- Oxidation Prevention: Specialized capsule polymers block oxygen and moisture transmission entirely. Quality control managers ensure multi-ingredient stability throughout extended global supply chains.
- Dosage Precision: Automated filling equipment guarantees exact milligram ratios of distinct bioactives. Clinical directors must deliver identical pharmacokinetic profiles to maintain practitioner confidence.
- Cross-Reactivity Control: Internal compartmentalization prevents chemical interactions between antagonistic ingredients. Formulators face rapid product degradation if they ignore these critical spatial separation requirements.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis By Health Focus
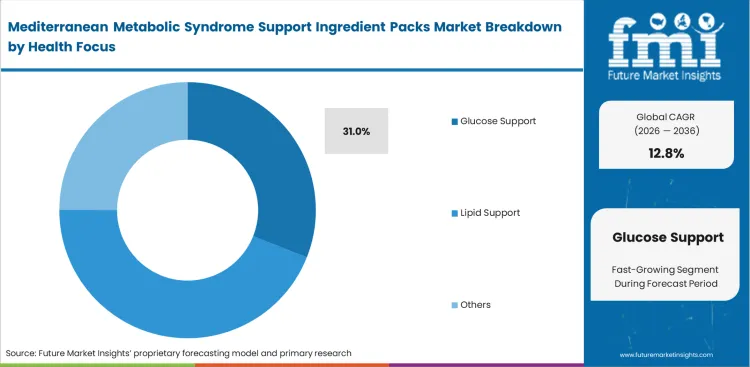
Targeted metabolic intervention requires specific physiological mechanism engagement propelling the glucose support to capture a 31.0% share as clinical focus intensifies on early-stage insulin resistance management, further driving the demand for specialized mediterranean ingredient packs for insulin resistance. Based on FMI's assessment, medical directors at functional food brands construct portfolios around these heat stable probiotics to address escalating pre-diabetic demographics. Reversing peripheral insulin insensitivity demands precise combinations of berberine and specific phenolic fractions. General wellness formulations completely fail to trigger the necessary enzymatic pathways required for measurable glycemic control. Achieving these endpoints requires precise pharmacokinetic timing that standard extract blending cannot deliver, forcing formulation scientists to carefully evaluate olive polyphenols vs berberine for metabolic syndrome support when designing synchronized matrices. Formulators that miss these physiological requirements produce supplements that do not deliver measurable outcomes, which weakens their standing with healthcare practitioners.
- Enzymatic Activation: Precise bioactive ratios trigger specific metabolic pathways controlling cellular glucose uptake. Clinical directors achieve measurable reductions in fasting blood sugar metrics.
- Practitioner Adoption: Validated glycemic control mechanisms convince healthcare professionals to integrate these supplements into primary care protocols. Brand managers secure highly sticky, recurring institutional revenue streams.
- Formulation Complexity: Synchronizing the absorption rates of distinct active compounds requires advanced matrix engineering. Procurement teams face chronic supply delays if their B2B partners lack this specific stabilization expertise.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis by End use
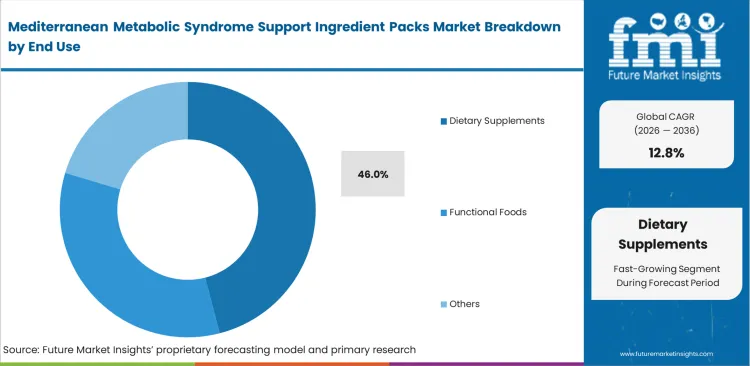
Speed to market defines procurement strategies across final product categories. FMI observes that product development heads leverage pre-formulated functional food ingredients to bypass years of internal stability testing leading the dietary supplements to account for 46.0% share because brand owners must rapidly deploy validated combinations to capture surging practitioner demand for advanced metabolic health supplements. Outsourcing the complex co-processing of antagonistic bioactives allows brands to focus entirely on marketing and distribution. Attempting to build multi-ingredient stability matrices internally severely delays product launches and drains R&D resources. General industry metrics obscure the fact that practitioner-tier formulations require specific clinical dossiers that bulk ingredient suppliers cannot provide. Brands using unvalidated ingredient blends risk regulatory rejection and loss of credibility within clinical networks.
- Rapid Deployment: Pre-stabilized blends eliminate extensive internal formulation trials. Product development heads accelerate time-to-market for complex multi-pathway metabolic interventions.
- Clinical Substantiation: Comprehensive dossiers accompanying these packs satisfy rigorous regulatory scrutiny. Compliance officers avoid costly product launch delays and marketing claim rejections.
- Resource Allocation: Outsourcing stabilization complexities allows brands to concentrate capital on clinical trials. Operations managers that spend resources on basic matrix stability lose time and capital that could go toward clinical validation and market development.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis By Distribution Channel
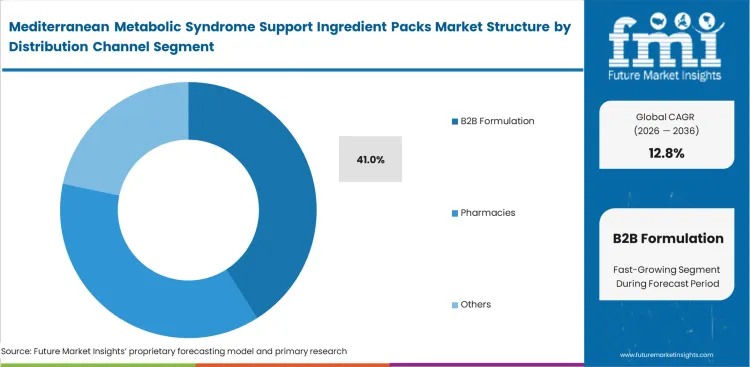
Technical complexity forces brands to restructure their entire supply chain architecture leading the B2B formulation to capture a 41.0% share as the technical barrier of stabilizing multi-ingredient matrices exceeds the capabilities of standard contract manufacturers. In FMI's view, supply chain directors rely on specialized botanical extracts partners and dedicated metabolic health premix manufacturers to guarantee exact active compound ratios. Ensuring batch-to-batch consistency across varying agricultural harvests requires sophisticated analytical infrastructure. Brands that source individual raw materials directly often run into blending inconsistencies that compromise active compound ratios. The critical tension involves balancing raw material cost against the enormous expense of discarded, unstable production runs. Selecting low-cost individual ingredients without proper co-processing technology significantly raises the risk of final product rejection.
- Analytical Infrastructure: Specialized B2B partners utilize advanced chromatography to verify exact active ratios. Supply chain directors eliminate costly raw material quarantines and production bottlenecks.
- Yield Optimization: Proprietary extraction techniques maximize the recovery of high-value phenolic compounds. Procurement heads must secure these efficient supply lines to maintain viable final product margins.
- Harvest Variability: Advanced blending protocols normalize natural fluctuations in botanical active levels. Brand owners that do not buffer against seasonal raw material variation risk batch rejections and compliance delays.
Mediterranean Metabolic Syndrome Support Ingredient Packs Market Drivers, Restraints, and Opportunities
Rigorous clinical endpoint requirements compel formulation directors to source pre-validated, multi-pathway glucose and lipid support nutraceutical blends immediately. Regulatory bodies across key regions now actively reject generic traditional diet claims, demanding precise pharmacokinetic data for cardiovascular and glycemic interventions. Delaying the integration of these highly standardized botanical ingredients forces brands out of the highly lucrative practitioner-recommended tier. Medical nutrition companies must secure proprietary extraction agreements to guarantee exact active ratios of polyphenols and specific lipid fractions within their metabolic health premix for supplements. The speed at which healthcare systems integrate these validated combinations into chronic care protocols dictates total commercial expansion.
Severe extraction and stabilization complexities stall rapid portfolio expansion for mid-tier supplement brands. Achieving batch-to-batch consistency across multiple active compounds requires analytical infrastructure that standard contract manufacturers simply do not possess. Unpredictable agricultural yields create massive variations in baseline active levels, necessitating sophisticated, high-cost normalization protocols. While emerging synthetic biology techniques offer potential workarounds, current regulatory frameworks heavily restrict their use in natural dietary supplement categories. This friction creates severe production bottlenecks for procurement teams attempting to scale new metabolic product lines.
- Precision Fermentation: Engineering specific microbial strains to produce targeted phenolic compounds bypasses agricultural variability. R&D directors secure infinitely scalable, highly consistent bioactive supply lines.
- Nanoparticle Delivery: Utilizing advanced lipid botanical co2 extracts encapsulation dramatically improves the bioavailability of poorly absorbed compounds. Formulation scientists uncover entirely new efficacy claims for legacy functional ingredients for metabolic syndrome.
- Personalized Diagnostic Integration: Linking specific bioactive ratios to individual biomarker testing protocols creates highly tailored metabolic interventions. Brand managers establish incredibly sticky, high-margin consumer subscriptions targeting specialized bergamot extract metabolic health demographics.
Regional Analysis
Based on regional analysis, Mediterranean Metabolic Syndrome Support Ingredient Packs market is segmented into North America, Latin America, Europe, East Asia, South Asia, and Middle East & Africa across 40 plus countries. Growth rates vary significantly across regions, shaped by local regulatory frameworks and proximity to key botanical raw materials. The region-by-region breakdown highlights how state mandates and agricultural heritage uniquely shape procurement and formulation strategies across the globe.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Egypt | 13.6% |
| Italy | 13.4% |
| Spain | 13.2% |
| Morocco | 13.0% |
| Turkey | 12.9% |
| Greece | 12.4% |
| France | 11.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Middle East & Africa Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis
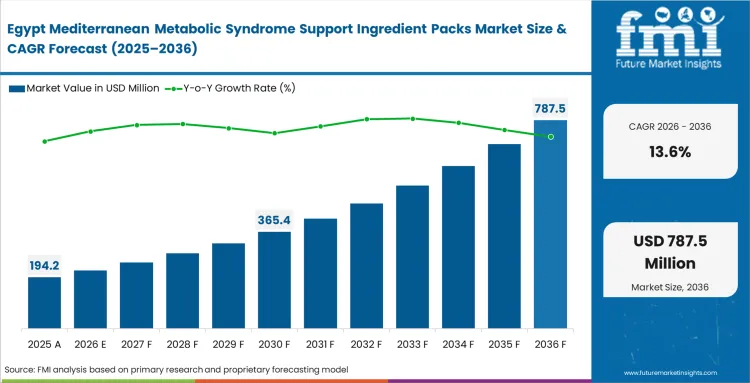
Across the Middle East and Africa, the push for metabolic health is increasingly driven by aggressive state-sponsored initiatives that are completely reshaping how brands procure their materials. With public health officials rushing to embed standardized nutritional interventions into subsidized chronic care programs, local extractors are under immense pressure. FMI's analysis indicates that to meet these new institutional demands, suppliers must rapidly elevate their analytical capabilities to match strict European benchmarks. Bidding on these massive public contracts with unverified, unstandardized raw materials now guarantees immediate disqualification. The entire regional focus, as a result, has shifted toward building highly reliable dietary supplements supply chains that can consistently deliver precise pharmacokinetic profiles.
- Egypt: Egypt is emerging as a faster-growing market as clinical nutrition and metabolic health suppliers respond to stronger public health attention around chronic disease management. Demand for metabolic syndrome support ingredient packs in Egypt is projected to grow at a 13.6% CAGR from 2026 to 2036. This is creating more demand for standardized, compliance-ready ingredients and encouraging buyers to secure longer-term extraction and supply agreements.
- Morocco: Morocco is gaining ground as local extraction capacity improves and suppliers position themselves to serve European buyers more effectively. Sales of metabolic syndrome support ingredient packs linked to Morocco are expected to rise at a 13.0% CAGR between 2026 and 2036. Investment in better processing and testing infrastructure is helping local producers meet stricter requirements for active content, consistency, and export quality.
- Turkey: Turkey is witnessing steady momentum as metabolic health formulations become more closely aligned with clinically supported care approaches. Revenue from metabolic syndrome support ingredient packs in Turkey is forecast to expand at a 12.9% CAGR from 2026 to 2036. Growth is being supported by increasing interest in validated phenolic combinations and by greater emphasis on documented efficacy in health-focused product development.
FMI's report includes South Africa and GCC Countries. Rapid urbanization across these emerging hubs is driving distinct shifts in regional disease profiles, fundamentally changing how practitioners approach metabolic interventions. Rising insulin resistance rates in urban populations are pushing formulators to develop bioactive combinations calibrated to local dietary patterns and clinical guidelines.
Europe Mediterranean Metabolic Syndrome Support Ingredient Packs Market Analysis
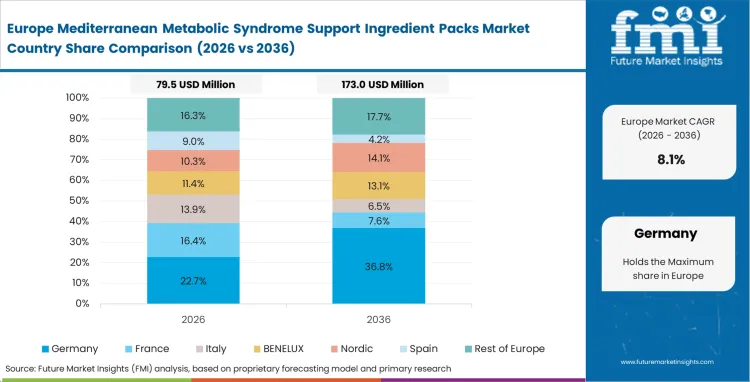
The formulation landscape across Europe is fundamentally dictated by the rigorously enforced guidelines of the European Food Safety Authority, creating a deeply interconnected continental supply chain. While intense regulatory scrutiny in western markets eliminates low-tier generic extract suppliers from practitioner networks, heritage olive producers in the east are aggressively pivoting toward high-value, standardized extract processing to meet this surging institutional demand. To capture lucrative, long-term contracts from premier medical nutrition brands, extraction facility managers are abandoning low-margin commodity oil production and investing heavily in advanced chromatography. In the end, securing these clinically validated pathways, backed by massive, endpoint-specific clinical dossiers, is the only real mechanism for brands to achieve reimbursable medical claims in this highly restricted environment.
- Italy: In Italy, regional cooperatives play an important role in the supply chain and are helping the market expand at a CAGR of 13.4%. Direct supply ties with medical nutrition brands are strengthening the link between raw material processing and finished product development. As extraction and formulation move closer together, Italian suppliers are building a more integrated model that is shaping broader nutraceutical activity across Europe.
- Spain: Spain’s market is growing at a CAGR of 13.2%, supported in part by practitioner-led adoption of more structured metabolic health formulations. This creates demand for ingredients that can deliver consistent performance and fit within closely monitored product frameworks. For that reason, formulators often work with established olive polyphenol suppliers that can offer both quality control and stronger documentation.
- Greece: In Greece, agricultural networks are moving further toward specialized extraction as demand rises for bioactive ingredients with clearer functional value. The market is projected to grow at a CAGR of 12.4%. To meet buyer expectations, producers are investing more in analytical testing and ingredient verification. That shift is helping Greek suppliers strengthen their position in a more quality-focused export market.
- France: France is expected to grow at a CAGR of 11.6%, with market development shaped by a more regulated product environment. Entry often depends on stronger clinical support, product stability data, and longer development timelines. For companies targeting medical nutrition channels, that raises the cost of entry, but it can also open access to more specialized and higher-value contracts once regulatory requirements are met.
FMI's report includes major economies like Germany and the United Kingdom alongside developing markets such as Poland and Russia. Across the continent, rapidly escalating healthcare costs are forcing public administrators to aggressively evaluate preventative nutritional interventions, leading national health systems to increasingly subsidize specialized metabolic packs.
Competitive Aligners for Market Players
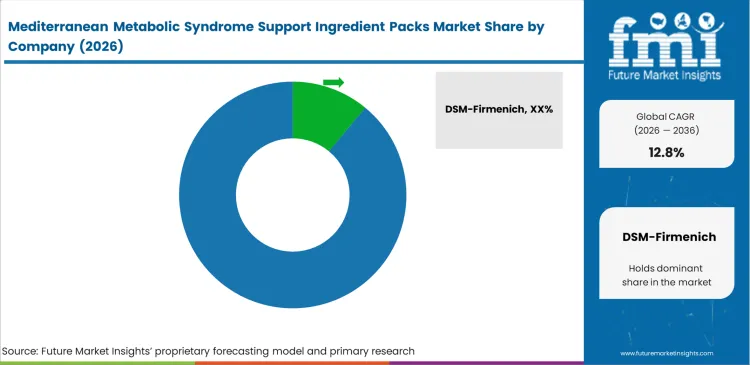
Formidable technical barriers define competitive interaction within this specialized extraction sector. DSM-Firmenich commands 13.0% share by mastering the complex co-processing required to stabilize highly reactive phenolic compounds alongside sensitive lipid fractions. Procurement directors at top-tier clinical nutrition brands evaluate suppliers strictly on their analytical infrastructure and their ability to guarantee exact batch-to-batch pharmacokinetic profiles. Securing these massive institutional supply agreements requires extensive clinical validation dossiers that smaller extractors simply cannot finance. The absolute necessity for verified clinical endpoints forces intense consolidation, as mid-tier suppliers fail to meet escalating European Food Safety Authority standards.
Established primary extractors possess deep, multi-generational relationships with regional agricultural cooperatives, ensuring preferential access to specific high-yield botanical harvests. Challengers must construct entirely new supply networks while simultaneously investing in advanced multi nutritional supplement analytical testing equipment. Suppliers that bypass established agricultural relationships face raw material shortages and significant variation in baseline active compound levels. Securing consistent agricultural input remains the critical bottleneck, severely limiting the speed at which new entrants can scale production of validated metabolic interventions.
Large functional food brands actively resist supply chain exclusivity by enforcing stringent dual-sourcing qualification protocols. Procurement heads require multiple extractors to use identical analytical verification methods so raw materials can be substituted without reformulation. As extraction technologies advance, specialized B2B formulators capture disproportionate value by offering proprietary stabilization matrices that generic extractors cannot replicate. As demand for clinically validated multi-ingredient formulations grows, primary agricultural suppliers face pressure to meet the extraction and standardization standards required by medical nutrition buyers.
Key Players in Mediterranean Metabolic Syndrome Support Ingredient Packs Market
- DSM-Firmenich
- Indena
- Euromed
- ADM
- IFF
- Glanbia Nutritionals
- Pharmactive
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 310 million to USD 1,030 million, at a CAGR of 12.8% |
| Market Definition | Pre-formulated, standardized combinations of bioactive compounds specifically calibrated to target insulin resistance and dyslipidemia. These combinations provide contract manufacturers with clinically validated, ready-to-encapsulate metabolic interventions. |
| Segmentation | By Ingredient type, By Pack format, By Health focus, By End use, By Distribution channel |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia, Middle East & Africa |
| Countries Covered | Egypt, Italy, Spain, Morocco, Turkey, Greece, France, Germany, United Kingdom, South Africa, GCC Countries, Poland, Russia |
| Key Companies Profiled | dsm-firmenich, Indena, Euromed, ADM, IFF, Glanbia Nutritionals, Pharmactive |
| Forecast Period | 2026 to 2036 |
| Approach | Validated active compound extraction volumes converted to standardized pack equivalents |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
By Ingredient Type:
- Polyphenols
- Omega-3s
- Probiotics
- Prebiotics
- Berberine
By Pack Format:
- Capsule packs
- Sachet packs
- Powder blends
- Stick packs
- Softgels
By Health Focus:
- Glucose support
- Lipid support
- Weight support
- Gut support
- Inflammation support
By End Use:
- Dietary supplements
- Functional foods
- Medical nutrition
- Personalized packs
- Practitioner packs
By Distribution Channel:
- B2B formulation
- Brand owners
- Pharmacies
- E-commerce
- Clinics
Regions:
- North America
- United States
- Asia Pacific
- Japan
- China
- India
- Europe
- Germany
- United Kingdom
- France
- Italy
- Greece
- Spain
- Middle-East
- United Arab Emirates
- Turkey
- Morocco
- Egypt
Bibliography
- Dama, A., Tarasiuk, A., DiNicolantonio, J. J., O’Keefe, J. H., McCarty, M. F., & Török, B. (2024). Targeting metabolic diseases: The role of nutraceuticals in oxidative stress and inflammation modulation. Nutrients, 16(4), 507.
- Vignesh, A., et al. (2024). A review on the influence of nutraceuticals and functional foods in human health. Food Bioscience.
- Wang, Q., Huang, H., Yang, Y., Yang, X., Li, X., Zhong, W., Wen, B., He, F., & Li, J. (2024). Reinventing gut health: Leveraging dietary bioactive compounds for the prevention and treatment of diseases. Frontiers in Nutrition, 11, 1491821.
- Mazumdar, S., et al. (2025). The use of functional foods and its association to chronic multimorbidity among adults. Scientific Reports.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Exact extraction infrastructure requirements necessary to guarantee consistent batch-to-batch phenolic ratios.
- Operational consequences of utilizing unstandardized botanical powders in clinical nutrition formulations.
- Specific enzymatic pathways activated by validated multi-ingredient metabolic interventions.
- Technical challenges surrounding the co-processing of antagonistic bioactives in single delivery formats.
- Regulatory scrutiny applied to European medical nutrition cardiovascular claims.
- Competitive advantages of proprietary stabilization matrices over generic bulk extract blending.
- Agricultural supply constraints impacting the scaling of high-yield botanical extraction.
- Practitioner adoption dynamics driving demand for multi-pathway glycemic control supplements.
Frequently Asked Questions
What ingredients are used in mediterranean metabolic syndrome support packs?What role does matrix stabilization play in product development?
These packs utilize precise, standardized blends of olive polyphenols, bergamot extracts, multistrain probiotics, and omega-3 fractions to target multiple physiological endpoints simultaneously.
How do olive polyphenols support metabolic health?
Standardized olive polyphenols actively improve endothelial function and prevent lipid oxidation, satisfying rigorous clinical requirements for cardiovascular health claims.
Are probiotic blends effective for metabolic syndrome support?
Specific multistrain probiotic matrices enhance gut barrier integrity and modulate systemic inflammation, acting as foundational microbiome ingredients for metabolic health.
What is the market size of mediterranean metabolic health ingredients?
The category reaches USD 310 million in 2026, driven by the escalating clinical requirement for fully standardized, multi-pathway metabolic interventions.
How do private label metabolic syndrome supplement packs compete with established brands?
Private label manufacturers leverage pre-validated B2B formulation matrices to bypass complex internal stability testing, enabling rapid deployment of practitioner-grade therapies.
Why compare bergamot olive polyphenols probiotics and omega-3 for metabolic health?
Formulators must balance these highly reactive compounds within a single matrix to address lipid oxidation, insulin resistance, and localized inflammation concurrently without active degradation.
What are the leading suppliers of mediterranean metabolic health ingredients?
The supply chain is dominated by specialized B2B formulators and primary extractors who possess the advanced analytical infrastructure required to guarantee exact batch-to-batch pharmacokinetic profiles.
Why choose a metabolic health premix for supplements over single ingredients?
Pre-stabilized premixes eliminate extensive internal formulation trials and prevent cross-reactivity between antagonistic bioactives, dramatically accelerating time-to-market for complex interventions.
How do these packs function as GLP-1 companion nutrition ingredients?
Clinically validated Mediterranean botanical blends preserve lean muscle mass and stabilize peripheral insulin sensitivity, providing critical nutritional support during aggressive medically induced weight loss phases.
Why do polyphenols command the highest share among ingredients?
Polyphenols hold 29.0% share because specific phenolic compounds successfully demonstrate measurable impacts on endothelial function, leading procurement directors to prioritize these networks to satisfy rigorous practitioner demands.
What drives the preference for capsule packs?
Capsule packs secure 38.0% share because their hermetically sealed environments offer superior protection against environmental degradation and prevent cross-reactivity between aggressive phenolic compounds and sensitive lipid fractions.
Why does glucose support lead the health focus segment?
Glucose support captures 31.0% share as clinical focus intensifies on early-stage insulin resistance management, which demands precise combinations of berberine and specific phenolic fractions that simple wellness formulations cannot replicate.
How do dietary supplements maintain their dominant position?
Dietary supplements account for 46.0% share because brand owners rapidly deploy validated combinations to capture surging practitioner demand while bypassing years of internal stability testing.
Why is B2B formulation the preferred distribution channel?
B2B formulation captures 41.0% share due to the enormous technical barrier of stabilizing multi-ingredient matrices, forcing supply chain directors to rely on specialized partners to guarantee exact active compound ratios.
What specific operational friction slows adoption for mid-tier brands?
Unpredictable agricultural yields create massive variations in baseline active levels, necessitating sophisticated, high-cost normalization protocols that standard contract manufacturers simply do not possess.
How do state mandates impact adoption in Egypt?
Egypt expands at 13.6% as aggressive public health mandates force clinical nutrition brands to procure massive volumes of standardized metabolic therapies.
What structural difference explains Italy's growth trajectory?
Italy tracks at 13.4% due to regional agricultural cooperatives establishing direct supply agreements with top-tier medical nutrition brands to secure preferential access to specific high-yield botanical harvests.
How does Spain navigate practitioner prescribing frameworks?
Spain advances at 13.2% as formulation scientists deliver exact pharmacokinetic replication of traditional dietary benefits to satisfy stringent European compliance networks driven by practitioner-led prescribing systems.
Why does France prioritize registered medical nutrition pathways?
France climbs at 11.6% because securing registered medical nutrition pathways unlocks exclusive, high-margin institutional contracts for specialized formulators after extensive multi-year clinical validation.
What drives the technical upgrade among Greek producers?
Greece registers 12.4% as heritage olive producers pivot aggressively toward high-value extract processing, implementing rigorous analytical testing to satisfy demanding international buyers and secure lucrative Western European contracts.
How do extraction complexities impact procurement strategies?
Securing consistent hydroxytyrosol and oleuropein ratios requires sophisticated extraction infrastructure, forcing formulation scientists to rely on proprietary B2B supply agreements to avoid severe degradation profiles during shelf-life testing.
What role does matrix stabilization play in product development?
Specialized matrices prevent rapid oxidation of sensitive phenolic compounds, helping brands avoid severe recall risks and immediate regulatory intervention if active levels drop below claimed minimums.
How does agricultural variability influence supply chains?
Advanced blending protocols normalize natural fluctuations in botanical active levels, requiring procurement teams to secure efficient supply lines to buffer against seasonal raw material inconsistencies.
What commercial advantage does precision fermentation offer?
Engineering specific microbial strains to produce targeted phenolic compounds bypasses agricultural variability completely, granting R&D directors infinitely scalable and highly consistent bioactive supply lines.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Polyphenols
- Omega-3s
- Others
- Polyphenols
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Pack Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Pack Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Pack Format, 2026 to 2036
- Capsule Packs
- Sachet Packs
- Others
- Capsule Packs
- Y to o to Y Growth Trend Analysis By Pack Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Pack Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Health Focus
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Health Focus, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Health Focus, 2026 to 2036
- Glucose Support
- Lipid Support
- Others
- Glucose Support
- Y to o to Y Growth Trend Analysis By Health Focus, 2021 to 2025
- Absolute $ Opportunity Analysis By Health Focus, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Dietary Supplements
- Functional Foods
- Others
- Dietary Supplements
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- B2B Formulation
- Pharmacies
- Others
- B2B Formulation
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Pack Format
- By Health Focus
- By End Use
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- DSM-Firmenich
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Indena
- Euromed
- ADM
- IFF
- Glanbia Nutritionals
- DSM-Firmenich
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Pack Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Health Focus, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Pack Format
- Figure 9: Global Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Health Focus
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Pack Format
- Figure 35: North America Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Health Focus
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Distribution Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Pack Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Health Focus
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Pack Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Health Focus
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Pack Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Health Focus
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Pack Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Health Focus
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Pack Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Health Focus
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Pack Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Pack Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Pack Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Health Focus, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Health Focus, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Health Focus
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

