Mediterranean Healthy-Aging Beverage Ingredients Market
The mediterranean healthy-aging beverage ingredients market is segmented by Ingredient Type (Polyphenols, Vitamins, Minerals, Omega fatty acids, Botanicals), Source (Plant extracts, Marine extracts, Fermented ingredients, Synthetic ingredients), Application (Functional beverages, RTD beverages, Powder mixes, Concentrates), Function (Antioxidants, Anti-inflammatory, Cognitive support, Heart health), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mediterranean Healthy-Aging Beverage Ingredients Market Size, Market Forecast, and Outlook By FMI
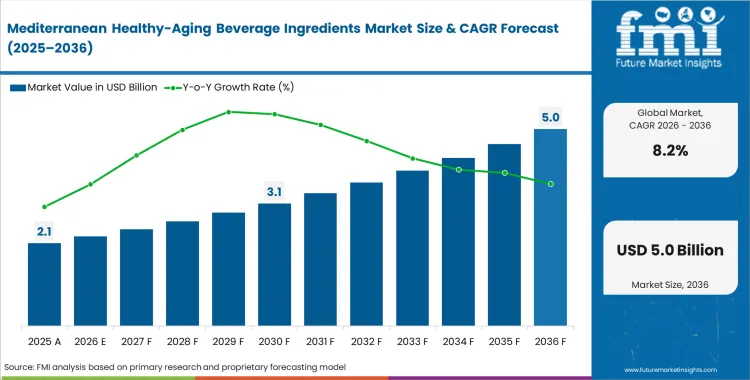
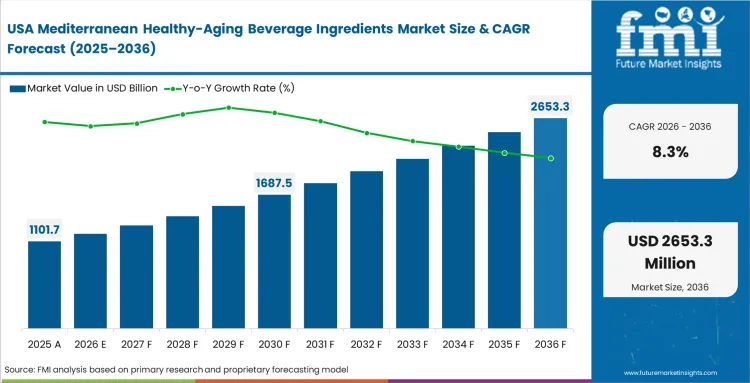
The Mediterranean healthy‑aging beverage ingredients segment crossed a valuation of USD 1.9 billion in 2025, supported by consistent demand for formulations positioned around aging‑related health benefits. Near‑term growth remains firm, with sales expected to pass USD 2.1 billion in 2026, as formulators continue to shift toward higher‑value ingredient systems rather than volume expansion alone. Ongoing investment in extraction capability, claim substantiation, and formulation reliability is expected to lift total market value to USD 4.6 billion by 2036, equivalent to an average annual increase of 8.2%. This path reflects growing reliance on locally sourced Mediterranean botanicals that can meet increasingly specific European health‑claim requirements.
Summary of Mediterranean Healthy-Aging Beverage Ingredients Market
- Market Snapshot
- The Mediterranean healthy‑aging beverage ingredients market stood at USD 1.9 billion in 2025 and is expected to reach USD 4.6 billion by 2036, reflecting steady expansion over the long term rather than short‑cycle demand spikes.
- Between 2026 and 2036, the sector is forecast to grow at an average annual rate of 8.2% CAGR, translating into approximately USD 2.50 billion in additional market value over the period.
- This market operates as a specification‑led ingredient segment within functional beverages, built around defined combinations of polyphenols, omega fatty acids, vitamins, minerals, and botanical extracts positioned to support age‑related health outcomes.
- Market direction is influenced by advances in healthy‑aging nutrition research, the regulatory relevance of olive‑derived polyphenol claims, and the continued commercial credibility of Mediterranean diet–associated ingredients in beverage formulations.
- Demand and Growth Drivers
- Demand is being supported by demographic shifts, as the global population aged 60 and above is expected to rise from 1.1 billion in 2023 to 1.4 billion by 2030, broadening the addressable audience for beverages positioned around healthy‑aging support.
- Ingredients rich in polyphenols are seeing wider use as Mediterranean dietary patterns continue to be associated with healthier aging outcomes, including antioxidant defense and improved quality‑of‑life indicators in middle‑aged and older populations.
- Olive‑derived components remain central to commercial formulations, largely because EFSA‑recognized claim pathways for olive‑oil polyphenols provide a regulatory foundation that supports premium positioning of Mediterranean antioxidant concepts in beverages.
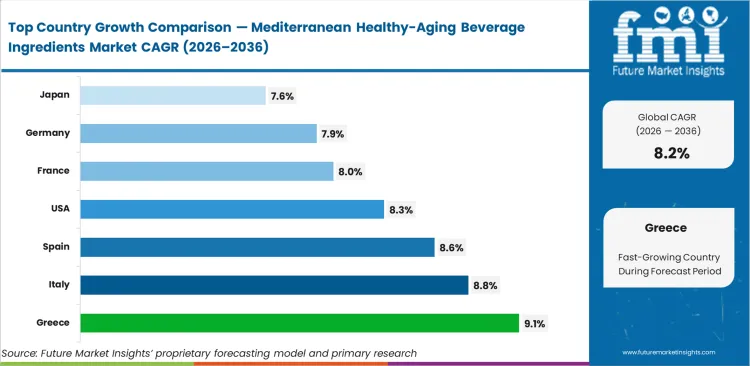
- Performance differs by country, with China recording the highest pace at 9.4%, followed by Spain at 9.1%, Japan at 8.9%, Italy at 8.8%, Greece at 8.5%, the United States at 8.4%, and Germany at 7.9%, reflecting variations in regulatory alignment, consumer acceptance, and formulation maturity.
- Adoption is constrained by practical formulation requirements, including the need for tighter ingredient standardization, effective taste‑masking strategies, reliable bioavailability, and the translation of Mediterranean diet health narratives into dosage formats suitable for beverage applications.
- Product and Segment View
- The market encompasses bioactive beverage ingredients sourced primarily from plant and marine extracts, fermentation‑based systems, and a limited set of synthetic nutrients, all used to support age‑related health, antioxidant activity, and cardiometabolic positioning in beverage formulations.
- These ingredients are incorporated across functional drinks, ready‑to‑drink beverages, powder mixes, and concentrates, with formulation decisions guided by practical considerations such as stability, taste impact, visual clarity, and achievable dosage levels.
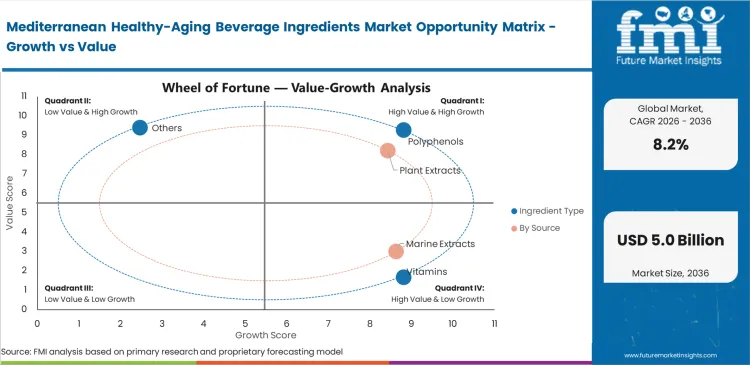
- Polyphenols represent the largest ingredient category, accounting for 34.5% of usage, as olive, acerola, bergamot, tea, and similar antioxidant systems align well with both Mediterranean dietary concepts and healthy‑aging positioning in beverages.
- By source, plant‑derived inputs hold a 41.2% share, reflecting the central role of olive, fruit, herbal, and botanical bioactives in Mediterranean‑inspired healthy‑aging formulations.
- Powders account for 44.1% of formats, largely because dry premixes are easier to handle in shelf‑stable beverage systems and are better suited to long‑distance distribution and export‑oriented ingredient trade.
- Functional beverages make up the largest application segment at 46.8%, consistent with the category’s focus on proactive wellness, routine consumption, and age‑support use cases rather than occasional or indulgent formats.
- From a functional standpoint, antioxidants account for 38.6% of demand, as oxidative‑stress management remains the most established and easily communicated link between Mediterranean diet research and beverage ingredient claims.
- The market definition covers standalone beverage ingredients and premix‑ready actives intended for healthy‑aging applications, while excluding finished dietary supplements, conventional juices without functional actives, and non‑beverage healthy‑food categories.
- Geography and Competitive Outlook
- China, Spain, and Japan currently show the strongest uptake at the national level, supported by a combination of consumer acceptance, regulatory clarity, and active formulation pipelines. The United States, meanwhile, serves as the largest established demand base, providing scale and commercial consistency rather than rapid market acceleration.
- Competitive positioning is determined largely by technical depth, including the ability to support evidence‑based ingredient use, assist with formulation challenges, address bioavailability constraints, and maintain close working relationships with beverage manufacturers during development and scale‑up.
- The supplier landscape includes firms such as DSM Firmenich, Indena, ADM, Kerry, Symrise, GELITA, and Glanbia Nutritionals. No single company dominates the segment, with the leading participant holding an estimated 17.5% share, indicating a market structure where scale and technical capability matter, but supplier concentration remains limited.
At the formulation level, product directors face immediate pressure to address solubility limitations associated with minimally processed botanicals. Postponing these solutions often forces brands to fall back on highly refined substitutes, which undermines product positioning and limits eligibility for premium functional beverage shelves where origin transparency is closely examined. Procurement teams attempt to mitigate risk by securing raw inputs ahead of harvest volatility, but this approach frequently overlooks constraints in localized extraction capacity. Access to raw olive leaf does not guarantee availability of water‑soluble extracts once contract manufacturing capacity becomes constrained.
Regulatory standardization is emerging as a key inflection point. As European authorities formalize claim language for citrus‑derived polyphenols, consumer trust barriers begin to fall, accelerating uptake across broader product lines. This validation pressure is prompting mid‑tier manufacturers to revisit legacy formulations, replacing generic antioxidant systems with ingredients capable of supporting approved claims. Competitive advantage is shifting away from simple botanical inclusion toward extraction efficiency, reproducibility, and claim‑ready performance.
Greece currently shows the fastest expansion at 9.1%, driven by domestic suppliers scaling extraction capacity around endemic crops. Italy follows at 8.8%, supported by sustained public investment in polyphenol research for beverage applications. Spain records 8.6% growth, underpinned by cooperative‑led citrus processing systems focused on higher‑value exports. In the United States, formulators are increasing procurement at 8.3% as Mediterranean ingredients are used to distinguish premium lines from synthetic alternatives. France advances at 8.0%, where regulatory scrutiny continues to favor naturally derived antioxidants, while Germany posts 7.9% growth, shaped by strict clinical efficacy standards for retail acceptance. Japan rounds out the group at 7.6%, with demand concentrated in narrowly defined cognitive and longevity formulations rather than broad anti‑aging positioning.
Segmental Analysis
Mediterranean Healthy-Aging Beverage Ingredients Market Analysis by Ingredient Type
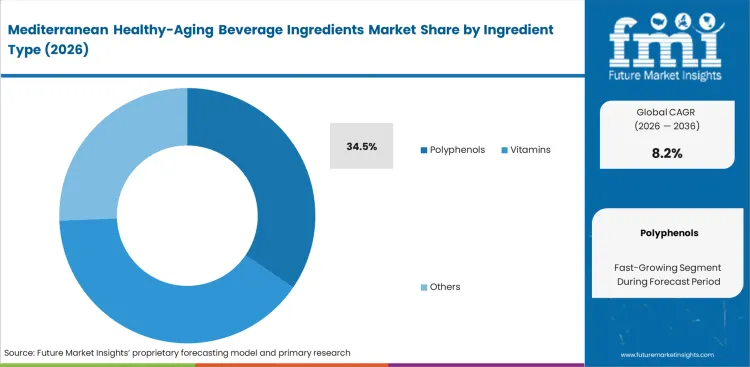
Clinical validation of cardiovascular outcomes helps explain why polyphenols lead to formulation specifications. Demand is increasingly shaped by clinically substantiated ingredients rather than broad antioxidant positioning alone. Polyphenols are estimated to hold for 34.5% share in 2026, as procurement directors prioritize olive‑polyphenols supported by EFSA‑approved health claims. Incorporating these high-density compounds presents challenges for flavor chemists. They must develop effective masking systems to reduce strong astringency without compromising the integrity of active compounds. Formulators who treat these ingredients as simple additions often face immediate rejection during consumer sensory testing. Another critical factor often overlooked in ingredient specifications is the rate of thermal degradation during standard UHT pasteurization. Relying solely on pre-processing concentration data can result in sub-therapeutic levels in the final product. Beverage brands that fail to account for these processing losses may also face regulatory risks.
- Initial specification trigger: Regulatory alignment directs R&D teams toward defined phenolic profiles to support specific cardiovascular claims. Formulators must address solubility challenges in water-based systems.
- Formulation qualification: Sensory testing evaluates the effectiveness of masking agents in managing natural botanical bitterness. Chemists often go through multiple formulation cycles, balancing functional dosing with acceptable mouthfeel for functional drinks ingredients.
- Commercial expansion: Marketing teams leverage clean-label positioning to broaden product portfolios, while procurement teams secure long-term supply agreements to manage price fluctuations linked to harvest variability.
Mediterranean Healthy-Aging Beverage Ingredients Market Analysis by Source
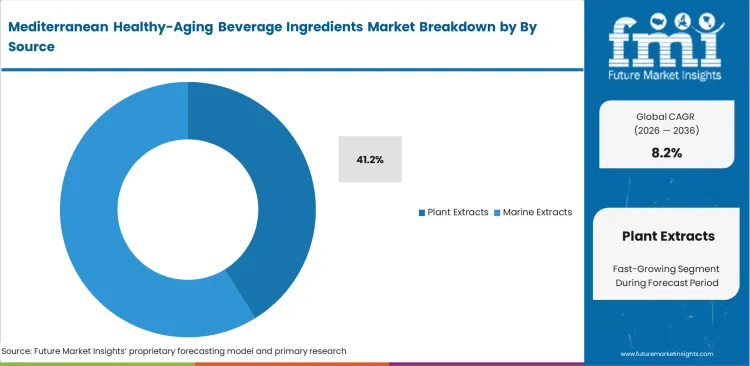
Consumer demand for ingredients with clearly identifiable natural origins often clashes with the realities of complex agricultural supply chains. Sourcing teams working with plant extracts must manage significant variation in bioactive content across harvest cycles. A less visible challenge in this segment is the reliance on third-party extraction providers. Many premium beverage brands do not have in-house extraction capabilities and depend on external facilities, often located in Europe. Plant extracts are anticipated to account for a 41.2% share, acting as key active ingredients in lifestyle-driven product offerings. Dependence on volume-based contracts, without standardized active compound assurances, can lead to inconsistent product performance. Without proper audits of these partners, companies risk batch-level issues, particularly related to solvent residue compliance.
- Upstream procurement savings: Streamlining agricultural supplier networks helps reduce variability in raw materials. Sourcing teams typically negotiate seasonal contracts to secure better pricing for plant extract beverage ingredients.
- Hidden processing costs: Third-party standardization can create margin pressure when crop yields of bioactive compounds fall below expected levels. Brands may need to increase dosing to maintain label claims, leading to higher costs.
- Lifecycle value realization: Consistent efficacy supports long-term brand value. Companies that invest in reliable extraction partnerships are better able to maintain quality and sustain a premium position in the market.
Mediterranean Healthy-Aging Beverage Ingredients Market Analysis by Application
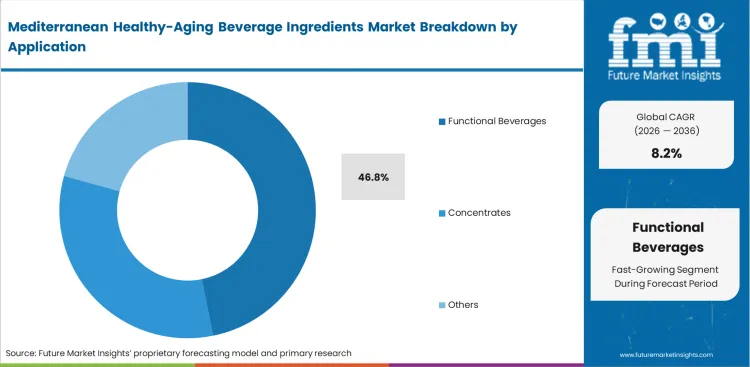
The shift toward active hydration is reshaping product development pipelines. Functional beverages lead the application segment set for 46.8% share, serving as the main delivery systems for complex botanical blends. Formulation scientists working with clean-label ingredients in liquid formats face significant stability challenges. Active compounds often precipitate over a typical six-month shelf life, making consistent dispersion difficult. Addressing this requires advanced hydrocolloid systems, which can increase unit costs. A key gap in this segment lies in the difference between lab-scale stability results and real-world distribution conditions. Temperature fluctuations during ambient transport can accelerate the degradation of botanical compounds. Brands that do not conduct thorough accelerated-aging tests risk product recalls due to visible sedimentation.
- Solubility failure: Weak emulsion systems can lead to precipitation of bioactive compounds. Quality teams may require product withdrawal when visible defects appear on retail shelves carrying healthy drink formulations.
- Sensory degradation: Extended storage at ambient conditions can intensify natural bitterness and astringency. Flavor chemists must develop masking systems that remain effective throughout the product’s shelf life.
- Efficacy retention: Ensuring that active compound levels remain stable after pasteurization is essential for regulatory compliance. R&D teams often implement ongoing batch testing to confirm that ready-to-drink beverages maintain their intended functional benefits until expiration.
Mediterranean Healthy-Aging Beverage Ingredients Market Analysis by Function
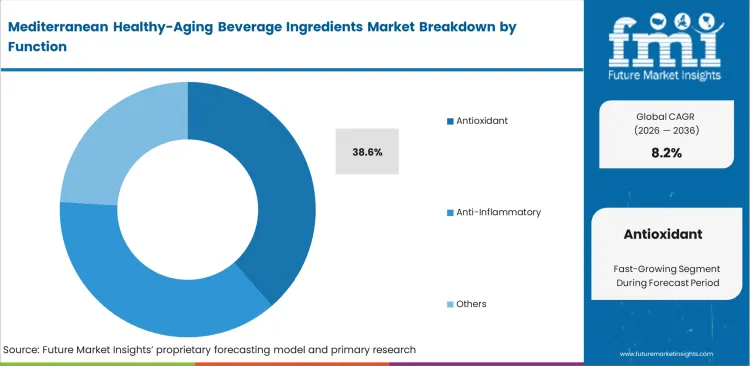
Consumer understanding of cellular protection benefits plays a key role in driving initial product trials across global markets. Incorporating highly reactive antioxidants creates challenges for packaging design. Engineers must use advanced oxygen-scavenging closures, as inadequate packaging can reduce ingredient effectiveness before the product reaches consumers. FMI analysis also highlights limitations in how antioxidant performance is measured. Total antioxidant capacity tests, such as ORAC, often do not reflect actual bioavailability in the human body. In vitro ORAC values do not directly translate to absorption in human serum. Antioxidants are expected to account for a 38.6% share, offering clear and widely recognized health positioning for marketing teams. Marketing strategies based only on these figures are increasingly questioned by informed retail buyers assessing beverage antioxidant claims.
- Early brand adopters: Premium niche brands use complex ingredients like grape seed extracts to differentiate their products. Higher material costs are typically managed through premium pricing strategies.
- Mainstream market fast-followers: Mid-tier beverage manufacturers adopt more standardized ingredients, such as citrus-derived antioxidants, to appeal to a broader audience. Procurement teams benefit from established food antioxidants supply chains that support cost efficiency.
- Laggard conversion: Mass-market hydration brands gradually introduce low-dose functional blends to remain competitive. Ongoing price pressure often leads these manufacturers to consider more cost-effective synthetic alternatives where regulations allow. Top of Form
Mediterranean Healthy-Aging Beverage Ingredients Market Drivers, Restraints, and Opportunities
Clinical validation requirements established by European regulatory bodies force beverage formulators to abandon generic botanical extracts. R&D directors require strictly standardized compounds matching EFSA-compliant health claims for premium shelf space. Strict documentation protocols prevent mid-tier manufacturers from utilizing cheaper substitutes, locking procurement teams into designated European supply networks. Delaying this transition excludes beverage brands from high-margin retail channels. Such delays leave companies competing in saturated conventional categories using inferior functional beverage formulation ingredients.
Formulation instability in complex liquid matrices acts as a persistent structural friction slowing rapid product commercialization. Active polyphenols naturally precipitate when exposed to standard commercial pasteurization. Flavor chemists struggle stabilizing these ingredients without artificial emulsifiers that contradict clean-label positioning. Advanced microencapsulation technologies offer partial stabilization, yet high deployment costs restrict usage to ultra-premium product tiers. Mainstream manufacturers continue battling ongoing shelf-life limitations when incorporating aging nutrition ingredients.
Opportunities in the Mediterranean Healthy-Aging Beverage Ingredients Market
- Microencapsulation deployment: Advanced coating technologies protect sensitive bioactives from thermal degradation. Formulation scientists achieve extended shelf stability without compromising clean-label requirements for longevity beverage ingredients.
- Synergistic botanical blending: Combining specific olive and citrus derivatives amplifies overall antioxidant capacity. R&D teams deliver higher functional efficacy utilizing lower individual dietary supplements dosages.
- Upcycled ingredient sourcing: Extracting high-value polyphenols from agricultural processing waste reduces base material costs. Procurement directors secure sustainable supply lines supporting compelling environmental marketing narratives.
Regional Analysis
Based on regional analysis, Mediterranean Healthy-Aging Beverage Ingredients Market is segmented into Europe, North America, and East Asia across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Greece | 9.1% |
| Italy | 8.8% |
| Spain | 8.6% |
| United States | 8.3% |
| France | 8.0% |
| Germany | 7.9% |
| Japan | 7.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Europe Mediterranean Healthy-Aging Beverage Ingredients Market Analysis
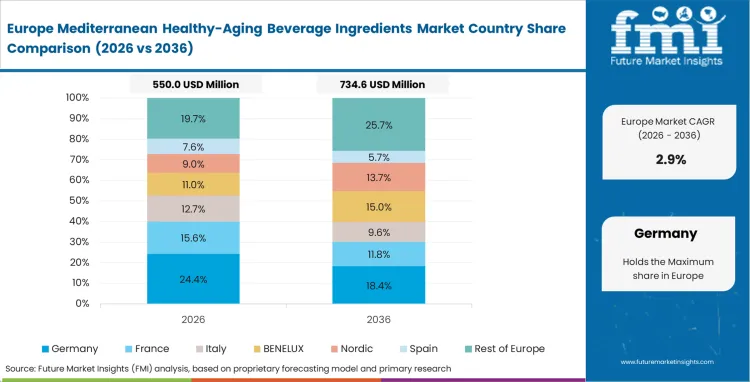
Vertical integration among agricultural cooperatives plays a critical role in determining supply availability across regional extraction facilities. Local processing capabilities enable European suppliers to manage the value chain from harvest through to standardized Mediterranean diet extracts. According to FMI’s assessment, proximity advantages help protect domestic buyers from transit-related degradation that often affects international sourcing. Strict origin-labeling regulations create barriers for imported botanical alternatives, supporting the growth of specialized polyphenol extract suppliers. When procurement teams at major European brands bypass international intermediaries and contract directly with local extraction facilities, they reduce layered margins that typically increase the cost of high-concentration bioactive compounds. This localized sourcing model ultimately determines which formulation teams can sustain premium retail positioning while maintaining cost efficiency.
- Greece: State‑backed investment in extraction facilities has eased processing limitations for local botanicals, most notably in olive leaf refinement. This has allowed formulation teams to secure high‑potency raw materials at stable prices before larger volumes move into export channels. Manufacturers are better able to manage costs while preserving consistency in bioactive content. These conditions support steady expansion of Greece’s botanical and polyphenol ingredients market, which is increasing at an annual rate of about 9.1% CAGR, driven by export demand and procurement practices that soften the impact of seasonal price fluctuations.
- Italy: Public investment in clinical nutraceutical research has improved the scientific grounding of functional claims associated with grape and citrus derivatives. In Italy, these clinically validated ingredients are incorporated into domestic R&D programs early, reducing the need for companies to build evidence internally. Local suppliers continue to influence quality standards across European markets. The functional bioactive ingredients segment is growing at 8.8% CAGR, supported by procurement teams that rely on established clinical datasets to move more efficiently through regulatory review processes.
- Spain: In Spain, large agricultural cooperatives are moving away from bulk commodity exports toward standardized bioactive extraction, reshaping supply availability at scale. This consolidation allows procurement teams to secure longer‑term contracts with more predictable pricing than is typically possible in fragmented sourcing environments. Greater consistency in batch quality has also reduced the need for repeated formulation adjustments during production. The bioactive compound and polyphenol market is expanding at an annual rate of around 8.6% CAGR, with national processing infrastructure continuing to influence global pricing for compounds such as olive‑derived hydroxytyrosol.
- France: Heightened regulatory oversight of natural label claims has led to the gradual removal of synthetic additives from domestic beverage formulations. In France, this stricter compliance environment has reshaped formulation priorities, particularly for manufacturers working within Mediterranean diet‑aligned product frameworks. The natural functional beverage ingredients market is advancing by about 8.0% CAGR, favoring producers that can manage complex botanical systems without relying on artificial stabilizers or masking agents. Within these limits, flavor chemists continue to face challenges in controlling astringency using only certified organic inputs, increasing the need for fully natural dispersion and stabilization approaches.
- Germany: Retail approval for functional hydration and antioxidant beverages depends on extensive clinical and analytical documentation, with procurement teams requiring clear evidence of efficacy before moving forward. Botanical extracts that lack peer‑reviewed bioavailability data are routinely excluded, particularly within Germany, where compliance teams operate under some of the most demanding regulatory pathways in the region. Suppliers are expected to maintain tight control over batch‑to‑batch active compound levels, often verified through independent laboratory testing rather than supplier specifications alone. The functional antioxidant ingredients market is expanding at an annual rate of about 7.9% CAGR, as strict enforcement practices make traceability and analytical precision baseline requirements rather than optional considerations. Random third‑party audits further reinforce these standards, directly affecting supplier selection and product continuity.
FMI's report includes broader European analysis beyond these highlighted regional leaders. Supply chain consolidation accelerates as smaller extraction tollers merge to meet the immense capacity requirements of multinational beverage brands. Portugal is emerging as a secondary growth market, where investments in olive by‑product valorization and improved solvent‑free extraction methods are expanding available polyphenol volumes.
North America Mediterranean Healthy-Aging Beverage Ingredients Market Analysis
Formulation differentiation in highly competitive functional hydration categories is pushing manufacturers toward more specialized ingredient profiles that are difficult for domestic competitors to replicate. Growing consumer fatigue with basic vitamin fortification is leading North American R&D teams to adopt more complex botanical blends, helping justify premium pricing in retail markets. Supply chain risks remain significant due to heavy reliance on trans-Atlantic sourcing. Incorporating natural food additives sourced primarily from Europe requires advanced inventory planning to avoid production disruptions during peak demand periods. Sourcing teams often face lead-time uncertainty caused by customs delays, which can affect the timely availability of key bioactive ingredients. This geographic dependency also forces companies to maintain higher inventory levels of sensitive extracts, increasing working capital requirements to ensure consistent production.
- United States: Competition in the functional beverage space is compressing innovation timelines and encouraging the rapid rollout of health‑oriented formulations that are not yet common in many other markets. To distinguish premium lines from lower‑cost synthetic alternatives, brand owners are increasingly sourcing Mediterranean botanical extracts despite higher input complexity. This dynamic is supporting growth of the functional beverage ingredients market at an annual rate of 8.3% CAGR in the United States, while also introducing new challenges for procurement teams. Exchange‑rate movements between the euro and the dollar can materially affect ingredient costs, leading purchasing functions to integrate currency risk management more closely into sourcing and margin‑stability decisions.
FMI's report includes a comprehensive analysis of cross‑border supply dynamics impacting North American operations. The persistent lack of domestic extraction equivalents forces heavy reliance on established European botanical supply chains. Mexico is also gaining attention, as investments in citrus peel recovery and botanical co‑processing are beginning to support regional extraction capacity closer to U.S. manufacturing hubs.
East Asia Mediterranean Healthy-Aging Beverage Ingredients Market Analysis
Well-established regulatory frameworks for functional foods define strict ingredient specifications and limit how health claims can be communicated across the region. Regulatory authorities require clear physiological evidence, which drives formulators to use highly standardized extracts instead of general whole-plant ingredients. In Japan, functional water brands are at the forefront of incorporating targeted cardiovascular health ingredients into mainstream retail products. These formats require high levels of purity and minimal sensory impact from active compounds. Suppliers face strong pressure to deliver botanical extracts with neutral taste profiles. Flavor chemists must remove most natural bitterness and astringency before these ingredients are considered for product development. Products that fail to meet these sensory expectations are unlikely to succeed with consumers who are highly sensitive to taste variations.
- Japan: The Foods for Specified Health Uses (FOSHU) framework provides a clearly defined route for commercializing scientifically substantiated ingredients, but compliance requirements are demanding. Product development teams operating in Japan must adhere to narrow dosage ranges and formulation parameters to avoid regulatory delays, often extending development timelines. The FOSHU‑regulated functional food and beverage ingredients market reflects an average annual increase of about 7.6% CAGR, reflecting a focus on ingredient purity and clinical validation rather than rapid product turnover. As a result, formulators typically spend longer in the qualification phase, carrying out detailed internal stability and compatibility testing. These conditions create high entry barriers, limiting access for smaller or unverified foreign suppliers and reinforcing the role of established domestic and global ingredient providers.
FMI's report includes an assessment of surrounding Asian markets adopting similar stringent regulatory postures. Ingredient suppliers engineer highly refined iterations specifically tailored to meet these extremely demanding regional organoleptic standards. South Korea is highlighted as a growing influence, where tightening functional food regulations and strong consumer sensitivity to taste are driving demand for ultra‑purified botanical actives.
Competitive Aligners for Market Players
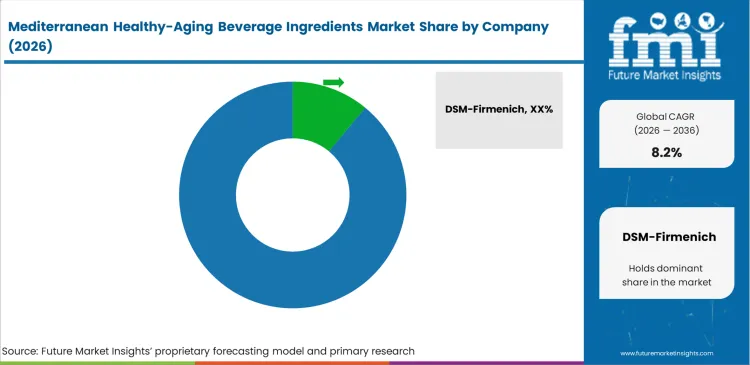
Supplier capability depends on effectively managing the gap between raw agricultural output and food-grade botanical extracts. Leading players such as DSM-Firmenich and Indena S.p.A. shape market conditions by ensuring reliable batch-to-batch consistency in bioactive content. Procurement teams at major beverage companies typically avoid working with beverage ingredient suppliers that lack strong clinical documentation to support their nutraceutical ingredients. Mid-tier brands that attempt to source directly from unverified agricultural cooperatives often encounter serious production issues due to variations in phenolic concentrations.
Market leadership is largely driven by proprietary extraction technologies and well-established clinical validation data. Companies such as BASF SE and Archer Daniels Midland Company hold extensive stability data that shows how specific beverage ingredients perform in complex liquid formulations under different thermal processing conditions. New entrants in this space must be able to replicate this level of application-specific data. Simply offering high-quality botanical extracts is not enough if suppliers cannot demonstrate stability during processes such as UHT pasteurization. Established players use their data capabilities as a strategic advantage, creating strong vendor dependency and limiting switching options for buyers.
Large functional beverage manufacturers are actively reducing reliance on single suppliers by requiring dual-qualification across their supply networks. Companies such as Kerry Group plc and Symrise AG are expanding their formulation support services, often placing flavor and application experts directly within client R&D teams. Buyers are also increasing their expectations around traceability, requiring clear sourcing back to specific Mediterranean growing regions. These changes are pushing ingredient suppliers to take on a more integrated sports nutrition, acting as formulation partners and addressing stability challenges early in the development process, before ingredients reach pilot-scale production. This collaborative model is supporting the broader adoption of Mediterranean nutraceutical compounds in global markets.
Key Players in Mediterranean Healthy-Aging Beverage Ingredients Market
- DSM-Firmenich
- Indena S.p.A.
- BASF SE
- Archer Daniels Midland Company
- Kerry Group plc
- Symrise AG
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 2.1 Billion to USD 4.6 Billion, at a CAGR of 8.20% |
| Market Definition | Mediterranean healthy-aging beverage ingredients encompass bio-active compounds extracted primarily from endemic regional flora, standardized for integration into liquid consumption formats. |
| Segmentation | Ingredient Type, Source, Application, Function, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | Greece, Italy, Spain, United States, France, Germany, Japan |
| Key Companies Profiled | DSM-Firmenich, Indena S.p.A., BASF SE, Archer Daniels Midland Company, Kerry Group plc, Symrise AG |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline established via commercial extraction facility output volumes. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Mediterranean Healthy-Aging Beverage Ingredients Market Analysis by Segments
Ingredient Type:
- Polyphenols
- Vitamins
- Minerals
- Omega fatty acids
- Botanicals
Source:
- Plant extracts
- Marine extracts
- Fermented ingredients
- Synthetic ingredients
Application:
- Functional beverages
- RTD beverages
- Powder mixes
- Concentrates
Function:
- Antioxidants
- Anti-inflammatory
- Cognitive support
- Heart health
Region:
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Greece
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- ANZ
- Rest of South Asia & Pacific
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of MEA
Bibliography
- Dobroslavska, P., Kyselova, Z., & colleagues. (2024). Mediterranean Dietary Pattern for Healthy and Active Aging: A Narrative Review of an Integrative and Sustainable Approach. Nutrients, 16(11), 1725.
- European Food Safety Authority. (2024, May 15). Flavouring Group Evaluation 413 (FGE.413): Naringenin. EFSA Journal.
- Zhong, J., and colleagues. (2024). Dietary flavonoid intake is negatively associated with accelerating aging: an American population-based cross-sectional study. Nutrition Journal, 23, Article 92.
- Toledo, R., and colleagues. (2024). An update on citrus polymethoxyflavones: chemistry, metabolic fate, and relevant bioactivities. European Food Research and Technology.
- Tessier, A. J., Cortese, M., Yuan, C., and colleagues. (2024). Consumption of Olive Oil and Diet Quality and Risk of Dementia-Related Death. JAMA Network Open, 7(5), e2410021.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Formulation scientists balancing active polyphenol concentrations against necessary organoleptic masking requirements.
- Procurement directors securing stable Mediterranean botanical supplies against harvest-driven price volatility.
- Regulatory compliance teams matching specific ingredient profiles to EFSA-approved cardiovascular health claims.
- Supply chain managers navigating extraction capacity bottlenecks for Mediterranean diet supplements.
- Quality assurance heads monitoring active compound degradation during thermal pasteurization protocols.
- Brand managers attempting differentiation using traceable regional healthy beverage additives.
- R&D directors evaluating advanced microencapsulation technologies preventing liquid matrix precipitation.
- Strategic planners assessing capacity limits of third-party European botanical extraction tollers.
Frequently Asked Questions
What is the baseline valuation for this sector?
Revenues reached USD 1.9 billion in 2025. This baseline reflects established integration of basic botanical extracts into premium hydration channels before regulatory shifts required highly standardized clinical dosages.
What structural condition sustains the 8.20% growth rate?
Strict regulatory frameworks require scientifically validated health claims, forcing manufacturers away from synthetic alternatives. This regulatory pressure guarantees sustained demand for authentic, standardized extracts.
What valuation does the forecast model project?
Total opportunity scales to USD 4.6 billion by 2036. This expansion relies entirely on suppliers solving complex solubility challenges in high-volume liquid manufacturing.
Why do polyphenols lead the ingredient type segment?
Extensive clinical documentation directly links specific compounds to cardiovascular health outcomes. Formulators prioritize polyphenols because they support highly defensible consumer health claims.
What operational consequence drives plant extract adoption?
Clean-label requirements force marketing teams to demand recognizable botanical names on ingredient panels. Plant extracts provide necessary functional efficacy without triggering consumer skepticism associated with chemical nomenclature.
What changes inside the organization when functional beverages dominate?
R&D departments fundamentally overhaul stability testing protocols. Transitioning to active liquid matrices requires continuous monitoring for ingredient precipitation and flavor degradation across entire shelf lives.
What does the share figure for antioxidants obscure?
Raw antioxidant capacity metrics frequently fail to translate into actual human bioavailability. Procurement teams purchasing solely on ORAC values often acquire ingredients providing zero physiological benefit post-pasteurization.
What hidden costs emerge in botanical formulation?
Organoleptic masking introduces significant secondary expenses. Neutralizing severe astringency inherent in olive and citrus extracts requires complex flavor engineering that drastically compresses final product margins.
What differentiates Greece's growth trajectory from Germany's?
Greece expands at 9.1% driven by aggressive upstream vertical integration and localized extraction infrastructure. Germany tracks at 7.9% based strictly on downstream retail compliance and clinical efficacy demands.
What failure mode must packaging engineers prevent?
Light and oxygen exposure rapidly degrade sensitive active compounds. Packaging teams must specify advanced UV-blocking materials and oxygen scavengers preventing complete efficacy loss before consumption.
Who adopts advanced microencapsulation first?
Ultra-premium clinical nutrition brands deploy encapsulation guaranteeing absolute dosage delivery. Mainstream brands avoid this technology until scaled production lowers prohibitive unit costs.
Where do performance gaps appear in liquid matrices?
Prolonged ambient storage exposes critical emulsion failures. Botanical blends frequently separate or precipitate when exposed to temperature cycling during standard commercial distribution.
What limits supply side expansion?
Toll extraction capacity remains highly restricted across regional basins. Securing raw agricultural material yields zero commercial advantage if third-party facilities lack available scheduling to process harvests.
How do large buyers resist vendor lock-in?
Multinational beverage conglomerates mandate strict dual-sourcing qualification protocols. Procurement teams force secondary suppliers to perfectly match precise organoleptic profiles and solubility metrics of primary incumbents.
What triggers initial purchase decisions for specific extracts?
Brand managers demand specific active profiles matching targeted competitor claims. Procurement must immediately source ingredients satisfying marketing narratives while fitting established cost parameters.
What residual risk remains after selecting premium extracts?
Thermal pasteurization unpredictably degrades active concentration. Brands risk regulatory action if final retail products fail to match ingredient levels stated on packaging.
Who produces standardized botanical derivatives?
Concentrated tiers of European extraction specialists control majority food-grade output. Incumbents maintain power through massive clinical data libraries that challengers cannot quickly replicate.
What drives renewal of long-term ingredient contracts?
Demonstrated batch-to-batch consistency in both efficacy and flavor profile secures ongoing supplier relationships. Beverage brands refuse switching vendors if integration requires reformulating established masking systems.
Where do cost savings originate in procurement?
Forward-contracting directly with agricultural cooperatives prior to harvest mitigates spot-market volatility. Sourcing directors secure stable pricing guaranteeing volume purchases regardless of seasonal yield variations.
What validates supplier selection during qualification?
Suppliers must prove extracts remain entirely stable and soluble under specific UHT processing conditions utilized by beverage manufacturer pilot plants.
How do compliance teams influence ingredient selection?
Regulatory officers veto any ingredient lacking comprehensive safety and efficacy documentation. Compliance units enforce strict adherence to EFSA guidelines shielding brands from legal liability.
What dictates the competitive trajectory toward 2036?
Competition moves away from simple extraction toward integrated formulation support. Suppliers delivering pre-masked, fully soluble ingredient systems systematically displace vendors selling basic raw extracts.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Observational and In-context Research
- Social and Community Interactions
- Primary Modes
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Polyphenols
- Vitamins
- Others
- Polyphenols
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Source
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Source, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Source, 2026 to 2036
- Plant Extracts
- Marine Extracts
- Plant Extracts
- Y to o to Y Growth Trend Analysis By By Source, 2021 to 2025
- Absolute $ Opportunity Analysis By By Source, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Functional Beverages
- Concentrates
- Others
- Functional Beverages
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Function, 2026 to 2036
- Antioxidant
- Anti-Inflammatory
- Others
- Antioxidant
- Y to o to Y Growth Trend Analysis By Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By By Source
- By Application
- By Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By By Source
- By Application
- By Function
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By By Source
- By Application
- By Function
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By By Source
- By Application
- By Function
- Competition Analysis
- Competition Deep Dive
- DSM-Firmenich
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Indena S.p.A.
- BASF SE
- Archer Daniels Midland Company
- Kerry Group plc
- DSM-Firmenich
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by By Source, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Function, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by By Source
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Function
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Ingredient Type
- Figure 29: North America Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by By Source
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Function
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 42: Latin America Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by By Source
- Figure 45: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Function
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by By Source
- Figure 58: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Application
- Figure 61: Western Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Function
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by By Source
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Application
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Function
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 81: East Asia Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by By Source
- Figure 84: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Application
- Figure 87: East Asia Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Function
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by By Source
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Function
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by By Source, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by By Source, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by By Source
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Function
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

