Heat-Stable Probiotics Market
The Heat-Stable Probiotics Market is segmented by Strain Type (Bacillus coagulans, Bacillus subtilis, Bacillus clausii, Other Spore-Forming), Application (Baked Goods, Hot Beverages, Shelf-Stable Supplements, Ready-to-Drink Beverages, Functional Foods), End Use (Food & Beverage Manufacturing, Dietary Supplements, Clinical Nutrition, Animal Feed), Distribution Channel (Pharmacies, Supermarkets, E-commerce, Food Service Distributors) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Heat-Stable Probiotics Market Size and Share Forecast Outlook By FMI
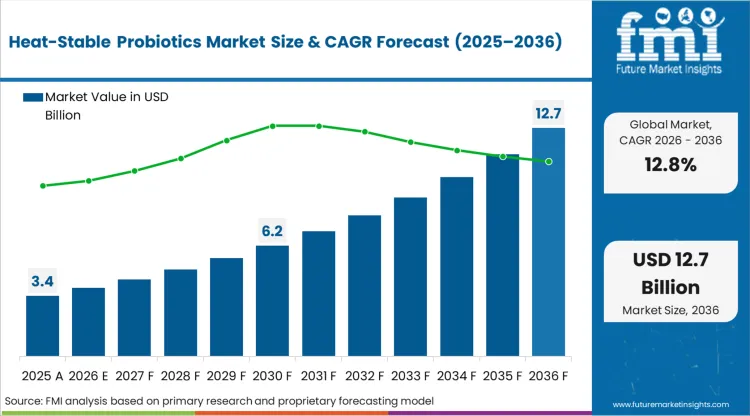
In 2025, the heat-stable probiotics market was valued at USD 3.4 billion. Based on Future Market Insights' analysis, demand for spore-forming probiotic strains is estimated to grow to USD 3.8 billion in 2026 and USD 12.7 billion by 2036. FMI projects a CAGR of 12.8% during the forecast period.
Absolute dollar growth of USD 8.9 billion over the decade reflects a structural shift from refrigeration-dependent probiotic delivery toward thermotolerant Bacillus species enabling ambient storage, pasteurisation survival, and expansion into baked goods and hot beverage applications previously impossible for traditional strains. Growth depends on strain-specific heat resistance validation demonstrating CFU viability through commercial baking cycles and 36-month room temperature shelf life.
Summary of Heat-Stable Probiotics Market
- Heat-Stable Probiotics Market Definition
- The market covers spore-forming Bacillus species surviving heat processing and ambient storage while germinating in the intestinal tract, enabling probiotic delivery through baked goods, hot beverages, and shelf-stable formats impossible for refrigerated strains.
- Demand Drivers in the Market
- Cold chain elimination delivering estimated 20 to 30% distribution cost savings, enabling vending machines and ambient e-commerce shipping impossible for Lactobacillus strains degrading 10 to 15% monthly at room temperature.
- Food application expansion into baked goods showing 50 to 90% spore survival through commercial baking and hot beverages achieving 99.76% viability after tea brewing.
- Protective endospore formation resisting heat, desiccation, and pH variability without requiring microencapsulation technologies adding formulation complexity and cost.
- Key Segments Analyzed in the FMI Report
- Strain Type: Bacillus coagulans dominates at 46.8% share through FDA GRAS status; Bacillus subtilis shows fastest growth at 15.2% CAGR through Japanese natto tradition.
- Application: Shelf-stable supplements lead at 38.4% share; baked goods demonstrate 17.6% CAGR as manufacturers pursue functional wellness differentiation.
- Geography: Asia Pacific dominates through traditional Bacillus fermented food consumption; North America shows highest growth through FDA regulatory clarity.
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, "In the updated Heat-Stable Probiotics Market Report for 2026 to 2036, CXOs will find that competitive advantage belongs to strain holders with FDA GRAS confirmation and published CFU viability data through commercial baking cycles, as food manufacturers will not invest in functional positioning without documented heat survival kinetics."
- Strategic Implications/Executive Takeaways
- Invest in strain-specific heat survival kinetics documentation to establish FDA GRAS eligibility and qualified health claim positioning.
- Develop application partnerships with baked goods and beverage manufacturers, providing CFU retention data validating functional food positioning.
- Establish ambient direct-to-consumer shipping capabilities, eliminating cold pack logistics for cost-competitive subscription models.
- Methodology
- Built on primary interviews with probiotic ingredient suppliers, thermal processing food scientists, and regulatory specialists across North America, Europe, and Asia Pacific.
- Benchmarked against overall probiotics market sizing, functional food ingredient trends, and cold chain logistics cost structures.
- Validated using spore survival kinetics research, thermal death time studies, and structured internal review as per FMI modelling standards.
Ingredient suppliers with FDA GRAS-confirmed strains and published clinical dossiers are commanding long-term formulation contracts with baked goods and beverage manufacturers. This contracting behaviour concentrates market value among a small number of validated strain holders, while suppliers offering marginally heat-tolerant non-spore strains face rapid displacement as food manufacturer technical teams apply standard baking viability testing protocols.
India (16.2% CAGR) leads through LactoSpore domestic origin and tropical climate necessitating heat-resistant formulations. Japan (14.8% CAGR) follows through natto tradition establishing Bacillus subtilis cultural acceptance. United States (13.4% CAGR) contributes through FDA GRAS framework and supplement market maturity. Australia (12.4% CAGR) benefits through sports nutrition adoption and hot climate logistics challenges. Mature markets including Germany (11.8%) and United Kingdom (10.6%) generate steady volume through pharmaceutical distribution, constrained by EU regulatory timelines for novel strain approvals.
Heat-Stable Probiotics Market Definition
The heat-stable probiotics market comprises spore-forming probiotic bacteria primarily from the Bacillus genus, including Bacillus coagulans, Bacillus subtilis, and Bacillus clausii, characterised by protective spore coatings enabling survival through heat processing above 200°F, pasteurisation, and ambient storage. These strains germinate in the intestinal tract delivering digestive and immune health benefits, serving food and beverage manufacturers, dietary supplement producers, clinical nutrition formulators, and animal feed applications.
Heat-Stable Probiotics Market Inclusions
The report includes global and regional market sizes (volume and value) and a 10-year forecast (2026 to 2036). It covers segmental breakdowns by strain type, application, end use, and distribution channel, alongside heat resistance validation data, cold chain cost analysis, FDA GRAS and EU QPS regulatory tracking across 40 plus countries.
Heat-Stable Probiotics Market Exclusions
The scope excludes traditional refrigeration-dependent Lactobacillus and Bifidobacterium strains unless formulated with microencapsulation enabling marginal heat tolerance. It omits finished consumer probiotic food products, focusing strictly on the spore-forming ingredient and its immediate food manufacturing applications. Pharmaceutical-grade probiotic drug products requiring clinical trial approval pathways are also excluded.
Heat-Stable Probiotics Market Research Methodology
- Primary Research: Interviews were conducted with probiotic ingredient suppliers, food scientists specialising in thermal processing, baking industry technical consultants, and regulatory affairs specialists navigating FDA GRAS determinations.
- Desk Research: Academic research on spore survival kinetics, thermal death time studies, FDA GRAS filings, EU QPS status documentation, and functional food ingredient trade association data were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was applied, reconstructing demand from functional food ingredient volumes and supplement market sizing, validated against strain supplier production capacities.
- Data Validation and Update Cycle: Outputs were cross-referenced with clinical CFU viability literature, regulatory approval timelines, and structured internal review as per FMI modelling standards.
Segmental Analysis
Heat-Stable Probiotics Market Analysis by Strain Type
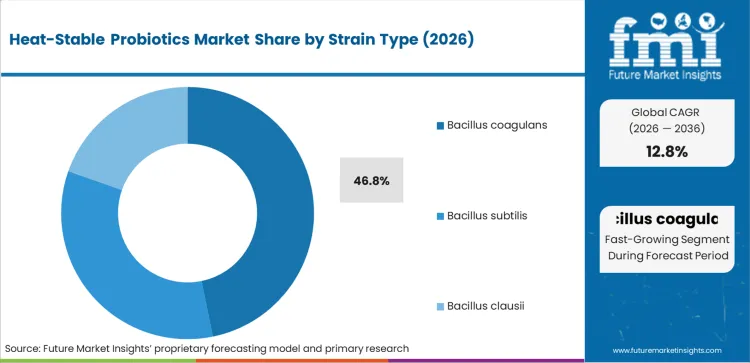
Based on FMI's heat-stable probiotics market report, Bacillus coagulans is estimated to hold 46.8% share in 2026. Dominance reflects FDA GRAS status for major commercial strains including GanedenBC30 and LactoSpore, extensive peer-reviewed clinical documentation, and superior heat resistance validated through commercial baking and brewing temperature studies that competing strains cannot match without equivalent trial investment.
- GRAS Confirmation: Ganeden Biotech's GanedenBC30 holds FDA GRAS status confirmed through self-affirmation filing, enabling food manufacturers to incorporate the strain into baked goods and beverages without pre-market approval requirements that create launch barriers for strains lacking equivalent regulatory standing [1].
- Clinical Documentation: Bacillus coagulans GBI-30 has accumulated 17 peer-reviewed publications documenting digestive health and immune function benefits, disclosed through Ganeden's ingredient marketing materials and supporting retailer buyer substantiation requirements [2].
- LactoSpore Stability: Sabinsa Corporation's LactoSpore demonstrated 36-month room temperature stability in published stability studies, validating ambient distribution economics for finished products and eliminating refrigeration infrastructure requirements [3].
Heat-Stable Probiotics Market Analysis by Application
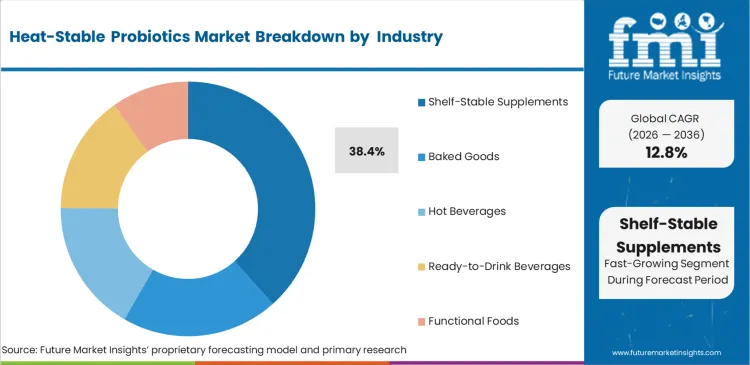
Based on FMI's heat-stable probiotics market report, shelf-stable supplements hold 38.4% share in 2026 through cost structure advantages eliminating refrigeration at every distribution stage. Ambient-temperature formats enable vending machine distribution and standard e-commerce shipping inaccessible to refrigerated probiotic capsules requiring cold pack logistics adding USD 3 to 8 per direct-to-consumer shipment.
- Baked Goods Entry: Lallemand Health Solutions disclosed baking application validation data in 2024 demonstrating its Bacillus coagulans strain maintaining greater than 80% CFU viability through standard muffin and cookie production at 275°F, providing bakery manufacturers ingredient certification required for functional wellness label positioning [4].
- Hot Beverage Validation: Research published in the Journal of Food Science in 2023 documented Bacillus coagulans achieving 99.76% viability after tea brewing at standard temperatures, providing beverage manufacturers peer-reviewed substantiation enabling probiotic tea and coffee product development [5].
- RTD Expansion: Kerry Group disclosed heat-stable probiotic ingredient supply agreements with ready-to-drink beverage manufacturers in its 2024 annual report, reflecting demand from beverage companies seeking pasteurisation-surviving strains for ambient-temperature probiotic RTD formats [6].
Heat-Stable Probiotics Market Drivers, Restraints, and Opportunities
Future Market Insights analysis confirms the heat-stable probiotics category is transitioning from specialty fermented food ingredient toward mainstream functional food enabler, driven by food manufacturers recognising that cold chain elimination creates commercial distribution advantages alongside functional positioning. The USD 3.8 billion 2026 valuation reflects early mainstream penetration where FDA GRAS confirmations for major commercial strains have removed the primary regulatory barrier preventing food company formulation investment.
As per FMI, the central tension sits between strain clinical documentation depth and food manufacturer formulation speed expectations. Licensed GRAS-confirmed strains with existing clinical dossiers enable twelve to eighteen-month product launch cycles versus three to five years for proprietary novel strain development, concentrating commercial momentum among established strain holders and limiting new entrant differentiation to application innovation.
- FDA GRAS Framework: GanedenBC30 and LactoSpore GRAS status through self-affirmation and expert panel review enables food manufacturers to incorporate heat-stable probiotics into baked goods and beverages without pre-market approval timelines, removing the regulatory barrier that previously prevented mainstream food company formulation investment [1].
- India Origin Advantage: Sabinsa Corporation's domestic LactoSpore manufacturing creates cost structure advantages for tropical markets where ambient temperatures above 30°C eliminate refrigerated probiotic viability, establishing heat-stable strains as the only commercially viable probiotic format for Indian convenience retail and e-commerce channels [3].
- EU QPS Pathway: EFSA's Qualified Presumption of Safety status for specific Bacillus coagulans and subtilis strains provides European food manufacturers a regulatory framework enabling ingredient use without full novel food authorisation, reducing market entry costs versus traditional novel ingredient approval pathways [7].
Regional Analysis
Based on the regional analysis, the heat-stable probiotics market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40 plus countries. The full report also offers market attractiveness analysis based on regional regulatory environments and cold chain infrastructure dynamics.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 16.2% |
| Japan | 14.8% |
| United States | 13.4% |
| Australia | 12.4% |
| Germany | 11.8% |
| United Kingdom | 10.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Heat-Stable Probiotics Market Analysis
Asia Pacific is the market's largest revenue region through Bacillus fermented food traditions in Japan and South Korea, domestic ingredient manufacturing in India, and tropical climate conditions across Southeast Asia making refrigerated probiotic logistics commercially unviable. Sabinsa Corporation leads South Asian ingredient supply through LactoSpore; Morinaga Milk Industry anchors Japanese functional food application; Kerry Group's regional division supplies RTD beverage manufacturers.
- India: Demand for heat-stable probiotics in India is projected to rise at 16.2% CAGR through 2036. Sabinsa Corporation's domestic LactoSpore production provides cost advantages unavailable to import-dependent competitors, with 2024 Bangalore facility expansion increasing capacity targeting domestic functional food manufacturer supply. India's Food Safety and Standards Authority updated its functional food permitted ingredient list in 2023 to include Bacillus coagulans strains with established safety dossiers, providing food manufacturers regulatory clearance for probiotic fortification of shelf-stable baked goods previously restricted by approval ambiguity [3].
- Japan: Demand for heat-stable probiotics in Japan is projected to rise at 14.8% CAGR through 2036. Japan's Consumer Affairs Agency expanded its FOSHU framework in 2023 to include digestive health claims for specific Bacillus subtilis strains in non-dairy food formats, enabling functional food manufacturers to carry authorised claims on ambient-temperature products previously restricted to refrigerated dairy. Morinaga Milk Industry's 2024 annual report disclosed R&D investment in Bacillus coagulans applications for shelf-stable snack formats targeting FOSHU claim eligibility [8].
FMI's analysis of the Asia Pacific heat-stable probiotics market covers India, Japan, South Korea, China, Australia, and Rest of Asia Pacific. Readers can find FOSHU framework analysis, domestic ingredient production data, and tropical market cold chain cost dynamics.
Europe Heat-Stable Probiotics Market Analysis
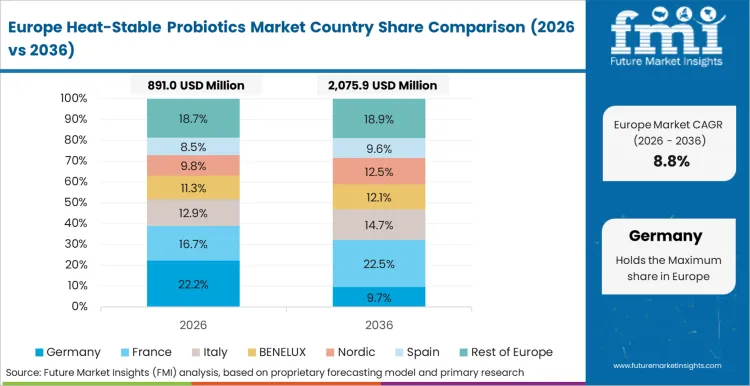
Europe functions as the market's regulatory validation environment where EFSA's QPS framework creates structured approval pathways rewarding established strain holders with regulatory certainty while raising barriers for new strain entrants. Chr. Hansen holds Bacillus strain libraries targeting food manufacturer formulation; Lallemand Health Solutions leads baking application technical support; BASF's nutrition division supplies fermentation culture inputs across EU markets.
- Germany: Demand for heat-stable probiotics in Germany is projected to rise at 11.8% CAGR through 2036. The German Federal Institute for Risk Assessment published updated Bacillus species safety guidance in 2023 covering permissible strain usage levels in food applications, enabling product development investment previously delayed by approval ambiguity. Chr. Hansen's 2024 annual report disclosed expanded Bacillus coagulans supply agreements with German functional food manufacturers targeting ambient-temperature fortified bread and cereal lines [10].
FMI's analysis of the heat-stable probiotics market in Europe covers Germany, United Kingdom, France, Netherlands, Sweden, and Rest of Europe. Readers can access EFSA QPS tracking, pharmaceutical distribution data, and functional food claim frameworks.
North America Heat-Stable Probiotics Market Analysis
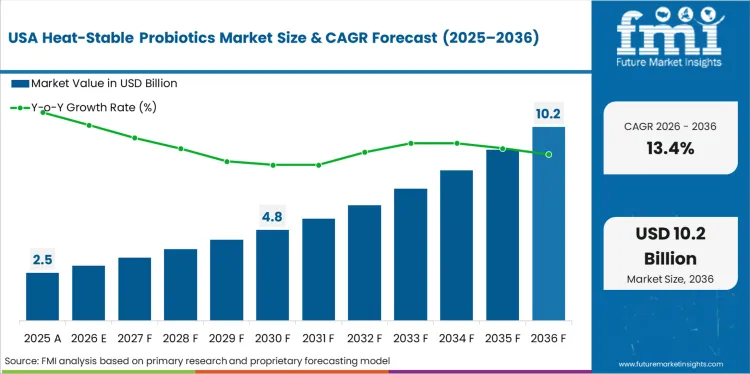
North America is the market's highest-growth developed region through FDA regulatory clarity for major commercial strains and supplement market maturity enabling premium convenience positioning. Ganeden Biotech leads through GanedenBC30 GRAS confirmation; Kerry Group supplies beverage manufacturer formulation demand; Lallemand Health Solutions anchors baking application technical development.
- United States: Demand for heat-stable probiotics in the United States is projected to rise at 13.4% CAGR through 2036. The FDA's GRAS self-affirmation pathway, under which GanedenBC30 established regulatory standing through expert panel review, continues enabling food manufacturers to launch heat-stable probiotic products without pre-market approval delays. NIH's National Center for Complementary and Integrative Health allocated USD 47 million in 2023 toward gut microbiome clinical research, generating publications that functional food brands cite in substantiation dossiers supporting pharmacy and retailer buyer presentations [9].
FMI's report includes detailed analysis of the North America heat-stable probiotics market covering the United States and Canada. Readers can find FDA GRAS framework analysis, supplement channel dynamics, and baked goods application development trends.
Competitive Aligners for Market Players
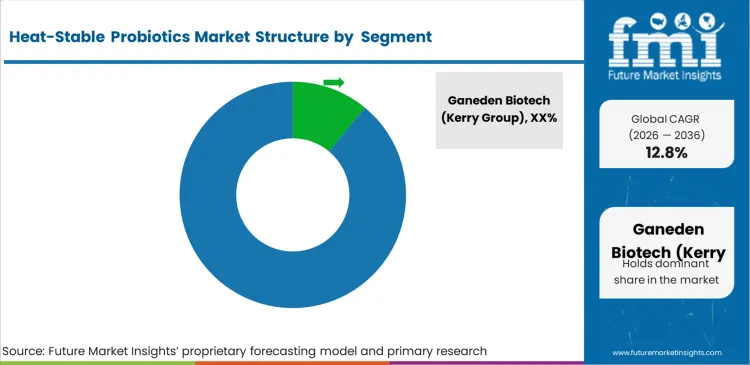
The heat-stable probiotics market is moderately concentrated among strain holders with FDA GRAS confirmation and EU QPS status, where the primary competitive variable is clinical documentation depth rather than production scale. GanedenBC30 and LactoSpore collectively hold the strongest commercial positions through published clinical dossiers that food manufacturer technical teams require before committing formulation investment, while smaller strain suppliers compete on price in animal feed and commodity supplement applications.
Ingredient suppliers with proprietary spore germination data and application-specific CFU retention studies hold structural advantages competitors cannot overcome through price alone. Food manufacturers require documented viability through their specific processing conditions including baking temperatures and pasteurisation cycles, creating technical qualification barriers favouring established strain suppliers over commodity producers offering generic Bacillus strains without process-specific performance data.
Buyer leverage among large food manufacturers is significant, as companies including Kerry Group and Ingredion evaluate multiple strain suppliers against standardised technical specifications before awarding formulation contracts, limiting price premium sustainability and rewarding suppliers offering technical application support and regulatory documentation packages.
Recent Developments
- In October 2024, Kerry Group disclosed expanded heat-stable probiotic ingredient supply agreements with ready-to-drink beverage manufacturers in its annual investor report, targeting pasteurisation-surviving strain applications for ambient-temperature RTD formats [6].
- In August 2024, Lallemand Health Solutions published baking application validation data confirming Bacillus coagulans CFU viability above 80% through standard commercial muffin production cycles, providing the functional baked goods category with peer-reviewed substantiation [4].
Key Players in Heat-Stable Probiotics Market
- Ganeden Biotech (Kerry Group)
- Sabinsa Corporation
- Lallemand Health Solutions
- Chr. Hansen Holding A/S
- Morinaga Milk Industry Co. Ltd
- Kerry Group plc
- Ingredion Incorporated
- BASF SE
- Unique Biotech Limited
- Probi AB
Scope of the Report
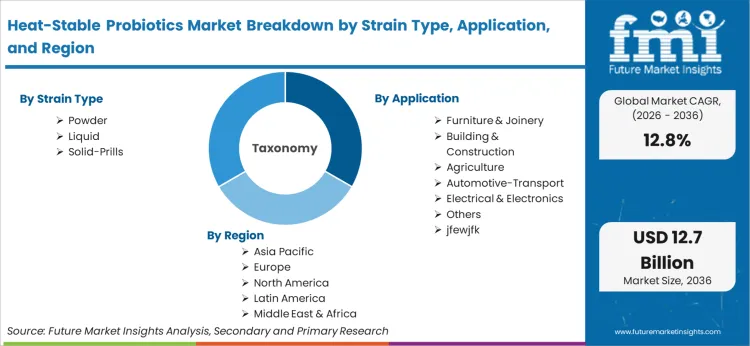
| Metric | Value |
|---|---|
| Quantitative Units | USD 3.8 billion (2026) to USD 12.7 billion (2036), at a CAGR of 12.8% |
| Market Definition | The heat-stable probiotics market comprises spore-forming Bacillus probiotic strains surviving heat processing, pasteurisation, and ambient storage while germinating in the intestinal tract, serving food and beverage manufacturing, dietary supplements, clinical nutrition, and animal feed applications. |
| Strain Type Segmentation | Bacillus coagulans, Bacillus subtilis, Bacillus clausii, Other Spore-Forming |
| Application Segmentation | Baked Goods, Hot Beverages, Shelf-Stable Supplements, Ready-to-Drink Beverages, Functional Foods |
| End Use Segmentation | Food & Beverage Manufacturing, Dietary Supplements, Clinical Nutrition, Animal Feed |
| Distribution Channel Segmentation | Pharmacies, Supermarkets, E-commerce, Food Service Distributors |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, India, Japan, South Korea, China, Australia and 40 plus countries |
| Key Companies Profiled | Ganeden Biotech, Sabinsa Corporation, Lallemand Health Solutions, Chr. Hansen, Morinaga Milk Industry, Kerry Group, Ingredion, BASF SE, Unique Biotech Limited, Probi AB |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up modelling validated through primary interviews with probiotic ingredient suppliers and thermal processing food scientists, supported by spore survival kinetics research and structured internal review |
Heat-Stable Probiotics Market Analysis by Segments
Strain Type
- Bacillus coagulans
- Bacillus subtilis
- Bacillus clausii
- Other Spore-Forming
Application
- Baked Goods
- Hot Beverages
- Shelf-Stable Supplements
- Ready-to-Drink Beverages
- Functional Foods
End Use
- Food & Beverage Manufacturing
- Dietary Supplements
- Clinical Nutrition
- Animal Feed
Distribution Channel
- Pharmacies
- Supermarkets
- E-commerce
- Food Service Distributors
Region
- Asia Pacific
- India
- Japan
- South Korea
- China
- Australia & New Zealand
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Netherlands
- Sweden
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of Middle East & Africa
Bibliography
- [1] Ganeden Biotech. (2018). GRAS Notice GRN 000770: Bacillus coagulans GBI-30, 6086. US Food and Drug Administration GRAS Notice Inventory, 2018.
- [2] Ganeden Biotech. (2024). GanedenBC30 clinical research summary: 17 peer-reviewed publications. Ganeden Ingredient Marketing Documentation, 2024.
- [3] Sabinsa Corporation. (2024). LactoSpore 36-month ambient stability data and Bangalore facility expansion. Sabinsa Corporate Communications, 2024.
- [4] Lallemand Health Solutions. (2024). Bacillus coagulans CFU viability through commercial baking cycles. Lallemand Technical Publication, August 2024.
- [5] Journal of Food Science. (2023). Bacillus coagulans viability after tea brewing at standard steeping temperatures. Wiley Online Library, 2023.
- [6] Kerry Group plc. (2024). Annual Report 2024: heat-stable probiotic ingredient supply agreements. Kerry Group Investor Relations, October 2024.
- [7] European Food Safety Authority. (2023). QPS assessment for Bacillus coagulans and Bacillus subtilis in food applications. EFSA Journal, 2023.
- [8] Morinaga Milk Industry Co. Ltd. (2024). Annual Report 2024: Bacillus coagulans R&D investment for FOSHU applications. Morinaga Investor Relations, 2024.
- [9] National Institutes of Health. (2023). NCCIH research funding: gut microbiome and mental health clinical trials. NIH Research Portfolio Online Reporting Tools, 2023.
- [10] Chr. Hansen Holding A/S. (2024). Annual Report 2023/24: Bacillus coagulans supply agreements with German functional food manufacturers. Chr. Hansen Investor Relations, 2024.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence for decision making across probiotic ingredient suppliers, food manufacturers, supplement brands, and cold chain logistics investors
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated strain supplier production data and functional food benchmarks
- Growth opportunity mapping across Bacillus strain types with emphasis on baked goods and hot beverage application unlocks
- Segment and regional revenue forecasts covering shelf-stable supplements, RTD beverages, and baked goods across North America, Europe, and Asia Pacific
- Competition strategy assessment including FDA GRAS advantages, clinical dossier depth, and food manufacturer formulation contract dynamics
- Regulatory compliance tracking including FDA GRAS framework, EU QPS status, FOSHU eligibility, and FSSAI permitted ingredient updates
- Regulatory impact analysis covering heat survival kinetics requirements and qualified health claim pathway access across key markets
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for heat-stable probiotics in the global market in 2026?
Demand for heat-stable probiotics in the global market is estimated to be valued at USD 3.8 billion in 2026.
What will be the market size of heat-stable probiotics by 2036?
The heat-stable probiotics market is projected to reach USD 12.7 billion by 2036.
What is the expected demand growth for heat-stable probiotics between 2026 and 2036?
Demand for heat-stable probiotics is expected to grow at a CAGR of 12.8% between 2026 and 2036.
Which strain type is poised to lead global sales in 2026?
Bacillus coagulans leads at 46.8% share through FDA GRAS status and extensive peer-reviewed clinical documentation.
Which application segment holds the largest share in the heat-stable probiotics market?
Shelf-stable supplements lead with 38.4% share in 2026 through cold chain elimination enabling ambient e-commerce and vending machine distribution.
Which region leads revenue in the heat-stable probiotics market?
Asia Pacific leads through traditional Bacillus fermented food consumption and domestic LactoSpore manufacturing in India.
What is driving heat-stable probiotics demand in the United States?
FDA GRAS confirmation for GanedenBC30 and NIH NCCIH's USD 47 million gut microbiome research funding are the primary drivers.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 13.4% during 2026 to 2036.
Does the report cover Japan in its regional analysis?
Japan is included within the Asia Pacific scope with individual CAGR and FOSHU framework analysis.
What is the main demand theme linked to Japan in Asia Pacific coverage?
Japan's demand is driven by the Consumer Affairs Agency's 2023 FOSHU expansion to include Bacillus subtilis digestive health claims in non-dairy ambient formats.
Does the report cover India in its regional analysis?
India is included within the Asia Pacific scope with individual CAGR and FSSAI ingredient approval assessment.
What is the main India demand theme in Asia Pacific coverage?
India's demand is driven by domestic LactoSpore production cost advantages and tropical climate eliminating refrigerated probiotic commercial viability.
Which distribution channel shows strongest growth in heat-stable probiotics?
E-commerce shows strongest growth through ambient shipping eliminating cold pack logistics costs of USD 3 to 8 per shipment.
What cold chain cost advantage do heat-stable probiotics provide?
Heat-stable probiotics deliver an estimated 20 to 30% distribution cost reduction versus refrigerated Lactobacillus strains.
What is the heat-stable probiotics market and what products does it cover?
The market covers spore-forming Bacillus probiotic ingredients incorporated into baked goods, hot beverages, supplements, RTD beverages, and functional foods.
What does the heat-stable probiotics market mean in this report?
The market refers to global production, trade, and industrial consumption of thermotolerant spore-forming Bacillus strains across food, supplement, clinical nutrition, and animal feed applications.
What is included in the scope of this report?
Scope covers strain types, applications, end use categories, distribution channels, market sizing, and regulatory tracking across 40 plus countries.
What is excluded from the scope of this report?
Refrigeration-dependent Lactobacillus strains, finished consumer probiotic food products, and pharmaceutical-grade probiotic drug products are excluded.
What does the market forecast mean on this page?
Market forecast represents a model-based projection built on defined assumptions for functional food ingredient adoption and cold chain cost reduction dynamics.
How does FMI build and validate the heat-stable probiotics market forecast?
The forecast uses a hybrid top-down and bottom-up model validated through primary interviews and spore survival kinetics research as per FMI modelling standards.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Strain Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Strain Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Strain Type , 2026 to 2036
- Bacillus coagulans
- Bacillus subtilis
- Bacillus clausii
- Bacillus coagulans
- Y to o to Y Growth Trend Analysis By Strain Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Strain Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Shelf-Stable Supplements
- Baked Goods
- Hot Beverages
- Ready-to-Drink Beverages
- Functional Foods
- Shelf-Stable Supplements
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Strain Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Strain Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Strain Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Strain Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Ganeden Biotech (Kerry Group)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sabinsa Corporation
- Lallemand Health Solutions
- Chr. Hansen Holding A/S
- Morinaga Milk Industry Co. Ltd
- Kerry Group plc
- Ganeden Biotech (Kerry Group)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Strain Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Strain Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Strain Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Strain Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Strain Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Strain Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Strain Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Strain Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Strain Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Strain Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Strain Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

