The liquid and lyophilised controls market is expected to grow from USD 918.6 million in 2026 to USD 1,757 million by 2036, reflecting a compound annual growth rate (CAGR) of 6.7%. These controls, used in laboratories and diagnostics to test the accuracy and reliability of medical instruments and assays, are essential in ensuring the quality of test results across various sectors, including clinical diagnostics, food safety, and environmental testing. The market’s growth is driven by the increasing demand for quality control and precision in laboratory testing, alongside the expansion of diagnostics in both clinical and research settings.
As regulatory standards for quality control become stricter and diagnostic tests become more complex, the need for reliable controls both liquid and lyophilised (freeze-dried) has grown significantly. Furthermore, advancements in medical technology, including automated and high-throughput testing platforms, continue to fuel the market demand for these controls. The growth is also supported by the rising healthcare spending, increasing investments in diagnostic technologies, and the expansion of the global diagnostics market.
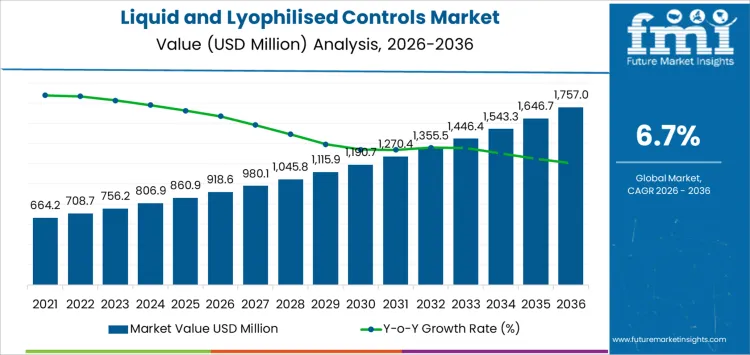
The saturation point analysis for the liquid and lyophilised controls market reveals consistent growth with a gradual slowdown as the market matures. Starting at USD 918.6 million in 2026, the market grows to USD 980.1 million in 2027 and USD 1,045.6 million in 2028, reflecting steady demand for liquid and lyophilised controls in various testing environments.
From 2028 to 2030, the market continues to expand, reaching USD 1,115.6 million in 2029 and USD 1,190.2 million in 2030. This period marks a gradual acceleration as more laboratories and diagnostic centers integrate advanced testing platforms that require more precise quality control tools. From 2030 to 2035, the market maintains steady growth, with the value reaching USD 1,269.8 million in 2031, USD 1,354.8 million in 2032, and USD 1,445.4 million in 2033.
By 2035, the market is projected to reach USD 1,542.1 million, with the final value of USD 1,757 million expected by 2036. The saturation point analysis suggests that while the market will experience consistent growth over the years, the rate of acceleration slows down towards the end of the forecast period. This slowdown reflects the point at which the market reaches more widespread adoption and saturation, as the demand for liquid and lyophilised controls becomes more stabilized within key industries like diagnostics, food safety, and environmental testing. Despite this, continued technological advancements will ensure steady growth as the market adapts to new diagnostic trends.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 918.6 million |
| Industry Forecast Value (2036) | USD 1,757 million |
| Industry Forecast CAGR (2026-2036) | 6.7% |
The global demand for liquid and lyophilised controls is driven by the need for reliable quality assurance in clinical diagnostics, pharmaceutical testing, and laboratory workflows. Controls are reference materials used alongside patient samples or test runs to verify that assays, instruments, and procedures deliver accurate and consistent results. In clinical laboratories, liquid and lyophilised controls are essential for routine calibration, proficiency testing, and compliance with regulatory and accreditation standards. Growth in diagnostic testing volume spurred by aging populations, rising chronic disease burden, and expanded screening programmes has increased reliance on controls to ensure lab performance and patient safety. Lyophilised controls, which are dehydrated for enhanced stability and extended shelf life, are particularly valued in markets where cold chain logistics are limited or where long term storage is needed. Liquid controls support real time assay validation and are widely used in automated and high throughput environments because they require minimal preparation and deliver consistent performance.
Future demand for liquid and lyophilised controls is expected to grow as healthcare systems prioritise quality management in diagnostics and pharmaceutical testing. Increasing adoption of advanced molecular assays, immunoassays, and multi parameter platforms will drive demand for specialised controls that match evolving technology requirements. Expansion of laboratory networks in emerging markets will also contribute to demand as facilities establish quality assurance programmes that rely on validated controls. Regulatory emphasis on standardisation and proficiency testing will continue to reinforce the use of reference materials to monitor performance and meet compliance thresholds. Technological developments that improve control stability, traceability, and ease of use including matrix matched and multi analyte controls will expand options for laboratories seeking greater efficiency. As diagnostics and therapeutic monitoring become more central to personalised medicine, the role of robust quality controls in delivering reliable results will support steady market growth worldwide.
The liquid and lyophilised controls market is segmented by product type and application. Liquid controls dominate the product type segment, accounting for 61.40% of the market share, due to their ease of use and immediate availability in laboratory testing. Clinical chemistry is the leading application segment, holding 28.70% of the market share, driven by the essential need for accurate, reliable control materials to ensure the precision of diagnostic testing. The market is experiencing growth as laboratories, especially those in healthcare and research, increasingly require high-quality control materials to maintain the accuracy, consistency, and reliability of diagnostic results across a wide range of testing fields, including clinical chemistry, immunoassay, molecular diagnostics, and more.
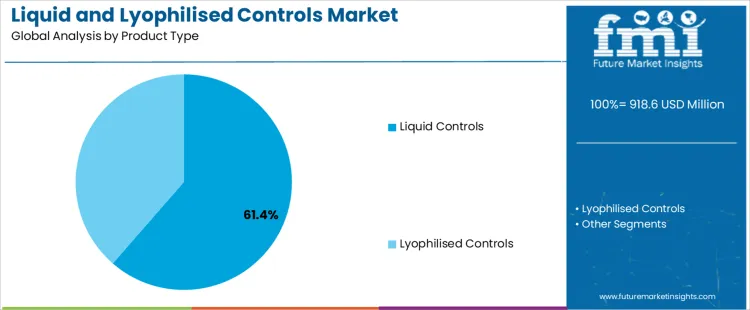
Liquid controls account for 61.40% of the market share, making them the dominant product type in the liquid and lyophilised controls market. These ready-to-use solutions offer significant advantages in terms of convenience, as they can be immediately utilized without preparation, making them highly suitable for high-demand environments such as hospitals, diagnostic laboratories, and clinical research settings. Liquid controls are stable, accurate, and provide a reliable means of calibrating and verifying laboratory testing instruments. Their role in quality assurance is critical across various diagnostic tests, including clinical chemistry, immunoassays, and molecular diagnostics. Liquid controls help laboratories maintain accuracy and consistency in their results, reducing the risk of errors and ensuring the reliability of test outcomes, which is essential for making accurate diagnoses. The increasing demand for automation and high-throughput testing in clinical settings is further driving the adoption of liquid controls, as these products integrate easily with automated systems, improving efficiency and workflow. As the healthcare sector continues to prioritize efficiency and accuracy, the demand for liquid controls is expected to grow significantly, securing their position as the leading product type in the market.
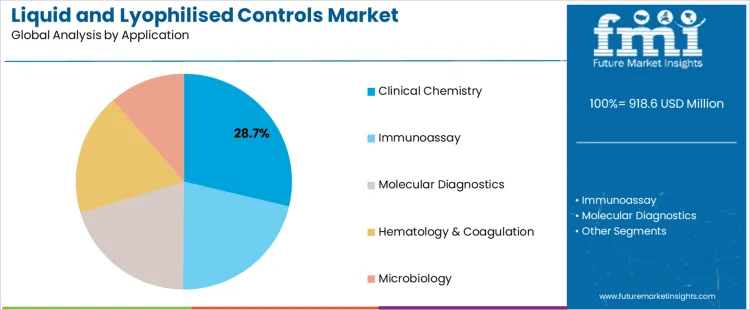
Clinical chemistry leads the application segment, representing 28.70% of the market share. Clinical chemistry plays a fundamental role in diagnostic medicine, focusing on the analysis of blood and other bodily fluids to detect a wide range of diseases, conditions, and infections. Accurate and consistent control materials are essential in clinical chemistry to ensure that laboratory instruments and testing platforms deliver reliable results. Liquid and lyophilised controls are used in clinical chemistry laboratories for calibration, performance monitoring, and verification of testing equipment, ensuring that diagnostic tests are accurate and consistent. These controls help to maintain the precision of tests for critical parameters such as glucose, cholesterol, and liver enzymes, all of which are vital for diagnosing and managing a variety of medical conditions. The need for reliable, high-quality controls in clinical chemistry is growing as more laboratories adopt automated systems and high-throughput testing, which increases the volume of tests performed and the need for ongoing calibration and validation. While immunoassay, molecular diagnostics, hematology & coagulation, and microbiology also contribute to market growth, clinical chemistry remains the largest application segment due to the widespread use of diagnostic tests in clinical and hospital settings. As medical advancements and the demand for more precise diagnostics increase, the role of control materials in clinical chemistry will continue to be a key driver in the market.
The global liquid and lyophilised controls market is expanding as clinical laboratories, diagnostics manufacturers, and research organisations seek reliable reference materials to ensure assay accuracy and consistency. Controls are used to verify the performance of in vitro diagnostics across infectious disease, oncology, endocrinology, and metabolic panels. Demand is shaped by increasing testing volumes, regulatory requirements for quality assurance, and the need to support standardised workflows. Preference for lyophilised formats grows where long shelf life and stability are priorities, while liquid controls offer convenience in high throughput environments.
Growing demand for clinical diagnostics and expanded screening programmes encourages use of high quality controls to monitor assay performance and reproducibility. Regulatory frameworks and accreditation standards require rigorous quality control documentation, which drives expenditure on characterised control materials. Growth in specialised testing areas such as molecular diagnostics and personalised medicine amplifies need for specific controls. Laboratories seeking to reduce variability and enhance confidence in results invest in validated liquid and lyophilised options. Expansion of lab infrastructure in emerging markets, digital reporting systems and automated platforms also support broader adoption of control materials.
One restraint is the cost associated with high quality, characterised control materials, which may reduce adoption in lower resource facilities. Some laboratories continue to use in house controls where regulatory and performance risks are perceived to be manageable, limiting purchases of commercial products. Transport and storage requirements can present challenges, especially for liquid formats that may need cold chain management. Lyophilised controls require reconstitution and validation steps that add procedural complexity compared with ready to use solutions. Variability in regional reimbursement policies and testing priorities influences investment in control materials across diagnostic segments.
A key trend is development of multi analyte and matrix matched control panels that support broad test menus and reduce inventory complexity. Manufacturers are expanding portfolios to include controls compatible with automated analyzers and high sensitivity platforms. Growth in digital quality management tools is linked with control usage, enabling electronic tracking, trend analysis and regulatory reporting. Increased focus on stability enhancements and extended shelf life influences product design, especially for lyophilised offerings. Collaboration between control developers and diagnostic companies helps align materials with evolving assay formats and performance expectations. Expansion of external quality assessment schemes enhances demand for standardised, traceable controls.
The Liquid and Lyophilised Controls market is experiencing steady growth, driven by increasing demand for quality control solutions in laboratory diagnostics and research. These controls are essential in ensuring the accuracy and reliability of diagnostic tests, particularly in medical laboratories and clinical settings. Liquid and lyophilised controls, which provide standardized samples for calibration and testing, are increasingly used in a variety of diagnostic and analytical applications. Countries such as India, China, and the USA are leading the market, fueled by advancements in healthcare infrastructure, growing laboratory services, and a focus on improving diagnostic precision. In the UK and Germany, the adoption of liquid and lyophilised controls is supported by robust healthcare systems and increasing emphasis on quality control in laboratory diagnostics. As the demand for accurate and reliable test results rises, the market for liquid and lyophilised controls is expected to grow across these regions.
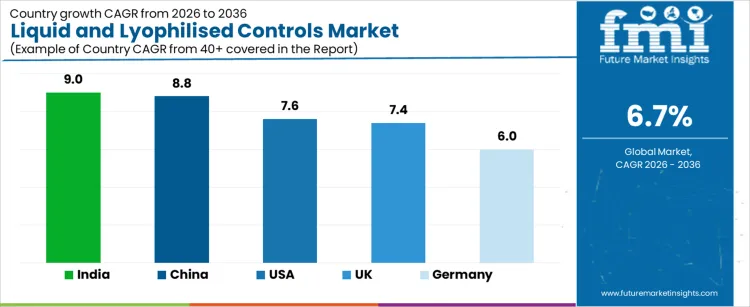
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9% |
| China | 8.8% |
| USA | 7.6% |
| UK | 7.4% |
| Germany | 6% |
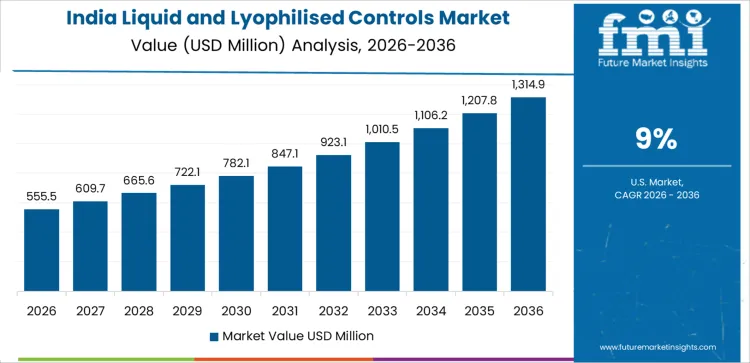
India’s Liquid and Lyophilised Controls market is projected to grow at a CAGR of 9%. The rapid expansion of healthcare services, particularly diagnostic laboratories, and an increasing focus on improving diagnostic accuracy are driving the demand for liquid and lyophilised controls. As the country’s healthcare infrastructure modernizes and more diagnostic facilities are established, the need for standardized and reliable testing methods becomes more pronounced. Liquid and lyophilised controls are essential for ensuring consistent and precise results in diagnostic tests. Furthermore, the growing adoption of advanced diagnostic technologies and increasing government investments in healthcare are expected to contribute to the growth of the market in India. As demand for reliable diagnostic tools continues to rise, India’s market for liquid and lyophilised controls is poised for continued expansion.
China’s Liquid and Lyophilised Controls market is expected to grow at a CAGR of 8.8%. The increasing focus on healthcare quality, along with rapid advancements in laboratory technology, is driving the adoption of liquid and lyophilised controls. China’s expanding healthcare sector, particularly in urban areas, has led to a greater emphasis on diagnostic accuracy and efficiency. The growing number of diagnostic laboratories, research institutions, and clinical facilities are increasing the demand for quality control solutions. Moreover, government efforts to improve healthcare standards and enhance medical infrastructure are contributing to the market’s growth. With a rising demand for precise and reliable diagnostic results, the need for liquid and lyophilised controls is expected to continue to rise in China.
The USA’s Liquid and Lyophilised Controls market is projected to grow at a CAGR of 7.6%. As the country’s healthcare system continues to expand, particularly in diagnostic services, there is increasing demand for accurate and reliable testing methods. Liquid and lyophilised controls are crucial for maintaining quality control in laboratory diagnostics, ensuring that diagnostic tests produce consistent and precise results. The adoption of advanced diagnostic technologies, coupled with the need for stringent regulatory standards, is driving the market in the USA. Additionally, the growing focus on personalized medicine, which requires accurate diagnostic tests, further contributes to the market’s growth. As healthcare facilities and research institutions continue to invest in quality control solutions, the demand for liquid and lyophilised controls in the USA is expected to rise steadily.
The UK’s Liquid and Lyophilised Controls market is projected to grow at a CAGR of 7.4%. The UK’s healthcare system places a strong emphasis on quality assurance in diagnostics, which is driving the adoption of liquid and lyophilised controls in medical laboratories. With increasing demands for accurate and reliable test results in clinical diagnostics, the need for effective quality control solutions has become essential. Liquid and lyophilised controls offer laboratories a reliable means to ensure diagnostic accuracy. Furthermore, advancements in laboratory automation and technology, combined with the UK government’s focus on improving healthcare standards, are contributing to the growth of the market. As the importance of diagnostic precision continues to rise, the demand for quality control solutions is expected to grow in the UK.
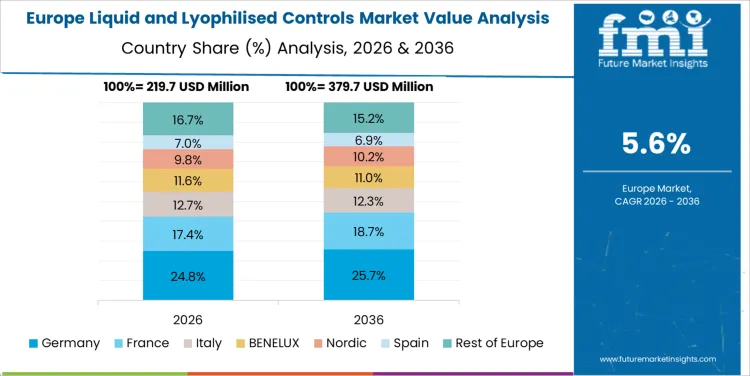
Germany’s Liquid and Lyophilised Controls market is expected to grow at a CAGR of 6%. Germany’s highly developed healthcare infrastructure, along with an increasing focus on improving diagnostic accuracy and reliability, is contributing to the market’s growth. Liquid and lyophilised controls are widely used in medical laboratories to maintain consistent test results, and their adoption is increasing as the demand for precise and accurate diagnostic testing rises. Germany’s strong emphasis on healthcare quality, coupled with advancements in laboratory technologies, is expected to further support market growth. As the healthcare sector continues to prioritize accuracy and standardization in diagnostics, the demand for liquid and lyophilised controls is likely to grow steadily in Germany.
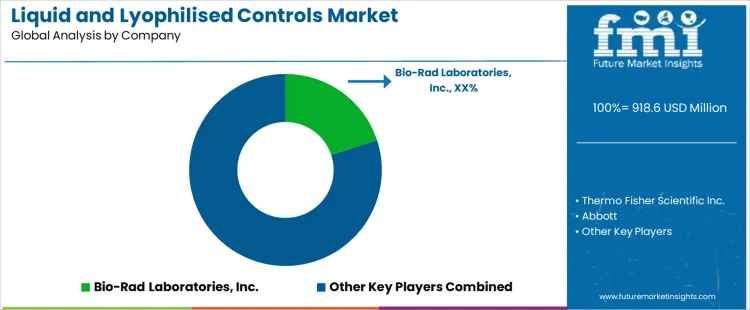
The Liquid and Lyophilised Controls Market is highly competitive, with several companies striving to meet the increasing demand for quality control solutions in diagnostics and laboratory testing. Bio-Rad Laboratories, Inc. leads the market, offering a comprehensive range of liquid and lyophilised controls that are essential for ensuring the accuracy and reliability of diagnostic testing. Their products are widely used in clinical laboratories, positioning Bio-Rad as a trusted brand in the market. Thermo Fisher Scientific Inc. follows closely with its diverse portfolio of control products, which include both liquid and lyophilised options. Their advanced technologies and strong global presence allow them to cater to a wide range of diagnostic applications.
Other key players, including Abbott, LGC Limited, and F. Hoffmann-La Roche, also play a significant role in the competitive landscape. Abbott provides high-quality liquid and lyophilised controls for a variety of diagnostic platforms, while LGC Limited focuses on specialized control products that support testing accuracy across multiple industries. F. Hoffmann-La Roche offers a wide selection of quality control materials, ensuring precision and consistency in diagnostics. These companies, alongside Randox Laboratories, Siemens Healthineers, and others, compete based on product performance, quality, and the ability to provide solutions that improve testing reliability in clinical and research environments.
Companies such as QuidelOrtho Corporation, Sysmex Corporation, and Bio-Techne bring innovations in liquid and lyophilised controls, each focusing on meeting the needs of their specific customer segments. Microbix Biosystems, Microbiologics, ZeptoMetrix, Fortress Diagnostics, and Streck, Inc. further contribute to the competitive dynamics with their specialized offerings, targeting niche applications within the market. The competition among these companies is driven by advancements in product quality, the expansion of product portfolios, and their ability to meet the increasing demand for high-precision control solutions in diagnostic testing.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product Type | Liquid Controls, Lyophilised Controls |
| Application | Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology & Coagulation, Microbiology |
| Matrix | Serum / Plasma, Urine, Whole Blood, Swab / Extract, Others (CSF, Tissue, etc.) |
| End User | Clinical Laboratories, Diagnostic Manufacturers, Hospitals, Research & Academic Labs |
| Companies | Bio-Rad Laboratories, Inc., Thermo Fisher Scientific Inc., Abbott, LGC Limited, F. Hoffmann-La Roche, Randox Laboratories, Siemens Healthineers, QuidelOrtho Corporation, Sysmex Corporation, Bio-Techne, Microbix Biosystems, Microbiologics, ZeptoMetrix, Fortress Diagnostics, Streck, Inc. |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar by sales by product type, application, matrix, end-user, and region. Includes market trends in liquid and lyophilised controls for clinical chemistry, immunoassays, molecular diagnostics, hematology & coagulation, and microbiology. Focus on the use of serum/plasma, urine, whole blood, swab/extract, and other matrices in diagnostic applications, cost-effectiveness, regulatory compliance, market share, and competitive positioning of key companies, and the role of these controls in improving diagnostic accuracy and consistency. |
How big is the liquid and lyophilised controls market in 2026?
The global liquid and lyophilised controls market is estimated to be valued at USD 918.6 million in 2026.
What will be the size of liquid and lyophilised controls market in 2036?
The market size for the liquid and lyophilised controls market is projected to reach USD 1,757.0 million by 2036.
How much will be the liquid and lyophilised controls market growth between 2026 and 2036?
The liquid and lyophilised controls market is expected to grow at a 6.7% CAGR between 2026 and 2036.
What are the key product types in the liquid and lyophilised controls market?
The key product types in liquid and lyophilised controls market are liquid controls and lyophilised controls.
Which application segment to contribute significant share in the liquid and lyophilised controls market in 2026?
In terms of application, clinical chemistry segment to command 28.7% share in the liquid and lyophilised controls market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.