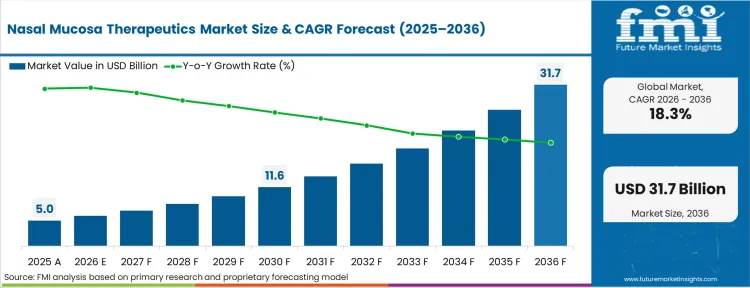
The nasal mucosa therapeutics market surpassed the valuation of USD 5.0 billion in 2025, propelling the industry to be poised to cross USD 5.9 billion in 2026 at a CAGR of 18.3% during the forecast period. The market is assessed at the estimation of USD 31.7 billion through 2036 as advanced formulations successfully bypass hepatic first-pass metabolism to deliver systemic molecules directly to the central nervous system.
Healthcare systems are actively deciding whether to transition acute crisis treatments from clinic-administered injectables to patient-administered nasal spray formats. Procurement directors must evaluate whether these mucosal delivery profiles offer sufficient absorption consistency to replace legacy emergency protocols. The commercial stakes for delay involve losing first-line prescription status in high-value indications, relegating traditional delivery methods to secondary options. Efficacy metrics alone do not dictate the winner, but rather the proprietary pairing of the molecule with the actuator device.
The exact moment this market accelerates is when fluid dynamics finally catches up to biochemistry, allowing large molecules to cross the mucosal barrier without triggering ciliary toxicity. Once device manufacturers achieve consistent plume geometry for high-viscosity biologics, the historical barrier to systemic delivery collapses. Pipeline candidates previously abandoned due to erratic absorption suddenly find immediate, viable pathways to commercialization.
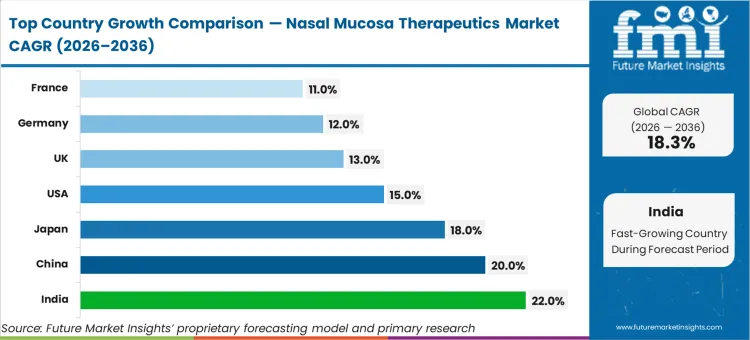
India is estimated to expand at 22.0%, followed by China at 20.0% and Japan tracking at 18.0%. The United States is poised to register 15.0%, while the United Kingdom garners 13.0%. Germany is projected to advance at 12.0% and France follows at 11.0%. The structural divergence across this range stems from the transition pace of domestic manufacturing infrastructure, as regions shift from producing basic localized respiratory solutions to manufacturing complex systemic delivery platforms.
The nasal mucosa therapeutics market encompasses pharmaceutical formulations and paired delivery mechanisms designed explicitly for absorption through the nasal cavity lining. The defining structural boundary is the intent of the delivery mechanism: it covers both therapies targeting local sinus pathology and formulations utilizing the highly vascularized mucosa as a rapid-onset pathway for systemic or central nervous system circulation.
This sector includes prescription and clinical-grade formulations administered via the nasal route, encompassing localized nasal polyposis treatment compounds and systemic agents. The scope covers the active pharmaceutical ingredients, the necessary absorption enhancers, and the specific primary delivery hardware required to achieve the validated dosing plume or droplet size necessary for mucosal adherence.
The market explicitly excludes over-the-counter saline washes, purely mechanical airway dilators, and traditional oral or intravenous formulations. These categories are excluded because they either do not rely on active pharmacological absorption across the mucosal barrier or they bypass the nasal delivery route entirely, placing them outside the specialized formulation and device engineering dynamics that govern this specific sector.
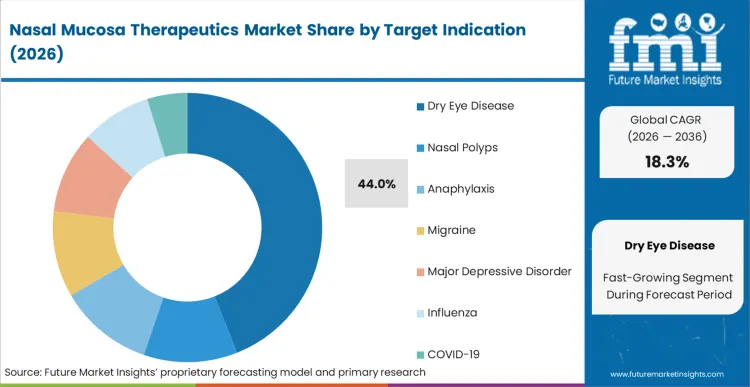
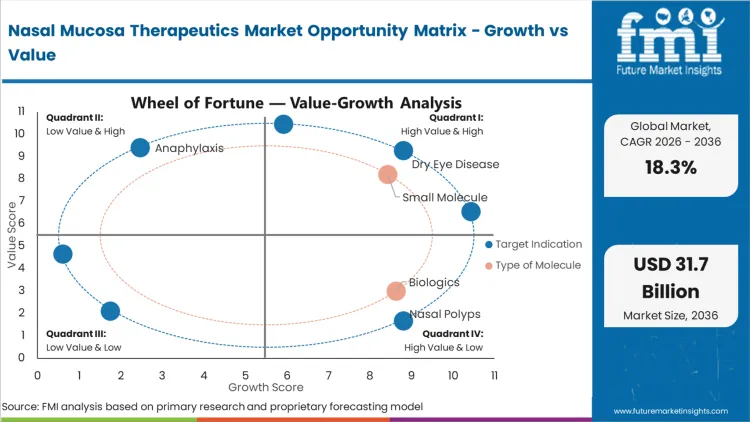
The dominant share of the Dry Eye Disease segment stems from a structural shift in treatment philosophy, addressing the pathology through neural pathways rather than topical application. FMI analysts opine that by targeting the trigeminal nerve, these therapeutics bypass the physical wash-out effect that limits traditional ocular drops. Clinical prescribers utilize this segment to achieve endogenous tear production, altering the fundamental management protocol for chronic patients. Formulators targeting this indication rely heavily on steroid-free nasal sprays to ensure long-term mucosal tolerability without the risk of localized atrophy. Development pipelines that ignore this neural pathway approach risk producing therapies with inferior patient adherence profiles.
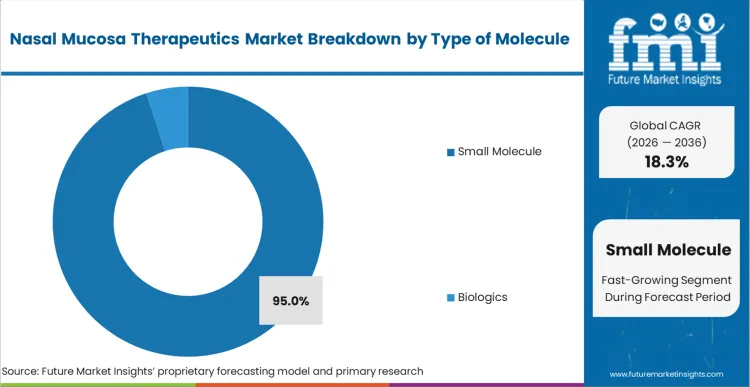
A failure to adequately permeate the mucosal lining results in immediate therapeutic failure, dictating the overwhelming reliance on small molecule architectures. Small molecules dominate with a 95.0% share in 2026 because their low molecular weight allows them to navigate the tight junctions of the nasal epithelium predictably. According to FMI's estimates, this predictability allows clinical pharmacologists to achieve systemic concentrations comparable to intravenous delivery within minutes. While the industry is attempting to adapt intranasal corticosteroids and other biologics for this route, the structural barriers of enzymatic degradation and molecular size remain severe. Developers clinging exclusively to large-molecule systemic therapies face prolonged regulatory scrutiny over absorption consistency.
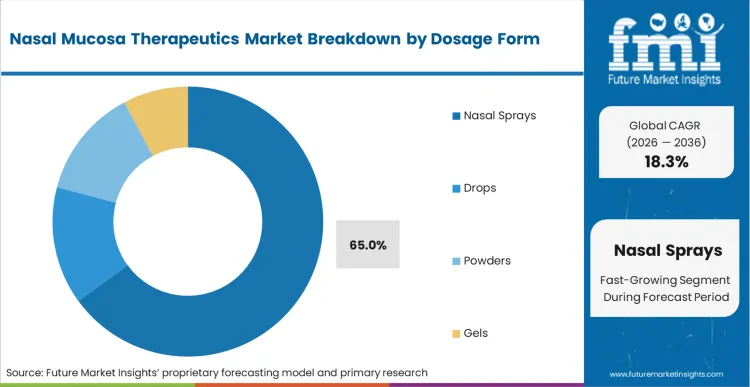
Traditional liquid drops fail to provide the spatial coating required for optimal absorption, forcing the transition to engineered spray mechanisms. Nasal sprays secure their leading position by delivering a calibrated mist that adheres to the highly vascularized turbinates rather than draining directly into the pharynx. Based on FMI's assessment, this precise plume geometry prevents the active ingredient from being lost to gastric degradation, maximizing the bioavailability of the formulation. The incorporation of advanced nasal allergy treatment compounds into these spray systems provides patients with reliable, metered dosing. Procurement networks that delay standardizing around advanced spray actuators face higher overall treatment costs due to wasted formulations and inconsistent clinical outcomes.
Moving emergency care out of the hospital and directly into the patient's hands is forcing healthcare networks to prioritize nasal delivery. Medical guidelines for severe migraines and anaphylaxis now actively favor intranasal administration because it completely eliminates the need for a trained professional to insert an IV line. This operational reality compels pharmaceutical companies to overhaul their older injectable drugs, transforming them into patient-friendly nasal congestion treatment devices and systemic sprays. The commercial outcome is clear: companies that successfully launch approved mucosal rescues capture the massive outpatient prescription market, while developers clinging to traditional injections are left fighting over a shrinking pool of severe hospital admissions.
Formulating for consistent mucosal absorption across varying patient physiologies without causing ciliary toxicity remains the primary structural friction slowing broader biologic adoption. The human nasal cavity is designed to clear foreign substances rapidly, meaning complex peptides often face enzymatic degradation or mucociliary clearance before reaching the bloodstream. While the introduction of permeation enhancers and mucoadhesive polymers provides a partial solution, these additives frequently irritate the nasal lining during chronic use, limiting their application to acute or infrequent dosing. This physiological barrier prevents a rapid, wholesale shift of the biologics pipeline into mucosal formats.
The regional analysis of the nasal mucosa therapeutics market highlights how diverse regulatory frameworks, healthcare infrastructure maturity, and demographic shifts shape adoption across global markets. FMI’s assessment reveals distinct structural trajectories dictating how domestic manufacturing capabilities and clinical procurement protocols are evolving in Asia Pacific, North America, and Europe, with the market being segmented into Asia Pacific, North America, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 22.0% |
| China | 20.0% |
| Japan | 18.0% |
| United States | 15.0% |
| United Kingdom | 13.0% |
| Germany | 12.0% |
| France | 11.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Contract manufacturing organizations are aggressively upgrading their cleanroom environments and device engineering capabilities to handle high-value neurological and systemic formulations. Manufacturing priorities are pivoting from pure volume production to achieving the strict bioequivalence metrics required for systemic mucosal absorption. The core industrial transition in this region is defined by the rapid evolution of domestic manufacturing infrastructure from basic respiratory generics to complex systemic delivery platforms. In FMI's view, this capability shift allows regional players to bypass the importation of expensive western delivery systems, establishing localized supply chains for advanced nasal packing devices and therapeutic sprays.
FMI's coverage of the Asia Pacific landscape indicates that the region is no longer just a high-volume generic hub, but is rapidly becoming a primary epicenter for advanced device-formulation pairing and localized consumption.
As per FMI's projection, this strict combination-product framework severely penalizes developers who treat the actuator as an afterthought, elevating the strategic importance of specialized mucosal atomization devices early in the clinical timeline. The regulatory environment in this region dictates that the delivery device and the active formulation must be approved as a single, inseparable combination product. The procurement landscape is by operational necessity dominated by alliances between tier-one drug developers and specialized engineering firms. This policy-led structure forces pharmaceutical companies to lock in their device partnerships years before commercialization, effectively preventing post-approval component swapping.
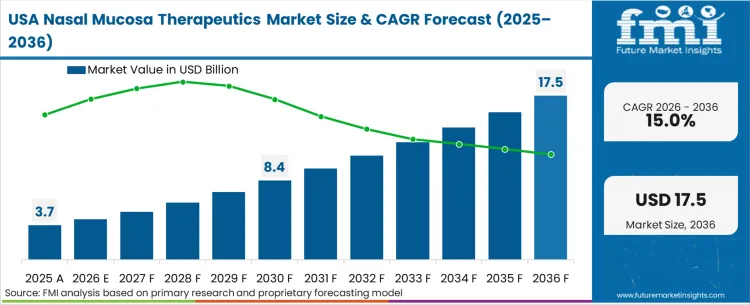
As a defining constraint per FMI’s assessment, the North American sector remains heavily gated by combination-product approvals, ensuring that only those who master both the molecule and the mechanical actuator can successfully navigate the region's acute crisis procurement pathways.
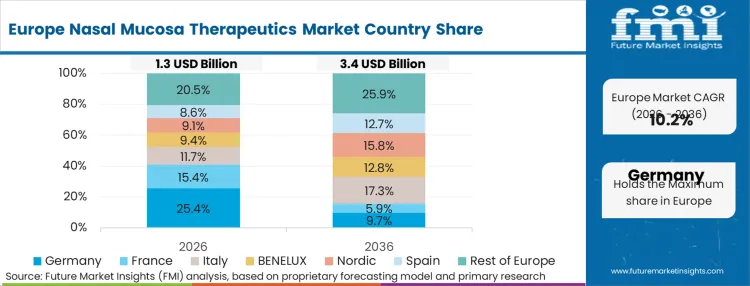
Health technology assessment bodies systematically evaluate the total lifecycle cost of therapies, heavily favoring patient-administered nasal formats over injectables that require nursing time and sterile clinical space. The ability to demonstrate reduced hospital visit frequency becomes the primary variable for securing favorable national formulary placement. Cost-containment mandates across nationalized healthcare systems drive the adoption of mucosal therapeutics as a mechanism to reduce clinical administration overhead. FMI analysts opine that this economics-led environment forces manufacturers to prove not just clinical efficacy, but broader health-system efficiency when submitting fragmentable nasal and ear dressing systems and advanced sprays for reimbursement.
FMI's report includes extensive analysis of neighboring regulatory environments across the broader European and Asian markets. The integration of continuous nasal implant technologies and advanced formulation monitoring systems across these secondary markets signals a shift toward standardized cross-border bioequivalence frameworks.
Succeeding in this space requires more than just mixing a potent chemical. Pharmaceutical giants must prove their mechanical hardware works flawlessly every single time a patient presses the nozzle. Procurement teams at major hospitals aren't just looking at the clinical data of the active ingredient; they are scrutinizing the physical reliability of the spray plume itself. This operational reality acts as a massive barrier to entry, allowing GlaxoSmithKline plc, Johnson & Johnson, and Pfizer Inc. to effectively shut out generic challengers who treat the plastic actuator as a cheap afterthought.
The actual moat protecting these market leaders is intellectual property tied to fluid dynamics, not just biology. Companies like AstraZeneca PLC and specialized hardware architects such as AptarGroup Inc. hold the precise mechanical blueprints required to keep thick, complex peptides from clogging the delivery nozzle. If a new player wants to steal institutional contracts, they cannot simply copy the molecule. They are forced to design an entirely new mechanical pathway around existing patents. Incumbents intentionally raise this engineering ceiling to keep rivals at bay, a strategy clearly visible as they pair advanced sinus irrigation systems with highly specialized biologics.
Hospital networks naturally fight this vendor lock-in. The moment patents expire on older, simple liquid sprays, institutional buyers immediately shift their volume to cheaper secondary suppliers. Yet, the broader push toward highly complex, systemic treatments guarantees that the real pricing power remains with the top-tier developers. As Novartis AG and Eli Lilly and Company steer clinical protocols toward proprietary neurological and emergency rescue platforms, the market is structurally splitting in two. Basic localized therapies are rapidly devolving into a fierce generic price war, while the high-stakes systemic treatments consolidate tightly among the few who have mastered both the medicine and the machine.

| Metric | Value |
|---|---|
| Quantitative Units | USD 5.9 billion to USD 31.7 billion, at a CAGR of 18.3% |
| Market Definition | The Nasal Mucosa Therapeutics Market encompasses pharmaceutical formulations and paired delivery mechanisms designed explicitly for absorption through the nasal cavity lining for both local pathology and systemic circulation. |
| Target Indication Segmentation | Dry Eye Disease, Nasal Polyps, Anaphylaxis, Migraine, Major Depressive Disorder, Influenza, COVID-19 |
| Type of Molecule Segmentation | Small Molecule, Biologics |
| Dosage Form Segmentation | Nasal Sprays, Drops, Powders, Gels |
| Regions Covered | Asia Pacific, North America, Europe |
| Countries Covered | India, China, Japan, United States, United Kingdom, Germany, France, and 40 plus countries |
| Key Companies Profiled | GlaxoSmithKline plc, Johnson & Johnson, Pfizer Inc., AstraZeneca PLC, AptarGroup Inc., Novartis AG, Eli Lilly and Company |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews were conducted with clinical formulation directors and device procurement heads. The baseline data anchors to proprietary device shipment volumes and prescription trends. Forecasts were cross-validated against regional hospital formulary contracts for acute crisis management. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Nasal Mucosa Therapeutics Market in 2026?
The market is valued at USD 5.9 billion in 2026, signalling the baseline commercial validation of mucosal pathways as viable alternatives to localized respiratory treatments thereby establishing the financial foundation for complex systemic delivery engineering.
What will it be valued at by 2036?
The industry will reach USD 31.7 billion by 2036, indicating that healthcare procurement networks have fundamentally reallocated emergency and neurological therapeutic budgets away from traditional injectables toward patient-administered mucosal options.
What CAGR is projected?
A CAGR of 18.3% is projected from 2026 to 2036, this aggressive rate reflects the rapid pipeline conversion of central nervous system therapeutics into nasal formats, rather than organic growth in basic allergy or sinus management.
Which Target Indication segment leads?
Dry Eye Disease accounts for 60.0% of the market as a result of intranasal neurostimulation effectively bypasses the physical washout limitations of topical ocular drops, allowing prescribers to trigger natural tear production directly via the trigeminal nerve.
Which Type of Molecule segment leads?
Small Molecule formulations capture 95.0% of the share, driven by the low molecular weight of these compounds, which navigate the tight junctions of the nasal epithelium predictably without requiring the complex permeation enhancers that biologics mandate.
Which Dosage Form segment leads?
Engineered spray actuators guarantee a calibrated mist that adheres precisely to the highly vascularized turbinates, preventing the formulation from draining into the pharynx and ensuring consistent systemic absorption with nasal sprays leading the dosage form category.
What drives rapid growth?
Clinical protocol shifts are forcing hospital formularies to transition acute crisis treatments out of the clinic as the mandate to provide patients with reliable, self-administered therapies for anaphylaxis and migraine eliminates the logistical bottleneck of requiring trained personnel for intravenous access.
What is the primary restraint?
The structural friction lies in formulating for consistent mucosal absorption without triggering ciliary toxicity with complex peptides often facing rapid enzymatic degradation or mucociliary clearance before reaching the bloodstream, limiting the immediate commercialization of large-molecule systemic sprays.
Which country grows fastest?
India expands at 22.0%, significantly outpacing the United States at 15.0%, this structural difference is India's massive existing generic manufacturing base rapidly pivoting its infrastructure to capture high-margin export contracts for complex device-formulation pairings, while the US focuses strictly on primary combination-product approvals.
How does the combination-product framework alter North American procurement?
The regulatory requirement to approve the device and drug as an inseparable unit forces pharmaceutical developers to lock in hardware partnerships years before clinical trials conclude, preventing post-approval component swapping and solidifying the market power of specialized atomization engineers.
What role do novel formulations play in expanding allergy applications?
Formulators engineering needle-free allergy therapy systems utilize the mucosal lining to achieve rapid systemic desensitization, entirely bypassing the clinical overhead of traditional subcutaneous immunotherapy, shifting the treatment paradigm to home-based management.
How do incumbents maintain their competitive advantage?
Leading pharmaceutical firms control the intellectual property surrounding both the mucoadhesive excipients and the high-viscosity atomization mechanics leading challengers to cannot simply synthesize a bioequivalent molecule; they must engineer around heavily defended actuator patents to achieve acceptable plume geometry.
Why are traditional clinical injectables losing formulary status?
Nationalized health systems utilize health technology assessments to measure total lifecycle costs while patient-administered nasal formats drastically reduce the clinical nursing time and sterile facility space required, making them economically superior to injectables despite higher initial unit costs.
How does seasonal demand impact mucosal device engineering?
During periods of high seasonal allergy prevalence, patient mucosal inflammation alters baseline absorption kinetics motivating device manufacturers to engineer actuators capable of delivering consistent doses even when the physical environment of the nasal cavity is compromised.
Why does China's centralized procurement favor advanced spray formats?
Centralized hospital networks mandate metered-dose mucosal systems to standardize therapeutic outcomes across vast populations, eliminating the dosage variability inherent in manual liquid drops, reducing secondary medical interventions caused by under-dosing.
What prevents biologics from immediately dominating the allergy immunotherapy segment?
Large biologic molecules cannot passively traverse the tight junctions of the nasal epithelium, until non-toxic, transient permeation enhancers are perfected, biologics remain stalled by poor bioavailability and the risk of chronic mucosal irritation.
How does the Japanese demographic profile shape device design?
An aggressively aging population necessitates self-administered therapeutics that require minimal motor skills when geriatric care protocols explicitly favor single-press intranasal actuators over complex auto-injectors to guarantee compliance among patients with declining dexterity.
How do surgical adjuncts fit into the mucosal therapeutic space?
Post-operative healing relies heavily on localized mucosal delivery as formulations paired with functional endoscopic sinus surgery systems ensure that anti-inflammatory agents remain exactly at the site of surgical intervention without being cleared by natural sinus drainage.
What makes the Dry Eye Disease application non-obvious to generalists?
Generalists assume nasal therapies only treat respiratory as practitioners understand that the nasal cavity provides direct access to the trigeminal nerve, allowing formulators to treat ocular pathology neurologically rather than topically.
Why are device manufacturers acquiring formulation capabilities?
Engineering sinus dilation devices without understanding the viscosity of the accompanying steroid leads to immediate clinical failure.
How does the UK's NHS protocol transition influence global markets?
The NHS is actively moving emergency anaphylaxis management toward stable intranasal formats as the NHS operates under strict cost-efficiency mandates, its successful integration of these systems provides the operational blueprint for other nationalized healthcare models globally.
What dictates the success of highly specific treatments like birch allergy treatment in mucosal formats?
The success of specific allergen formulations depends on the mucoadhesive polymer's ability to hold the antigen against the mucosa long enough for immune recognition, and without advanced polymer science, the antigen is swallowed before desensitization can occur.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.