Olopatandine API Market
Olopatandine API Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Demand for Olopatandine API Market Forecast and Outlook 2026 to 2036
The olopatandine API market is expected to grow from USD 159.9 million in 2026 to USD 253 million by 2036, reflecting a compound annual growth rate (CAGR) of 4.7%. Olopatandine, an antihistamine used primarily in the treatment of allergic conjunctivitis and other ocular conditions, is a widely used active pharmaceutical ingredient (API. Its demand is driven by the rising prevalence of allergic conditions and the growing market for over-the-counter and prescription eye care products. The market for Olopatandine APIs is also influenced by the broader growth in the pharmaceutical sector, with increasing production of generic formulations and more affordable options contributing to expanded accessibility.
The growth of this market is supported by the increasing awareness of eye health and rising rates of ocular allergies worldwide. As more pharmaceutical companies develop formulations based on Olopatandine, the demand for its API will continue to increase, driven by both the rising incidence of allergic eye conditions and the expanding availability of cost-effective treatments.
Quick Stats for Olopatandine API Market
- Olopatandine API Market Value (2026): USD 159.9 million
- Olopatandine API Market Forecast Value (2036): USD 253 million
- Olopatandine API Market Global Forecast CAGR (2026 to 2036): 4.7%
- Top API Grade by Share in Olopatandine API Market: Ophthalmic Grade (USP/EP/JP) (61.7%)
- Key Growth Regions in Olopatandine API Market: India, China, USA, UK, Germany
- Top Players in Olopatandine API Market: Kyowa Kirin, Teva API, Aurobindo Pharma, Dr. Reddy's Laboratories, MSN Laboratories, Hetero, Jubilant Pharmova, Zhejiang HuahAI Pharma, Zhejiang Hisun, Amoli Organics, Alkem Laboratories, Ipca Laboratories, Lupin, Cadila (Zydus Lifesciences)
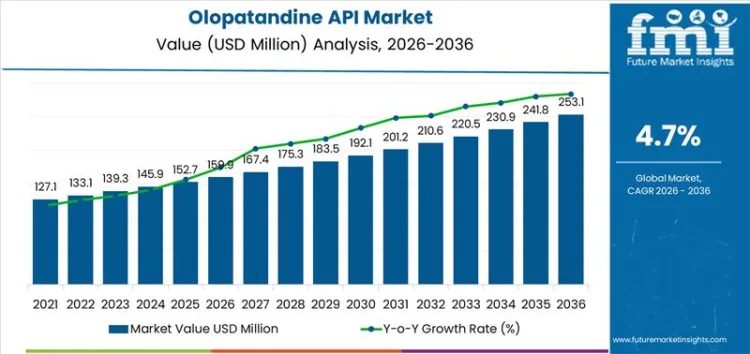
What is the Growth Forecast for the Olopatandine API Market through 2036?
The early vs late growth curve comparison for the Olopatandine API market shows gradual growth in the early years, followed by acceleration as the market matures. Starting at USD 159.9 million in 2026, the market grows to USD 167.4 million in 2027 and USD 175.2 million in 2028, reflecting steady but modest growth as pharmaceutical companies continue to incorporate Olopatandine into their offerings.
From 2028 to 2030, the market continues to expand, reaching USD 183.5 million in 2029 and USD 192.1 million in 2030. This period marks an acceleration as the market for eye care products continues to grow and the demand for Olopatandine API increases with the rise of related pharmaceutical treatments. Between 2030 and 2035, the market sees further acceleration, reaching USD 201.1 million in 2031, USD 210.5 million in 2032, and USD 220.4 million in 2033.
By 2035, the market is projected to reach USD 230.8 million, and by 2036, it will reach USD 253 million. The early vs late growth curve comparison reveals that while the market grows steadily in the initial years, it begins to accelerate as the adoption of Olopatandine-based treatments becomes more widespread. This acceleration is fueled by increased production of generics, wider market access, and continued advancements in eye care treatments, driving greater demand for Olopatandine API in the later part of the forecast period.
Olopatandine API Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 159.9 million |
| Industry Forecast Value (2036) | USD 253 million |
| Industry Forecast CAGR (2026-2036) | 4.7% |
What Is Driving the Demand for the Olopatadine API Market Globally?
The global demand for olopatadine active pharmaceutical ingredient (API) is driven by its widespread use in allergy and ophthalmic therapeutic products. Olopatadine is an antihistamine with mast cell stabilising properties, commonly formulated for allergic conjunctivitis and seasonal allergic symptoms affecting the eyes and nasal passages. Growing prevalence of allergic conditions worldwide influenced by environmental allergens, air pollution, and lifestyle factors has led to increased use of anti allergy therapies. Ophthalmic solutions, nasal sprays, and combination formulations containing olopatadine API are prescribed to reduce itching, redness, and inflammation, making the API a key input for branded and generic manufacturers of allergy medications. Expansion of healthcare access and rising awareness of allergy management, particularly in North America, Europe, and parts of Asia Pacific, support higher consumption of olopatadine based products. In addition, preference for convenient and non sedating therapies drives patient and clinician choice toward antihistamine APIs like olopatadine, further bolstering demand as part of pharmaceutical portfolios addressing seasonal and chronic allergic symptoms.
Future demand for the olopatadine API market is expected to grow as allergy prevalence continues and as pharmaceutical companies expand product lines with olopatadine based formulations. Growth in generic drug manufacturing and regulatory approvals for new delivery formats such as preservative free ophthalmic drops and fixed dose combinations will support uptake. Improvements in diagnosis and self care practices for allergic diseases may increase demand for over the counter and prescription treatments containing olopatadine, broadening the API’s end use base. Demographic shifts such as ageing populations and urbanisation in emerging economies may also contribute to higher incidence of allergic conditions, sustaining long term market expansion. Regulatory environments that facilitate generic entry and competition can influence pricing and availability, affecting how widely olopatadine API is sourced and used. Continued investment in research on allergy therapeutics and patient education about symptom management will shape demand patterns. Overall, as healthcare systems emphasise effective allergy treatment and patients seek rapid relief with minimal side effects, the olopatadine API market is expected to see steady global growth.
What Are the Key Segments in Olopatandine API Market?
The Olopatandine API market is segmented by API grade and synthesis route. Ophthalmic grade (USP/EP/JP) leads the API grade segment with 61.70% of the market share, reflecting the primary use of Olopatandine in treating eye-related conditions such as allergic conjunctivitis. Classical chiral resolution dominates the synthesis route segment with 46.80%, driven by its effective ability to separate chiral compounds in the production of Olopatandine. The market is growing as demand increases for effective and precise APIs used in the production of antihistamine treatments, particularly for eye care and allergy management.
Which API Grade Drives Olopatandine API Market Demand Globally?
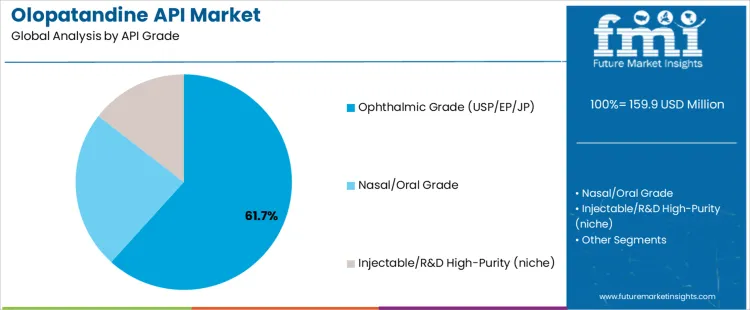
Ophthalmic grade (USP/EP/JP) accounts for 61.70% of the market share. Olopatandine is widely used as an antihistamine in ophthalmic formulations for the treatment of allergic conjunctivitis and other eye-related allergic conditions. Ophthalmic-grade APIs are required to meet stringent quality and safety standards as set by pharmacopeias such as USP (United States Pharmacopeia), EP (European Pharmacopoeia), and JP (Japanese Pharmacopoeia), ensuring purity and safety in eye care products. As the demand for effective treatments for eye allergies rises, especially in regions with high environmental pollution and allergens, ophthalmic-grade Olopatandine APIs are critical in providing patients with reliable, safe, and effective medications. The growing prevalence of allergic conditions, particularly in urban populations, is a key driver in the continued demand for ophthalmic-grade APIs, making it the leading segment in the Olopatandine API market.
Which Synthesis Route Drives Olopatandine API Market Demand Globally?
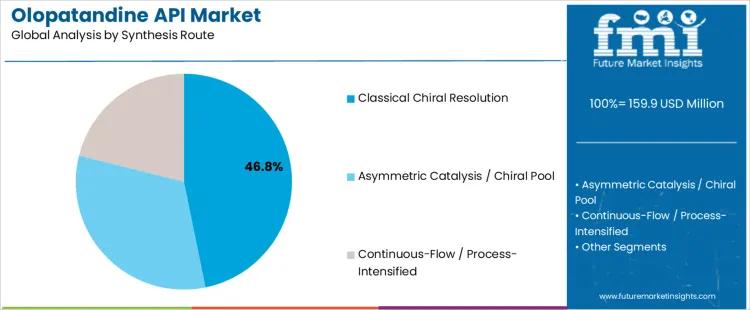
Classical chiral resolution leads the synthesis route segment with 46.80% of the market share. This route involves the separation of chiral molecules to produce high-purity Olopatandine, an essential process in creating the API for pharmaceutical formulations. Chiral resolution ensures the production of a single enantiomer of the compound, which is crucial for the efficacy and safety of the drug, particularly in ophthalmic and nasal treatments. The demand for classical chiral resolution is driven by its proven effectiveness in producing high-quality, pure Olopatandine for use in clinical treatments. As the pharmaceutical industry continues to focus on developing high-purity APIs for more precise therapeutic action, classical chiral resolution remains the preferred synthesis method, supporting its dominance in the market. While other synthesis routes such as asymmetric catalysis / chiral pool and continuous-flow / process-intensified also contribute to the market, classical chiral resolution remains the most widely used approach due to its established efficiency and reliability in API production.
What is the Summary of the Global Olopatadine API Market?
The global olopatadine API market is growing as demand increases for antihistamine treatments that address allergic conditions such as allergic rhinitis and conjunctivitis. Olopatadine is used in ophthalmic and nasal formulations to manage symptoms like itching, sneezing and irritation. Growth in allergy prevalence, broader access to healthcare, and rising adoption of convenient treatments support market expansion. Pharmaceutical manufacturers are producing generic and branded olopatadine APIs to supply topical and systemic products. Demand spans retail, clinical and over the counter channels across diverse regions with seasonal and perennial allergies.
What are the Key Drivers for the Global Olopatadine API Market?
Rising global prevalence of allergic diseases encourages use of effective antihistamine agents including olopatadine. Physicians and patients increasingly seek targeted therapies for eye and nasal allergy symptoms, supporting continued requirement for high quality API supply. Expansion of healthcare infrastructure, easier access to medical consultations and growing awareness of allergy management options promote uptake. Availability of olopatadine in multiple dosage forms such as eye drops and nasal sprays expands application opportunities. Generic entry and competitive pricing also broaden usage in cost sensitive markets. Pharmaceutical companies investing in respiratory and allergy portfolios maintain steady procurement of olopatadine API to support product pipelines.
What are the Restraints for the Global Olopatadine API Market?
One restraint is regulatory complexity associated with active pharmaceutical ingredient approval in different regions, which can slow product registration and commercialisation. Compliance with stringent quality and safety standards requires ongoing investment in manufacturing systems and documentation. Competition from alternative antihistamines or multi mechanism therapies may limit growth if prescribers favour other agents for certain patient groups. Supply chain disruptions for precursor chemicals or manufacturing inputs can affect continuity of API availability. Pricing pressures in markets with strong generic competition may reduce profitability for API producers, influencing investment decisions and capacity expansion.
What is the Key Trends in the Global Olopatadine API Market?
An important trend is expansion of olopatadine API production through strategic capacity investments and contract manufacturing partnerships to meet rising demand from pharmaceutical formulators. Manufacturers are focusing on ensuring consistent quality and compliance with global standards such as GMP to support exports and dossier submissions. Development of sustained release and combination formulations that include olopatadine encourages deeper integration into allergy care portfolios. Growth of over the counter allergy products featuring olopatadine drives stable procurement of API. Adoption of continuous manufacturing and quality by design practices enhances production efficiency and reduces batch variability, supporting reliable supply chains for formulators worldwide.
What is the Country-Wise Outlook for the Olopatandine API Market?
The Olopatandine API (Active Pharmaceutical Ingredient) market is growing steadily, driven by increasing demand for treatments for allergic conditions, including allergic rhinitis and conjunctivitis. Olopatandine, an antihistamine and mast cell stabilizer, is commonly used in eye drop formulations and oral medications. Countries such as India, China, and the USA are witnessing growth in this market due to rising healthcare needs, an increasing prevalence of allergies, and expanding pharmaceutical production. In markets like the UK and Germany, the demand for Olopatandine APIs is supported by a well-established pharmaceutical industry and a focus on improving healthcare access. As the global demand for allergy medications rises, the Olopatandine API market is expected to grow across these regions.
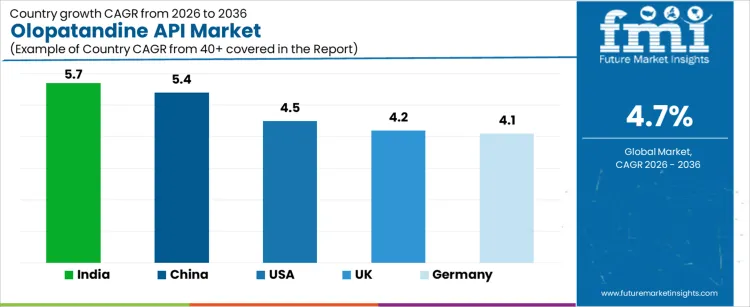
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 5.7% |
| China | 5.4% |
| USA | 4.5% |
| UK | 4.2% |
| Germany | 4.1% |
What is Driving the Growth of the Olopatandine API Market in India?
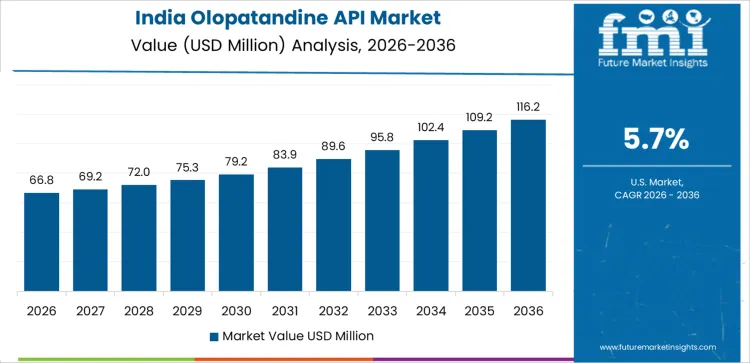
India’s Olopatandine API market is projected to grow at a CAGR of 5.7%. The country’s rapidly expanding pharmaceutical industry, coupled with increasing cases of allergies and other respiratory conditions, is driving demand for Olopatandine APIs. As a major player in generic drug production, India is witnessing growing exports of allergy-related medications, including Olopatandine-based products. Additionally, the rising prevalence of allergic rhinitis and conjunctivitis in India, particularly among urban populations, is further fueling market growth. India’s cost-effective manufacturing capabilities and increased access to affordable healthcare solutions are also contributing to the demand for Olopatandine APIs. With advancements in pharmaceutical research and increasing awareness of allergic conditions, India’s Olopatandine API market is expected to continue its upward trajectory.
What is Contributing to the Growth of the Olopatandine API Market in China?
China’s Olopatandine API market is expected to grow at a CAGR of 5.4%. The growing prevalence of allergies, particularly in urban areas, is driving demand for antihistamine treatments such as Olopatandine. As China continues to invest in improving healthcare infrastructure and access to medications, the pharmaceutical market is expanding, creating greater opportunities for API suppliers. The country’s rapid urbanization, combined with increased exposure to allergens and environmental factors, is contributing to the rising number of allergy cases. Moreover, China’s pharmaceutical industry is growing in both production and export, positioning the country as a major player in the global Olopatandine API market. With a growing focus on improving the accessibility of allergy treatments, the demand for Olopatandine is expected to rise steadily.
What is Driving the Olopatandine API Market Growth in the USA?
The USA’s Olopatandine API market is projected to grow at a CAGR of 4.5%. The increasing prevalence of allergic conditions, including allergic rhinitis and eye allergies, is driving the demand for Olopatandine-based treatments. With a high level of healthcare access and awareness, consumers in the USA are turning to antihistamine and mast cell stabilizer treatments to manage their symptoms. Additionally, as the demand for allergy medications continues to rise, pharmaceutical companies in the USA are seeking high-quality APIs to develop and manufacture over-the-counter and prescription products. The strong healthcare infrastructure, coupled with the demand for effective treatments, supports continued growth in the Olopatandine API market.
What is Influencing the Growth of the Olopatandine API Market in the UK?
The UK’s Olopatandine API market is projected to grow at a CAGR of 4.2%. Rising cases of allergic conditions, such as allergic rhinitis and conjunctivitis, are contributing to the growing demand for effective treatments like Olopatandine. The UK’s strong pharmaceutical industry, which focuses on producing both branded and generic medications, is increasingly adopting APIs like Olopatandine to meet market needs. The demand for allergy medications is also being driven by increased awareness of the importance of treating allergic symptoms and the availability of OTC allergy treatments. With a focus on healthcare improvement and the growing use of antihistamine-based treatments, the UK market for Olopatandine APIs is expected to see steady growth.
What is Contributing to the Growth of the Olopatandine API Market in Germany?
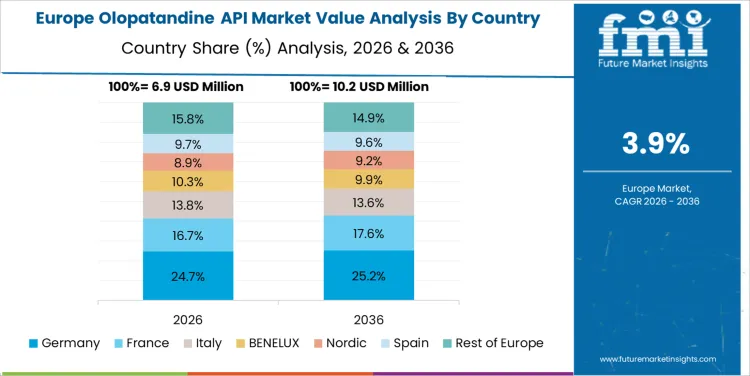
Germany’s Olopatandine API market is expected to grow at a CAGR of 4.1%. The rise in allergy-related health issues, particularly in urban populations, is increasing the demand for antihistamine treatments like Olopatandine. Germany’s pharmaceutical sector, which is one of the largest in Europe, is driving the demand for APIs as part of its focus on producing high-quality treatments for various conditions, including allergies. With a growing emphasis on improving healthcare access and offering effective treatments, the German market for Olopatandine APIs is benefiting from increased awareness and demand for allergy medications. As Germany’s pharmaceutical production continues to expand, the market for Olopatandine APIs is expected to grow steadily.
What is the Competitive Landscape for the Olopatandine API Market?
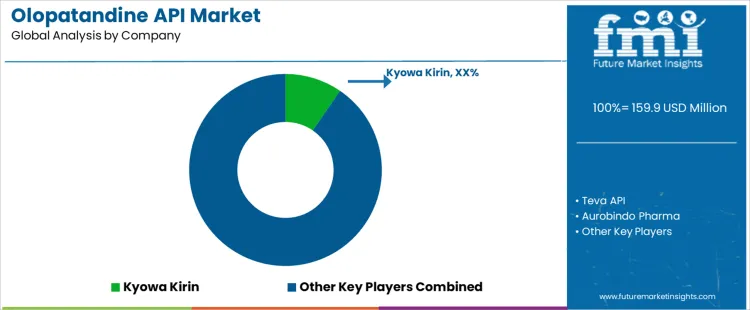
The Olopatandine API Market is experiencing significant competition, with several companies striving to meet the demand for this antihistamine API used in the treatment of allergic conjunctivitis. Kyowa Kirin leads the market, known for its high-quality olopatandine production that adheres to strict regulatory standards. Their established reputation and advanced manufacturing capabilities give them a strong competitive advantage in both domestic and global markets. Teva API follows closely with its reliable and cost-effective olopatandine production, making it a key player in the generic drug sector. The company’s broad distribution network and focus on affordability position it as a strong competitor.
Aurobindo Pharma, Dr. Reddy's Laboratories, and MSN Laboratories also contribute to the competitive landscape, each with a significant presence in the olopatandine API market. Aurobindo Pharma provides high-quality, competitively priced APIs that meet the needs of various markets. Dr. Reddy's Laboratories follows a similar strategy, focusing on high-efficiency manufacturing and meeting the increasing demand for generic APIs. MSN Laboratories is known for its cost-effective manufacturing processes, allowing the company to offer competitive pricing in the market. Other notable players, such as Hetero, Jubilant Pharmova, and Zhejiang HuahAI Pharma, provide strong competition by focusing on production scale, quality, and global distribution.
Zhejiang Hisun, Amoli Organics, Alkem Laboratories, Ipca Laboratories, Lupin, and Cadila (Zydus Lifesciences) round out the competitive field, each leveraging unique strengths to compete in the global market. These companies focus on cost leadership, regulatory compliance, and maintaining strong supplier relationships to ensure steady market presence. The competition within the olopatandine API market is driven by product quality, regulatory adherence, and the ability to scale production to meet the needs of both developed and emerging markets.
Key Players of the Olopatandine API Market
- Kyowa Kirin
- Teva API
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- MSN Laboratories
- Hetero
- Jubilant Pharmova
- Zhejiang HuahAI Pharma
- Zhejiang Hisun
- Amoli Organics
- Alkem Laboratories
- Ipca Laboratories
- Lupin
- Cadila (Zydus Lifesciences)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| API Grade | Ophthalmic Grade (USP/EP/JP), Nasal/Oral Grade, Injectable/R&D High-Purity (niche) |
| Synthesis Route | Classical Chiral Resolution, Asymmetric Catalysis / Chiral Pool, Continuous-Flow / Process-Intensified |
| End Use (Dosage Pull) | Ophthalmic Solutions / Eye Drops, Nasal Sprays, Oral Tablets/Syrups |
| Customer Type | Generic Formulators, Branded/Originator, CMOs/CDMOs (API/Fill-Form), Institutional/Other |
| Companies | Kyowa Kirin, Teva API, Aurobindo Pharma, Dr. Reddy's Laboratories, MSN Laboratories, Hetero, Jubilant Pharmova, Zhejiang HuahAI Pharma, Zhejiang Hisun, Amoli Organics, Alkem Laboratories, Ipca Laboratories, Lupin, Cadila (Zydus Lifesciences) |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar by sales by API grade, synthesis route, end-use dosage form, customer type, and region. Includes market trends in ophthalmic, nasal/oral, and injectable APIs, focusing on the demand for generic formulations, branded/originator products, and niche high-purity applications. Highlights regulatory compliance, process-intensified synthesis routes, market share, and the competitive positioning of key companies, along with the impact of various customer types on market growth. |
Olopatandine API Market Segmentation
API Grade
- Ophthalmic Grade (USP/EP/JP)
- Nasal/Oral Grade
- Injectable/R&D High-Purity (niche)
Synthesis Route
- Classical Chiral Resolution
- Asymmetric Catalysis / Chiral Pool
- Continuous-Flow / Process-Intensified
End Use (Dosage Pull)
- Ophthalmic Solutions / Eye Drops
- Nasal Sprays
- Oral Tablets/Syrups
Customer Type
- Generic Formulators
- Branded/Originator
- CMOs/CDMOs (API/Fill-Form)
- Institutional/Other
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the olopatandine api market in 2026?
The global olopatandine api market is estimated to be valued at USD 159.9 million in 2026.
What will be the size of olopatandine api market in 2036?
The market size for the olopatandine api market is projected to reach USD 5,788,912,970.0 million by 2036.
How much will be the olopatandine api market growth between 2026 and 2036?
The olopatandine api market is expected to grow at a 4.7% CAGR between 2026 and 2036.
What are the key product types in the olopatandine api market?
The key product types in olopatandine api market are ophthalmic grade (usp/ep/jp), nasal/oral grade and injectable/r&d high-purity (niche).
Which synthesis route segment to contribute significant share in the olopatandine api market in 2026?
In terms of synthesis route, classical chiral resolution segment to command 46.8% share in the olopatandine api market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By API Grade
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By API Grade , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By API Grade , 2026 to 2036
- Ophthalmic Grade (USP/EP/JP)
- Nasal/Oral Grade
- Injectable/R&D High-Purity (niche)
- Y to o to Y Growth Trend Analysis By API Grade , 2021 to 2025
- Absolute $ Opportunity Analysis By API Grade , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Synthesis Route
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Synthesis Route, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Synthesis Route, 2026 to 2036
- Classical Chiral Resolution
- Asymmetric Catalysis / Chiral Pool
- Continuous-Flow / Process-Intensified
- Y to o to Y Growth Trend Analysis By Synthesis Route, 2021 to 2025
- Absolute $ Opportunity Analysis By Synthesis Route, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use (Dosage Pull)
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use (Dosage Pull), 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use (Dosage Pull), 2026 to 2036
- Ophthalmic Solutions / Eye Drops
- Nasal Sprays
- Oral Tablets/Syrups
- Y to o to Y Growth Trend Analysis By End Use (Dosage Pull), 2021 to 2025
- Absolute $ Opportunity Analysis By End Use (Dosage Pull), 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- Generic Formulators
- Branded/Originator
- CMOs/CDMOs (API/Fill-Form)
- Institutional/Other
- Y to o to Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By API Grade
- By Synthesis Route
- By End Use (Dosage Pull)
- By Customer Type
- Competition Analysis
- Competition Deep Dive
- Kyowa Kirin
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Teva API
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- MSN Laboratories
- Hetero
- Jubilant Pharmova
- Zhejiang Huahai Pharma
- Zhejiang Hisun
- Amoli Organics
- Alkem Laboratories
- Ipca Laboratories
- Lupin
- Cadila (Zydus Lifesciences)
- Others
- Kyowa Kirin
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by API Grade , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Synthesis Route, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by End Use (Dosage Pull), 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by API Grade
- Figure 6: Global Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Synthesis Route
- Figure 9: Global Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 12: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Customer Type
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by API Grade
- Figure 29: North America Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Synthesis Route
- Figure 32: North America Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 34: North America Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 35: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Customer Type
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by API Grade
- Figure 42: Latin America Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Synthesis Route
- Figure 45: Latin America Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 48: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Customer Type
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by API Grade
- Figure 55: Western Europe Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Synthesis Route
- Figure 58: Western Europe Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 61: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by API Grade
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Synthesis Route
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by API Grade
- Figure 81: East Asia Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Synthesis Route
- Figure 84: East Asia Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 87: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Customer Type
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by API Grade
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Synthesis Route
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by API Grade , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by API Grade , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by API Grade
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Synthesis Route, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Synthesis Route, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Synthesis Route
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by End Use (Dosage Pull), 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by End Use (Dosage Pull), 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by End Use (Dosage Pull)
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

