Oxcarbazepine API Market
Oxcarbazepine API Market Analysis – Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Oxcarbazepine API Market Forecast and Outlook 2025 to 2035
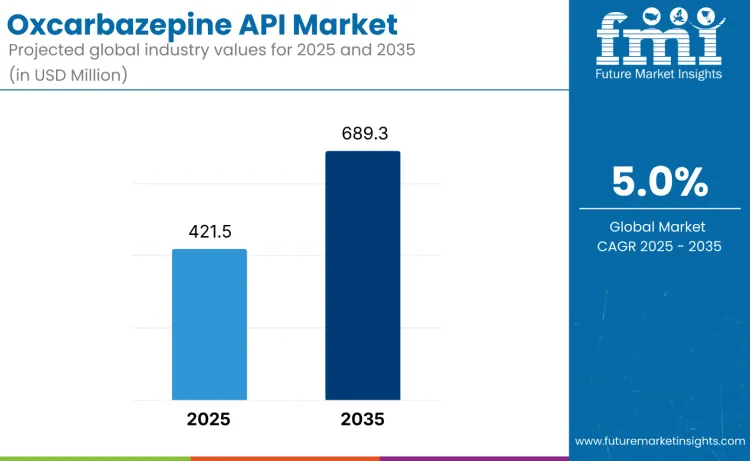
The global oxcarbazepine API market is projected to reach USD 689.3 million by 2035, expanding from USD 421.5 million in 2025 at a CAGR of 5.0% and achieving 1.6x growth over the forecast period. This expansion is driven by sustained clinical demand for oxcarbazepine in epilepsy and bipolar disorder management, rising neurological disease prevalence, and broader access to affordable antiepileptic therapies in emerging economies. Continued inclusion in public reimbursement lists and cost advantages over newer branded anticonvulsants reinforce their role as a long-term therapy option. Nevertheless, pricing pressure among generic suppliers, strict regulatory compliance requirements, and periodic supply disruptions linked to quality audits and DMF upkeep remain ongoing challenges.
Market growth reflects stable prescribing patterns across neurology clinics and hospital pharmacies, where oxcarbazepine is widely used for partial seizures and mood stabilization. Finished-dosage manufacturers increasingly prioritize API suppliers offering consistent impurity control, reliable particle-size distribution, and reproducible dissolution characteristics to ensure bioequivalence and maintain competitive positioning in tenders. Strong acceptance within government-funded healthcare systems and resource-sensitive treatment settings continues to support predictable volume demand, particularly in Asia, Latin America, and parts of Eastern Europe.
Quick Stats for Oxcarbazepine API Market
- Oxcarbazepine API Market Value (2025): USD 421.5 million
- Oxcarbazepine API Market Forecast Value (2035): USD 689.3 million
- Oxcarbazepine API Market Forecast CAGR: 5.0%
- Leading Market Segment: Pharmacopoeial Grade API (69.7% share)
- Key Growth Regions: Asia Pacific (India, China), North America, Europe
- Top Players: Sun Pharmaceutical Industries, Aurobindo Pharma, Hetero Drugs, Zhejiang Huahai Pharmaceutical, Teva API
Advancements in API manufacturing technology are strengthening supply reliability and regulatory readiness. Producers are improving crystallization techniques, solvent-recovery systems, and impurity-monitoring frameworks while reinforcing data integrity and quality-assurance practices aligned with FDA, EMA, and pharmacopoeial standards. At the same time, government healthcare spending, national epilepsy programs, and domestic manufacturing incentives, especially in India and China, are supporting long-term demand. Expanded hospital pharmacy distribution, tender-based procurement, and tele-neurology access models further enhance utilization across diverse clinical infrastructures.
Oxcarbazepine API Market Year-over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the oxcarbazepine API market is projected to rise from USD 421.5 million to USD 539.0 million, generating an incremental value of USD 117.5 million and accounting for 43.9 percent of total decade growth. Expansion is driven by increasing epilepsy and bipolar disorder diagnoses, wider formulary inclusion of cost-effective antiepileptic drugs, and tender-based procurement in emerging economies. Manufacturers strengthen competitiveness through improved crystallization control, advanced impurity profiling, solvent recovery optimization, and capacity expansion to meet GMP requirements across regulated and semi-regulated pharmaceutical supply channels.
From 2030 to 2035, the market is expected to expand from USD 539.0 million to USD 689.3 million, adding USD 150.3 million and representing 56.1 percent of overall ten-year growth. This phase reflects stronger vertical integration, preference for regionally sourced APIs to reduce supply risk, and tighter regulatory scrutiny on data integrity, DMF maintenance, and cGMP documentation. Growth is reinforced by expanding neurology care infrastructure, broader access to long-term therapies, and sustained demand for bioequivalent oxcarbazepine, driving investment in process efficiency, compliance systems, and durable supplier partnerships.
Oxcarbazepine API Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 421.5 million |
| Market Forecast Value (2035) | USD 689.3 million |
| Forecast CAGR (2025 to 2035) | 5.0% |
Why Is the Oxcarbazepine API Market Growing Worldwide?
The oxcarbazepine API market expands by supporting rising global treatment demand for epilepsy and bipolar disorder, where long-term therapy continuity and affordability remain critical to patient outcomes. Neurologists and psychiatric specialists increasingly favor oxcarbazepine due to its favorable tolerability and cost efficiency, sustaining steady procurement of related formulations. Clinical guidelines across multiple regions recognize the molecule for partial-onset seizure control and mood stabilization, driving utilization across hospital, retail, and public treatment channels. Growing epilepsy prevalence, improved diagnostic reach in developing economies, and heightened mental health awareness continue to reinforce consistent API demand across regulated and semi-regulated pharmaceutical markets.
Government essential-drug policies, broader insurance coverage, and public neurology programs expand access to generic anticonvulsants, enabling predictable API volume planning for producers. Rapid growth of pharmaceutical manufacturing hubs in India and China, combined with process optimization in impurity management, crystallization control, and GMP compliance, strengthens global supply reliability. These capabilities support competitive API exports to North America, Europe, and Latin America. Long-term demand remains favorable due to sustained focus on medication accessibility and chronic neurological disease management. Intense generic price competition, strict pharmacovigilance requirements, and periodic regulatory audits continue to influence margins and capacity deployment decisions.
How Is the Oxcarbazepine API Market Segmented by Product Type, Application, End User and Region?
The oxcarbazepine API market is segmented by product type, application, and end user. By product type, the market is categorized into pharmacopoeial grade API, technical/industrial grade API, and custom or contract-manufactured API. Based on application, the market is divided into epilepsy and seizure disorders, bipolar disorder, and neuropathic pain & other indications. End-user categories include generic drug manufacturers, branded formulation companies, and CDMOs & API exporters. Regionally, the market is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Product Type, Pharmacopoeial Grade API Accounts for a Dominant Market Share
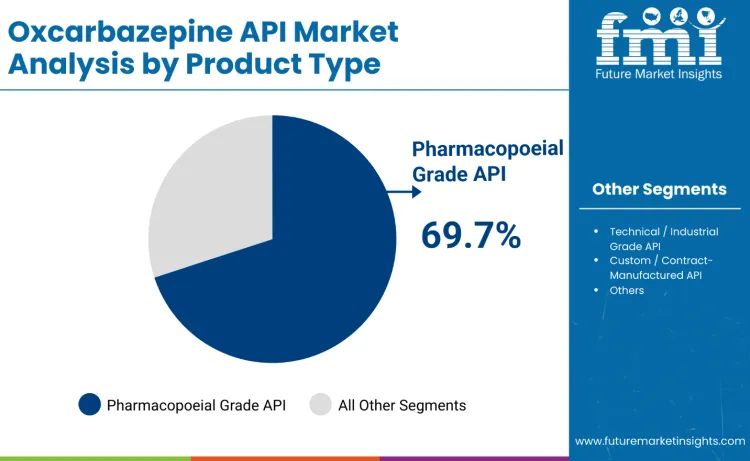
Pharmacopoeial grade API (USP/BP/Ph. Eur.) dominates revenue in 2025, holding about 69.7% share, driven by high-purity production under validated cGMP conditions, strict impurity control, stability validation, and DMF-ready regulatory compliance. Its leadership reflects strong adoption in regulated dosage manufacturing for seizure therapies requiring proven bioequivalence and long-term safety. Technical or industrial grade API accounts for 11.6%, serving cost-focused regional and pilot applications, while custom or contract-manufactured API contributes 18.7%, supported by rising outsourcing and tailored impurity requirements from multinational formulators.
By Application, Epilepsy & Seizure Disorders Represent the Largest Market Share
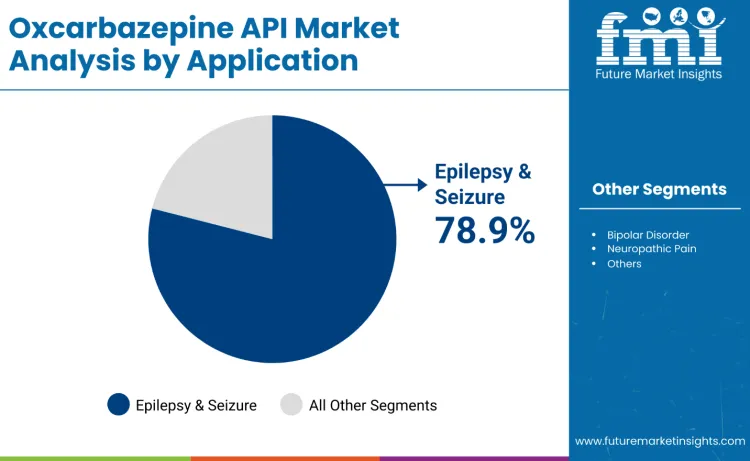
Epilepsy and seizure disorders dominate oxcarbazepine API demand in 2025, accounting for 78.9% share, reflecting its established role as a primary and adjunctive therapy for partial-onset seizures across adult and pediatric patients. Guideline endorsement, favorable tolerability, and sustained formulary inclusion within public neurology programs support consistent procurement cycles globally. Bipolar disorder applications contribute 14.6%, driven by psychiatric use in cost-efficient mood stabilization therapies. Neuropathic pain and other indications represent 6.5%, supported by off-label use. Leadership in epilepsy is reinforced by chronic treatment needs, pediatric safety preference, and government-backed essential-drug initiatives.
By End User, Generic Drug Manufacturers Hold the Largest Market Share
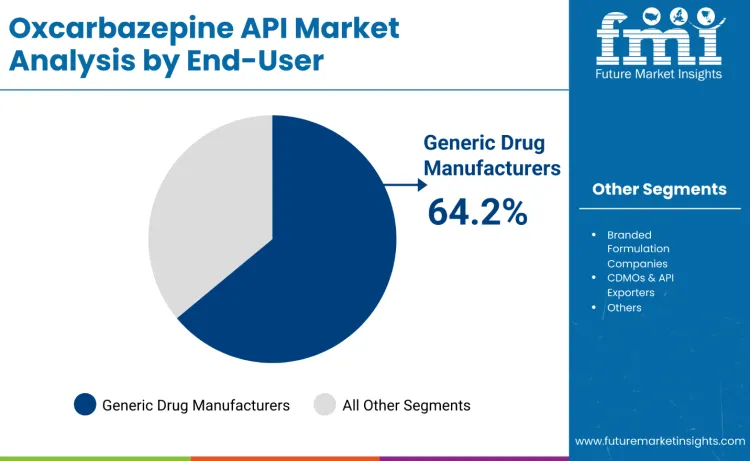
Generic drug manufacturers lead end-user demand in 2025 with a 64.2% share, driven by large-scale production of cost-effective oxcarbazepine formulations and steady procurement through tenders, reimbursement schemes, and hospital networks. Their dominance reflects DMF-based market entry, price-competitive sourcing, and scale advantages across established hubs in India and China. CDMOs and API exporters account for 23.9%, supported by contract manufacturing and export-oriented supply chains, while branded formulators hold 11.9% through legacy positioning. Leadership of generics is reinforced by chronic therapy volumes, public financing participation, mature infrastructure, and long-term institutional supply agreements.
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Oxcarbazepine API Market?
The oxcarbazepine API market is driven by rising neurological disease prevalence, expanding generic drug usage, and long-term treatment dependence. With 65-70 million people living with epilepsy globally, demand is reinforced by wider treatment access through national neurology programs and WHO essential medicine inclusion. Growing prescription volumes for oxcarbazepine in bipolar disorder management further strengthens baseline demand, as clinicians favor established mood stabilizers with predictable safety profiles. Expansion of generic manufacturing and DMF filings in India and China improves API availability, supporting global supply through cost-efficient, pharmacopoeia-compliant production.
What Are the Major Restraints Limiting the Oxcarbazepine API Market?
Market restraints include intense pricing pressure across generic antiepileptic APIs, driven by hospital tenders, public reimbursement systems, and competitive procurement practices that compress margins. Regulatory complexity and stringent GMP audits require continuous investment in documentation, stability testing, and impurity management, creating barriers for smaller manufacturers and prolonging site approvals. Supply chain vulnerabilities related to solvent availability, energy price volatility, and logistics disruptions can affect production continuity, particularly for export-oriented suppliers managing multi-region contracts and just-in-time delivery expectations across regulated pharmaceutical markets.
What Emerging Trends Are Shaping the Future of the Oxcarbazepine API Market?
Key trends include accelerated quality enhancement and regulatory optimization within major API hubs in India and China, where manufacturers are strengthening data-integrity systems and upgrading process controls to meet evolving FDA and EMA standards. Adoption of continuous manufacturing, precision crystallization, and solvent-recycling technologies is improving yield consistency, impurity control, and sustainability metrics. Competitive dynamics may shift with the emergence of alternative antiepileptic molecules, modified-release neurology formulations, and policy-driven domestic manufacturing incentives, requiring API producers to diversify supply chains and maintain rigorous regulatory alignment to sustain global competitiveness.
How Are Leading Countries Driving Growth in the Oxcarbazepine API Market?
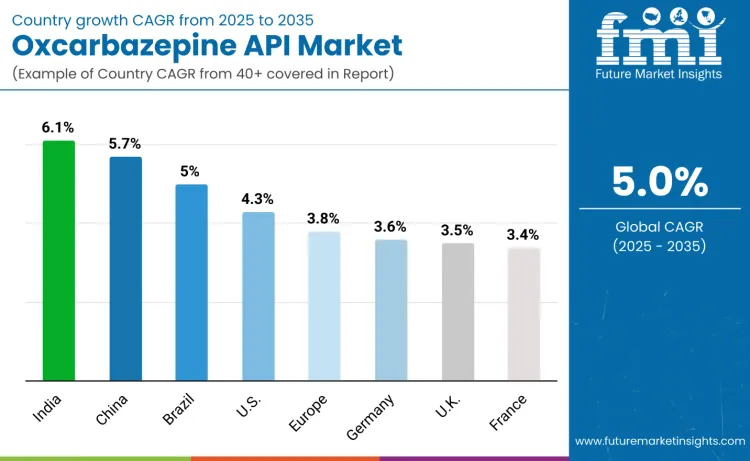
| Country | CAGR (2025 to 2035) |
|---|---|
| USA | 4.3% |
| Brazil | 5.0% |
| China | 5.7% |
| India | 6.1% |
| Europe | 3.8% |
| Germany | 3.6% |
| France | 3.4% |
| UK | 3.5% |
The oxcarbazepine API market advances globally, with India emerging as the fastest-growing region at a 6.1% CAGR, supported by large-scale manufacturing leadership, strong DMF presence, and expanding exports to regulated markets. China follows at 5.7%, driven by vertical integration and compliance upgrades. Brazil records 5.0% growth as public neurology programs expand generic access. The United States grows steadily at 4.3% through broad prescriber adoption of bioequivalent generics. Europe advances at 3.8%, with Germany (3.6%), the UK (3.5%), and France (3.4%) representing mature, reimbursement-driven markets prioritizing GMP-certified supply.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Is Neurology Treatment Infrastructure Accelerating Market Growth in China?
China shows strong momentum in the oxcarbazepine API landscape with a CAGR of 5.7% through 2035, supported by expanding neurology care infrastructure and large-scale pharmaceutical manufacturing capacity. National policies promoting epilepsy care access, generic substitution, and hospital formulary expansion drive sustained API demand. Growth concentrates in Zhejiang, Jiangsu, Shandong, and Guangdong, where GMP-certified manufacturers and CDMOs supply domestic and export markets. Wider insurance coverage, improved neurologist availability, and alignment with international pharmacopoeial standards strengthen long-term epilepsy management and reinforce consistent API consumption.
Why Are Neurology Treatment & Pharmaceutical Capacity Programs Enhancing Market Adoption in India?
India records the fastest growth at a 6.1% CAGR through 2035, driven by cost-competitive generic neurology drug production and expanding public healthcare coverage. National initiatives such as Ayushman Bharat and essential-medicine programs increase epilepsy and psychiatric treatment reach, while domestic API manufacturers scale GMP-compliant capacity. Pharmaceutical hubs including Hyderabad, Ahmedabad, and Visakhapatnam anchor production and exports. Strong DMF filings, expanding CDMO participation, and rising neurology prescriptions across public and private systems reinforce sustained oxcarbazepine API demand and long-term manufacturing planning.
How Does Regulatory and Quality-Driven Pharmaceutical Excellence Sustain Market Presence in Germany?
Germany maintains a stable oxcarbazepine API landscape with moderate growth of 3.6% through 2035, underpinned by rigorous regulatory standards and structured neurology care systems. University hospitals and specialty clinics emphasize evidence-based epilepsy management, supporting consistent demand for EU-GMP- and Ph. Eur.-compliant APIs. Strong reimbursement frameworks, disciplined formularies, and audit-focused procurement favor suppliers with proven quality and traceability. While volume growth is measured, Germany sustains premium API requirements and predictable offtake patterns aligned with long-term therapy continuity.
Why Is Expanding Public Neurology Access Fueling Market Demand in Brazil?
Brazil leads Latin America with a 5.0% CAGR through 2035, driven by expanding access to antiepileptic therapies under the Unified Health System. Public neurology programs, generic drug substitution, and essential-medicine procurement strengthen oxcarbazepine adoption across major urban centers. ANVISA-regulated formulation infrastructure and public tenders support stable API demand, while partnerships between local formulators and global suppliers ensure supply security. Government-backed epilepsy awareness and treatment initiatives further increase diagnosis rates and long-term therapy adherence across diverse population segments.
How Do Established Neurology Treatment Systems Strengthen Market Performance in United States?
The USA market demonstrates steady growth at a 4.3% CAGR through 2035, supported by mature neurology treatment networks and consistent epilepsy and bipolar disorder prevalence. Broad insurance coverage and strong preference for bioequivalent generics sustain oxcarbazepine utilization across hospitals, clinics, and pharmacy networks. FDA regulatory rigor drives demand for high-quality, fully compliant APIs, favoring suppliers with strong DMF maintenance and cGMP adherence. Established prescribing patterns and nationwide distribution infrastructure ensure predictable, long-term API offtake.
How Is Neurology Care Expansion Supporting Market Growth in United Kingdom?
The United Kingdom records stable growth of 3.5% through 2035, supported by strengthened NHS neurology services and standardized epilepsy care pathways. Clinicians prioritize clinically validated, cost-effective antiepileptic therapies, sustaining oxcarbazepine formulary inclusion across primary and specialist care. NHS procurement systems and reimbursement mechanisms ensure broad patient access while supporting competitive sourcing of compliant APIs. Academic-clinical collaboration and improved diagnostic pathways enhance therapy continuity, reinforcing steady API demand within a highly regulated and quality-focused healthcare environment.
How Are Neurology Care Investments Advancing Market Expansion Across Europe?
The oxcarbazepine API market in Europe is projected to grow from USD 45.9 million in 2025 to USD 67.6 million by 2035, registering a CAGR of 3.9%. Germany leads with a 24.2% share in 2025, easing to 23.8% by 2035, supported by advanced neurology care, centralized hospital procurement, and stringent GMP and regulatory compliance across major clinical hubs.
France follows with a 20.8% share in 2025, rising to 21.1% by 2035, driven by strong epileptology programs. The UK grows from 18.6% to 18.9% under NHS support, while Italy holds 14.1% throughout. Spain advances from 9.7% to 9.9%. The Netherlands remains at 6.3%, and Rest of Europe edges from 6.3% to 6.4% by 2035.
High-Precision Quality Standards Drive Pharmaceutical Requirements in Japan
The oxcarbazepine API market in Japan reflects a mature, precision-driven pharmaceutical environment defined by strict adherence to JP and Ph. Eur.-aligned specifications and advanced impurity-control frameworks. Neurology treatment systems across university hospitals, specialty centers, and long-term care facilities emphasize therapeutic reliability and long-term safety, sustaining demand for premium-grade APIs that meet rigorous performance expectations. Strong PMDA oversight reinforces quality-centric procurement and GMP-compliant manufacturing standards.
Japan’s market strength is further supported by advanced post-marketing surveillance and disciplined prescribing practices across Tokyo, Osaka, and other metropolitan hubs. Clinicians prioritize formulations offering controlled titration, favorable tolerability, and consistent seizure-management outcomes aligned with evidence-based epilepsy and psychiatric care protocols. These quality-driven clinical preferences ensure stable API demand, while validated crystallization consistency and lifecycle impurity documentation remain central to supplier selection and long-term supply partnerships.
International Suppliers Strengthen Neurology Care Support in South Korea
The oxcarbazepine API market in South Korea features a growing presence of international suppliers operating within a highly regulated pharmaceutical environment. Demand is supported by expanding epilepsy and mood-disorder management programs across Seoul, Busan, Incheon, and Daegu, where tertiary hospitals and specialty neurology centers prioritize clinically validated antiepileptic therapies aligned with national treatment guidelines and integrated electronic prescribing systems.
Major generic manufacturers and multinational API producers strengthen positioning through GMP-compliant supply, validated impurity-control frameworks, and technical dossiers meeting MFDS requirements. Hospital procurement networks and pharmacy chains emphasize documentation quality and secure supply partnerships. Increasing collaboration between global pharmaceutical firms and Korean formulators creates hybrid sourcing models that combine international manufacturing standards with strong local distribution, pharmacovigilance support, and clinician-focused medical education, reinforcing sustained oxcarbazepine API adoption.
How Competitive Is the Global Oxcarbazepine API Market and What Defines Its Structure?
The global oxcarbazepine API landscape shows moderate concentration, with a limited group of established producers holding a dominant supply position through extensive DMF coverage, vertically integrated manufacturing, and mature regulatory compliance frameworks. Competition centers on consistent quality delivery, regulatory readiness, impurity-control depth, and supply reliability rather than aggressive pricing, reflecting the expectations of regulated-market formulators and hospital procurement bodies. Manufacturers based in India and China retain strategic leadership due to strong export orientation and sustained adherence to global pharmacopoeial and documentation standards.
Leading suppliers such as Sun Pharmaceutical Industries, Aurobindo Pharma, and Hetero Drugs maintain advantage through multi-site production networks, strong regulatory track records across major regions, and well-developed quality-management systems supporting long-term neurology portfolios. Ongoing investment in impurity profiling, crystallization optimization, solvent recovery, and lifecycle stability programs enables these companies to defend positions while expanding presence across emerging therapeutic markets.
Challenger firms including Jubilant Pharmova, MSN Laboratories, and Granules India compete through efficient production economics, robust dossier support, and growing penetration in semi-regulated regions. Product-focused players and global innovators strengthen the landscape through compliance excellence, strategic partnerships, dependable logistics, and customer support spanning stability data, impurity lifecycle management, and assured long-term supply continuity.
Key Players in the Oxcarbazepine API Market
- Sun Pharmaceutical Industries
- Jubilant Pharmova (APIs)
- Aurobindo Pharma
- MSN Laboratories
- Zhejiang Huahai Pharmaceutical
- Hetero Drugs
- Teva API
- Granules India
- Alembic Pharmaceuticals
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 421.5 million |
| Product Type | Pharmacopoeial Grade API (USP/BP/Ph. Eur.), Technical / Industrial Grade API, Custom / Contract-Manufactured API |
| Application | Epilepsy & Seizure Disorders, Bipolar Disorder, Neuropathic Pain & Other Indications |
| End User | Generic Drug Manufacturers, Branded Formulation Companies, CDMOs & API Exporters |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | Sun Pharmaceutical Industries, Jubilant Pharmova (APIs), Aurobindo Pharma, MSN Laboratories, Zhejiang Huahai Pharmaceutical, Hetero Drugs |
| Additional Attributes | API sales by pharmacopoeial grade and epilepsy-mood disorder applications, regional procurement across Asia Pacific, Europe, and North America, competitive landscape of DMF-active manufacturers, regulatory and impurity benchmarks, GMP integration, and innovations in crystallization, solvent recovery, and stability-validated API grades. |
Oxcarbazepine API Market by Segments
Product Type:
- Pharmacopoeial Grade API (USP/BP/Ph. Eur.)
- Technical / Industrial Grade API
- Custom / Contract-Manufactured API
Application:
- Epilepsy & Seizure Disorders
- Bipolar Disorder
- Neuropathic Pain & Other Indications
End User:
- Generic Drug Manufacturers
- Branded Formulation Companies
- CDMOs & API Exporters
Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the oxcarbazepine API market in 2025?
The global oxcarbazepine API market is valued at USD 421.5 million in 2025.
What will be the size of the oxcarbazepine API market in 2035?
The oxcarbazepine API market is projected to reach USD 689.3 million by 2035.
How much will the oxcarbazepine API market grow between 2025 and 2035?
The oxcarbazepine API market is expected to grow at a 5.0% CAGR between 2025 and 2035.
Which product type leads the oxcarbazepine API market in 2025?
Pharmacopoeial grade oxcarbazepine API leads the market with an estimated 69.7% share in 2025.
Which end-user segment contributes the largest share in the oxcarbazepine API market?
Generic drug manufacturers account for the largest share, contributing 64.2% of total demand in the oxcarbazepine API market.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assessment
- Market Trade Assessment
- Products Market Assessment
- Investment Feasibility Matrix
- Value Chain Analysis
- Participants and their Roles
- Profit Margin Analysis
- Operating Margins By Region
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Product Launches & Development
- Technological Innovation
- Market Dynamics
- Regional Level Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size USD Million Analysis, 2020 to 2024
- Current and Future Market Size USD Million Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Product Type, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Product Type, 2025 to 2035
- Pharmacopoeial Grade API (USP/BP/Ph. Eur.)
- Technical / Industrial Grade API
- Custom / Contract-Manufactured API
- Y-o-Y Growth Trend Analysis By Product Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Application, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Application, 2025 to 2035
- Epilepsy & Seizure Disorders
- Bipolar Disorder
- Neuropathic Pain & Other Indications
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By End User, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By End User, 2025 to 2035
- Generic Drug Manufacturers
- Branded Formulation Companies
- CDMOs & API Exporters
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size USD Million Analysis By Region, 2020 to 2024
- Current Market Size USD Million Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Sun Pharmaceutical Industries
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Jubilant Pharmova (APIs)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Aurobindo Pharma
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- MSN Laboratories
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Zhejiang Huahai Pharmaceutical
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Hetero Drugs
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Teva API
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Granules India
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Alembic Pharmaceuticals
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Sun Pharmaceutical Industries
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million), by Product Type, 2020 to 2035
- Table 2: Global Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 3: Global Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 4: Global Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 5: Global Market Size (USD Million), by Application, 2020 to 2035
- Table 6: Global Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 7: Global Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 8: Global Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 9: Global Market Size (USD Million), by End User, 2020 to 2035
- Table 10: Global Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 11: Global Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 12: Global Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 13: North America Market Size (USD Million), by Product Type, 2020 to 2035
- Table 14: North America Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 15: North America Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 16: North America Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 17: North America Market Size (USD Million), by Application, 2020 to 2035
- Table 18: North America Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 19: North America Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 20: North America Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 21: North America Market Size (USD Million), by End User, 2020 to 2035
- Table 22: North America Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 23: North America Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 24: North America Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 25: USA Market Size (USD Million), by Product Type, 2020 to 2035
- Table 26: USA Market Size (USD Million), by Application, 2020 to 2035
- Table 27: USA Market Size (USD Million), by End User, 2020 to 2035
- Table 28: Canada Market Size (USD Million), by Product Type, 2020 to 2035
- Table 29: Canada Market Size (USD Million), by Application, 2020 to 2035
- Table 30: Canada Market Size (USD Million), by End User, 2020 to 2035
- Table 31: Mexico Market Size (USD Million), by Product Type, 2020 to 2035
- Table 32: Mexico Market Size (USD Million), by Application, 2020 to 2035
- Table 33: Mexico Market Size (USD Million), by End User, 2020 to 2035
- Table 34: Latin America Market Size (USD Million), by Product Type, 2020 to 2035
- Table 35: Latin America Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 36: Latin America Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 37: Latin America Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 38: Latin America Market Size (USD Million), by Application, 2020 to 2035
- Table 39: Latin America Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 40: Latin America Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 41: Latin America Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 42: Latin America Market Size (USD Million), by End User, 2020 to 2035
- Table 43: Latin America Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 44: Latin America Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 45: Latin America Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 46: Brazil Market Size (USD Million), by Product Type, 2020 to 2035
- Table 47: Brazil Market Size (USD Million), by Application, 2020 to 2035
- Table 48: Brazil Market Size (USD Million), by End User, 2020 to 2035
- Table 49: Argentina Market Size (USD Million), by Product Type, 2020 to 2035
- Table 50: Argentina Market Size (USD Million), by Application, 2020 to 2035
- Table 51: Argentina Market Size (USD Million), by End User, 2020 to 2035
- Table 52: Rest of Latin America Market Size (USD Million), by Product Type, 2020 to 2035
- Table 53: Rest of Latin America Market Size (USD Million), by Application, 2020 to 2035
- Table 54: Rest of Latin America Market Size (USD Million), by End User, 2020 to 2035
- Table 55: East Asia Market Size (USD Million), by Product Type, 2020 to 2035
- Table 56: East Asia Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 57: East Asia Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 58: East Asia Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 59: East Asia Market Size (USD Million), by Application, 2020 to 2035
- Table 60: East Asia Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 61: East Asia Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 62: East Asia Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 63: East Asia Market Size (USD Million), by End User, 2020 to 2035
- Table 64: East Asia Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 65: East Asia Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 66: East Asia Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 67: China Market Size (USD Million), by Product Type, 2020 to 2035
- Table 68: China Market Size (USD Million), by Application, 2020 to 2035
- Table 69: China Market Size (USD Million), by End User, 2020 to 2035
- Table 70: Japan Market Size (USD Million), by Product Type, 2020 to 2035
- Table 71: Japan Market Size (USD Million), by Application, 2020 to 2035
- Table 72: Japan Market Size (USD Million), by End User, 2020 to 2035
- Table 73: South Korea Market Size (USD Million), by Product Type, 2020 to 2035
- Table 74: South Korea Market Size (USD Million), by Application, 2020 to 2035
- Table 75: South Korea Market Size (USD Million), by End User, 2020 to 2035
- Table 76: South Asia Market Size (USD Million), by Product Type, 2020 to 2035
- Table 77: South Asia Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 78: South Asia Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 79: South Asia Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 80: South Asia Market Size (USD Million), by Application, 2020 to 2035
- Table 81: South Asia Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 82: South Asia Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 83: South Asia Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 84: South Asia Market Size (USD Million), by End User, 2020 to 2035
- Table 85: South Asia Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 86: South Asia Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 87: South Asia Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 88: India Market Size (USD Million), by Product Type, 2020 to 2035
- Table 89: India Market Size (USD Million), by Application, 2020 to 2035
- Table 90: India Market Size (USD Million), by End User, 2020 to 2035
- Table 91: ASEAN Countries Market Size (USD Million), by Product Type, 2020 to 2035
- Table 92: ASEAN Countries Market Size (USD Million), by Application, 2020 to 2035
- Table 93: ASEAN Countries Market Size (USD Million), by End User, 2020 to 2035
- Table 94: Australia & New Zealand Market Size (USD Million), by Product Type, 2020 to 2035
- Table 95: Australia & New Zealand Market Size (USD Million), by Application, 2020 to 2035
- Table 96: Australia & New Zealand Market Size (USD Million), by End User, 2020 to 2035
- Table 97: Rest of South Asia Market Size (USD Million), by Product Type, 2020 to 2035
- Table 98: Rest of South Asia Market Size (USD Million), by Application, 2020 to 2035
- Table 99: Rest of South Asia Market Size (USD Million), by End User, 2020 to 2035
- Table 100: Western Europe Market Size (USD Million), by Product Type, 2020 to 2035
- Table 101: Western Europe Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 102: Western Europe Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 103: Western Europe Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 104: Western Europe Market Size (USD Million), by Application, 2020 to 2035
- Table 105: Western Europe Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 106: Western Europe Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 107: Western Europe Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 108: Western Europe Market Size (USD Million), by End User, 2020 to 2035
- Table 109: Western Europe Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 110: Western Europe Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 111: Western Europe Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 112: Germany Market Size (USD Million), by Product Type, 2020 to 2035
- Table 113: Germany Market Size (USD Million), by Application, 2020 to 2035
- Table 114: Germany Market Size (USD Million), by End User, 2020 to 2035
- Table 115: UK Market Size (USD Million), by Product Type, 2020 to 2035
- Table 116: UK Market Size (USD Million), by Application, 2020 to 2035
- Table 117: UK Market Size (USD Million), by End User, 2020 to 2035
- Table 118: France Market Size (USD Million), by Product Type, 2020 to 2035
- Table 119: France Market Size (USD Million), by Application, 2020 to 2035
- Table 120: France Market Size (USD Million), by End User, 2020 to 2035
- Table 121: Italy Market Size (USD Million), by Product Type, 2020 to 2035
- Table 122: Italy Market Size (USD Million), by Application, 2020 to 2035
- Table 123: Italy Market Size (USD Million), by End User, 2020 to 2035
- Table 124: Spain Market Size (USD Million), by Product Type, 2020 to 2035
- Table 125: Spain Market Size (USD Million), by Application, 2020 to 2035
- Table 126: Spain Market Size (USD Million), by End User, 2020 to 2035
- Table 127: BENELUX Market Size (USD Million), by Product Type, 2020 to 2035
- Table 128: BENELUX Market Size (USD Million), by Application, 2020 to 2035
- Table 129: BENELUX Market Size (USD Million), by End User, 2020 to 2035
- Table 130: Nordic Countries Market Size (USD Million), by Product Type, 2020 to 2035
- Table 131: Nordic Countries Market Size (USD Million), by Application, 2020 to 2035
- Table 132: Nordic Countries Market Size (USD Million), by End User, 2020 to 2035
- Table 133: Rest of Western Europe Market Size (USD Million), by Product Type, 2020 to 2035
- Table 134: Rest of Western Europe Market Size (USD Million), by Application, 2020 to 2035
- Table 135: Rest of Western Europe Market Size (USD Million), by End User, 2020 to 2035
- Table 136: Eastern Europe Market Size (USD Million), by Product Type, 2020 to 2035
- Table 137: Eastern Europe Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 138: Eastern Europe Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 139: Eastern Europe Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 140: Eastern Europe Market Size (USD Million), by Application, 2020 to 2035
- Table 141: Eastern Europe Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 142: Eastern Europe Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 143: Eastern Europe Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 144: Eastern Europe Market Size (USD Million), by End User, 2020 to 2035
- Table 145: Eastern Europe Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 146: Eastern Europe Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 147: Eastern Europe Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 148: Russia Market Size (USD Million), by Product Type, 2020 to 2035
- Table 149: Russia Market Size (USD Million), by Application, 2020 to 2035
- Table 150: Russia Market Size (USD Million), by End User, 2020 to 2035
- Table 151: Hungary Market Size (USD Million), by Product Type, 2020 to 2035
- Table 152: Hungary Market Size (USD Million), by Application, 2020 to 2035
- Table 153: Hungary Market Size (USD Million), by End User, 2020 to 2035
- Table 154: Poland Market Size (USD Million), by Product Type, 2020 to 2035
- Table 155: Poland Market Size (USD Million), by Application, 2020 to 2035
- Table 156: Poland Market Size (USD Million), by End User, 2020 to 2035
- Table 157: Rest of Eastern Europe Market Size (USD Million), by Product Type, 2020 to 2035
- Table 158: Rest of Eastern Europe Market Size (USD Million), by Application, 2020 to 2035
- Table 159: Rest of Eastern Europe Market Size (USD Million), by End User, 2020 to 2035
- Table 160: Middle East & Africa Market Size (USD Million), by Product Type, 2020 to 2035
- Table 161: Middle East & Africa Market Size (USD Million), by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Table 162: Middle East & Africa Market Size (USD Million), by Technical / Industrial Grade API, 2020 to 2035
- Table 163: Middle East & Africa Market Size (USD Million), by Custom / Contract-Manufactured API, 2020 to 2035
- Table 164: Middle East & Africa Market Size (USD Million), by Application, 2020 to 2035
- Table 165: Middle East & Africa Market Size (USD Million), by Epilepsy & Seizure Disorders, 2020 to 2035
- Table 166: Middle East & Africa Market Size (USD Million), by Bipolar Disorder, 2020 to 2035
- Table 167: Middle East & Africa Market Size (USD Million), by Neuropathic Pain & Other Indications, 2020 to 2035
- Table 168: Middle East & Africa Market Size (USD Million), by End User, 2020 to 2035
- Table 169: Middle East & Africa Market Size (USD Million), by Generic Drug Manufacturers, 2020 to 2035
- Table 170: Middle East & Africa Market Size (USD Million), by Branded Formulation Companies, 2020 to 2035
- Table 171: Middle East & Africa Market Size (USD Million), by CDMOs & API Exporters, 2020 to 2035
- Table 172: Saudi Arabia Market Size (USD Million), by Product Type, 2020 to 2035
- Table 173: Saudi Arabia Market Size (USD Million), by Application, 2020 to 2035
- Table 174: Saudi Arabia Market Size (USD Million), by End User, 2020 to 2035
- Table 175: Other GCC Countries Market Size (USD Million), by Product Type, 2020 to 2035
- Table 176: Other GCC Countries Market Size (USD Million), by Application, 2020 to 2035
- Table 177: Other GCC Countries Market Size (USD Million), by End User, 2020 to 2035
- Table 178: Türkiye Market Size (USD Million), by Product Type, 2020 to 2035
- Table 179: Türkiye Market Size (USD Million), by Application, 2020 to 2035
- Table 180: Türkiye Market Size (USD Million), by End User, 2020 to 2035
- Table 181: South Africa Market Size (USD Million), by Product Type, 2020 to 2035
- Table 182: South Africa Market Size (USD Million), by Application, 2020 to 2035
- Table 183: South Africa Market Size (USD Million), by End User, 2020 to 2035
- Table 184: Rest of MEA Market Size (USD Million), by Product Type, 2020 to 2035
- Table 185: Rest of MEA Market Size (USD Million), by Application, 2020 to 2035
- Table 186: Rest of MEA Market Size (USD Million), by End User, 2020 to 2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 2: Global Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 3: Global Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 4: Global Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 5: Global Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 6: Global Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 7: Global Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 8: Global Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 9: Global Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 10: Global Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 11: Global Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 12: Global Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 13: North America Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 14: North America Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 15: North America Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 16: North America Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 17: North America Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 18: North America Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 19: North America Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 20: North America Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 21: North America Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 22: North America Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 23: North America Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 24: North America Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 25: USA Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 26: USA Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 27: USA Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 28: USA Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 29: USA Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 30: USA Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 31: Canada Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 32: Canada Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 33: Canada Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 34: Canada Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 35: Canada Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 36: Canada Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 37: Mexico Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 38: Mexico Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 39: Mexico Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 40: Mexico Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 41: Mexico Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 42: Mexico Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 43: Latin America Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 44: Latin America Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 45: Latin America Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 46: Latin America Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 47: Latin America Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 48: Latin America Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 49: Latin America Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 50: Latin America Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 51: Latin America Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 52: Latin America Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 53: Latin America Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 54: Latin America Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 55: Brazil Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 56: Brazil Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 57: Brazil Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 58: Brazil Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 59: Brazil Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 60: Brazil Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 61: Argentina Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 62: Argentina Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 63: Argentina Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 64: Argentina Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 65: Argentina Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 66: Argentina Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 67: Rest of Latin America Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 68: Rest of Latin America Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 69: Rest of Latin America Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 70: Rest of Latin America Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 71: Rest of Latin America Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 72: Rest of Latin America Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 73: East Asia Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 74: East Asia Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 75: East Asia Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 76: East Asia Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 77: East Asia Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 78: East Asia Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 79: East Asia Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 80: East Asia Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 81: East Asia Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 82: East Asia Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 83: East Asia Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 84: East Asia Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 85: China Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 86: China Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 87: China Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 88: China Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 89: China Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 90: China Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 91: Japan Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 92: Japan Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 93: Japan Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 94: Japan Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 95: Japan Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 96: Japan Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 97: South Korea Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 98: South Korea Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 99: South Korea Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 100: South Korea Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 101: South Korea Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 102: South Korea Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 103: South Asia Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 104: South Asia Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 105: South Asia Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 106: South Asia Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 107: South Asia Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 108: South Asia Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 109: South Asia Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 110: South Asia Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 111: South Asia Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 112: South Asia Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 113: South Asia Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 114: South Asia Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 115: India Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 116: India Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 117: India Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 118: India Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 119: India Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 120: India Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 121: ASEAN Countries Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 122: ASEAN Countries Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 123: ASEAN Countries Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 124: ASEAN Countries Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 125: ASEAN Countries Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 126: ASEAN Countries Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 127: Australia & New Zealand Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 128: Australia & New Zealand Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 129: Australia & New Zealand Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 130: Australia & New Zealand Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 131: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 132: Australia & New Zealand Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 133: Rest of South Asia Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 134: Rest of South Asia Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 135: Rest of South Asia Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 136: Rest of South Asia Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 137: Rest of South Asia Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 138: Rest of South Asia Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 139: Western Europe Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 140: Western Europe Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 141: Western Europe Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 142: Western Europe Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 143: Western Europe Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 144: Western Europe Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 145: Western Europe Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 146: Western Europe Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 147: Western Europe Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 148: Western Europe Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 149: Western Europe Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 150: Western Europe Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 151: Germany Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 152: Germany Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 153: Germany Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 154: Germany Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 155: Germany Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 156: Germany Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 157: UK Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 158: UK Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 159: UK Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 160: UK Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 161: UK Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 162: UK Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 163: France Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 164: France Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 165: France Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 166: France Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 167: France Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 168: France Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 169: Italy Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 170: Italy Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 171: Italy Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 172: Italy Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 173: Italy Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 174: Italy Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 175: Spain Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 176: Spain Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 177: Spain Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 178: Spain Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 179: Spain Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 180: Spain Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 181: BENELUX Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 182: BENELUX Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 183: BENELUX Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 184: BENELUX Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 185: BENELUX Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 186: BENELUX Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 187: Nordic Countries Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 188: Nordic Countries Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 189: Nordic Countries Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 190: Nordic Countries Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 191: Nordic Countries Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 192: Nordic Countries Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 193: Rest of Western Europe Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 194: Rest of Western Europe Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 195: Rest of Western Europe Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 196: Rest of Western Europe Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 197: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 198: Rest of Western Europe Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 199: Eastern Europe Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 200: Eastern Europe Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 201: Eastern Europe Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 202: Eastern Europe Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 203: Eastern Europe Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 204: Eastern Europe Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 205: Eastern Europe Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 206: Eastern Europe Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 207: Eastern Europe Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 208: Eastern Europe Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 209: Eastern Europe Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 210: Eastern Europe Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 211: Russia Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 212: Russia Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 213: Russia Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 214: Russia Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 215: Russia Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 216: Russia Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 217: Hungary Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 218: Hungary Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 219: Hungary Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 220: Hungary Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 221: Hungary Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 222: Hungary Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 223: Poland Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 224: Poland Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 225: Poland Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 226: Poland Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 227: Poland Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 228: Poland Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 229: Rest of Eastern Europe Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 230: Rest of Eastern Europe Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 231: Rest of Eastern Europe Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 232: Rest of Eastern Europe Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 233: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 234: Rest of Eastern Europe Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 235: Middle East & Africa Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 236: Middle East & Africa Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 237: Middle East & Africa Market (USD Million) Forecast, by Technical / Industrial Grade API, 2020 to 2035
- Figure 238: Middle East & Africa Market (USD Million) Forecast, by Custom / Contract-Manufactured API, 2020 to 2035
- Figure 239: Middle East & Africa Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 240: Middle East & Africa Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 241: Middle East & Africa Market (USD Million) Forecast, by Bipolar Disorder, 2020 to 2035
- Figure 242: Middle East & Africa Market (USD Million) Forecast, by Neuropathic Pain & Other Indications, 2020 to 2035
- Figure 243: Middle East & Africa Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 244: Middle East & Africa Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 245: Middle East & Africa Market (USD Million) Forecast, by Branded Formulation Companies, 2020 to 2035
- Figure 246: Middle East & Africa Market (USD Million) Forecast, by CDMOs & API Exporters, 2020 to 2035
- Figure 247: Saudi Arabia Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 248: Saudi Arabia Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 249: Saudi Arabia Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 250: Saudi Arabia Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 251: Saudi Arabia Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 252: Saudi Arabia Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 253: Other GCC Countries Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 254: Other GCC Countries Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 255: Other GCC Countries Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 256: Other GCC Countries Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 257: Other GCC Countries Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 258: Other GCC Countries Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 259: Türkiye Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 260: Türkiye Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 261: Türkiye Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 262: Türkiye Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 263: Türkiye Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 264: Türkiye Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 265: South Africa Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 266: South Africa Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 267: South Africa Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 268: South Africa Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 269: South Africa Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 270: South Africa Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 271: Rest of MEA Market (USD Million) Forecast, by Product Type, 2020 to 2035
- Figure 272: Rest of MEA Market (USD Million) Forecast, by Pharmacopoeial Grade API (USP/BP/Ph. Eur.), 2020 to 2035
- Figure 273: Rest of MEA Market (USD Million) Forecast, by Application, 2020 to 2035
- Figure 274: Rest of MEA Market (USD Million) Forecast, by Epilepsy & Seizure Disorders, 2020 to 2035
- Figure 275: Rest of MEA Market (USD Million) Forecast, by End User, 2020 to 2035
- Figure 276: Rest of MEA Market (USD Million) Forecast, by Generic Drug Manufacturers, 2020 to 2035
- Figure 277: Sun Pharmaceutical Industries - Company Snapshot
- Figure 278: Sun Pharmaceutical Industries - SWOT Analysis
- Figure 279: Jubilant Pharmova (APIs) - Company Snapshot
- Figure 280: Jubilant Pharmova (APIs) - SWOT Analysis
- Figure 281: Aurobindo Pharma - Company Snapshot
- Figure 282: Aurobindo Pharma - SWOT Analysis
- Figure 283: MSN Laboratories - Company Snapshot
- Figure 284: MSN Laboratories - SWOT Analysis
- Figure 285: Zhejiang Huahai Pharmaceutical - Company Snapshot
- Figure 286: Zhejiang Huahai Pharmaceutical - SWOT Analysis
- Figure 287: Hetero Drugs - Company Snapshot
- Figure 288: Hetero Drugs - SWOT Analysis
- Figure 289: Teva API - Company Snapshot
- Figure 290: Teva API - SWOT Analysis
- Figure 291: Granules India - Company Snapshot
- Figure 292: Granules India - SWOT Analysis
- Figure 293: Alembic Pharmaceuticals - Company Snapshot
- Figure 294: Alembic Pharmaceuticals - SWOT Analysis

