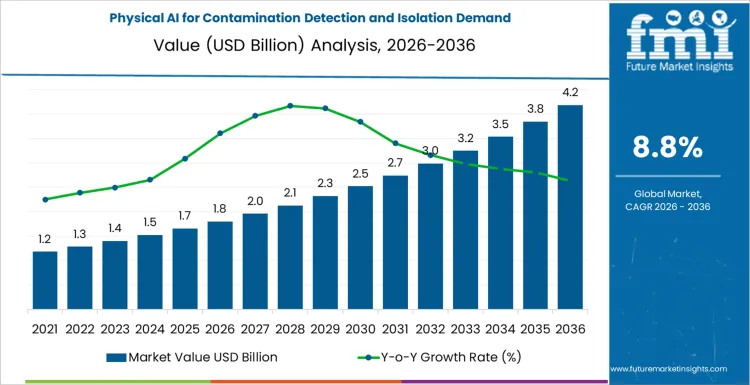
The demand for physical AI systems for contamination detection and isolation is estimated to reach USD 1.8 billion in 2026 and expand to USD 4.2 billion by 2036, growing at a CAGR of 8.8%. Enterprise spending on physical AI for contamination detection and isolation is allocated across several critical areas to ensure optimal performance, accuracy, and regulatory compliance. A significant portion of the budget is dedicated to advanced sensor networks and detection hardware, which are essential for identifying contaminants across multiple spectrums including chemical, biological, and particulate matter in real-time environments.
These systems play a crucial role in enabling the AI to detect contamination events with precision while minimizing false positives that can disrupt operations. Another substantial expenditure is directed toward the integration of machine learning algorithms and pattern recognition systems within the control infrastructure, allowing the system to not only identify known contaminants but also learn from new contamination signatures and adapt detection parameters based on environmental conditions and historical data.
The physical isolation and containment mechanisms also receive considerable investment, as these automated systems, such as valve controls, barrier deployment, and atmospheric isolation chambers, enable the system to contain contamination autonomously and prevent spread to clean areas. Additionally, ongoing calibration and validation of both the detection systems and the isolation mechanisms are essential to ensure their accuracy and effectiveness over time, requiring a steady flow of resources for regulatory compliance and performance verification. Training and certification programs also form a critical part of the expenditure, as staff must be trained to manage the complex integration of AI-driven detection with physical containment systems and understand contamination protocols and emergency procedures.
| Metric | Value |
|---|---|
| Demand Value (2026) | USD 1.8 billion |
| Demand Forecast Value (2036) | USD 4.2 billion |
| Forecast CAGR 2026 to 2036 | 8.8% |
Over the next one to two years, enterprise spending on physical AI for contamination detection and isolation is expected to focus on critical production environments where contamination events can result in significant product losses, regulatory violations, or safety incidents. Plants will typically fund an initial deployment that integrates detection sensors, AI analysis systems, and automated isolation mechanisms within one high-risk production area or cleanroom facility, then expand to additional zones once detection accuracy and response effectiveness meet operational requirements. Procurement will bundle hardware with software development, system integration, and validation services, since these systems must be calibrated for specific contaminant types and validated against regulatory standards for detection sensitivity and response time.
Operating budgets will increase for automated machine learning alongside capital purchases as buyers add continuous monitoring, algorithm refinement, isolation system maintenance, spare sensor inventory, and performance analytics to ensure detection reliability remains high. Compliance and validation spend will increase in parallel, driven by regulatory requirements for contamination control documentation and system qualification, so hazard analysis, detection threshold definition, and emergency response systems become part of the purchase. Standards development work referenced by FDA, EPA, and ISO will continue influencing buying behavior, pushing enterprises to invest in validation protocols and traceability systems that verify detection accuracy and isolation effectiveness. Near term spending will favor proven detection packages that can be deployed incrementally, validated against existing contamination records, and scaled across production areas with predictable performance outcomes.
Physical AI systems for contamination detection and isolation serve a critical function in maintaining product quality, preventing contamination spread, and reducing human exposure to hazardous substances. Adoption is influenced by detection sensitivity requirements, response time specifications, integration challenges, and regulatory compliance standards. Segmentation by technology type, detection function, and application reveals how manufacturers select specific AI-driven detection architectures to meet contamination control targets, response speed specifications, and safety requirements across different production environments.

Multi-spectral contamination detection systems account for 42.0%, driven by their ability to identify diverse contaminant types through simultaneous analysis across multiple wavelengths and detection spectrums. AI-powered particle analysis platforms hold 28.0%, supporting automated identification of particulate contamination through real-time size, shape, and composition analysis. Chemical signature recognition systems represent 18.0%, favored for their capability to detect molecular-level contamination through AI-enhanced spectroscopic analysis. Automated biological contamination detection contributes 8.0%, used where microbial contamination poses significant product or safety risks. Other detection technologies account for 4.0%.
Key Points:
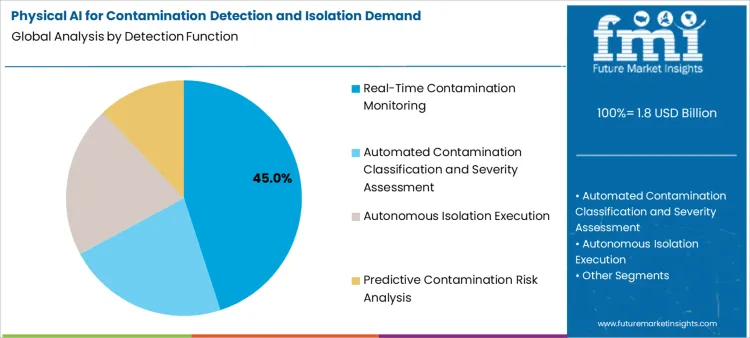
Real-time contamination monitoring represents 45.0%, reflecting priority on continuous assessment of environmental conditions and immediate contamination detection. Automated contamination classification and severity assessment account for 26.0%, essential for accurate threat evaluation and appropriate response selection. Autonomous isolation execution holds 18.0%, supporting physical containment actions without human intervention in safe operating conditions. Predictive contamination risk analysis represents 11.0%, addressing contamination prevention through environmental data analysis and trend monitoring.
Key Points:
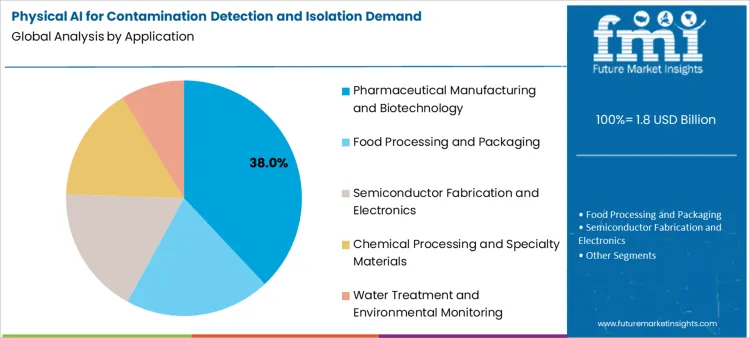
Pharmaceutical manufacturing and biotechnology lead with 38.0%, requiring stringent contamination control for product safety and regulatory compliance in sterile production environments. Food processing and packaging account for 24.0%, using AI detection systems for pathogen identification and foreign object detection throughout production lines. Semiconductor fabrication and electronics represent 16.0%, relying on contamination detection for yield protection and product quality in cleanroom environments. Chemical processing and specialty materials hold 14.0%, focused on preventing cross-contamination and maintaining product purity in multi-product facilities. Water treatment and environmental monitoring account for 8.0%, where contamination detection protects public health and environmental safety.
Key Points:
Current deployments indicate an adoption horizon driven by progressive validation milestones rather than immediate full-scale implementation. A typical early milestone is detection sensitivity validation in controlled conditions, where the system demonstrates reliable contamination identification across known contaminant types, concentration levels, and environmental variations while maintaining acceptable false positive rates. A second milestone is isolation system validation, covering automated containment capabilities, safety system integration, emergency override procedures, and documented performance in simulated contamination scenarios. Following validation, teams proceed to limited production deployment, where detection algorithms and isolation mechanisms are refined through real operating conditions while maintaining human oversight and manual confirmation authority.
A subsequent milestone is autonomous response expansion using robotics, where detection confidence levels and isolation success rates justify reduced human supervision and increased autonomous decision authority. The point where adoption accelerates is system standardization: validated detection models, proven isolation procedures, and reusable configuration templates that reduce deployment time for similar production environments. Full deployment across facilities follows once detection performance is documented, isolation effectiveness is proven, and operational cost benefits are quantified through reduced contamination incidents and improved product quality.
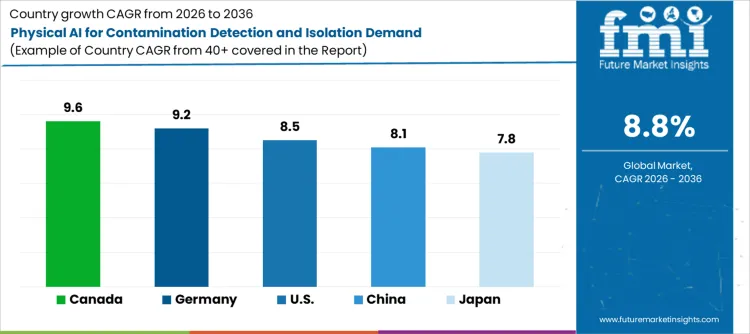
Global demand for physical AI systems for contamination detection and isolation is expanding as manufacturers seek to reduce contamination risks while improving product quality and regulatory compliance. Growth reflects rising adoption of artificial intelligence, edge computing capabilities, and automated containment technologies across pharmaceutical, food processing, and high-tech manufacturing sectors. Technology selection focuses on detection algorithms, isolation mechanisms, and safety systems that operate reliably in production environments with minimal human supervision. USA. records 8.5% CAGR, Germany records 9.2% CAGR, China records 8.1% CAGR, Japan records 7.8% CAGR, and Canada records 9.6% CAGR. Adoption remains driven by regulatory compliance requirements and product quality protection rather than technology advancement alone.
| Country | CAGR (%) |
|---|---|
| Canada | 9.6% |
| Germany | 9.2% |
| USA. | 8.5% |
| China | 8.1% |
| Japan | 7.8% |
Demand for physical AI systems for contamination detection and isolation in Canada is set to expand as pharmaceutical and biotechnology companies integrate advanced contamination control technologies. Growth at 9.6% CAGR reflects rising adoption of AI-driven detection systems in life sciences manufacturing, precision chemicals, and medical device production. Regulatory compliance requirements under Swiss and EU pharmaceutical guidelines drive selection of validated contamination detection systems with documented performance and traceability. Quality standards demand detection systems delivering contamination prevention and product protection at pharmaceutical-grade reliability levels. Leading biotechnology companies prioritize systems compatible with existing cleanroom infrastructure and established quality management systems. Demand concentrates within pharmaceutical manufacturing facilities, biotechnology research centers, and medical device production sites requiring advanced predictive maintenance capabilities.
Physical AI systems for contamination detection and isolation demand in Germany is positioned to grow as advanced manufacturing sectors integrate automated contamination control technologies. Growth at 9.2% CAGR reflects strong activity in automotive electronics, precision machinery, and chemical processing. Complex production processes require continuous contamination monitoring and rapid isolation capabilities for product quality protection and process consistency. Engineering excellence drives adoption of cutting-edge detection algorithms and automated isolation systems. Leading industrial companies invest in AI-powered contamination systems for operational efficiency and quality assurance. Demand remains centered on high-precision manufacturing applications serving automotive, aerospace, and industrial technology markets.
Demand for physical AI systems for contamination detection and isolation in the USA. is poised to strengthen as manufacturers integrate automated contamination control across pharmaceutical, semiconductor, and food processing sectors. Growth at 8.5% CAGR reflects rising adoption in biotechnology manufacturing, electronics fabrication, and food safety applications. FDA regulations and safety requirements drive selection of validated detection systems with proven isolation capabilities and documented performance. Advanced research institutions and technology companies lead development of next-generation AI contamination detection algorithms. Large corporations prioritize contamination systems supporting both operational efficiency and regulatory compliance requirements. Demand remains strongest within industries facing stringent contamination control requirements and product liability concerns.
Physical AI systems for contamination detection and isolation demand in China is anticipated to grow as manufacturers integrate automated contamination control across electronics, pharmaceutical, and food processing sectors. Growth at 8.1% CAGR reflects adoption in consumer electronics manufacturing, pharmaceutical production, and food safety applications. Export quality requirements drive integration of contamination detection systems with existing production control systems and quality management platforms. Manufacturing excellence standards influence selection of cost-effective detection technologies and isolation mechanisms. Established manufacturing companies invest in AI contamination capabilities for quality improvement and export competitiveness. Demand is driven by product quality requirements and international compliance standards rather than domestic regulations alone.
Demand for physical AI systems for contamination detection and isolation in Japan is positioned to rise as precision manufacturing and technology sectors adopt automated contamination control technologies. Growth at 7.8% CAGR reflects integration in electronics manufacturing, precision instruments, and pharmaceutical production. Quality control standards drive adoption of contamination detection systems ensuring consistent production outcomes and product reliability. Established manufacturing industry provides foundation for advanced AI contamination system deployment. Industrial companies prioritize systems supporting both automation efficiency and worker safety in controlled environment conditions. Demand remains focused on applications requiring high precision and long-term reliability rather than rapid deployment.
Key companies and organizations active in the ecosystem for physical AI contamination detection and isolation include major analytical instrument manufacturers like Thermo Fisher Scientific, Danaher Corporation, Agilent Technologies, and Waters Corporation, which offer integrated detection platforms with AI-powered analysis capabilities. Software and AI providers such as IBM, NVIDIA, and specialized contamination analysis companies provide machine learning algorithms and pattern recognition platforms supporting automated contamination detection. Sensor and detection hardware companies like Honeywell, Emerson Electric, and specialized contamination monitoring firms offer sensing hardware crucial for contamination data collection.
System integrators, both large multinational firms and specialized cleanroom automation consultancies, focus on implementing contamination detection systems within existing production infrastructure. Standards organizations like ISO/TC 209 and ASTM guide contamination control requirements for automated detection systems. Research institutions and industry consortia play key roles in advancing detection algorithm development and establishing best practices for automated contamination control in manufacturing environments.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Technology Type | Multi-Spectral Contamination Detection Systems; AI-Powered Particle Analysis Platforms; Chemical Signature Recognition Systems; Automated Biological Contamination Detection; Others |
| Detection Function | Real-Time Contamination Monitoring; Automated Contamination Classification and Severity Assessment; Autonomous Isolation Execution; Predictive Contamination Risk Analysis |
| Application | Pharmaceutical Manufacturing and Biotechnology; Food Processing and Packaging; Semiconductor Fabrication and Electronics; Chemical Processing and Specialty Materials; Water Treatment and Environmental Monitoring |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | Canada, Germany, USA., China, Japan, and 40+ countries |
| Key Companies Profiled | Thermo Fisher Scientific Inc.; Danaher Corporation; Agilent Technologies Inc.; Waters Corporation; PerkinElmer Inc.; Others |
| Additional Attributes | Dollar sales by technology type, detection function, and application; performance in contamination detection accuracy and isolation success rates across pharmaceutical, food processing, and semiconductor industries; detection speed, isolation reliability, and regulatory compliance under automated operation conditions; impact on contamination incidents, product quality, and operational safety during unmanned operation; compatibility with existing cleanroom control systems and quality management platforms; procurement dynamics driven by regulatory certification requirements, validation protocols, and long-term service partnerships. |
How big is the physical ai for contamination detection and isolation demand in 2026?
The global physical ai for contamination detection and isolation demand is estimated to be valued at USD 1.8 billion in 2026.
What will be the size of physical ai for contamination detection and isolation demand in 2036?
The market size for the physical ai for contamination detection and isolation demand is projected to reach USD 4.2 billion by 2036.
How much will be the physical ai for contamination detection and isolation demand growth between 2026 and 2036?
The physical ai for contamination detection and isolation demand is expected to grow at a 8.8% CAGR between 2026 and 2036.
What are the key product types in the physical ai for contamination detection and isolation demand?
The key product types in physical ai for contamination detection and isolation demand are multi-spectral contamination detection systems, ai-powered particle analysis platforms, chemical signature recognition systems, automated biological contamination detection and others.
Which detection function segment to contribute significant share in the physical ai for contamination detection and isolation demand in 2026?
In terms of detection function, real-time contamination monitoring segment to command 45.0% share in the physical ai for contamination detection and isolation demand in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.