AI Fetal Ultrasound View Detection Software Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The AI Fetal Ultrasound View Detection Software is segmented by Deployment (Cloud, On-premise, Hybrid), Function (View detection, Anatomy labeling, Quality scoring, Report automation), Workflow stage (Real-time, Post-scan, QA review), End user (Hospitals, Imaging centers, Maternity clinics, Tele-ultrasound hubs), Integration (OEM-integrated, Platform add-on, Standalone SaaS), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
AI Fetal Ultrasound View Detection Software Market Size, Market Forecast and Outlook By FMI
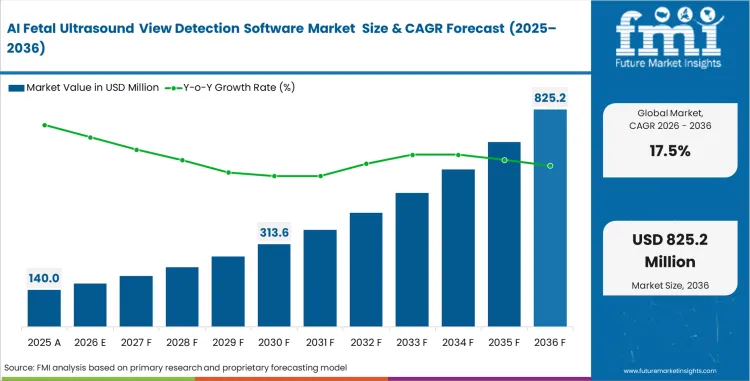
The AI fetal ultrasound view detection software market was valued at USD 119.1 million in 2025 and is projected to reach USD 140 million in 2026. The industry outlook points to 17.5% CAGR from 2026 to 2036, taking total valuation to USD 700 million by the end of the forecast period.
Summary of AI Fetal Ultrasound View Detection Software Market
- The market is forecast to reach USD 700 million by 2036.
- The market is expected to grow at a CAGR of 17.5% from 2026 to 2036.
- The market was estimated at USD 119.1 million in 2025.
- The forecast period represents an incremental opportunity of USD 560 million.
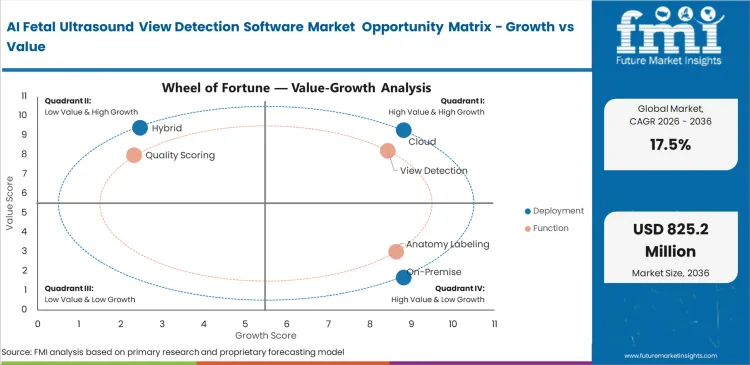
- Cloud deployment leads the segment with a 58% share, supported by easy integration into existing clinical workflows.
- View detection dominates the function segment with a 36% share, as standard-plane recognition remains the primary commercial application.
- Real-time workflow support accounts for 62% share, reflecting growing demand for in-scan guidance rather than post-scan analysis.
- OEM-integrated solutions hold a 54% share, driven by direct embedding into ultrasound platforms.
- Hospitals lead the end-user segment with a 48% share, due to higher prenatal imaging volumes and centralized procurement.
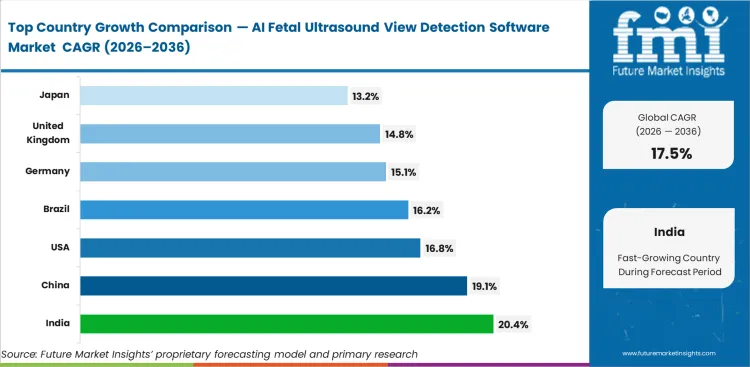
- India, China, and Brazil are the fastest-growing markets, while the United States and Germany remain key high-value adoption regions.
- Market growth is driven by the need for standardized imaging quality, reduced operator dependency, and increasing adoption of AI-assisted prenatal screening tools.
Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 140 million |
| Industry Value (2036) | USD 700 million |
| CAGR (2026 to 2036) | 17.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Clinical screening protocols continue to place strong weight on accurate plane acquisition, which keeps automated detection software relevant in routine fetal imaging. Products that fit directly into established ultrasound workflows remain better placed than standalone tools because installation friction stays low and day-to-day use is easier to sustain.
Radiology and prenatal imaging departments continue to work under specialist shortages that slow examination flow and delay confirmation of critical fetal views. Manual dependence on experienced maternal-fetal specialists creates scheduling pressure, especially where scan volumes are high and staffing depth is limited. Embedding automated standard plane detection into the scanning pathway helps reduce that bottleneck by supporting faster image validation during the exam itself. Delayed adoption can leave facilities with heavier reporting backlogs, longer patient wait times, and greater exposure to documentation risk in anomaly screening. Workflow fit remains central, so software that works within existing hardware environments is advancing faster than products that require separate operating layers.
Adoption is likely to strengthen further once reimbursement frameworks begin to recognize algorithm-assisted scan support more clearly. Quality-focused clinical guidance is already pushing the sector toward tighter imaging consistency, and that creates a practical opening for software that improves repeatability without requiring a larger specialist team. Diagnostic capacity can expand more efficiently when departments improve scan standardization through software rather than depending only on additional highly trained staff.
Rising at a 20.4% CAGR through 2036, India remains the fastest-rising country market as maternal care services extend further into rural territories where skill-bridging tools are becoming more relevant. China is also on a strong upward path, with the sector expected to advance at 19.1% CAGR as regional medical centers continue to upgrade under state-backed healthcare improvement programs. Private clinic efficiency programs keep the United States on a positive trend, and the market there is projected to expand at a CAGR of 16.8% through 2036. Brazil, where prenatal screening access is widening, is forecast to register 16.2% CAGR over the study period. Germany continues to benefit from a compliance-driven imaging environment, and the industry outlook in the country points to 15.1% CAGR through 2036. In the United Kingdom, software adoption is rising as imaging services work to manage specialist capacity constraints, with the market set to record 14.8% CAGR during the forecast period. Japan is expected to post 13.2% CAGR by 2036, supported by the need to raise productivity across an aging clinical workforce. Local clinician availability continues to shape adoption patterns more directly than economic scale alone.
Segmental Analysis
AI Fetal Ultrasound View Detection Software Market Analysis by Deployment
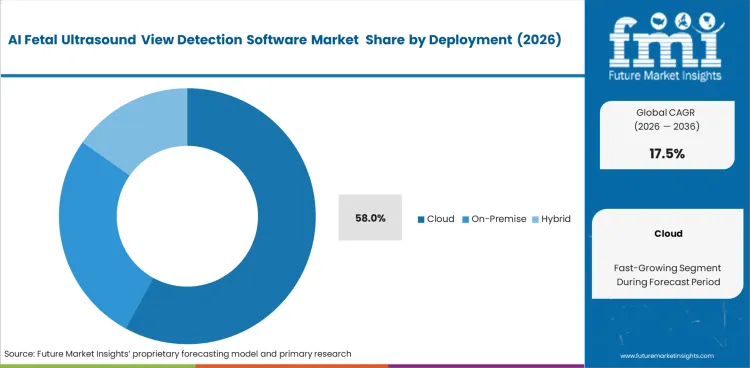
Hospital IT directors migrate clinical applications toward centralized architectures. The cloud segment is expected to account for 58.0% share in 2026. Centralized models dominate because multi-site health systems require unified quality benchmarking across all affiliated clinics to maintain compliance. Chief Medical Information Officers prioritize remote data access over isolated workstation installations. Pushing algorithmic updates simultaneously to dozens of distant scanning locations eliminates version control nightmares. Local hardware limitations accelerate this shift, as older ultrasound systems cannot process complex deep learning models locally. Cloud processing bypasses hardware refresh cycles entirely. Cloud deployment can support multi-site standardization, but adoption depends on privacy, connectivity, and local IT readiness.
- Initial selection: IT administrators prioritize SOC2 compliance and data encryption standards during vendor evaluation.
- Technical validation: Network engineers measure latency rates ensuring real-time image processing does not lag clinical workflows.
- Enterprise expansion: Health system executives mandate system-wide rollouts after pilot clinics demonstrate measurable throughput gains.
AI Fetal Ultrasound View Detection Software Market Analysis by Function
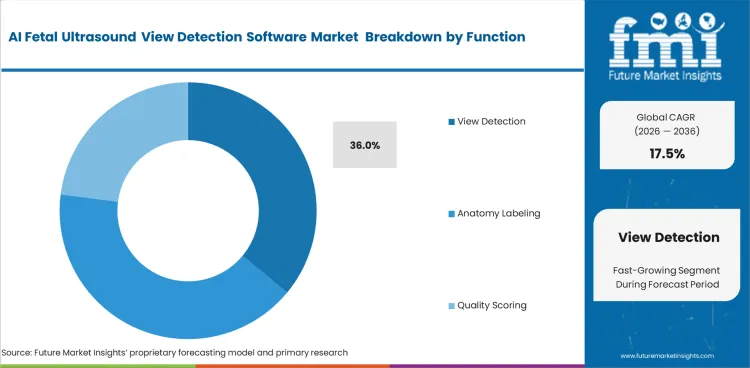
Maternal-fetal specialists prioritize immediate anatomical plane verification above all other features. View detection is projected to secure 36.0% share in 2026. This function dominates because professional society guidelines mandate strict image criteria for anomaly screening. Lead sonographers rely on these algorithms to confirm difficult cardiac views instantly, which prevents manual senior physician overreads during routine scans. Advanced anatomy labeling features often distract novice users from fundamental plane acquisition. Reliable view detection can improve scan consistency and reduce incomplete-image capture, though clinical outcomes still depend on operator skill and workflow quality. Clinics failing to standardize view acquisition suffer high patient recall rates for incomplete examinations. Integrating these capabilities alongside diagnostic imaging services elevates baseline care quality dramatically.
- Error prevention: Real-time plane verification prevents sonographers from saving inadequate diagnostic images.
- Residual vulnerability: Algorithms still struggle with high maternal BMI patients requiring manual optimization techniques.
- Maximum utilization: Clinical directors achieve full value only when enforcing mandatory algorithmic sign-off for every saved image.
AI Fetal Ultrasound View Detection Software Market Analysis by Workflow stage
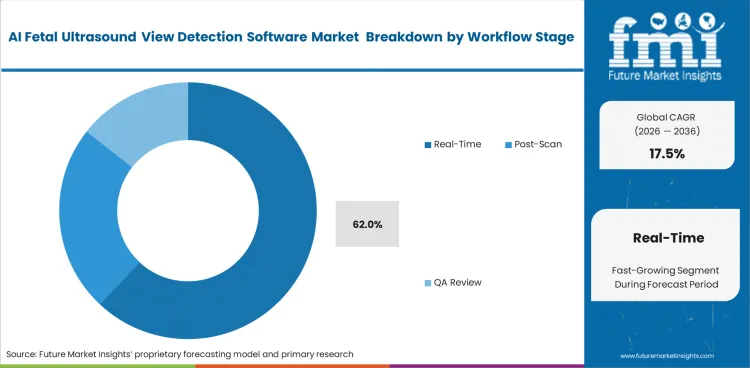
Clinical administrators target operational bottlenecks occurring directly at patient bedsides. Real-time integration is likely to represent 62.0% of the market in 2026. Instant feedback loops correct acquisition errors before patients ever leave examination rooms. Sonography department managers champion these tools to eliminate costly repeat appointments. Receiving immediate on-screen guidance accelerates junior staff training curves effectively. Post-scan analysis identifies errors too late to prevent patient callbacks, highlighting problems instead of preventing them actively. Facilities relying solely on post-scan review experience significantly higher patient dissatisfaction rates. Modern ultrasound devices processing algorithms instantaneously win overwhelming clinical preference.
- Savings origination: Eliminating repeat scanning appointments opens calendar slots for new billable patients.
- Hidden expenses: Integrating real-time processing requires upgrading local network bandwidth to handle continuous data streams.
- Lifecycle analysis: Long-term financial models prove real-time error prevention yields a higher return on investment than cheaper post-scan analytics.
AI Fetal Ultrasound View Detection Software Market Analysis by End user
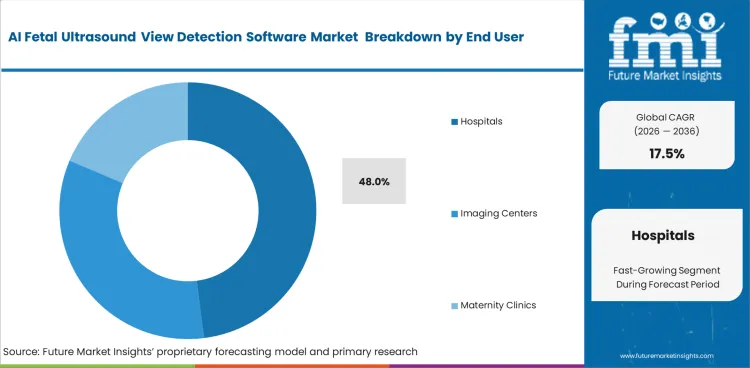
Tertiary care centers manage high volumes of complex high-risk pregnancies demanding absolute precision. Large medical institutions command capital budgets necessary for comprehensive algorithmic deployments. Hospitals are expected to contribute 48.0% of total market share in 2026. Chief of Obstetrics roles mandate standardized screening protocols across dozens of employed sonographers. High staff turnover within hospital imaging departments makes automated skill-bridging tools indispensable. Massive research hospitals often lag smaller regional centers in adoption speed due to complex cybersecurity approval committees. Freestanding clinics deploy these solutions months faster than bureaucratic hospital networks. Delaying implementation leaves major institutions vulnerable to diagnostic discrepancy lawsuits. Deploying tools built for fetal and neonatal care equipment ensures compliance.
- Early adopters: Academic research hospitals validate clinical efficacy through rigorous peer-reviewed algorithmic testing protocols.
- Mainstream followers: Regional medical centers adopt proven software to match academic institution diagnostic quality standards.
- Late converters: Rural critical access clinics finally implement solutions when cloud pricing models become financially accessible.
AI Fetal Ultrasound View Detection Software Market Analysis by Integration
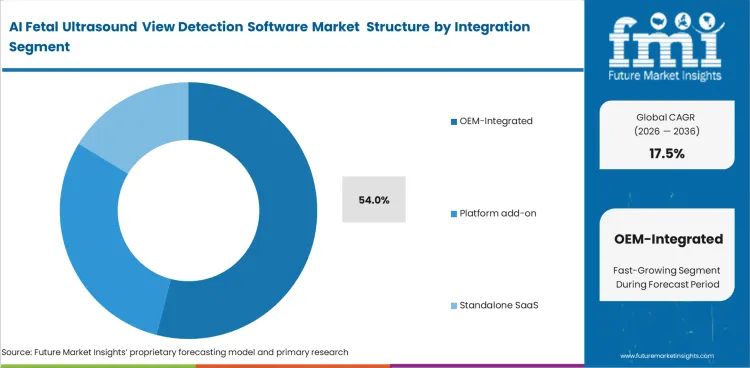
Hardware purchasers strongly prefer algorithms bundled natively with their scanning machines. OEM-integrated solutions are anticipated to emerge with 54.0% market share in 2026. Clinical users actively resist interacting with secondary tablets or external monitors during examinations. Radiology IT directors appreciate deploying single-vendor systems that minimize complex interoperability challenges. Purchasing boards negotiate software licenses directly into multi-year hardware fleet upgrade contracts. Standalone software vendors face meaningful integration barriers to achieve seamless user interface parity with native OEM integrations. Third-party applications frequently suffer from slight display latency that ruins sonographer trust entirely. Clinics applying fragmented software add-ons face continuous technical support disputes between hardware and software vendors. Modern portable cardiology ultrasound systems demonstrate how native integration drives rapid clinical acceptance.
- Baseline expectations: Clinical users demand zero-latency processing matching native hardware frame rates perfectly.
- Edge conditions: Native integrations perform flawlessly offline while cloud add-ons fail completely during network outages.
- Acceptability standards: Hospital purchasing boards require strict FDA clearance documentation for all integrated software modules prior to contract signing.
AI Fetal Ultrasound View Detection Software Market Drivers, Restraints, and Opportunities
Missed congenital anomalies trigger intense clinical and legal consequences for imaging facilities. Radiology directors implement automated view detection to support more consistent adherence to screening protocols when used within established clinical workflows. Human sonographers experience severe visual fatigue during high-volume clinic days. Algorithmic support can reduce variability in plane assessment, especially in high-volume settings, but does not remove the need for trained clinical judgment. Standardized documentation may strengthen quality assurance processes, though liability impact should not be overstated without specific evidence. Facilities lacking automated verification face rising premiums and heightened legal exposure. This financial penalty accelerates obstetric ultrasound AI software market adoption faster than pure clinical benefits alone.
Integrating third-party algorithms into legacy imaging fleets creates deep interoperability nightmares. Hospital IT managers spend months attempting to route proprietary video signals into external processing portals. Older machines output analog or encrypted digital streams that block direct software integration entirely. Upgrading entire hardware fleets requires capital expenditure that delays software adoption by years. Software vendors attempt developing universal hardware adapters to bypass this constraint. These physical workarounds introduce unacceptable display latency during fast-paced examinations.
Opportunities in the AI Fetal Ultrasound View Detection Software Market
- Rural deployment: Extending automated analysis tools into remote clinics enables minimally trained technicians to capture diagnostic-quality maternal-fetal images confidently.
- Telemedicine integration: Connecting real-time algorithmic quality scoring with remote physician oversight allows instant expert consultation utilizing ai radiology worklist orchestration platforms.
- Training acceleration: Educational institutions deploy feedback software to accelerate student sonographer competency metrics dramatically.
Regional Analysis
Based on regional analysis, AI Fetal Ultrasound View Detection Software is segmented into North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, and Middle East and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 20.4% |
| China | 19.1% |
| United States | 16.8% |
| Brazil | 16.2% |
| Germany | 15.1% |
| United Kingdom | 14.8% |
| Japan | 13.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
South Asia and Pacific AI Fetal Ultrasound View Detection Software Market Analysis
Large population centers across South Asia and the Pacific continue to work with limited maternal-fetal specialist capacity, which keeps pressure on frontline screening systems. Health ministries are extending diagnostic capability beyond major cities into rural primary care networks, where software support can help reduce variation in scan quality. Low-cost bundled systems from regional equipment makers are helping widen access, especially where buyers need practical tools that fit basic ultrasound setups and tight budget conditions. High patient throughput also matters across this territory, as clinics need to move routine prenatal examinations faster without weakening image review standards. Software that supports automated view detection fits that requirement well, and its connection with wearable pregnancy devices is adding further operational value to remote maternal monitoring pathways.
- India: State health programs are using algorithm-based tools to bring more consistency to prenatal screening across large rural care networks. Referral pressure remains a major issue, so local administrators are placing value on systems that help primary centers complete more reliable first-line assessments before cases move upward. The market in India is projected to expand at a CAGR of 20.4% through 2036. Vendors that secure public-sector rollout programs are likely to strengthen their position quickly because scale, service coverage, and execution capacity matter heavily in this market.
FMI’s report includes detailed analysis of pricing direction, deployment patterns, end-user adoption, and software integration priorities across South Asia & Pacific. It also tracks how rural screening expansion and device accessibility are shaping the regional industry outlook.
East Asia AI Fetal Ultrasound View Detection Software Market Analysis
East Asia benefits from a stronger clinical and manufacturing base, but hospitals across the region still face heavy imaging volumes and rising pressure to keep quality standards consistent. Medical authorities are tightening obstetric imaging protocols, which is pushing providers toward software that can support standardization across busy departments. Domestic technology competition also remains strong, and that is helping accelerate model refinement, hardware integration, and product localization. Buyers in this region prefer solutions that work smoothly with existing hospital systems and broader health-data infrastructure, since workflow compatibility often carries more weight than software novelty alone.
- China: China is likely to post 19.1% CAGR in the sector by 2036. The country’s strong local hardware base also supports wider software embedding across both premium and more accessible systems. Large hospitals are using view-detection software to reduce pressure on experienced sonographers while improving training support for junior staff. That matters in a market where patient volume is high and departments need a practical way to scale imaging capacity without slowing clinical throughput.
- Japan: Imaging departments in Japan are working around an experienced but aging sonography workforce, which keeps the focus on productivity and clinical consistency. Hospitals are giving preference to tools that help maintain scanning standards while easing dependence on a limited pool of highly trained users. Japan is expected to record 13.2% CAGR in the market during the forecast period. This also gives domestic medical device companies a disciplined home market in which to refine software before pushing it into broader international use.
FMI’s report includes detailed tracking of deployment trends, hospital adoption patterns, domestic hardware-software integration, and workflow standardization across East Asia. It also examines how staffing pressure and local product development are influencing the regional market outlook.
North America AI Fetal Ultrasound View Detection Software Market Analysis
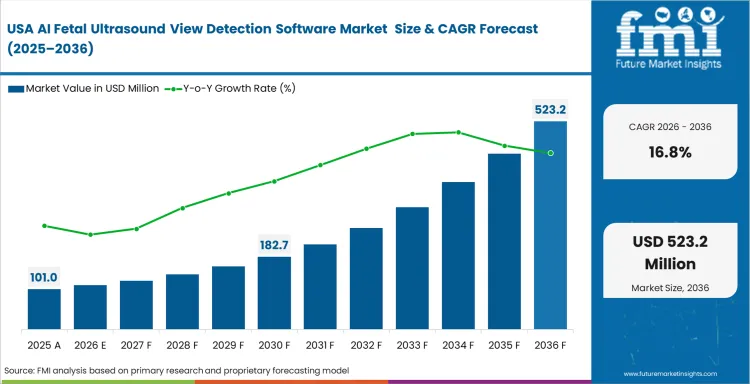
North America remains one of the most commercially important regions because software adoption is closely tied to workflow efficiency, documentation quality, and return on installed ultrasound systems. Private imaging networks and hospital groups both place clear value on tools that can reduce scan time, limit repeat examinations, and improve consistency across multi-site operations. FDA clearance remains a major barrier for new entrants, which strengthens the position of companies already tied to established hardware platforms or proven clinical software environments. Documentation requirements and reimbursement discipline also support adoption, especially where providers are expected to show consistent imaging quality across high-volume prenatal workflows.
- United States: The industry outlook in the United States points to 16.8% CAGR through 2036. Better standardization also supports compliance and reduces the operational cost of repeat imaging or avoidable reporting variation. Large imaging groups are standardizing AI-enabled tools across broad clinic networks to improve consistency and simplify operating models after acquisition-led expansion. Clinical leadership teams want the same scan logic, documentation quality, and workflow standard across every site, especially where scale makes variation expensive.
FMI’s report includes detailed analysis of FDA-led commercialization, multi-site deployment strategy, reimbursement-linked workflow adoption, and enterprise ultrasound integration across North America. It also evaluates how installed base strength continues to support software expansion in the region.
Latin America AI Fetal Ultrasound View Detection Software Market Analysis
Latin America is advancing from a lower digital base, but software adoption is improving where healthcare systems need better prenatal screening coverage without a matching increase in specialist capacity. Hospitals and diagnostic networks are placing value on tools that improve scan consistency and support earlier identification of cases that need specialist review. Budget discipline remains a defining factor, so solutions that work with existing equipment and require limited workflow disruption are better placed in the region. Public health expansion and wider maternal-care access are keeping the category relevant across key urban and secondary-care markets.
- Brazil: Brazil is forecast to register 16.2% CAGR in this market over the study period. Adoption in the country is supported by the need to improve coverage and consistency at the same time. Large healthcare providers in Brazil are placing more value on software that helps standardize prenatal imaging across mixed-quality care settings. Clinical teams need practical tools that improve first-line assessment quality while easing pressure on referral centers and specialist workloads.
FMI’s report includes detailed evaluation of adoption barriers, hospital digitization patterns, equipment compatibility needs, and maternal screening expansion across Latin America. It also tracks how cost control and uneven specialist coverage are shaping the regional market.
Europe AI Fetal Ultrasound View Detection Software Market Analysis
Europe remains a compliance-led market where adoption depends heavily on clinical standardization, documentation quality, and smooth fit with established imaging workflows. Publicly funded health systems across the region are under pressure to use specialist capacity more efficiently, which supports interest in software that can reduce variability in prenatal scans. Buyers also expect compatibility with existing hospital information systems, reporting formats, and governance requirements before deployment moves forward. Commercial progress tends to be steadier here than in faster-volume markets, but the installed clinical base and disciplined workflow environment keep Europe important for long-term software adoption.
- Germany: Germany is set to advance at a CAGR of 15.1% through 2036 in this market. That keeps the country among the key high-value adoption bases in Europe. German providers place strong value on software that supports consistent prenatal imaging quality without creating unnecessary workflow disruption. Hospitals and imaging departments prefer tools that fit established equipment environments and documentation processes, especially where implementation discipline is high.
- United Kingdom: NHS workflow pressure and sonography capacity constraints are supporting interest in software that can improve scan consistency across high-volume prenatal pathways. Buyers are more likely to adopt solutions that help standardize routine imaging while fitting existing reporting and governance management. The United Kingdom market is on a positive trend at 14.8% CAGR through 2036. Stable integration and practical clinical value remain more important than feature breadth alone.
FMI’s report includes detailed tracking of adoption patterns across Germany and the United Kingdom. It also examines how compliance requirements, public-system workflow discipline, and specialist capacity constraints continue to influence software expansion across Europe.
Competitive Aligners for Market Players
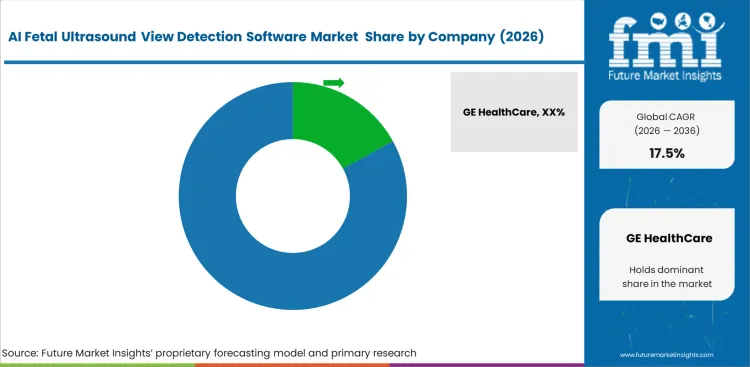
Medical imaging conglomerates control software distribution by restricting third-party access to proprietary hardware interfaces. GE HealthCare leads by embedding advanced view detection algorithms directly into its premium console architecture. Samsung Healthcare and Philips utilize their installed bases to push software subscriptions seamlessly to existing clinical users. Independent software developers face massive technical hurdles attempting to extract real-time image data from these closed systems. This hardware control forces pure software companies to pursue complex partnership agreements rather than selling directly to hospitals.
Incumbents possess proprietary datasets consisting of millions of annotated fetal ultrasound images. This data advantage allows companies like GE HealthCare to train highly accurate neural networks that smaller competitors cannot replicate. Sonio and BrightHeart counter this advantage by developing vendor-neutral cloud platforms capable of analyzing images from any manufacturer. They build specific deep learning ai dermatology imaging crossover techniques to enhance image processing efficiency. Regulatory clearance libraries represent another barrier. Companies holding FDA 510(k) clearances for specific anatomical view detection prevent unverified startups from entering clinical workflows.
Large hospital networks resist vendor lock-in by mandating DICOM compatibility for all purchased software tools. Chief Information Officers actively reject algorithms that only function on specific hardware brands. Butterfly Network capitalizes on this frustration by coupling view detection with ultra-portable hardware at a fraction of traditional costs. Ultrasound AI targets specialized maternity clinics demanding highly specific anomaly detection modules. Purchasing power shifts toward organizations offering true hardware-agnostic clinical intelligence. Independent clinics increasingly favor flexible SaaS deployments over rigid OEM contracts.
Key Players in AI Fetal Ultrasound View Detection Software Market
- GE HealthCare
- Sonio
- Samsung Healthcare
- BrightHeart
- Butterfly Network
- Ultrasound AI
- Philips
Scope of the Report
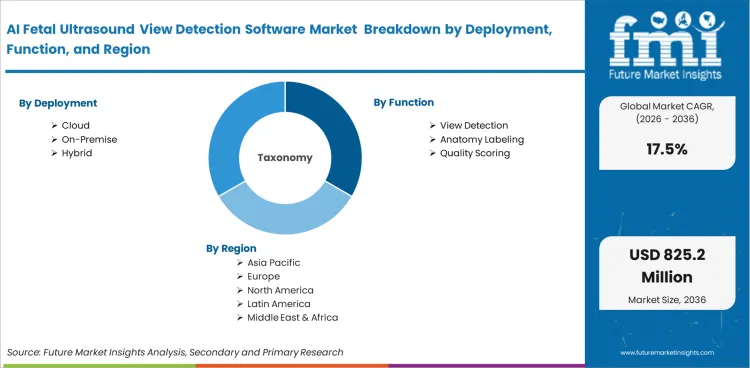
| Metric | Value |
|---|---|
| Quantitative Units | USD 140 million to USD 700 million, at a CAGR of 17.5% |
| Market Definition | Specialized algorithmic tools automatically identify standard diagnostic planes during obstetric scans. These systems verify image quality instantly against clinical guidelines and provide real-time feedback. |
| Segmentation | Deployment, Function, Workflow stage, End user, Integration |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East and Africa |
| Countries Covered | India, China, United States, Brazil, Germany, United Kingdom, Japan |
| Key Companies Profiled | GE HealthCare, Sonio, Samsung Healthcare, BrightHeart, Butterfly Network, Ultrasound AI, Philips |
| Forecast Period | 2026 to 2036 |
| Approach | Clinical software deployment contracts at major hospital networks. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
AI Fetal Ultrasound View Detection Software Market Analysis by Segments
Deployment:
- Cloud
- On-premise
- Hybrid
Function:
- View detection
- Anatomy labeling
- Quality scoring
- Report automation
Workflow stage:
- Real-time
- Post-scan
- QA review
End user:
- Hospitals
- Imaging centers
- Maternity clinics
- Tele-ultrasound hubs
Integration:
- OEM-integrated
- Platform add-on
- Standalone SaaS
Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- South Asia and Pacific
- East Asia
- Middle East and Africa
Bibliography
- USA Food and Drug Administration. (2026). Artificial Intelligence-Enabled Medical Devices.
- United Nations, Department of Economic and Social Affairs, Population Division. (2024). World Population Prospects 2024: Summary of Results.
- Khalil, A., et al. (2024). ISUOG Practice Guidelines: performance of third-trimester obstetric ultrasound scan. Ultrasound in Obstetrics & Gynecology.
- Han, X., Yu, J., Yang, X., Chen, C., Zhou, H., Qiu, C., Cao, Y., Zhang, T., Peng, M., & Zhu, G. (2024). Artificial intelligence assistance for fetal development: Evaluation of an automated software for biometry measurements in the mid-trimester. BMC Pregnancy and Childbirth, 24, 158.
- Lim, A., Abdalla, M., et al. (2024). Automatic standard plane and diagnostic usability classification in obstetric ultrasounds. WFUMB Ultrasound Open.
- Venturini, L., Budd, S. F., Farruggia, A., Wright, R., Matthew, J., Day, T. G., Kainz, B., Razavi, R., & Hajnal, J. V. (2025). Whole examination AI estimation of fetal biometrics from 20-week ultrasound scans. npj Digital Medicine, 8.
- D’Alberti, E., Patey, O., Smith, C., Šalović, B., Hernandez-Cruz, N., Noble, J. A., & Papageorghiou, A. T. (2025). Artificial intelligence-enabled prenatal ultrasound for the detection of fetal cardiac abnormalities: a systematic review and meta-analysis. eClinicalMedicine, 84, 103250.
- British Medical Ultrasound Society, & Society of Radiographers. (2024). Recruiting international sonographers and those without a medical background.
- Society for Maternal-Fetal Medicine. (2025, January 30). New study highlights AI's potential to help doctors detect congenital heart defects.
- University of North Carolina Institute for Global Health and Infectious Diseases. (2024, August 2). AI-enabled ultrasound revolutionizes gestational age estimation in low-resource settings.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Sonographer shortage solutions utilizing automated diagnostic verification.
- Malpractice premium reductions tied directly to strict guideline adherence.
- Capital budget allocation shifts prioritizing OEM-integrated software modules.
- Cloud deployment acceleration driving regional health system standardization.
- Real-time feedback implementation eliminating costly patient scanning callbacks.
- Competitive positioning strategies for standalone algorithm developers facing incumbents.
- Rural clinic expansion models utilizing deeply discounted software bundles.
- Regulatory clearance hurdles delaying unverified startups from clinical entry.
Frequently Asked Questions
What is the revenue outlook for AI fetal ultrasound view detection software in 2026?
The industry reaches USD 140 million in 2026 as hospital networks prioritize automated quality assurance tools immediately.
What valuation will this technology achieve by 2036?
Total revenue is projected to hit USD 700 million by 2036, driven by strict regulatory screening mandates globally.
Which deployment architecture leads software integration?
Cloud deployments command 58.0% share in 2026 because multi-site health systems require centralized data aggregation and unified benchmarking.
Why does view detection dominate software functionality?
View detection captures 36.0% share in 2026 because specialists depend absolutely on instant anatomical plane verification for compliance.
What workflow stage generates highest clinical demand?
Real-time integration holds 62.0% share in 2026, instantly identifying acquisition errors to reduce avoidable repeat scans by identifying acquisition issues during the exam.
Which end user dictates algorithm purchasing trends?
Hospitals represent 48.0% share in 2026, utilizing enterprise-wide deployments to standardize screening protocols across large employed sonographer teams.
How does integration type affect hospital purchasing?
OEM-integrated solutions secure 54.0% share in 2026 as purchasing boards negotiate software licenses directly into hardware fleet contracts.
Why is India expanding faster than other analyzed nations?
India advances at 20.4% CAGR. Government health ministries subsidize algorithmic tools to standardize prenatal screening across rural centers.
How does China approach algorithmic imaging integration?
China climbs at 19.1% CAGR. Major urban hospitals apply automated view detection to rapidly train junior sonographer cohorts.
What shapes United States adoption patterns?
United States revenue expands at 16.8% CAGR as private equity-backed imaging networks standardize tools to reduce malpractice premiums.
Why do standalone software developers struggle against incumbents?
Major equipment manufacturers restrict access to proprietary hardware interfaces, blocking independent developers from extracting real-time image data effectively.
What prevents hospital networks from switching software vendors?
Incumbents hold FDA clearances for specific anatomical views. Switching vendors requires workflow retraining that disrupts daily examination throughput.
How do clinics reduce malpractice liability using AI?
Automated quality scoring provides structured quality documentation may strengthen auditability, though liability impact depends on local practice, governance, and evidence standards.
What hidden costs impact real-time software deployment?
Processing high-resolution video streams instantaneously requires high network bandwidth, forcing heavy IT infrastructure investments before deploying algorithmic feedback.
Why do academic hospitals sometimes lag rural clinics in adoption?
Research institutions utilize complex cybersecurity approval committees that delay implementation. Freestanding clinics bypass these bureaucratic hurdles deploying immediately.
How does hardware capability limit algorithm expansion?
Older ultrasound machines lack processing power for deep learning models, forcing facilities to purchase new scanning fleets entirely.
What clinical problem do anatomy labeling features introduce?
Advanced labeling modules distract novice sonographers from fundamental plane acquisition. More reliable view detection can improve scan consistency, but diagnostic yield still depends on operator skill, patient factors, and downstream review quality
How do professional societies influence software purchasing?
Organizations publish standardized quality guidelines creating clinical frameworks. Algorithms mathematically guarantee compliance with these published standards during examinations
Why do sonographers reject third-party software applications?
Third-party software may face workflow adoption barriers where latency, interface consistency, or interoperability fall short of native OEM integration. This micro-delay ruins clinical trust and disrupts scanning.
What allows portable hardware manufacturers to compete effectively?
Companies coupling view detection with ultra-portable scanning devices bypass traditional capital budgets, targeting clinics demanding affordable clinical intelligence.
How do independent developers overcome proprietary dataset advantages?
Smaller companies develop vendor-neutral cloud platforms capable of analyzing any image, circumventing annotated datasets held exclusively by incumbents.
What forces late adopters to finally implement view detection?
Critical access clinics transition when cloud pricing becomes financially accessible, as rising care standards make manual verification legally unjustifiable.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment , 2026 to 2036
- Cloud
- On-Premise
- Hybrid
- Cloud
- Y to o to Y Growth Trend Analysis By Deployment , 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Function, 2026 to 2036
- View Detection
- Anatomy Labeling
- Quality Scoring
- View Detection
- Y to o to Y Growth Trend Analysis By Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow Stage, 2026 to 2036
- Real-Time
- Post-Scan
- QA Review
- Real-Time
- Y to o to Y Growth Trend Analysis By Workflow Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Imaging Centers
- Maternity Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Integration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Integration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Integration, 2026 to 2036
- OEM-Integrated
- Platform add-on
- Standalone SaaS
- OEM-Integrated
- Y to o to Y Growth Trend Analysis By Integration, 2021 to 2025
- Absolute $ Opportunity Analysis By Integration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Deployment
- By Function
- By Workflow Stage
- By End User
- By Integration
- Competition Analysis
- Competition Deep Dive
- GE HealthCare
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sonio
- Samsung Healthcare
- BrightHeart
- Butterfly Network
- Ultrasound AI
- Philips
- GE HealthCare
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Integration, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Deployment , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Workflow Stage, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Integration, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Deployment
- Figure 6: Global Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Function
- Figure 9: Global Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Workflow Stage
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Integration
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Deployment
- Figure 32: North America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Function
- Figure 35: North America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Workflow Stage
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Integration
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Deployment
- Figure 48: Latin America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Function
- Figure 51: Latin America Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Workflow Stage
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Integration
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Deployment
- Figure 64: Western Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Function
- Figure 67: Western Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Workflow Stage
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Integration
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Function
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Workflow Stage
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Integration
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Deployment
- Figure 96: East Asia Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Function
- Figure 99: East Asia Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Workflow Stage
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Integration
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Function
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Workflow Stage
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Integration
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Deployment , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Function
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Workflow Stage, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow Stage, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Workflow Stage
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Integration, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Integration, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Integration
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

