Poly Peel Sheets Market
Poly Peel Sheets Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Poly Peel Sheets Market Forecast and Outlook 2026 to 2036
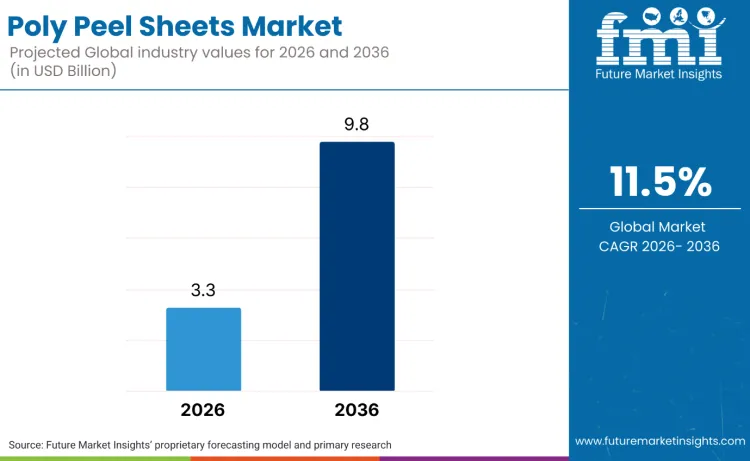
The global poly peel sheets market is valued at USD 3.3 billion in 2026 and is projected to reach USD 9.8 billion by 2036, expanding at an 11.5% CAGR. Growth quality is being governed by three compliance systems that directly influence material choice and supplier selection. In Europe, the Packaging and Packaging Waste Regulation entered into force on 11 February 2025 and will apply from 12 August 2026, tightening design-for-recycling and placing structural pressure on multi-material peel structures that are hard to sort or recycle. In the United States, FDA’s food contact substances framework links new polymer, adhesive, and coating chemistries to documented authorisation pathways, reducing tolerance for rapid formulation changes without regulatory alignment. In regulated packs, validation discipline extends into medical device packaging where ISO 11607 validation requirements are recognised within FDA’s consensus standards framework, reinforcing repeatable sealing and peel performance controls.
Supplier consolidation is a second structural driver. Amcor completed its combination with Berry Global on 30 April 2025, explicitly positioning the combined platform as broader and more complete for customers, with CEO Peter Konieczny stating: ‘This combination delivers on our strategy to become a stronger company with a broader, more complete offering for customers.’ This scale effect matters in peel sheets because global food and pharma accounts increasingly require harmonised specifications across regions, alongside audit-ready documentation for substrates, sealants, and migration performance.
Poly Peel Sheets Market Key Takeaways
| Items | Values |
|---|---|
| Market Value (2026) | USD 3.3 Billion |
| Market Value (2036) | USD 9.8 Billion |
| CAGR (2026 to 2036) | 11.5% |
Summary of the Poly Peel Sheets Market
What Is Growth Outlook for Poly Peel Sheets Market as per FMI Projection?
FMI projects the poly peel sheets market to grow at an 11.5% CAGR from USD 3.3 billion in 2026 to USD 9.8 billion by 2036.
FMI Research Approach: Forecasting anchored in food and healthcare packaging compliance cycles, sealant redesign, and converter capacity concentration.
How Do FMI Analysts Perceive Poly Peel Sheets Market to Evolve?
FMI sees the market shifting toward peel structures engineered for recyclability and validation, where seal performance, opening force stability, and regulatory documentation become the primary procurement filters rather than film price alone.
FMI Research Approach: Mapping governance signals such as EU packaging rules, FDA food-contact authorisations, and sterile packaging validation expectations.
Which Country Holds Largest Share in Global Poly Peel Sheets Market?
China holds a significant share, supported by scale in packaged food production, rapid e-commerce fulfilment, and national standards that formalise food contact material compliance for plastic packaging.
FMI Research Approach: Country modelling aligned with packaged food throughput, export-linked packaging demand, and national standards adoption.
How Large Will Poly Peel Sheets Market Be by 2036?
The poly peel sheets market is expected to reach USD 9.8 billion by 2036.
FMI Research Approach: Pricing and volume triangulation across food lidding, pharma blister-lidding systems, and sterile barrier packs.
What Is Definition of Poly Peel Sheets Market?
Poly peel sheets are polymer-based peelable sheet and film structures used in packaging systems to deliver controlled opening, seal integrity, and compatibility with food, pharmaceutical, medical device, and consumer goods packaging lines.
FMI Research Approach: Market definition anchored in packaging system function and compliance boundaries rather than end-brand categories.
What Are the Globally Unique Trends in Poly Peel Sheets Market?
The global trend is governance-led redesign toward recyclable structures and auditable compliance, with suppliers building scale and documentation capability to serve regulated and export-facing packaging programs.
FMI Research Approach: Regulatory signal tracking combined with company capacity actions and portfolio simplification patterns.
Which Forces Are Accelerating Poly Peel Sheets Adoption Through 2036?
Demand is being structurally pulled by compliance and validation requirements that force packaging owners to standardise peel performance across regions. In the EU, PPWR’s entry into force and staged application timeline is pushing converters to redesign peel structures around recyclability constraints, shifting sourcing toward suppliers that can deliver compliant mono-material or simplified laminates. In the USA, FDA’s food contact substances framework links polymer and adhesive choices to documented authorisation pathways, limiting rapid substitution and favouring suppliers with stable, approved chemistries. In India, FSSAI’s packaging regulations formalise packaging material requirements for food business operators, raising the value of documented compliance in flexible packs used for processed foods.
How is the Poly Peel Sheets Market Segmented?
The poly peel sheets market is segmented by material type, product type, end use industry, distribution channel, end user, and region to reflect how peel performance and compliance requirements shift by pack system and buyer. Material selection is governed by recyclability and migration constraints. Product architecture is governed by line speed, seal reliability, and opening force control. End use mix is anchored in high-throughput food packaging, regulated pharma packs, and sterile barrier applications. Distribution structures reflect converter procurement models where qualification documentation and inventory continuity matter as much as price.
Why Does Polyethylene Lead the Material Type Segment in Poly Peel Sheets Market?
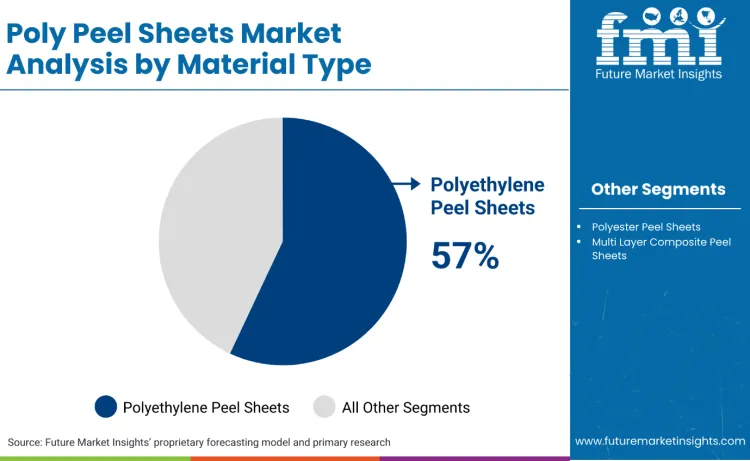
Polyethylene leads the material type segment with a 57% share because regulatory and procurement logic increasingly rewards structures that can be positioned within established recycling and compliance frameworks while still delivering controlled peel performance. In Europe, PPWR creates a governance pull toward recyclable packaging designs and reduces tolerance for complex, hard-to-recycle composite structures in high-volume applications.
Polyethylene-based peel sheets benefit because they can be engineered as simplified laminates and paired with compliant sealant layers without forcing a full material system change at the converter. In the United States, FDA’s food contact substances system ties packaging chemistries to documented authorisations, which increases the value of widely used polymer families with established regulatory histories and stable supplier documentation. For large food accounts, polyethylene also supports scale manufacturing economics through broad resin availability and mature extrusion capacity, which stabilises supply during frequent package redesign cycles triggered by recycled content targets and downgauging programs.
What Keeps Single Sided Peel Sheets Dominant in the Product Type Segment?
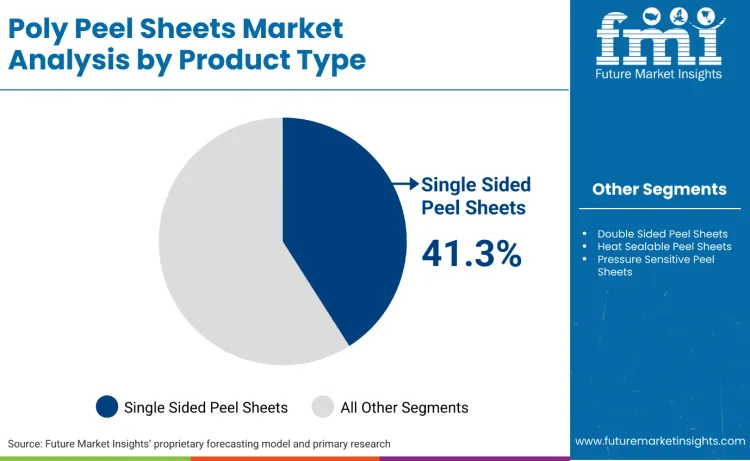
Single sided peel sheets account for 41.3% of the market because packaging line economics favour architectures that concentrate peel functionality at the lidding interface without adding unnecessary converting steps or qualification complexity. In food packaging, single sided peel designs allow converters to standardise seal initiation temperature windows and opening force outcomes while maintaining high line speeds across tray-seal and pouch formats, which is critical for volume categories that operate on overall equipment effectiveness targets.
In regulated environments, procurement teams also prefer structures that simplify documentation and change control. FDA’s framework for food contact substances and its notification-linked authorisation pathways raise the cost of frequent material substitutions, which pushes brands to use repeatable, validated configurations rather than experimenting with multi-interface peel layers. For medical device packs, sterile barrier validation discipline further rewards controlled, repeatable sealing outcomes, reinforcing demand for peel designs that are easier to validate and revalidate when equipment or process parameters change.
Why Does Food Packaging Lead End-Use Demand for Poly Peel Sheets?
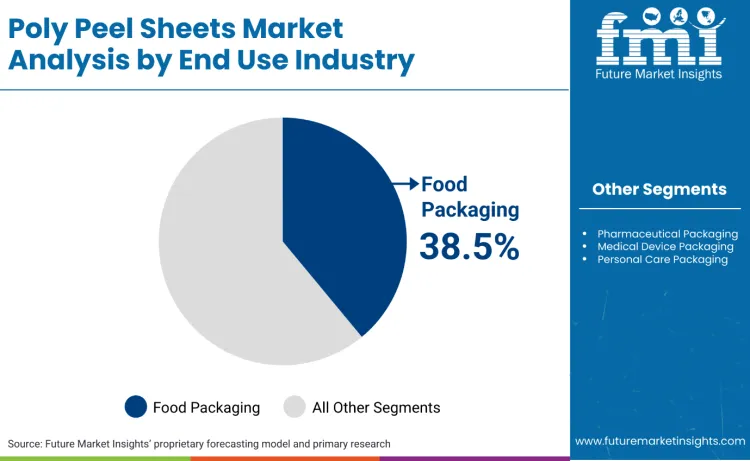
Food packaging leads with a 38.5% share because it concentrates the highest volume of seal-and-open events and operates inside strict compliance boundaries that reward validated, documented material systems. In the United States, FDA’s packaging and food contact substances framework establishes the regulatory infrastructure for packaging materials and components that contact food, which increases the value of suppliers that can support documented compliance for peelable sealants, coatings, and polymer layers.
In India, FSSAI’s packaging regulations require food business operators to ensure packaging materials align with prescribed requirements, reinforcing compliance as a procurement filter for flexible packaging used in processed and packaged foods. Food categories also run high SKU counts and frequent pack refresh cycles driven by branding and shelf-life optimisation, which increases recurring demand for peel sheet qualification and supply continuity. Unlike discretionary consumer goods packs, food packs are replenishment-driven and linked to stable throughput volumes, keeping demand resilient across cycles.
What Are the Trends and Restraints Shaping Poly Peel Sheets Demand?
A primary trend is governance-driven redesign toward recyclable peel structures and scale consolidation among suppliers that can fund compliance testing and multi-region documentation. PPWR’s staged implementation is accelerating specification churn in Europe, pushing converters to redesign peelable packaging with fewer incompatible layers and clearer end-of-life pathways. In parallel, the USA regulatory framework for food contact substances continues to function as a gating mechanism for new chemistries in packaging, reinforcing demand for suppliers that can support authorised formulations and stable compliance dossiers.
A key restraint is the rising cost and operational friction of packaging compliance under tightening waste governance, which can slow adoption of higher-specification peel solutions in cost-sensitive segments. India’s Plastic Waste Management regime assigns extended producer responsibility obligations across plastic packaging, increasing the compliance workload and cost stack for brand owners and pack spec changes. In the UK, packaging extended producer responsibility guidance formalises reporting and obligation structures, increasing administrative overhead that can delay packaging transitions for smaller manufacturers and contract packers. These constraints shift demand toward fewer, standardised peel formats rather than broad proliferation.
Poly Peel Sheets Market Analysis by Key Countries
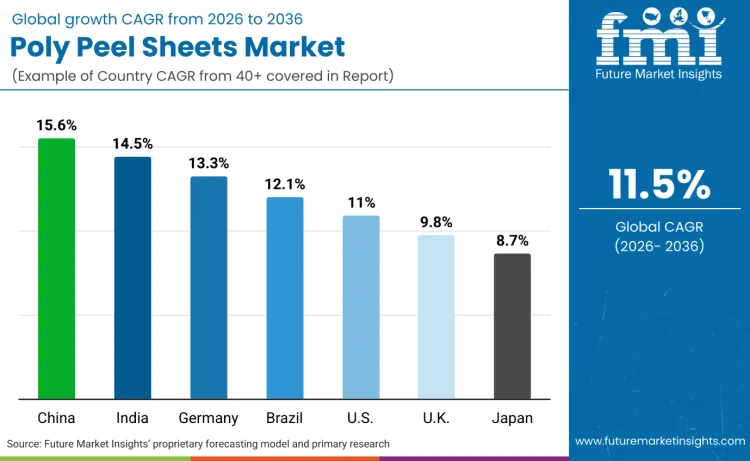
| Country | CAGR |
|---|---|
| China | 15.6% |
| India | 14.5% |
| Germany | 13.3% |
| Brazil | 12.1% |
| USA | 11.0% |
| UK | 9.8% |
| Japan | 8.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Does China Sustain the Highest CAGR for Poly Peel Sheets Through 2036?
China’s 15.6% CAGR is anchored in formalised national standards for food contact materials and a policy push to standardise and green the express packaging system that moves packaged food and consumer goods at scale. The National Health Commission’s 2023 announcement issuing multiple food safety national standards includes standards relevant to food-contact materials, tightening the compliance floor for plastic packaging used in domestic distribution and export-linked supply chains.
Parallel governance is visible in state-led action to reduce and regulate express packaging waste, which forces packaging designers to balance performance with material discipline across the fulfilment system. These mechanisms increase demand for peel sheet structures that can meet seal integrity and opening performance while aligning with tightening material and waste governance. Scale effects in Chinese converting capacity then accelerate adoption once specifications are qualified, because high-throughput food and consumer goods programs can roll compliant peel structures across multiple SKUs rapidly.
What Makes India a Fast-Scaling Market for Poly Peel Sheets Over 2026 to 2036?
India’s 14.5% CAGR reflects the interaction between formalised food packaging rules and the rising compliance load of plastic waste governance, which pushes brand owners toward documented, standardised packaging structures that can be deployed nationally. FSSAI’s packaging regulations require food business operators to ensure packaging materials align with defined requirements, increasing the value of compliant peel structures for packaged foods expanding across modern trade and e-commerce.
On the waste side, India’s Plastic Waste Management framework assigns extended producer responsibility obligations across plastic packaging, raising the importance of packaging designs that can be reported, rationalised, and scaled with fewer variations. These governance forces push demand toward qualification-ready peel sheet formats that reduce change-control frequency while maintaining performance. As food manufacturers expand multi-plant sourcing and contract packing, procurement shifts toward suppliers that can support compliance documentation and stable supply across regions, sustaining high growth.
Why Does Germany Remain a High-Compliance Growth Engine for Poly Peel Sheets?
Germany’s 13.3% CAGR is driven by packaging governance that formalises producer responsibility and tightens the operational need for traceable, compliant packaging specifications across supply chains. Germany’s packaging law framework and the Central Agency Packaging Register system reinforce registration and compliance discipline for packaging placed on the market, increasing the value of standardised packaging formats and controlled supplier documentation.
In parallel, EU-level packaging reform through PPWR adds pressure to redesign packaging for recyclability, accelerating specification churn in flexible formats where peel functionality historically relied on complex layer stacks. This governance combination increases demand for peel sheet solutions that can deliver opening performance while simplifying material structures and supporting audit-ready documentation. German industry procurement also favours supplier reliability and validated performance, which supports premiumisation in regulated food and pharma packaging where failures carry high cost.
How Do Brazil’s Governance and Healthcare Packaging Needs Support Growth?
Brazil’s 12.1% CAGR is supported by compliance-driven packaging demand across food and healthcare supply chains, where formal regulatory pathways for food-contact and pharma-adjacent packs increase the value of qualified materials and traceable supplier documentation. ANVISA’s legislative framework includes rules governing plastic packaging and materials intended to come into contact with food, which creates compliance gates that favour suppliers capable of supporting documented formulations and test evidence.
As packaged food penetration rises in urban centres and healthcare distribution expands, converters and brand owners increasingly prefer packaging structures that reduce failure risk and can be qualified across plants. Peel sheets benefit in these systems because controlled opening and seal integrity are operationally linked to product safety, shelf-life, and consumer usability. These drivers support steady adoption of peelable packaging in food packs, pharmaceutical secondary packaging, and medical device packaging formats where process stability and compliance readiness matter.
Why Does the United States Maintain Strategic Importance for Poly Peel Sheets?
The United States grows at an 11.0% CAGR because regulatory authorisation pathways and sterile packaging validation requirements industrialise packaging material selection, favouring suppliers that can operate inside defined compliance systems. FDA’s packaging and food contact substances program links materials used in manufacturing, packing, and packaging food to documented authorisations and inventories, reducing tolerance for undocumented chemistry shifts in peelable sealants and coatings.
In regulated healthcare packs, FDA-recognised consensus standards include ISO 11607 validation requirements for medical device packaging forming and sealing processes, reinforcing demand for peel structures that behave predictably across validated processes. These governance mechanisms support continued demand growth in high-throughput food packaging, pharmaceutical packaging, and sterile medical device packs, while also driving supplier consolidation as compliance overhead increases the advantage of scaled operators.
What Sustains the United Kingdom’s Moderate but Durable Growth Path?
The UK expands at a 9.8% CAGR as packaging governance formalises reporting and responsibility structures, pushing the market toward standardised, traceable packaging formats and disciplined supplier documentation. UK guidance on extended producer responsibility for packaging defines reporting and obligation expectations, increasing administrative overhead for packaging placed on the market and incentivising packaging simplification.
This governance pressure supports peel sheet demand where controlled opening and seal integrity remain essential in food and healthcare packaging, but it also limits uncontrolled SKU proliferation because compliance reporting and specification maintenance costs rise. The net effect is steady growth concentrated in packaging programs where performance is non-discretionary, and where suppliers can support documentation and transition management during packaging redesign cycles.
Why Is Japan’s CAGR Lower Despite High Packaging Quality Standards?
Japan’s 8.7% CAGR reflects high compliance discipline and mature packaging quality standards that reduce rapid format churn while sustaining steady demand in regulated food and healthcare packs. Japan’s food-contact governance includes the positive list system for utensils, containers, and packaging, which structures material selection and slows aggressive chemistry substitution without documented compliance.
This mechanism supports reliable demand for qualified peel sheet structures but moderates growth because packaging changes require careful regulatory and technical alignment. Japan also has mature converter ecosystems and strong incumbent procurement relationships, which can limit fast supplier switching even when global suppliers hold leadership elsewhere. As a result, global scale leadership does not always translate directly into share acceleration in Japan, where qualification discipline and domestic supply alignment shape adoption.
How is the Competition in the Poly Peel Sheets Market?
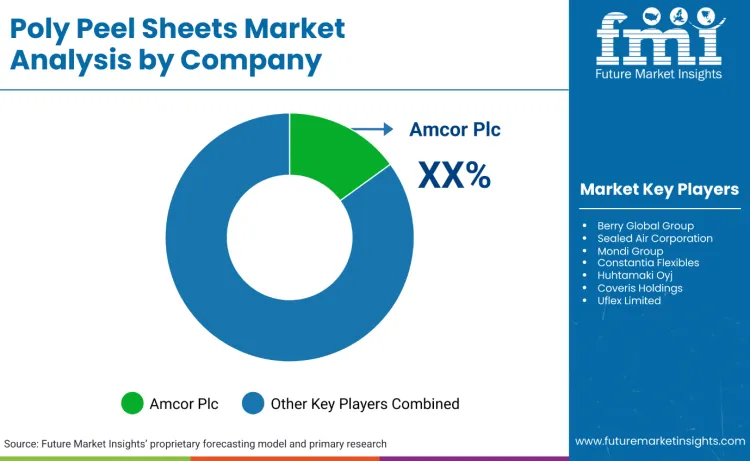
Competition is defined by ability to supply peelable polymer sheet and film structures used in food, pharmaceutical, medical device, and consumer goods packaging systems, including single sided and double sided peel formats, pressure-sensitive peel layers, and heat-sealable peel structures. Excluded are non-peel flexible films without controlled opening functionality, paper-only lidding, aluminium foil structures that do not rely on polymer peel layers, and packaging machinery. Amcor is the largest player globally at 12.8% share, strengthened by its completed combination with Berry Global on 30 April 2025, which expanded scale and breadth across flexible packaging and engineered materials.
North America remains scale-led by Amcor, Berry legacy assets, and regional specialists such as ProAmpac and Winpak. Europe is shaped by governance pressure from PPWR and by strong converter platforms including Mondi, Constantia Flexibles, and Coveris, where redesign capacity and compliance documentation are decisive. Asia is split between global platforms and regional leaders such as Uflex, while Japan behaves differently because its positive list governance and qualification discipline increase the advantage of established regulatory operators, limiting simple scale translation from global leaders.
Recent Developments
- Amcor completed its combination with Berry Global on 30 April 2025, expanding scale in flexible packaging and engineered material solutions relevant to peel sheet supply programs.
- The EU Packaging and Packaging Waste Regulation entered into force on 11 February 2025 and will apply from 12 August 2026, accelerating redesign cycles for peel structures in European packaging.
- Constantia Flexibles completed its acquisition of Aluflexpack on 4 March 2025, expanding its flexible packaging footprint across food and pharma-adjacent structures where peel systems are frequently specified.
Key Players
- Amcor Plc
- Berry Global Group
- Sealed Air Corporation
- Mondi Group
- Constantia Flexibles
- Huhtamaki Oyj
- Coveris Holdings
- Uflex Limited
- Winpak Ltd.
- ProAmpac
Market Definition
Poly peel sheets are peelable polymer sheet and film structures used to create controlled-open packaging systems across food, pharmaceutical, medical device, personal care, and industrial packs. They enable predictable opening force, seal integrity, and process stability on packaging lines. The market includes polyethylene, polypropylene, polyester, and composite peel sheet designs, including heat-sealable and pressure-sensitive peel formats. It is supplier- and compliance-driven, not brand-driven. Amcor is the largest global player at 12.8% share, while regional leadership differs due to governance and converter ecosystems, and global leadership does not always translate into Japan, where qualification discipline shapes supplier choice.
Market Inclusion
Included are polymer peel sheets and peelable film structures supplied as lidding, sealant, or peel-interface layers used in packaging systems that require controlled opening and verified sealing. This covers single-sided and double-sided peel sheets, heat-sealable peel structures, pressure-sensitive peel layers, and multilayer composites where peel behaviour is a designed functional requirement. Included end uses span food packaging, pharmaceutical packaging, medical device sterile barrier systems, personal care packaging, and industrial packaging where peel access is specified. Included channels cover direct sales to converters and brand owners, and distributor-led supply programs that provide documentation, inventory continuity, and qualification support.
Market Exclusion
Excluded are flexible films and sheets that do not provide engineered peel-open functionality, including standard barrier films without controlled peel layers. Excluded are paper-only lids and closures, aluminium foil packs where opening is driven by foil tear mechanics rather than polymer peel interfaces, and rigid packaging formats not using peel sheet structures. Packaging machinery, sealing equipment, and tooling are excluded even when used with peel materials. Labels, tapes, and non-packaging industrial release liners are excluded unless they function as peel interfaces in a defined packaging system. Services such as testing labs and regulatory consulting are excluded from market value estimates.
Scope of Report
| Attributes | Details |
|---|---|
| Quantitative Units | Value (USD Billion), CAGR (2026 to 2036) |
| Forecast Value (2036) | USD 9.8 Billion |
| CAGR (2026 to 2036) | 11.5% |
| Application Segments | Material Type, Product Type, End Use Industry, Distribution Channel, End User |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan |
| Key Companies Profiled | Amcor, Berry, Sealed Air, Mondi, Constantia Flexibles, Huhtamaki, Coveris, Uflex, Winpak, ProAmpac |
| Additional Attributes | Governance and compliance mapping, recyclability constraint analysis, food-contact authorisation and validation discipline signals, converter capacity concentration assessment, channel dependency and distributor qualification role, corporate consolidation tracking |
Poly Peel Sheets Market by Key Segments
By Material Type:
Polyethylene Peel Sheets, Polypropylene Peel Sheets, Polyester Peel Sheets, Multi Layer Composite Peel Sheets
By Product Type:
Single Sided Peel Sheets, Double Sided Peel Sheets, Heat Sealable Peel Sheets, Pressure Sensitive Peel Sheets
By End Use Industry:
Food Packaging, Pharmaceutical Packaging, Medical Device Packaging, Personal Care Packaging, Industrial Packaging
By Distribution Channel:
Packaging Distributors, Direct Sales, Online B2B Platforms
By End User:
Food Manufacturers, Pharmaceutical Companies, Medical Device Manufacturers, Consumer Goods Companies
Bibliography
- Agência Nacional de Vigilância Sanitária. (2025). Resolução RDC 961/2025 on plastic materials intended for food contact (Legislative database).
- Amcor plc. (2025, April 30). Amcor completes combination with Berry Global [Press release].
- Electronic Code of Federal Regulations. (n.d.). Submission of a premarket notification for a food contact substance (21 CFR 170.100).
- European Commission. (2025). Packaging and Packaging Waste Regulation: Entry into force and application timeline.
- Food Safety and Standards Authority of India. (2025). Compendium: Food safety and standards (packaging) regulations, 2018 (Updated).
- National Health Commission of the People’s Republic of China. (2023). Announcement issuing food safety national standards (2023 No. 6).
- USA Food and Drug Administration. (2024). Packaging and food contact substances (FCS).
Frequently Asked Questions
How big is the poly peel sheets market in 2026 and 2036?
The poly peel sheets market is valued at USD 3.3 billion in 2026 and is projected to reach USD 9.8 billion by 2036, expanding at an 11.5% CAGR.
Which material type leads the poly peel sheets market?
Polyethylene peel sheets lead with a 57% share, reflecting recyclability-driven redesign and broad compliance acceptance for food-contact packaging systems.
Which product type holds the largest share in poly peel sheets?
Single sided peel sheets lead with a 41.3% share, supported by line-efficiency economics and simplified validation and change-control burden.
Which end use industry dominates demand for poly peel sheets?
Food packaging leads with a 38.5% share, as high-throughput food packs require repeatable seal integrity and controlled opening within regulated food-contact frameworks.
What is the key commercial constraint for poly peel sheets scale-up?
Packaging EPR obligations and tightening recyclability rules increase redesign frequency and documentation load, which can slow adoption in cost-sensitive segments even when performance benefits are clear.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Sustainability Filings
- Packaging Industry Journals and Technical Publications
- Corporate Websites and Product Specification Sheets
- Investor Presentations and Earnings Transcripts
- Regulatory Filings and Standards Documentation
- Technical White Papers and Validation Notes
- Trade Journals and Packaging Magazines
- Conference Proceedings and Industry Webinars
- Government Statistics and Customs Data
- Press Releases and Verified Media Coverage
- Specialist Packaging Newsletters
- Sector Databases and Materials Repositories
- FMI Internal Knowledge Base
- Subscription-based Industry Databases
- Supply Chain and Trade Intelligence Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Validation Approach
- Why Primary Evidence is Used
- Field Techniques
- Converter and Brand-owner Interviews
- Procurement and Compliance Surveys
- Packaging Engineer Workshops
- Operational Observations
- Standards and Audit Interactions
- Stakeholder Universe Engaged
- C-suite Executives
- Packaging Procurement Heads
- Quality and Compliance Directors
- R&D and Materials Engineers
- Regulatory Affairs Specialists
- Packaging Validation Experts
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Material Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Material Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Material Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Y to o to Y Growth Trend Analysis By Product Type
- Absolute $ Opportunity Analysis By Product Type
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use Industry
- Y to o to Y Growth Trend Analysis By End Use Industry
- Absolute $ Opportunity Analysis By End Use Industry
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Y to o to Y Growth Trend Analysis By Distribution Channel
- Absolute $ Opportunity Analysis By Distribution Channel
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Y to o to Y Growth Trend Analysis By End User
- Absolute $ Opportunity Analysis By End User
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material Type
- By Product Type
- By End Use Industry
- By Distribution Channel
- By End User
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Poly Peel Sheets Market Key Takeaways
- Global Poly Peel Sheets Market Size, 2021 to 2036
- Poly Peel Sheets Market Size by Material Type
- Poly Peel Sheets Market Size by Product Type
- Poly Peel Sheets Market Size by End Use Industry
- Poly Peel Sheets Market Size by Distribution Channel
- Poly Peel Sheets Market Size by End User
- Poly Peel Sheets Market Size by Region
- Poly Peel Sheets Market CAGR by Country
- Scope of Report
List of Figures
- Poly Peel Sheets Market Growth Trajectory, 2026 to 2036
- Governance-led Packaging Redesign Framework
- Material Type Share Analysis
- Product Type Share Distribution
- End Use Industry Mix Analysis
- Regional Market Attractiveness Analysis
- Competitive Landscape Overview

