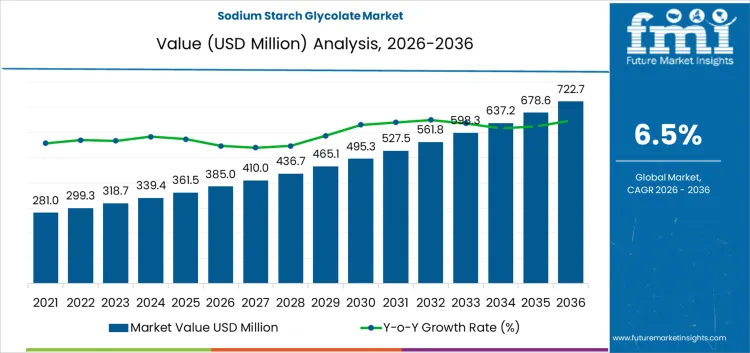
The sodium starch glycolate market is likely to be valued at USD 385.0 million in 2026 and is projected to reach USD 720.0 million by 2036 at a CAGR of 6.5%. Value behavior is shaped by its function as a critical super-disintegrant within solid oral dosage manufacturing rather than a commodity excipient. Demand formation reflects reliance on rapid and reproducible tablet disintegration to meet bioavailability and dissolution requirements. Capital allocation by pharmaceutical manufacturers prioritizes excipients with consistent swelling capacity and compatibility with high-speed compression processes. Purchasing decisions align with pharmacopeial acceptance, supplier qualification depth, and sensitivity of finished dosage performance to disintegration variability. Demand persistence concentrates within immediate-release tablet portfolios and high-volume generic production where formulation robustness directly affects regulatory outcomes.
Manufacturing and supply economics are influenced by variability in starch feedstock characteristics and control over chemical modification processes. Inconsistent substitution levels affect swelling index and disintegration time reproducibility, increasing formulation risk. Tight regulatory oversight requires extensive validation, documentation, and change control, raising compliance costs for suppliers. Multi-pharmacopeia alignment increases analytical testing burden across regions. Moisture sensitivity during storage necessitates controlled packaging and logistics conditions. Agricultural input price fluctuations introduce cost volatility. Limited pools of approved suppliers restrict sourcing flexibility for regulated products. Reformulation barriers discourage rapid supplier changes, slowing scalability across geographically distributed pharmaceutical manufacturing networks.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 385.0 million |
| Market Forecast Value (2036) | USD 720.0 million |
| Forecast CAGR 2026 to 2036 | 6.5% |
Demand for sodium starch glycolate is increasing as pharmaceutical manufacturers emphasize formulation performance for orally disintegrating tablets and immediate release products. This excipient enhances water uptake and promotes rapid disintegration, supporting consistent dissolution profiles required for a range of active pharmaceutical ingredients. Regulatory expectations for bioavailability performance and reproducible manufacturing outcomes drive formulators to select reliable super-disintegrants that integrate with high speed processing equipment. Contract development and manufacturing organizations incorporate sodium starch glycolate in formulation libraries to address diverse client projects that require robust excipient functionality. Growth in chronic disease treatment portfolios and broader access to generic medications maintain ongoing requirements for performance optimized tablet formulations that benefit from this excipient’s properties.
Demand from nutritional supplement producers supports market expansion because product developers seek tools that improve consumer convenience without compromising ingredient stability. Veterinary pharmaceutical sectors use sodium starch glycolate in solid dosage forms designed for animals, where rapid disintegration supports ease of administration. Upstream suppliers scale production capacity and refine quality control systems to meet stringent pharmacopeial standards demanded by global markets. Research labs evaluating novel disintegrant combinations require consistent batches to compare performance metrics. Expanded distribution networks and supply chain resilience planning help ensure availability for formulation activities across regions.
Demand for sodium starch glycolate increases due to expanding solid dosage production and emphasis on consistent drug release performance. Formulation developers prioritize excipients that ensure rapid disintegration without compromising tablet integrity. Regulatory scrutiny drives preference for well characterized functional additives across regulated manufacturing environments. Growth in oral dosage pipelines and scale up of generic production further reinforce sustained utilization across industrial formulation processes.
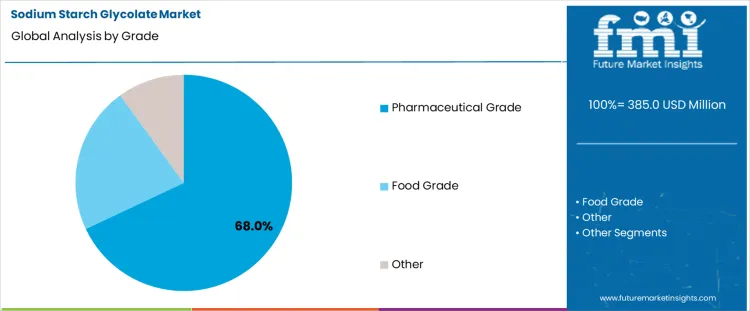
Pharmaceutical grade accounts for a 68.0% share, reflecting dominant use within regulated solid dosage manufacturing. This grade meets strict purity, particle size, and performance specifications required for drug formulations. Consistent swelling behavior ensures predictable tablet breakup under gastrointestinal conditions. Manufacturers rely on validated excipient quality to support regulatory filings and batch reproducibility. High volume tablet production amplifies dependence on pharmaceutical grade material across multiple therapeutic categories.
Key Points
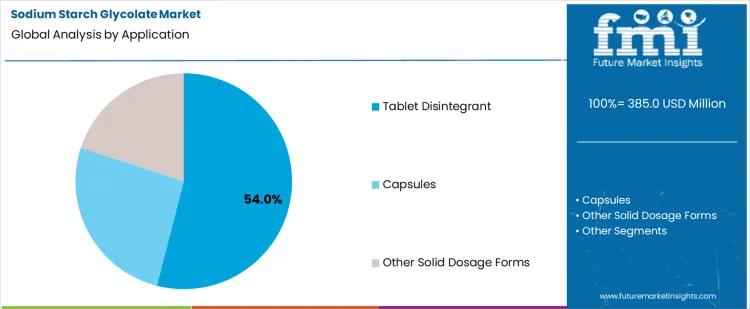
Tablet disintegrant applications hold a 54.0% share due to central importance in oral solid dosage performance. Sodium starch glycolate enables rapid water uptake and swelling within compressed matrices. Effective disintegration supports timely drug release and bioavailability expectations. Compatibility with diverse active ingredients sustains widespread formulation adoption. High tablet production volumes across prescription and generic portfolios reinforce sustained demand concentration within this application.
Key Points
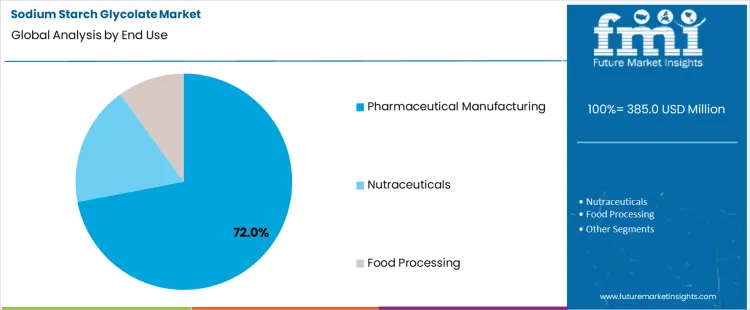
Pharmaceutical manufacturing represents a 72.0% share, reflecting concentration of solid dosage production activity. Large scale facilities require consistent excipient supply to maintain throughput and quality control. Compliance driven processes prioritize excipients with established performance histories. Integration into standardized formulation workflows sustains recurring consumption. Expansion of generic drug manufacturing further reinforces reliance on sodium starch glycolate within pharmaceutical production environments.
Key Points
Demand dynamics in the sodium starch glycolate market reflect formulation performance requirements within pharmaceutical solid dosage manufacturing. Global activity centers on excipient reliability, regulatory compliance, and tablet disintegration efficiency. Value formation depends on particle morphology, swelling capacity, and batch consistency. Purchasing decisions align with pharmacopeial acceptance, supplier qualification depth, and compatibility with high-speed tablet compression processes.
Adoption increases as pharmaceutical manufacturers prioritize rapid and predictable tablet disintegration across immediate-release formulations. Demand strengthens where high drug loading and low active solubility require efficient superdisintegrant functionality. Sodium starch glycolate supports uniform dispersion through controlled swelling behavior under gastric fluid exposure. Process robustness under direct compression and wet granulation enhances suitability for large-scale production. Regulatory filings favor excipients with extensive pharmacopeial monographs and historical safety data. Compatibility with commonly used binders, lubricants, and active ingredients reduces formulation redevelopment risk. Manufacturing efficiency improves through reduced compression force sensitivity and stable performance across humidity ranges, supporting consistent tablet quality during high-throughput production campaigns.
Market scalability remains constrained by variability in starch feedstock quality and chemical modification control. Demand encounters pressure where substitution consistency affects swelling index and disintegration time reproducibility. Tight regulatory oversight requires extensive documentation, validation, and change control for excipient suppliers. Compliance with multiple pharmacopeias increases testing and certification cost. Moisture sensitivity during storage and handling elevates packaging and logistics requirements. Price exposure to agricultural input fluctuations impacts cost stability. Limited approved supplier pools restrict sourcing flexibility for regulated drug products. Reformulation barriers discourage rapid supplier switching, slowing volume expansion across geographically distributed manufacturing sites.
Global demand for sodium starch glycolate is increasing due to expanding solid oral dosage manufacturing and formulation standardization across pharmaceutical supply chains. Growth reflects reliance on superdisintegrants to ensure bioavailability, batch consistency, and regulatory compliance. Capacity additions in generics, contract manufacturing expansion, and tablet complexity are reinforcing recurring consumption. India records 7.8% CAGR, China records 7.2% CAGR, the USA records 6.1% CAGR, Germany records 5.6% CAGR, and Japan records 5.2% CAGR. Adoption remains tied to pharmacopeial acceptance, excipient quality assurance, and scaling of high volume tablet production globally.
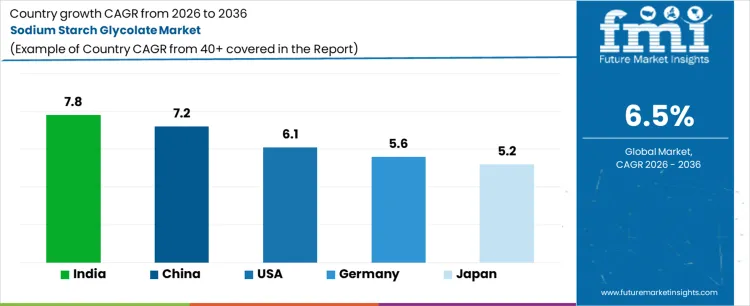
| Country | CAGR (%) |
|---|---|
| India | 7.8% |
| China | 7.2% |
| USA | 6.1% |
| Germany | 5.6% |
| Japan | 5.2% |
Demand for sodium starch glycolate in India is rising as generic drug manufacturing expands across domestic and export oriented facilities. Growth at 7.8% CAGR reflects increased tablet production volumes requiring reliable disintegration performance. Contract manufacturing organizations scale capacity, reinforcing standardized excipient procurement. Regulatory scrutiny on dissolution and bioequivalence strengthens preference for pharmacopeia compliant grades. Local availability of starch feedstock supports cost competitive production. Export demand to regulated markets drives quality documentation and consistency requirements. Adoption concentrates in immediate release formulations across antibiotics, analgesics, and chronic therapy drugs. Replacement demand aligns with formulation optimization and line expansion activities.
Sodium starch glycolate demand in China is expanding as pharmaceutical manufacturing modernizes and quality standards tighten. Growth at 7.2% CAGR aligns with increased production of oral solid dosage forms for domestic consumption. Regulatory reforms elevate expectations for excipient performance and traceability. Local manufacturers invest in upgraded processing and quality systems. Adoption supports formulation consistency across large batch operations. Export focused producers adopt internationally accepted grades to meet overseas audit requirements. Demand strengthens within hospitals and retail medicine supply chains. Integration with automated tableting lines increases usage intensity across industrial scale facilities.
Sales of sodium starch glycolate in the USA are increasing as pharmaceutical companies emphasize formulation reliability and regulatory adherence. Growth of 6.1% CAGR reflects sustained production of branded generics and specialty tablets. Procurement prioritizes excipients with consistent particle size and rapid disintegration profiles. Strong regulatory oversight reinforces validated supplier relationships. Demand emerges from reformulation efforts addressing bioavailability and dissolution challenges. Limited domestic manufacturing sustains import reliance. Contract development and manufacturing organizations drive recurring consumption. Replacement cycles align with portfolio refresh and lifecycle management strategies across mature drug products.
Sodium starch glycolate demand in Germany is shaped by pharmaceutical quality standards and controlled manufacturing practices. Expansion at 5.6% CAGR reflects steady production of prescription and specialty medicines. Buyers emphasize documentation, traceability, and compliance with European pharmacopeial requirements. Adoption favors high purity grades suitable for regulated markets. Limited tolerance for variability constrains rapid volume scaling. Demand concentrates within established pharmaceutical manufacturers and CDMOs. Sustainability and supply chain transparency influence supplier selection. Replacement demand follows equipment modernization and incremental formulation optimization rather than capacity driven expansion.
Sodium starch glycolate market demand in Japan is advancing through disciplined pharmaceutical manufacturing expansion. Growth at 5.2% CAGR reflects focus on high quality oral dosage formulations and patient safety. Manufacturers prioritize excipients with proven stability and reproducibility. Regulatory frameworks emphasize rigorous validation and consistency. Domestic production remains limited, supporting import dependence. Demand concentrates in chronic therapy medications and aging population treatments. Adoption remains incremental due to conservative formulation change practices. Replacement demand aligns with periodic quality upgrades and compliance driven process reviews across pharmaceutical plants.
Demand for sodium starch glycolate is driven by solid oral dosage growth, generic drug production, and formulation performance requirements. Buyers evaluate disintegration efficiency, particle size control, moisture sensitivity, compendial compliance, and consistency across manufacturing batches. Procurement behavior prioritizes qualified suppliers with validated regulatory documentation, reproducible quality, and reliable global supply continuity. Purchasing decisions emphasize audit readiness, change control discipline, technical formulation support, and predictable long term pricing structures. Trend in the sodium starch glycolate market reflects increased tablet complexity, higher excipient functionality expectations, and tighter quality standardization across regulated pharmaceutical manufacturing environments.
Roquette leads competitive positioning through vertically integrated starch processing, pharmaceutical grade portfolios, and extensive regulatory acceptance. DFE Pharma supports demand with co processed excipient expertise, application driven development, and strong relationships with generic manufacturers. JRS Pharma competes through cellulose and starch based excipient specialization, technical service depth, and consistent batch performance. Colorcon maintains relevance by combining formulation support services with excipient distribution and coating system integration. Ashland participates through specialty excipient offerings aligned with controlled release and performance focused pharmaceutical applications. Competitive differentiation centers on quality assurance rigor, regulatory credibility, supply reliability, and formulation collaboration capability.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Grade | Pharmaceutical Grade; Food Grade; Other |
| Application | Tablet Disintegrant; Capsules; Other Solid Dosage Forms |
| End Use | Pharmaceutical Manufacturing; Nutraceuticals; Food Processing |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, Japan, and 40+ countries |
| Key Companies Profiled | Roquette; DFE Pharma; JRS Pharma; Colorcon; Ashland |
| Additional Attributes | Dollar sales by grade, application, end use, and sales channel; disintegration efficiency and swelling capacity influencing tablet performance; particle size distribution and flow properties affecting compression behavior; compliance with pharmacopeial standards across regulated markets; sourcing consistency and batch-to-batch uniformity; procurement patterns spanning direct pharmaceutical manufacturer contracts, excipient distributors, and formulation development partnerships. |
How big is the sodium starch glycolate market in 2026?
The global sodium starch glycolate market is estimated to be valued at USD 385.0 million in 2026.
What will be the size of sodium starch glycolate market in 2036?
The market size for the sodium starch glycolate market is projected to reach USD 722.7 million by 2036.
How much will be the sodium starch glycolate market growth between 2026 and 2036?
The sodium starch glycolate market is expected to grow at a 6.5% CAGR between 2026 and 2036.
What are the key product types in the sodium starch glycolate market?
The key product types in sodium starch glycolate market are pharmaceutical grade, food grade and other.
Which application segment to contribute significant share in the sodium starch glycolate market in 2026?
In terms of application, tablet disintegrant segment to command 54.0% share in the sodium starch glycolate market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.