Suture Sleeve Kit Market
The suture sleeve kit market is segmented by End user (Hospitals, Ambulatory Surgical Centers, Diagnostics Laboratories, Clinics), Procedure Type (Pacemakers, Defibrillators, CRT Devices), Product Type (Standard Sleeves, Silicone Sleeves, Split Sleeves), Sales Channel (OEM Bundles, Direct Tenders, Distributors), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Suture Sleeve Kit Market Size, Market Forecast and Outlook By FMI
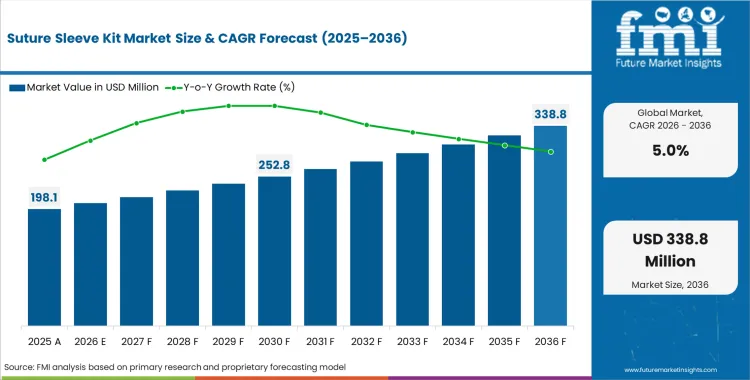
The suture sleeve kit market stood at USD 188.7 million in 2025 and is estimated at USD 198.1 million in 2026. The market is projected to rise at a CAGR of 5.0% from 2026 to 2036 and reach USD 322.7 million by the end of the forecast period. The outlook is being supported by tighter fixation expectations in implant procedures, a steady base of replacement activity, and growing emphasis on reducing avoidable lead-related complications.
Summary of Suture Sleeve Kit Market
- Market Snapshot
- The Suture Sleeve Kit Market is valued at USD 188.7 million in 2025 and is projected to reach USD 322.7 million by 2036.
- The industry is expected to expand at a 5.0% CAGR from 2026 to 2036, with the market reaching USD 198.1 million in 2026 as the forecast base year.
- This remains a procedure-linked cardiac consumables market where purchasing is shaped by implant volume, kit standardization, physician preference, and compatibility with established cardiac rhythm management workflows.
- Demand stays closely tied to pacemaker, defibrillator, and CRT implantation procedures, since suture sleeve kits are used to secure leads during device placement.
- Demand and Growth Drivers
- Growth is being supported by the steady expansion of cardiac implantable electronic device procedures, which creates recurring demand for lead-fixation accessories used during implantation.
- Hospitals continue to account for the largest share of usage because most pacemaker and defibrillator procedures still move through hospital electrophysiology labs and cardiac intervention settings.
- Demand is also supported by broader adoption of standardized implant kits, where providers prefer accessories that reduce handling variation during lead positioning and fixation.
- Market expansion remains moderate rather than aggressive because suture sleeve kits are not independently adopted capital products; they rise with procedure throughput and device placements.
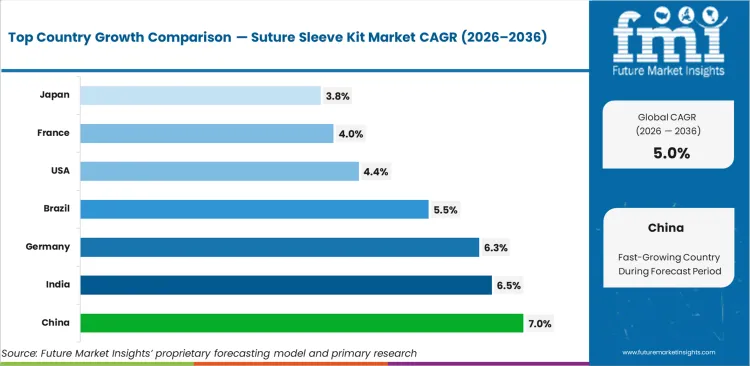
- Among key countries, China leads at 7.0% CAGR, followed by India at 6.5%, Germany at 6.3%, and Brazil at 5.5%, while France, the United States, and Japan reflect a slower but commercially relevant expansion profile.
- Product and Segment View
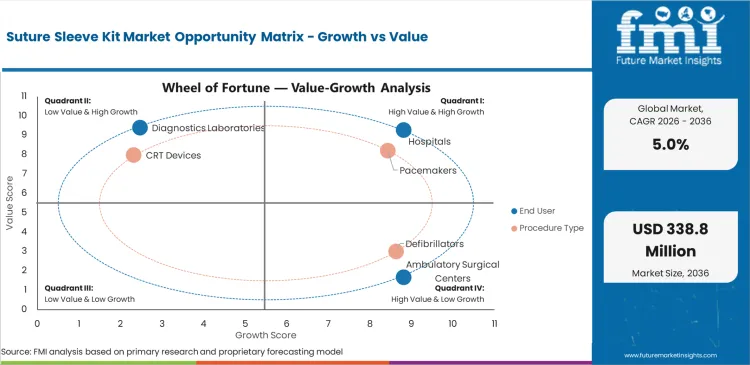
- The market is led by Hospitals, which hold 60.0% share in 2026 as cardiac implant procedures remain concentrated in hospital-based care environments with established CRM infrastructure.
- By procedure type, Pacemakers account for 46.0% share, reflecting their broad procedural base and the routine use of lead-securing accessories during implantation.
- By product type, Standard Sleeves hold 58.0% share because they fit conventional implant workflows and remain the most widely accepted format across routine lead-placement procedures.
- By sales channel, OEM Bundles lead with 64.0% share, indicating that procurement is often tied to broader device-system purchasing rather than stand-alone accessory sourcing.
- The remaining market structure includes ambulatory surgical centers, diagnostics laboratories, and clinics on the end-user side, along with defibrillators and CRT devices, silicone and split sleeves, and direct tenders and distributors across product and channel categories.
- Regional coverage includes North America, Europe, East Asia, South Asia, Latin America, Oceania, and the Middle East & Africa, with North America representing the most mature installed base.
- Geography and Competitive Outlook
- North America remains the mature value center of the market because of its established cardiac implant ecosystem, broad specialist access, and steady replacement procedure base.
- East Asia and South Asia present the faster expansion opportunity, supported by rising procedure access, broader hospital capacity, and improving penetration of implantable cardiac care.
- China and India stand out as the strongest-growth national markets, while Germany and Brazil add important regional demand depth through specialized cardiac treatment infrastructure and broader healthcare access.
- The United States, France, and Japan remain commercially important markets even with lower growth rates, as demand is supported by mature implant volumes and established hospital procurement channels.
- Competition is shaped by device portfolio strength, physician relationships, hospital access, implant workflow integration, and bundled sales capability, rather than by suture sleeve kits alone as isolated products.
- Key companies operating in the market include Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, and MicroPort CRM
Hospitals with large cardiovascular procedure volumes are placing greater emphasis on accessory standardization across implant programs. Cardiac rhythm management teams are under pressure to limit product variation because mixed kit usage complicates stocking, increases handling inconsistency, and creates avoidable friction inside electrophysiology labs. Differences in sleeve fit, material behavior, and compatibility can affect procedural reliability over time, especially when multiple implant platforms are used across the same network. Buyers are responding by favoring products that support clinical consistency and easier integration into established operating-room protocols rather than focusing only on small purchase-price differences.
Large institutions often set the pattern for broader market adoption once internal sizing norms and approved-product pathways are established. Regional centers tend to follow those specifications when contracts are renewed, physician preferences consolidate, or operating teams push for more predictable case preparation. That transition supports cleaner inventory planning, lowers substitution risk, and improves day-to-day execution in cardiac implantable electronic device procedures. Market momentum is therefore being shaped by standardization at the hospital-network level rather than by sharp changes in procedure demand alone.
Country-level growth remains differentiated, reflecting clear differences in procedure access, reimbursement support, implant penetration, and the degree of standardization across hospital networks. China stays at the front of the growth curve as electrophysiology infrastructure continues to expand, with demand expected to rise at a 7.0% CAGR through 2036. India follows with 6.5% CAGR, supported by broader cardiac care capacity and wider access to implant-based treatment across large hospital systems. Germany is projected to grow at 6.3%, where established reimbursement and consistent procedure volumes keep demand on firm footing. Brazil is set to record 5.5% CAGR as diagnostic access improves and referral networks become more reliable across the care pathway. The United States is expected to post 4.4%, with growth shaped more by replacement demand than by a meaningful rise in first-time procedures. France and Japan are forecast to expand at 4.0% and 3.8% respectively, reflecting mature treatment pathways and a slower build in incremental procedure volumes.
Segmental Analysis
Suture Sleeve Kit Market Analysis by End User
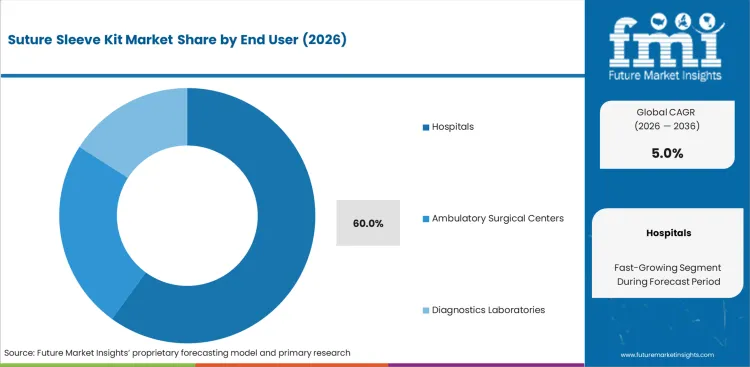
Cardiology departments evaluate fixation supplies through procedural volume, inventory standardization, and revision-risk control. Hospitals handle the widest base of routine cardiac implant procedures, which gives them a clear purchasing advantage in this market. High-throughput settings also need dependable replenishment cycles to avoid delays in scheduled interventions. The hospitals segment is estimated to account for 60.0% share in 2026. Acquisition teams at tertiary care centers usually favor unified vendor contracts because they simplify validation across multiple pacing platforms and support ambulatory surgical center workflows where speed and compatibility matter. Weak anchoring performance raises the probability of lead revision, and that cost exposure carries both financial and clinical consequences under tighter care-quality benchmarks. Hospitals are expected to remain the volume center of demand, while outpatient cardiac settings continue to influence product design by pushing suppliers toward faster deployment and easier handling.
- Decision trigger: Clinical administrators move toward bulk purchasing after quarterly audits show elevated revision rates or sleeve-related handling concerns.
- Validation metric: Electrophysiology lab managers test sleeve fit against existing catheter profiles during vendor review, and failed compatibility typically removes a supplier from long-term consideration.
- Renewal condition: Supply executives extend contracts only when manufacturers can assure uninterrupted delivery, since shortages force an immediate shift toward alternate vendors serving ambulatory surgical center demand.
Suture Sleeve Kit Market Analysis by Procedure Type
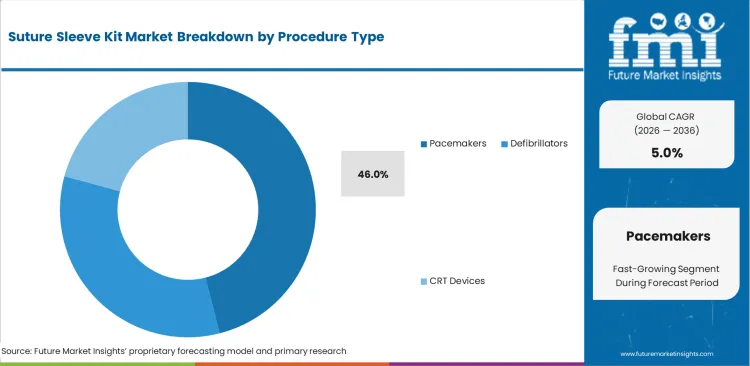
Pacemakers remain the procedural base of this market because bradycardia management continues to represent the largest routine implantation volume across aging patient populations. Electrophysiologists performing standard implant procedures usually stay with familiar fixation methods because established workflows reduce procedural variability and support predictable outcomes. That preference keeps demand concentrated around conventional sleeve usage in high-volume pacing cases. Pacemakers are projected to account for 46.0% share in 2026. The volume lead of pacemakers should not obscure the commercial importance of adjacent segments, since temporary pacing leads and more complex interventions often require different sleeve formats with better margin potential. Suppliers focused only on routine pacemaker demand can miss higher-value opportunities in defibrillator and CRT-related accessory requirements. The segment stays volume-led, but profitability often shifts toward more specialized procedure categories.
- Procurement savings: Bulk sourcing for standard pacemaker procedures helps supply teams negotiate lower unit costs across large bradycardia-focused purchasing programs.
- Hidden operations: Complex dual-chamber procedures and temporary pacing leads often consume more anchoring components than baseline inventory models assume.
- Lifecycle calculation: Low-cost fixation parts may underperform over time, pushing cardiovascular teams toward more durable silicone options when assessing long-term implanted-device support needs.
Suture Sleeve Kit Market Analysis by Product Type
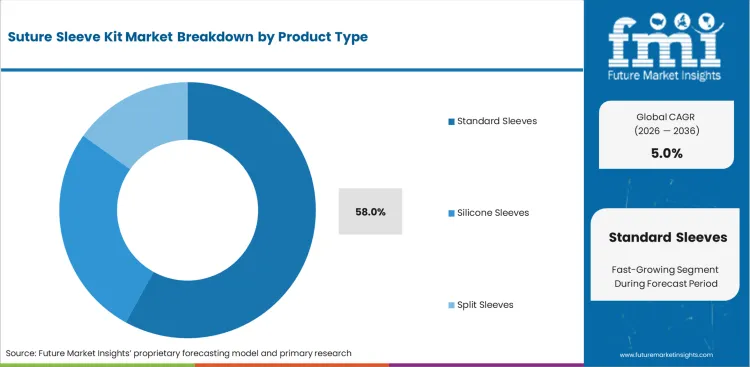
Clinical familiarity continues to slow the shift toward newer split-format designs. Senior electrophysiologists who trained on conventional tie-down methods often stay with standard sleeves because those techniques are deeply embedded in routine operating practice. Training teams also face resistance when they try to update fixation protocols across established surgical staff. Poorly managed product transitions can lead to inconsistent anchoring technique and raise the risk of early lead movement. Standard designs still benefit from broad acceptance, established usage patterns, and easier purchasing justification in conservative hospital settings. Newer split-silicone formats are gaining attention because they may reduce tissue adherence during later extraction procedures. For that reason, Standard Sleeves are anticipated to command 58.0% share in 2026. That leadership reflects current workflow preference more than a settled long-term technical advantage. Hospitals weighing future extraction burden alongside current implantation practice are likely to keep this product debate active, especially where angiographic catheters and related follow-up procedures expose downstream handling issues.
- Prevention capability: Standard sleeve configurations are widely used to reduce acute mechanical dislodgement risk during the immediate post-operative period.
- Residual risk: Excessive suturing force can still damage lead insulation, which keeps quality teams focused on technique consistency across operating shifts.
- Benefit capture: Maximum anchoring performance often depends on matching sleeve dimensions with the right tying tools, so purchase decisions frequently extend beyond the sleeve itself, particularly in procedures involving angiographic catheters.
Suture Sleeve Kit Market Analysis by Sales Channel
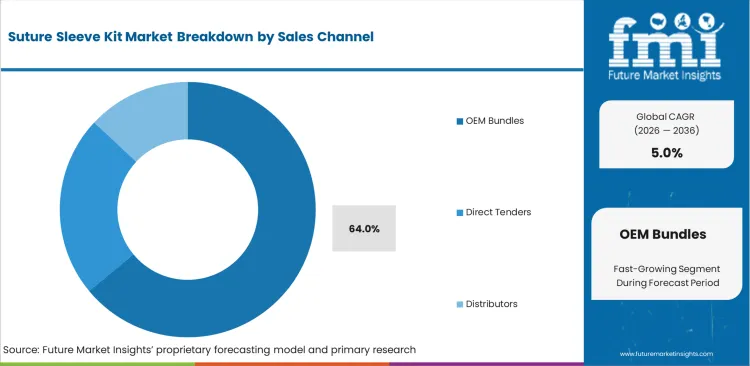
Sales channel dynamics are shaped by a basic tension between hospital sourcing preferences and OEM control over accessory ecosystems. Sourcing teams often want pricing flexibility and broader supplier choice, while large pacing manufacturers push bundled purchasing through existing generator contracts. Bundled supply remains attractive because it reduces qualification effort and lowers the chance of compatibility disputes during implantation. It also gives manufacturers a strong grip over repeat accessory demand and design specifications. OEM Bundles are likely to capture 64.0% share in 2026. That position reflects the commercial strength of integrated contracting rather than a purely product-level preference. Hospitals that step outside bundled arrangements usually face more administrative work, secondary validation steps, and added compatibility review for cardiovascular devices used alongside anchoring components. Independent suppliers can still compete, but most need distributor access or a clear technical advantage to overcome entrenched OEM purchasing patterns.
- Production control: Leading pacemaker manufacturers retain tight control over sleeve specifications and often adjust dimensions to match proprietary platform changes.
- Supply constraint: Independent producers remain more exposed to silicone shortages and raw material bottlenecks when they lack vertically integrated sourcing.
- Landscape evolution: Direct hospital tenders are expected to expand gradually as digital sourcing systems mature and buyers seek more flexibility across cardiovascular devices supply networks.
Suture Sleeve Kit Market Drivers, Restraints, and Opportunities
Rising revision surgery financial penalties compel hospital administrators to mandate premium anchoring solutions immediately. Delaying this critical quality upgrade exposes cardiovascular departments to unacceptable clinical complication rates. Electrophysiology directors now demand strong fixation mechanisms capable of surviving decades without degradation. Cheap components failing mid-lifecycle cause devastating surgical interventions. Administrators prioritizing patient safety proactively ban generic surgical ties from specialized electrophysiology suites entirely. Upgrading basic implantable defibrillator protocols represents low-cost insurance against massive long-term liability claims. This dynamic acts as one of the primary suture sleeve kit market growth drivers.
Platform-specific lead designs continue to restrict cross-compatibility in mixed-vendor hospital settings, leaving buyers with limited flexibility when assembling procedure kits. Hospitals often respond by carrying multiple sleeve SKUs for different OEM platforms, which adds avoidable pressure to inventory control and shelf-life monitoring. That burden becomes heavier as lead specifications change and accessory validation must be repeated. Independent suppliers face a clear disadvantage here, since each design revision can trigger fresh fit, safety, and performance testing. Larger pacing manufacturers retain an advantage by controlling lead dimensions within their own product ecosystems, making third-party compatibility harder to sustain over time. Smaller vendors rarely have the capital or testing continuity needed to keep pace with that cycle. Once compatibility risk enters the buying decision, demand tends to consolidate around OEM-bundled options, narrowing room for independent sleeve suppliers across the broader electrophysiology market.
Opportunities in the Suture Sleeve Kit Market
- Biodegradable integration: Material science advances allow temporary anchoring during initial fibrotic encapsulation phases. R&D directors developing resorbable sleeves eliminate long-term mechanical tissue irritation entirely, presenting a clear suture sleeve kit opportunity analysis pathway.
- Ambulatory expansion: Outpatient cardiac centers require highly streamlined procedural kits. Packaging engineers designing single-use sterile bundles specifically for operating room supplies workflows capture high-margin accounts providing suture sleeve kits for outpatient implant centers.
- Sensor-embedded tracking: Next-generation fixations incorporate passive telemetry capabilities. Clinical managers utilizing smart sleeves detect microscopic dislodgements before pacing failures occur, modernizing general electrophysiology lab consumables.
Regional Analysis
Based on regional analysis, Suture Sleeve Kit is segmented into North America, Europe, East Asia, South Asia, Latin America, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.0% |
| India | 6.5% |
| Germany | 6.3% |
| Brazil | 5.5% |
| United States | 4.4% |
| France | 4.0% |
| Japan | 3.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Suture Sleeve Kit Market Analysis
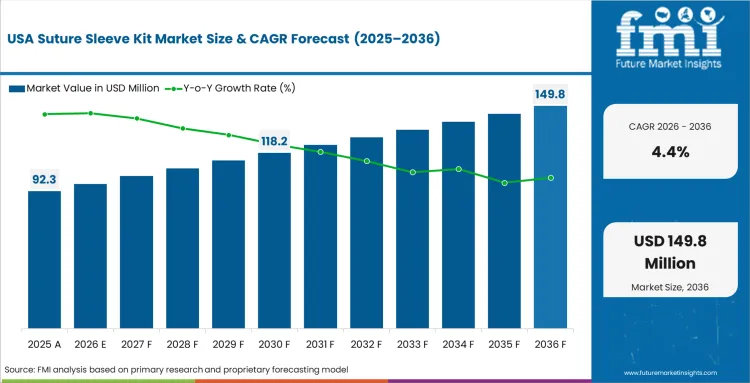
High operator density across major metropolitan medical centers guarantees stable demand year-round. Mature implant penetration limits first-time pacing expansion opportunities, shifting the focus to routine replacements. FMI analysts note that regional purchasing directors concentrate almost exclusively on securing favorable bulk pricing for these resupply cycles. Surgical teams rely heavily on familiar legacy OEM bundles, supported by established reimbursement frameworks. Delaying sourcing modernization slowly erodes operating margins for regional clinics.
- United States: Purchasing directors negotiating massive replacement contracts secure significant cost advantages across mature healthcare networks. Demand for suture sleeve kits in the United States is anticipated to rise at a CAGR of 4.4% through 2036. Advanced hospital networks completely dominate regional external pacemakers intervention volume. This established procedural throughput prioritizes premium component upgrades, solidifying the suture sleeve kit market in the United States.
FMI's report includes Canada. Comprehensive healthcare systems across northern territories prioritize unified national acquisition contracts.
Europe Suture Sleeve Kit Market Analysis
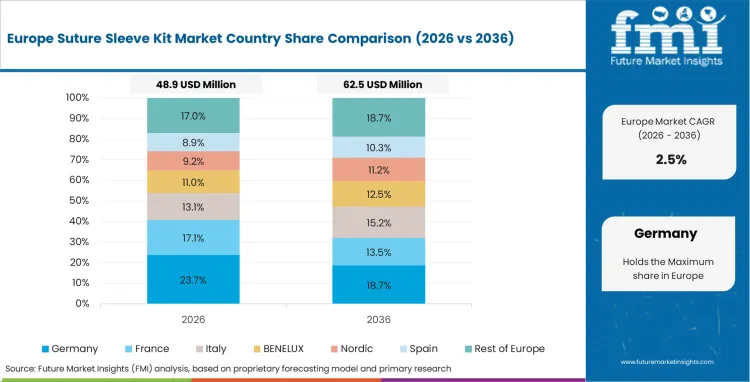
Strict clinical outcome tracking by regional hospital administrators effectively bans low-quality generic fixation devices from public health systems. Universal healthcare coverage ensures steady patient access to complex rhythm management therapies, while strong reimbursement structures dictate procedural throughput. Established domestic manufacturers heavily dominate regional supply chains because electrophysiology lab managers strongly prefer locally produced accessories. Navigating highly concentrated regulatory approvals remains critical for foreign entrants evaluating the pacemaker accessories market. FMI observes that these regulations inherently slow novel product introductions.
- Germany: Strict national reimbursement protocols reward cardiovascular departments demonstrating exceptionally low revision surgery rates. Clinical directors adopting premium silicone anchors achieve superior long-term patient outcomes to secure public funding. The suture sleeve kit industry in Germany is expected to grow at a CAGR of 6.3% during the study period. Sustained procedural excellence continually attracts lucrative cross-border cardiac rhythm management devices medical tourism.
- France: France is forecast to record steady growth in suture sleeve kits at a CAGR of 4.0% through 2036. Centralized public health purchasing mandates deep volume discounts across all cardiac accessory categories. Supply chain managers navigating these strict national tenders lock out smaller independent vendors completely. Dominant pacing conglomerates hold absolute commercial leverage, forcing hospitals to rely exclusively on established proprietary bundles.
FMI's report includes United Kingdom, Italy, and Spain. Expanding elderly populations maintain consistent demand for routine bradycardia interventions across southern territories.
East Asia Suture Sleeve Kit Market Analysis
Provincial health administrators prioritize aggressively expanding first-time implant access over premium device upgrades as previously underserved regional hospitals rapidly build new electrophysiology labs. According to FMI's estimates, these high patient volumes severely strain existing distribution networks. Domestic manufacturers rapidly scale production capacities to meet this exploding demand while technology transfer initiatives slowly elevate local manufacturing quality standards. Foreign conglomerates face pricing pressure from agile domestic producers, where securing provincial tender wins guarantees massive volume commitments.
- China: Provincial health directors building new cardiac centers rapidly consume massive domestic production volumes to support local care infrastructure. The suture sleeve kit segment in China is poised to expand at a CAGR of 7.0% through 2036. Agile domestic manufacturers capturing early regional automated external defibrillator tenders establish insurmountable commercial dominance. This exploding electrophysiology capacity creates unprecedented demand for basic cardiovascular accessories.
- Japan: Senior electrophysiologists fiercely defend established procedural workflows against disruptive novel technologies during complex multi-lead implantations. Highly advanced geriatric care networks demand ultra-reliable fixation components above all other considerations. Suture sleeve demand in Japan is likely to post a CAGR of 3.8% by 2036. Premium quality components completely monopolize domestic hospital purchasing budgets, leaving minimal opportunity for unproven independent vendor entry.
FMI's report includes South Korea. Advanced robotic surgery adoption accelerates demand for highly specialized automated deployment sleeves.
South Asia Suture Sleeve Kit Market Analysis
Growing middle-class populations demand sophisticated rhythm management therapies, forcing expanding hospital cardiac programs to rapidly decentralize advanced care access beyond major metropolitan hubs. As per FMI's projection, regional purchase directors aggressively balance premium OEM bundles against cost-effective domestic alternatives. Favorable government policies encourage local medical device manufacturing, though price sensitivity remains exceptionally high across private clinic networks. International pacing conglomerates must formulate highly localized pricing strategies, as local distribution partnerships dictate overall commercial success rates.
- India: Suture sleeve kit sales in India are poised to grow at a CAGR of 6.5% during the assessment period. Widening private hospital cardiac programs rapidly democratize access to advanced rhythm management procedures. Acquisition managers balancing tight budgets increasingly evaluate high-quality domestic silicone alternatives. Independent accessory manufacturers securing major private network contracts of the cardiac monitoring and cardiac rhythm management devices, unlock massive regional growth potential within this framework.
FMI's report includes ASEAN countries. Rising medical tourism across developing hubs create demand for internationally certified cardiovascular components.
Latin America Suture Sleeve Kit Market Analysis
Public health infrastructure investments gradually expand regional electrophysiology capabilities, slowly uncovering massive untreated bradycardia patient populations. Based on FMI's assessment, macroeconomic volatility forces supply chain managers to maintain highly flexible inventory protocols. Unpredictable currency fluctuations severely complicate international purchase contracts, causing regional cardiovascular centers to rely heavily on established global distribution networks. Navigating complex import regulations challenges independent accessory manufacturers because local production capabilities remain relatively constrained.
- Brazil: Clinical administrators managing surging patient volumes demand highly reliable bulk supply chains to support modernization. The suture sleeve kit segment in Brazil is anticipated to gain momentum, with demand rising at a CAGR of 5.5% over the forecast period. Global manufacturers navigating complex local import electrophysiology frameworks establish highly lucrative long-term supply monopolies. This expanding access funnels untreated patient backlogs directly into major urban centers.
FMI's report includes Mexico. Cross-border manufacturing investments slowly build strong local medical device production ecosystems.
Competitive Aligners for Market Players
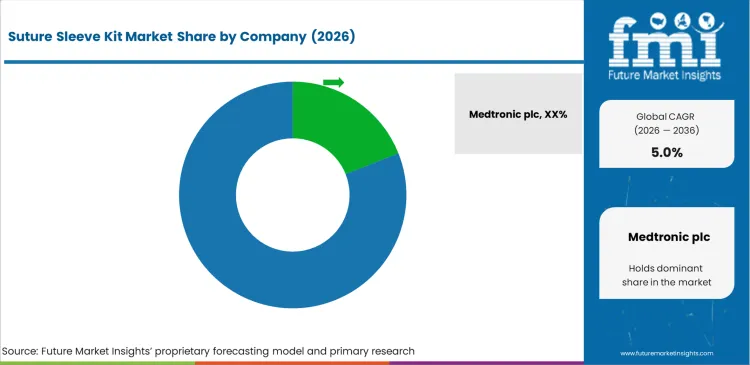
Major pacing manufacturers dictate competitive dynamics by tightly controlling peripheral accessory distribution networks. Top-tier conglomerates utilize massive pulse generator contracts to mandate proprietary sleeve usage across global hospital systems. Medtronic plc and Abbott Laboratories seamlessly embed fixation components within comprehensive catheter packages. Independent accessory vendors compete fiercely on unit price but struggle against established OEM bundling agreements. Hospital procurement directors consistently prioritize guaranteed technical compatibility over marginal cost savings. Supply chain consolidation further isolates independent producers lacking broad cardiovascular portfolios, limiting opportunities for specialized cardiac lead fixation accessory vendors.
Extensive clinical trial libraries constitute critical incumbent protection mechanisms. Established manufacturers possess decades of proven in-vivo stability data. New entrants cannot easily validate alternative silicone blends without conducting multi-year longitudinal studies. Clinical directors hesitate to risk patient safety on unproven anchoring technologies lacking substantial published evidence. Regulatory pathways demand rigorous predicate device comparisons. Independent producers must invest heavily in complex biomechanical cardiovascular diagnostics testing to achieve basic commercial viability. Strong intellectual property portfolios effectively block competitors from replicating optimal sleeve geometries, constantly challenging newer cardiac implant accessories suppliers.
Large cardiovascular networks combat vendor lock-in by aggressively demanding standardized split-sleeve geometries across all purchasing agreements. This resistance forces major manufacturers to slowly adopt universal dimensions despite heavily preferring closed product ecosystems. Purchase directors utilizing digital bidding platforms systematically dismantle opaque pricing structures. Independent producers finding niche traction in specialized pediatric applications will inevitably face aggressive acquisition strategies from dominant conglomerates dominating the suture sleeve kit landscape. Consolidation actively shrinks available distribution channels for standalone accessory manufacturers, prompting buyers to evaluate Boston Scientific vs BIOTRONIK pacing leads and weigh Medtronic vs Abbott pacing lead accessories.
Key Players in Suture Sleeve Kit Market
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- BIOTRONIK SE & Co. KG
- MicroPort CRM
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 198.1 million to USD 322.7 million, at a CAGR of 5.0% |
| Market Definition | Mechanical anchoring solutions designed exclusively for securing implantable cardiac leads at vascular entry points define scope. These critical components maintain permanent stability while preventing tissue damage over decades of implantation. |
| Suture Sleeve Kit Market Segmentation | By End user, By Procedure Type, By Product Type, By Sales Channel, and Region |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, United States, France, Japan |
| Key Companies Profiled | Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, MicroPort CRM |
| Forecast Period | 2026 to 2036 |
| Approach | Annual cardiac rhythm device implantation volumes |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Suture Sleeve Kit Market Analysis by Segments
End user
- Hospitals
- Ambulatory Surgical Centers
- Diagnostics Laboratories
- Clinics
Procedure Type
- Pacemakers
- Defibrillators
- CRT Devices
Product Type
- Standard Sleeves
- Silicone Sleeves
- Split Sleeves
Sales Channel
- OEM Bundles
- Direct Tenders
- Distributors
Region:
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
Bibliography
- Altaf, S., Witharana, T., Tindale, A., & Jones, D. G. (2025). Permanent pacemaker implantation complicated by anchoring sleeve embolization through cephalic vein: Case report and discussion of management strategies. European Heart Journal - Case Reports, 9(9), ytaf416.
- Bakytzhanuly, A., Otegen, T., & Nuralinov, O. (2024). New-generation leadless pacemaker implantation: First procedures in Central Asia. Interventional Cardiology, 19, e19.
- Biffi, M., Quaranta, A., Martignani, C., Carecci, A., & Boriani, G. (2026). Tips and tricks for extravascular implantable cardioverter-defibrillator implantation: A single-centre experience. Europace, 28(2).
- Chang, S., Jiang, Z., Liu, X., Tang, Y., Bai, M., Xu, J., Wang, H., Chen, Y., Li, C., Chen, Y., Dong, J., Luo, J., Li, J., Fu, G., Wang, S., Huang, H., Zhao, Y., Zhuang, X., Jilaihawi, H., & Song, G. (2024). Permanent pacemaker reduction using temporary-permanent pacemaker as a 1-month bridge after transcatheter aortic valve replacement: A prospective, multicentre, single-arm, observational study. EClinicalMedicine, 72, 102603.
- Gunes, M. T., Duman, S., Demir, D., & Simsek, E. (2025). Comparison of two suturing techniques in terms of lead stabilizing efficiency in acute and chronic terms. Journal of Interventional Cardiac Electrophysiology, 68(6), 1295-1306.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
Frequently Asked Questions
What is a suture sleeve kit?
A suture sleeve kit consists of mechanical anchoring solutions designed exclusively for securing implantable cardiac leads at vascular entry points. These critical components maintain permanent stability while preventing tissue damage over decades of implantation.
What is a suture sleeve used for in pacemaker implantation?
During pacemaker implantation, surgeons use suture sleeves to protect delicate lead insulation from crushing forces when tying the lead to the vein. The sleeve ensures the wire remains anchored securely without compromising its integrity.
How big is the suture sleeve kit market?
The overall industry valuation reached USD 188.7 million in 2025. Demand projections indicate the total valuation will expand to USD 322.7 million by 2036.
Explain the suture sleeve kit market to me?
This sector revolves around specialized silicone components used to secure pacemaker and defibrillator wires inside patients. Hospitals purchase these either independently or bundled directly with the expensive pulse generators from major medical device manufacturers.
Summarize the suture sleeve kit market outlook 2026 to 2036?
Sales are expected to reach USD 198.1 million in 2026 at a CAGR of 5.0%. Consistent investment propels cumulative revenue to USD 322.7 million by 2036 as stringent cardiovascular implant qualification cycles mandate tighter fixation tolerances.
Which countries are growing fastest in the suture sleeve kit market?
China tracks at 7.0% CAGR through rapidly expanding electrophysiology capacity. India follows closely at 6.5% as hospital cardiac programs widen. Germany registers 6.3% on strong reimbursement structures.
Who are the leading suture sleeve kit manufacturers?
Top-tier conglomerates including Medtronic plc, Abbott Laboratories, and Boston Scientific Corporation dominate production. These major pacing manufacturers dictate competitive dynamics by tightly controlling peripheral accessory distribution networks.
Why are hospitals the largest end users of suture sleeve kits?
Cardiology department heads weigh procedural volume against inventory complexity when approving fixation supplies. Hospitals capture massive routine patient loads, demanding ultra-reliable supply chains to prevent surgical scheduling disruptions.
Why does hospital demand dominate the suture sleeve kit market?
Massive baseline pacemaker implantation throughput occurs entirely within hospital settings. High-volume environments require universal compatibility and stable supply chains, making hospitals the absolute center of purchasing gravity.
Compare Medtronic Abbott and Boston Scientific in suture sleeve kit exposure?
These three giants seamlessly embed fixation components within comprehensive catheter packages. They utilize massive pulse generator contracts to mandate proprietary sleeve usage across global hospital systems, locking out independent vendors.
How do OEM bundles affect suture sleeve kit sales?
Global pacing conglomerates utilize massive generator contracts to strictly mandate proprietary accessory usage. Accepting OEM bundles guarantees absolute technical compatibility but effectively eliminates competitive pricing pressure for routine resupply orders.
Are suture sleeve kits sold separately from pacing leads?
Independent accessory vendors compete fiercely on unit price by selling sleeves separately. Hospital purchase directors consistently prioritize guaranteed technical compatibility through bundled contracts over marginal cost savings.
What increases suture sleeve kit demand in China?
Provincial health administrators rapidly build new basic cardiac intervention facilities across previously underserved territories. Exploding local electrophysiology capacity creates unprecedented demand for basic cardiovascular accessories.
How reimbursement affects suture sleeve kit demand?
Strict national reimbursement protocols reward cardiovascular departments demonstrating exceptionally low revision surgery rates. Clinical directors adopting premium silicone anchors achieve superior long-term patient outcomes, directly influencing purchasing tiers.
What is the suture sleeve kit procurement strategy for hospitals?
Supply chain directors calculate acceptable unit costs based directly upon historical silicone degradation failure data. Procurement must purchase matching accessory tools simultaneously to ensure flawless technical integration.
Explain Electrophysiology lab expansion market impact?
Enhancing hospital cardiac programs rapidly decentralize advanced care access beyond major metropolitan hubs. This infrastructure buildout directly translates into surging volume requirements for specialized anchoring components.
What is RFQ for pacing lead accessories?
Direct hospital tenders increasingly bypass traditional intermediaries as digital sourcing transparency matures. Strategic sourcing directors anticipate massive disintermediation across established cardiovascular supply networks when issuing new requests for quotation.
Explain hospital tender for suture sleeve kits.
Centralized public health purchasing mandates deep volume discounts across all cardiac accessory categories. Supply chain managers navigating strict national tenders lock out smaller independent vendors completely.
What is the pacemaker lead accessories market size?
The broader ecosystem encompassing these specific components closely tracks global pacing volumes. Industry data anchors firmly on the USD 188.7 million baseline established for dedicated sleeve kits.
What are cardiac catheterization suite supplies?
Specialized surgical environments require highly specific dimensional tolerances for cardiac leads. Administrators prioritizing patient safety proactively ban generic surgical ties from these suites entirely.
Explain pacemaker sleeves vs ICD sleeves?
Standard sleeves effectively prevent acute mechanical dislodgement during standard pacing procedures. Complex dual-chamber ICD implantations frequently demand specialized split sleeves to prevent mechanical wire cross-friction within the vein.
What is pacing lead suture sleeve market?
This specific segment commands majority volume due to large aging populations requiring standard bradycardia therapy. Routine cardiac rhythm maintenance relies heavily on proven historical fixation techniques.
What is suture sleeve kit market report?
Comprehensive evaluations incorporate direct primary interviews with cardiology directors to reveal hidden operational frictions. Baseline volume models anchor on verified annual cardiac rhythm device implantation figures to ensure accuracy.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User , 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Diagnostics Laboratories
- Hospitals
- Y to o to Y Growth Trend Analysis By End User , 2021 to 2025
- Absolute $ Opportunity Analysis By End User , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Procedure Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procedure Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procedure Type, 2026 to 2036
- Pacemakers
- Defibrillators
- CRT Devices
- Pacemakers
- Y to o to Y Growth Trend Analysis By Procedure Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Procedure Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Standard Sleeves
- Silicone Sleeves
- Split Sleeves
- Standard Sleeves
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- OEM Bundles
- Direct Tenders
- Distributors
- OEM Bundles
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End User
- By Procedure Type
- By Product Type
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Medtronic plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- Boston Scientific Corporation
- BIOTRONIK SE & Co. KG
- MicroPort CRM
- Medtronic plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Procedure Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End User
- Figure 6: Global Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Procedure Type
- Figure 9: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Product Type
- Figure 12: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Sales Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End User
- Figure 29: North America Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Procedure Type
- Figure 32: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product Type
- Figure 35: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Sales Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Latin America Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Procedure Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by End User
- Figure 55: Western Europe Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Procedure Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Product Type
- Figure 61: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by End User
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Procedure Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by End User
- Figure 81: East Asia Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Procedure Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Product Type
- Figure 87: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Procedure Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by End User , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by End User , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Procedure Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Procedure Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Procedure Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

