The demand for acetaminophen API in the UK is projected to reach a valuation of USD 110.0 million in 2026. This sector is anticipated to grow to USD 156.4 million by 2036, advancing at a compound annual growth rate of 3.6%.
This stable expansion is fundamentally anchored in acetaminophen's entrenched position as a first-line analgesic and antipyretic, its extensive use in both prescription and OTC medications, and its critical role in combination therapies, particularly with opioids for moderate to severe pain management.
The consistent need for high-quality API is driven by the vast and steady consumption of pain relief medications across the National Health Service (NHS) and retail channels. This creates a resilient, volume-driven market focused on reliable supply chains, stringent regulatory compliance, and cost-effective manufacturing to serve both generic and branded pharmaceutical formulations.

The UK's demand for Acetaminophen API stems from its comprehensive public healthcare system and a well-developed self-care culture. The NHS's extensive formulary and protocols for pain management, both in hospitals and primary care, guarantee a large, consistent baseline demand for prescription-grade API. Concurrently, the high consumer trust and accessibility of OTC pain relief medications ensure robust parallel demand through retail pharmacies and supermarkets.
Furthermore, the UK's stringent regulatory framework, enforced by the Medicines and Healthcare products Regulatory Agency (MHRA), mandates the highest standards of quality, safety, and traceability for APIs. This creates a market dominated by established, compliant manufacturers and complex supply chains, with a significant portion of demand linked to combination drugs used for post-operative and chronic pain management within clinical settings.
The acetaminophen API landscape in UK is categorized by the type of final drug product, its point of sale, its regulatory status, and its method of delivery. This segmentation reveals a market split between high-volume OTC consumer health and critical prescription pain management, with distinct supply chains and regulatory pathways for each.

Acetaminophen API combined with opioids holds a dominant 52.0% share. This prominence is driven by the clinical reliance on these combination drugs such as co-codamol and co-dydramol for managing moderate to severe pain within the NHS and palliative care. The opioid component enhances analgesic effect, making this a critical therapeutic class for hospital and prescription use, driving demand for specially formulated and tightly controlled API batches.
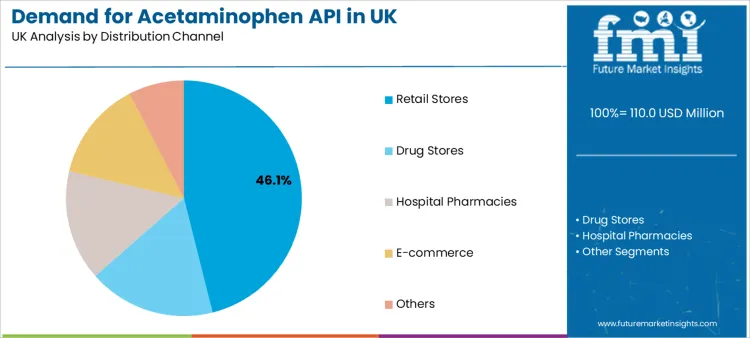
Retail stores constitute the leading distribution channel, with a 46.1% share. This includes pharmacies, drug stores, and supermarkets where consumers directly purchase OTC acetaminophen products.
This channel's dominance underscores the massive scale of self-medication for common ailments like headaches, muscle pain, and fever. Hospital pharmacies represent a critical channel for prescription formulations and inpatient use.
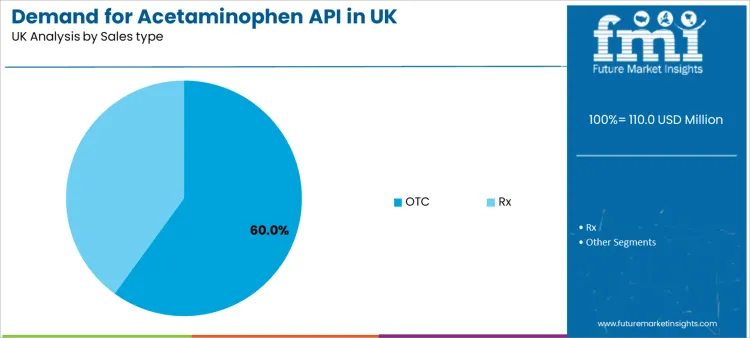
OTC sales represent the leading type at 60.0%. This reflects the widespread public use of acetaminophen as a front-line, accessible remedy without a prescription. The prescription (Rx) segment, while smaller in volume share, is crucial for higher-dose formulations, opioid combinations, and specific patient populations under medical supervision, representing a high-value, regulated stream of demand.

The oral route of administration commands a 70.0% share. This includes tablets, capsules, liquids, and soluble powders, offering convenience, cost-effectiveness, and patient preference for self-administration. Injectable formulations, used in hospital settings for rapid effect or where oral administration is not possible, represent a specialized and essential niche within the market.
The primary growth driver is the persistent, high prevalence of pain-related conditions and febrile illnesses, ensuring continuous consumption. The growing awareness and cultural acceptance of responsible self-medication for minor ailments sustain OTC volume. Furthermore, the ongoing focus on multimodal pain management protocols in healthcare, which often include acetaminophen as a foundation to reduce opioid use, supports stable prescription demand.
A significant restraint is the intense price pressure from NHS procurement bodies and generic competition, which squeezes API manufacturer margins. Increasingly stringent regulatory requirements for API sourcing, manufacturing practices, and environmental discharge increase compliance costs. Furthermore, supply chain vulnerabilities for key starting materials and geopolitical factors affecting global API trade can disrupt the steady supply required for this essential medicine.
Substantial opportunities are emerging from the development of novel, patent-protected combination drugs that include acetaminophen for specific therapeutic areas. The growth of pediatric and geriatric-friendly formulations such as pleasant-tasting liquids and orally disintegrating tablets, addresses specific demographic needs. Technological advancements in manufacturing for more consistent purity and the development of acetaminophen prodrugs with improved safety profiles present avenues for differentiation.
The sector faces threats from potential regulatory actions limiting pack sizes or strengthening warnings about hepatotoxicity at overdose, which could impact OTC volumes. A major public health shift towards alternative first-line analgesics could gradually erode market share.
Global supply chain concentration for API manufacturing in specific regions creates strategic vulnerability. Any significant safety concerns leading to drug recalls would have immediate disruptive effects.
Growth is influenced by regional population density, healthcare infrastructure, and prescribing patterns. The projected compound annual growth rates from 2026 to 2036 illustrate these geographic distinctions.

| Region | CAGR (2026-2036) |
|---|---|
| England | 3.9% |
| Scotland | 3.5% |
| Wales | 3.3% |
| Northern Ireland | 2.9% |
England leads with a projected CAGR of 3.9%. This is driven by its large, dense population which drives high absolute consumption through both retail and NHS channels. The concentration of major hospital trusts, primary care networks, and the headquarters of leading pharmacy chains and drug wholesalers creates the most concentrated demand center. It is the primary hub for national procurement and distribution logistics for both OTC and prescription pharmaceuticals.
Scotland exhibits a CAGR of 3.5%. Demand is supported by the nation's distinct public health policies and prescribing guidelines within NHS Scotland. The presence of significant pharmaceutical manufacturing and research facilities, alongside a population with specific demographic health challenges, creates a stable demand profile. Regional initiatives on pain management and public health directly influence consumption patterns and API demand.
Wales shows stable growth with a CAGR of 3.3%. The operational framework of NHS Wales and its integrated care systems underpins this. Demand follows population health needs and local formulary decisions, with a focus on equitable access to essential medicines. Public health campaigns on the safe use of analgesics within the Welsh population also shape the market environment.
Northern Ireland, with a projected CAGR of 2.9%, reflects growth aligned with its population size and health service structure. Demand is influenced by the health policies of the Department of Health in Northern Ireland and procurement practices across its health and social care trusts. Cross-border considerations with the Republic of Ireland may also play a minor role in supply chain dynamics.
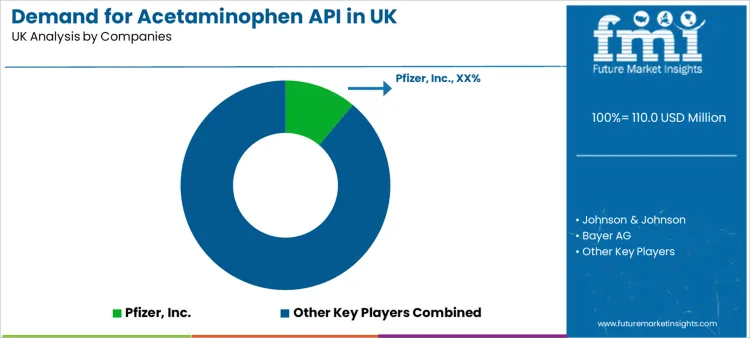
The competitive environment is mature and cost-sensitive, characterized by rivalry on the basis of production scale, cost efficiency, regulatory track record, and supply chain reliability. Large, vertically integrated multinational pharmaceutical companies compete with dedicated global API manufacturers and generic drug producers.
Success depends not only on achieving the lowest cost per kilogram while meeting pharmacopoeial standards but also on demonstrating impeccable regulatory compliance and audit readiness. Building long-term, strategic supply agreements with major finished-dose manufacturers and wholesalers is critical. Investment in continuous manufacturing processes and green chemistry initiatives can provide a competitive edge by reducing costs and environmental impact.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Drug Class | Acetaminophen API with Opioids, Acetaminophen API, Others |
| Distribution Channel | Retail Stores, Drug Stores, Hospital Pharmacies, E-commerce, Others |
| Sales Type | Over-the-Counter (OTC), Prescription (Rx) |
| Route of Administration | Oral, Injectable, Others |
| Regions Covered | England, Scotland, Wales, Northern Ireland |
How big is the demand for acetaminophen api in uk in 2026?
The demand for acetaminophen api in uk is estimated to be valued at USD 110.0 million in 2026.
What will be the size of acetaminophen api in uk in 2036?
The market size for the acetaminophen api in uk is projected to reach USD 156.4 million by 2036.
How much will be the demand for acetaminophen api in uk growth between 2026 and 2036?
The demand for acetaminophen api in uk is expected to grow at a 3.6% CAGR between 2026 and 2036.
What are the key product types in the acetaminophen api in uk?
The key product types in acetaminophen api in uk are cetaminophen api with opioids, acetaminophen api and others.
Which distribution channel segment is expected to contribute significant share in the acetaminophen api in uk in 2026?
In terms of distribution channel, retail stores segment is expected to command 46.1% share in the acetaminophen api in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.