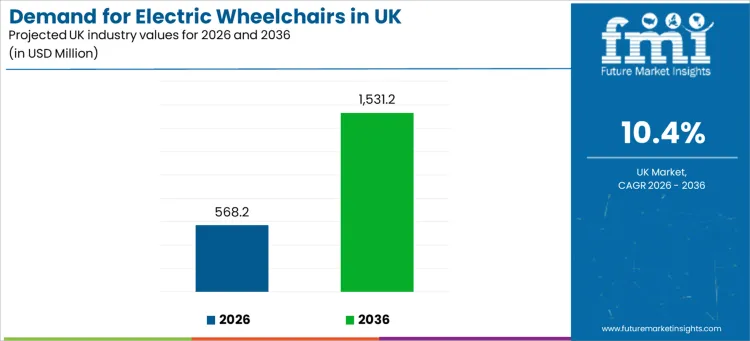
Demand for electric wheelchairs in the UK is projected at USD 568.2 million in 2026 and is expected to reach USD 1,531.2 million by 2036, expanding at a 10.4% CAGR. Growth is linked to the rising need for independent mobility support across aging populations, long-term disability care, and post-acute recovery pathways that require consistent assisted movement. Electric wheelchairs are positioned as essential daily-living equipment where manual propulsion is not feasible due to strength limitations, fatigue, neurological conditions, or complex clinical requirements.
Electric wheelchairs include rear wheel drive, front wheel drive, standing electric wheelchairs, and other formats designed for different mobility environments. Stakeholders evaluate selection through stability, turning radius, battery range, seating compatibility, controller usability, service reliability, and repair turnaround time. In procurement environments, the device is assessed as part of an overall mobility solution, including seating, posture support, safety controls, and long-term maintenance readiness.
For healthcare leaders, demand is shaped by how quickly users can be assessed, fitted, trained, and supported through service cycles. For manufacturers and distribution partners, value is tied to durable product engineering, adaptable configurations, and service infrastructure that can sustain uptime through daily wear, transport exposure, and battery lifecycle demands.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 568.2 million |
| Industry Forecast Value (2036) | USD 1,531.2 million |
| Forecast CAGR (2026 to 2036) | 10.4% |
Electric wheelchair demand in the UK is shaped by mobility as a clinical outcome, not only a comfort feature. Health systems increasingly recognise that appropriate mobility support influences independence, safety, caregiver load, and long-term health stability. When mobility is restricted, people face higher risks linked to isolation, falls, reduced physical conditioning, pressure injuries, and avoidable complications that raise care costs.
Service delivery frameworks are also evolving. NHS England introduced a Wheelchair Quality Framework to support integrated care boards and wheelchair service providers in delivering higher-quality provision, focusing on improved access, outcomes, and experience. When service leaders aim to reduce delays and improve fitting quality, procurement and replacement activity rises because devices must match real-world user needs and usage environments.
Demand is also connected to how users access the right mobility category. NHS guidance explains how wheelchairs and mobility support can be accessed through health and care pathways, including NHS support and broader equipment routes. User organisations and charities also highlight that powered wheelchair eligibility depends on local wheelchair services, reinforcing the importance of assessment and clinical justification.
A policy layer is emerging around how powered mobility devices are used in public space. In January 2026, the UK government opened a consultation on changing rules for powered mobility devices, including powered wheelchairs and mobility scooters. This type of review can influence user adoption confidence, safety expectations, and design priorities such as speed control, braking systems, and stability.
Technology design priorities add another growth lever. Users increasingly want compact turning performance, better suspension, longer battery life, and improved seating integration. Product teams shaping long-term portfolios often keep category context aligned with broader mobility equipment themes such as electric wheelchair and complementary assistive formats such as mobility aids and transportation equipment when prioritising innovation and service models.
How users move in daily environments, how complex their seating needs are, and where the device is funded and supported define segmentation. Product type choice is shaped by stability, manoeuvrability, user control needs, and indoor-outdoor usage patterns. End-user segmentation reflects where the device is initially prescribed, fitted, used, and maintained.
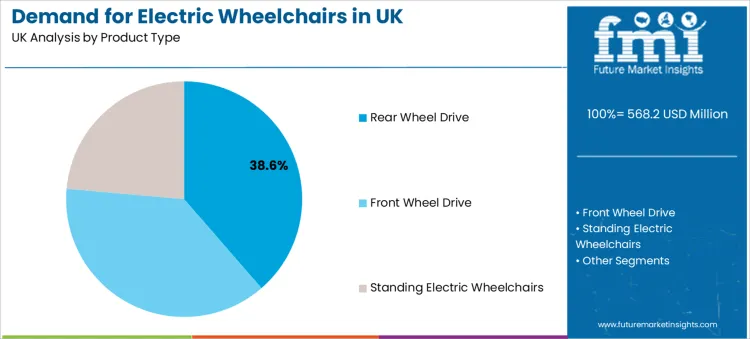
Rear wheel drive holds a 38.6% share, placing it as the leading product type. This dominance reflects performance characteristics valued by both clinicians and experienced users. Rear wheel drive designs often provide predictable stability at higher travel speeds and support smoother outdoor performance on uneven terrain. They are also commonly selected for users who prioritise straight-line control and need a configuration that handles longer daily travel distances reliably.
Hospitals and clinics often prefer product types that provide dependable handling, clear training routines, and a range of seating compatibility options. Rear wheel drive configurations can align with these priorities when the goal is stable mobility paired with clinically appropriate seating and posture support.
Front wheel drive and standing electric wheelchairs play important roles in supporting specific user requirements and mobility environments. Front wheel drive is often evaluated for indoor manoeuvrability, turning behaviour in tighter spaces, and navigation across household layouts. Standing electric wheelchairs align with users who require standing support as part of daily function needs, pressure management routines, or therapy-led mobility goals. These device categories are driven by user outcomes rather than volume standardisation.
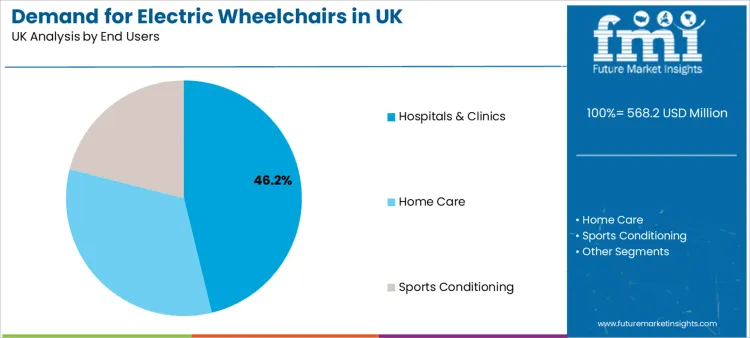
Hospitals & clinics account for 46.2% of demand, reflecting their role as key access points for assessment, prescription, fitting, and training. Many users enter the pathway through clinical care systems where mobility is tied to diagnosis progression, rehabilitation planning, or long-term condition management. In these settings, electric wheelchairs are treated as clinical equipment, not only a consumer product.
Hospital-based procurement also reflects the need for consistent device performance, predictable servicing, and supplier support that can handle repair workflows under clinical scheduling pressure.
Home care demand grows as users prioritise independent movement in residential settings and need configurations that support daily living tasks. In home environments, selection places weight on ease of transfer, battery reliability, safe navigation across doorways, and predictable control interface performance. Sports conditioning reflects specialised use cases where users require mobility solutions aligned with training routines, community participation, and adapted sport engagement.
Service quality and access priorities drive replacement and upgrade cycles. NHS England’s Wheelchair Quality Framework positions wheelchair provision as a pathway where outcomes and experience are central, which increases the importance of fit-for-purpose powered mobility.) Global guidance reinforces the value of appropriate wheelchair provision and service models, including training and fitting principles that support better outcomes.
Regulatory expectations for medical device monitoring also strengthen focus on product reliability and field performance. The MHRA published guidance to help manufacturers prepare for new post-market surveillance requirements in Great Britain, which came into force on 16 June 2025.
Provision delays and service variability can slow device uptake, particularly where fitting capacity is constrained. Repairs and parts availability can also limit user confidence and create downtime risks. Cost sensitivity remains a real barrier in some pathways because powered mobility devices are high-value equipment with long service life expectations.
Compliance and documentation requirements also create procurement complexity. As post-market surveillance expectations strengthen, manufacturers and suppliers must demonstrate robust monitoring and reporting readiness.
The strongest opportunities sit in reliability engineering and service enablement. Buyers value suppliers that can support faster repair cycles, consistent battery availability, and modular component replacement that reduces downtime. Device control systems also offer innovation space, including improved joystick ergonomics, programmable driving profiles, and safety logic that supports public-space operation confidence.
Standard-aligned product development supports competitiveness. ISO standards address wheelchair power and control systems for electrically powered wheelchairs and scooters, helping shape safety and performance requirements. Electromagnetic compatibility also matters for powered wheelchairs and battery chargers, influencing safe operation in real-world environments.
Technology planners often align feature roadmaps with adjacent assistive formats such as smart wheelchair and broader user demand patterns tied to wheelchairs when setting investment direction for connected mobility and premium performance tiers.
Supply chain disruptions can delay deliveries or limit availability of specialised components such as controllers, motors, and battery packs. Service ecosystem strain is another threat, since delayed repairs reduce user trust and may drive out-of-pocket purchase outside standard pathways. Policy change can reshape usage conditions in public environments, which affects product requirements and user adoption confidence.
| Region | CAGR 2026 to 2036 |
|---|---|
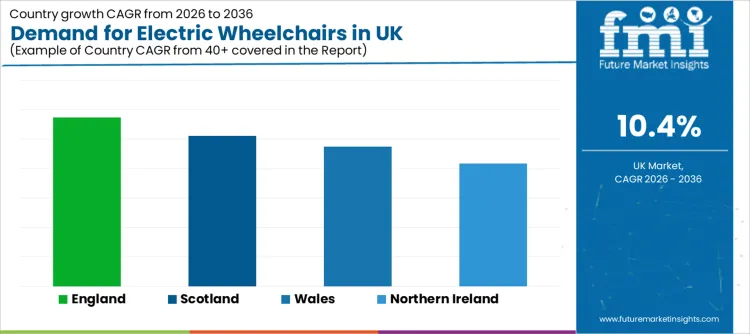
| England | 11.5% |
| Scotland | 10.2% |
| Wales | 9.5% |
| Northern Ireland | 8.3% |
England grows at 11.5%, reflecting larger population concentration, higher service volumes, and broader clinical infrastructure reach. Rehabilitation pathways, long-term condition management, and the need for reliable mobility solutions that reduce care burden support demand. Service models shaped by quality and access priorities influence both new provision and replacement cycles.
Scotland advances at 10.2%, shaped by sustained demand for independent mobility solutions and consistent care delivery focus. Procurement and provision priorities often centre on correct device fit, safe usage training, and long-term support needs that help users stay active and reduce reliance on caregiver assistance.
Wales increases at 9.5%, supported by growing need for home-based mobility solutions and continued clinical provision for users requiring powered assistance. Device selection is shaped by residential layout constraints, outdoor surface variation, and the need for stable battery performance in daily routines.
Northern Ireland rises at 8.3%, reflecting stable demand linked to mobility support needs and service-driven provision cycles. Buyers place weight on long service life, dependable supplier support, and clear maintenance pathways that reduce the risk of prolonged downtime.
Competition is shaped by three core factors: user outcomes, device reliability, and service capability. Buyers evaluate manufacturers not only on product engineering, but also on the availability of parts, repair responsiveness, and the ability to tailor seating and controls to specific clinical needs. Procurement teams also assess suppliers through documentation readiness, product traceability, and field performance monitoring.
Post-market surveillance expectations are becoming more influential in supplier evaluation. MHRA guidance highlights the importance of preparing for strengthened medical device post-market surveillance requirements in Great Britain, influencing how quality systems and reporting readiness are assessed.
Invacare Corporation competes through broad wheelchair portfolios and established distribution networks. Permobil AB competes through premium powered mobility positioning and seating integration strength. Pride Mobility Products Corp. competes through product breadth and consumer-facing availability. Sunrise Medical LLC competes through mobility device engineering and clinical-facing product families. Drive DeVilbiss Healthcare competes through accessible equipment portfolios and wide channel reach.
Product strategy teams often link positioning choices to broader mobility categories such as personal mobility devices and emerging premium formats such as smart wheelchair when aligning portfolio direction with independence and performance expectations.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Rear Wheel Drive; Front Wheel Drive; Standing Electric Wheelchairs |
| End Users | Hospitals & Clinics; Home Care; Sports Conditioning |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Invacare Corporation; Permobil AB; Pride Mobility Products Corp.; Sunrise Medical LLC; Drive DeVilbiss Healthcare |
How big is the demand for electric wheelchairs in uk in 2026?
The demand for electric wheelchairs in uk is estimated to be valued at USD 568.2 million in 2026.
What will be the size of electric wheelchairs in uk in 2036?
The market size for the electric wheelchairs in uk is projected to reach USD 1,531.2 million by 2036.
How much will be the demand for electric wheelchairs in uk growth between 2026 and 2036?
The demand for electric wheelchairs in uk is expected to grow at a 10.4% CAGR between 2026 and 2036.
What are the key product types in the electric wheelchairs in uk?
The key product types in electric wheelchairs in uk are rear wheel drive, front wheel drive and standing electric wheelchairs.
Which end users segment is expected to contribute significant share in the electric wheelchairs in uk in 2026?
In terms of end users, hospitals & clinics segment is expected to command 46.2% share in the electric wheelchairs in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.