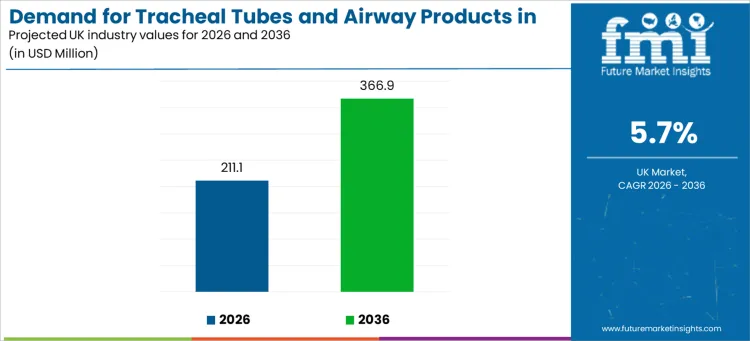
Demand for tracheal tubes and airway products in the UK is projected at USD 211.1 million in 2026 and is expected to reach USD 366.9 million by 2036, expanding at a 5.7% CAGR. This demand profile reflects how hospitals, surgical units, and homecare providers prioritize airway control, ventilation safety, and standardized intubation protocols across acute and non-acute care environments.
Airway devices are treated as safety-critical consumables that support oxygen delivery, anesthesia administration, and respiratory stabilization. Clinical leaders evaluate these products through sealing reliability, resistance to kinking, cuff integrity, biocompatibility, and compatibility with ventilator circuits. Procurement teams link purchasing decisions to infection control policies, regulatory compliance, and the need for predictable supply continuity.
Hospital executives focus on airway device reliability because intubation failure or cuff leakage carries clinical and legal risk. Surgical leaders prioritize device uniformity because standard tube formats reduce training burden and procedural variability. Respiratory care managers emphasize material safety, patient comfort, and long-term stability during prolonged ventilation.
Technology service providers and device manufacturers influence adoption outcomes through material engineering, production scale, and quality assurance discipline. Buyers value product lines that maintain dimensional accuracy, consistent wall thickness, and dependable cuff inflation performance across batch volumes.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 211.1 million |
| Industry Forecast Value (2036) | USD 366.9 million |
| Forecast CAGR 2026 to 2036 | 5.7% |
Demand for tracheal tubes and airway products in the UK rises as healthcare systems expand surgical throughput, intensive care capacity, and emergency response readiness. Airway management remains a core clinical function across anesthesia delivery, trauma care, respiratory therapy, and post-operative monitoring. Hospitals maintain high device consumption volumes because airway access must remain immediately available in operating rooms, emergency departments, and critical care units.
Clinical teams rely on standardized tracheal tubes to reduce procedural variation and training risk. National airway management guidance emphasizes the importance of consistent device selection, routine equipment checks, and predictable cuff performance during intubation and ventilation. These expectations support steady replacement demand even when overall procedure volumes remain stable.
Procurement strategies increasingly align airway device sourcing with infection prevention protocols. Single-use formats dominate because they reduce cross-contamination risk and simplify sterilization workflows. Hospital leaders view reliable airway supply as part of operational resilience planning.
Homecare growth also contributes to long-term volume expansion. Patients receiving long-term ventilation or tracheostomy support require stable access to compatible tubes, suction accessories, and airway maintenance components. Care providers prioritize materials that balance flexibility, durability, and patient comfort during extended use.
This segmentation reflects how airway devices are deployed across procedural intensity, ventilation duration, and institutional care settings.
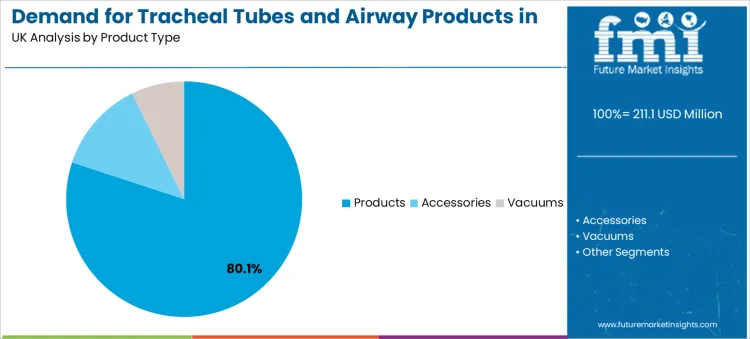
Products account for 80.1% of total demand, making them the dominant product category. This position reflects the fact that tracheal tubes remain the primary airway access device across anesthesia, emergency intubation, and mechanical ventilation. Hospitals consume large volumes because tubes are treated as disposable items in most procedural environments.
Standard tube formats simplify clinical workflows. Anesthesiology teams prefer predictable tube geometry, standardized connectors, and uniform cuff inflation behavior. These characteristics reduce procedural complexity and support rapid airway access during time-critical interventions.
Accessory products such as suction devices and vacuums support airway maintenance and secretion management. Their demand follows tube deployment volumes but remains secondary in revenue contribution.
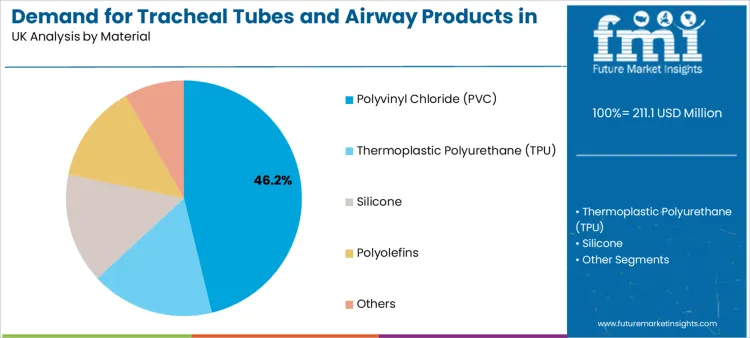
Polyvinyl chloride represents 46.2% of total demand by material. PVC dominates because it offers cost efficiency, process consistency, and reliable mechanical performance. Manufacturers value PVC because it supports precise extrusion control and stable dimensional tolerances.
Clinicians prefer PVC tubes because they balance flexibility with structural integrity. These tubes maintain lumen patency under bending stress and provide dependable cuff inflation behavior. Procurement teams also favor PVC because it supports high-volume sourcing with predictable pricing.
Thermoplastic polyurethane and silicone serve specialized applications where enhanced flexibility or biocompatibility is required. Polyolefins support selected designs focused on lightweight construction. These materials contribute to portfolio diversification but do not displace PVC in mainstream procedural use.
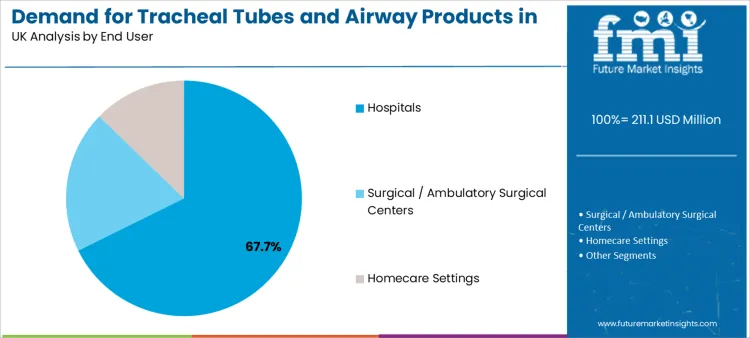
Hospitals represent 67.7% of total demand by end user. This dominance reflects their central role in surgery, trauma care, intensive care, and emergency response. Hospitals maintain large airway inventories because device availability must remain continuous.
Surgical and ambulatory surgical centers contribute through procedural volume growth. These settings prioritize standardized tube formats that align with anesthesia protocols and equipment compatibility.
Homecare settings support long-term ventilation and tracheostomy care. Their contribution remains smaller but structurally important because it drives demand for patient-comfort-oriented materials and long-term durability features.
Airway device demand expands as surgical throughput rises across elective and emergency procedures. Hospitals increase airway inventory levels because intubation readiness must remain uninterrupted across operating rooms, emergency departments, and intensive care units. Clinical governance teams enforce airway safety checklists that require immediate access to standardized tube sizes and compatible suction components.
Critical care capacity expansion also strengthens volume consumption. Prolonged mechanical ventilation cases require tube replacement due to secretion buildup, cuff degradation, or migration risk. Respiratory care teams prioritize tube reliability because airway failure events carry direct patient harm exposure and institutional liability.
Infection prevention policy alignment further supports single-use airway formats. Hospitals favor disposable tracheal tubes and suction accessories because sterilization workflows increase cost, labor burden, and cross-contamination exposure. Procurement leaders treat airway products as infection-control assets rather than routine consumables.
Training standardization contributes to repeatable purchasing behavior. Anesthesia and emergency medicine teams favor consistent tube brands and geometries because uniform handling reduces procedural error risk. These preferences create supplier stickiness when clinical familiarity becomes embedded into airway protocols.
Capital allocation pressure shapes purchasing timing. Hospital procurement departments must prioritize across diagnostic imaging equipment, surgical robotics, and patient monitoring infrastructure. Airway devices remain essential but do not always receive budget preference during capital review cycles.
Supply chain exposure remains a structural restraint. Tube extrusion capacity and material sourcing disruptions can create lead-time instability. Hospitals respond by stockpiling, which temporarily suppresses forward procurement activity after buffer inventory is built.
Workforce constraints also influence consumption pacing. Staffing shortages reduce elective surgical throughput, which lowers short-term airway device usage volumes. Intensive care staffing availability affects ventilator utilization rates, indirectly influencing tube replacement frequency. Regulatory compliance burden can slow new product onboarding. Material safety certification, labeling compliance, and packaging validation requirements delay hospital approval of alternative suppliers.
Material engineering programs that improve cuff compliance and reduce tracheal wall pressure offer clinical value differentiation. Hospitals show interest in tubes that lower mucosal injury risk during prolonged ventilation. Portfolio rationalization creates operational efficiency. Hospitals prefer narrower SKU ranges that simplify training, stocking, and emergency cart configuration. Suppliers that align tube portfolios to standardized airway kits strengthen adoption probability.
Traceability systems support recall readiness and quality monitoring. Device labeling formats that enable lot-level tracking improve risk management for hospital procurement teams. Homecare channel expansion offers structural growth. Long-term ventilation patients require reliable tube replacement access and compatible suction accessories. Providers favor suppliers that support stable homecare logistics.
Procurement consolidation increases price pressure. Group purchasing organizations exert downward pricing leverage that compresses supplier margins and restricts portfolio breadth. Regulatory tightening around plasticizers and material additives creates compliance risk for PVC-based tubes. Suppliers face reformulation cost exposure if material restrictions expand.
Manufacturing quality lapses carry reputational damage risk. Tube dimensional inconsistency or cuff leakage events undermine clinical trust and accelerate supplier substitution. Substitution risk exists where supraglottic airway devices replace intubation in selected procedures, reducing tube usage in low-risk anesthesia cases.
| Region | CAGR 2026 to 2036 |
|---|---|
| England | 6.3% |
| Scotland | 5.6% |
| Wales | 5.2% |
| Northern Ireland | 4.5% |
England grows at 6.3%, driven by its concentration of tertiary hospitals, trauma centers, and surgical hubs. High elective surgery volumes generate consistent tracheal tube turnover. Emergency departments also sustain elevated airway device consumption due to trauma response readiness requirements.
Critical care bed expansion strengthens long-term tube demand. Prolonged ventilation cases increase tube replacement frequency and suction accessory usage. Hospitals maintain larger airway inventories to prevent procedural delays during admission surges.
Regional procurement networks favor standardized airway formats to support staff rotation across hospital trusts. Uniform tube geometries reduce training friction and simplify emergency airway cart stocking. These practices reinforce dominant supplier positioning.
Scotland advances at 5.6%, shaped by steady surgical throughput and consistent anesthesia service utilization. Hospitals emphasize reliable airway product availability to support procedural scheduling stability.
Respiratory care teams maintain disciplined replacement protocols for tubes used in prolonged ventilation. Clinical governance structures reinforce standardized device use across health boards.
Procurement leaders favor long-term supplier contracts that guarantee material consistency and stable pricing. Training programs embed preferred tube brands into airway protocols, reinforcing repeat purchasing behavior.
Wales grows at 5.2%, driven by investments in surgical access expansion and emergency care readiness. Hospitals increase airway stock levels to reduce procedural cancellation risk linked to supply gaps. Regional service planners prioritize standardized airway kits to support consistent intubation practices across district hospitals.
Procurement teams prefer simplified SKU portfolios that reduce stock management complexity. Critical care utilization contributes incremental demand growth as ventilator access expands and respiratory support capacity rises.
Northern Ireland rises at 4.5%, reflecting budget discipline and staged airway inventory expansion. Hospitals align airway procurement with broader respiratory equipment planning. Elective surgery pacing remains controlled, which moderates tube consumption growth. Emergency care readiness still sustains baseline airway usage volumes. Procurement teams favor conservative supplier onboarding, prioritizing established brands with proven quality consistency and service responsiveness.
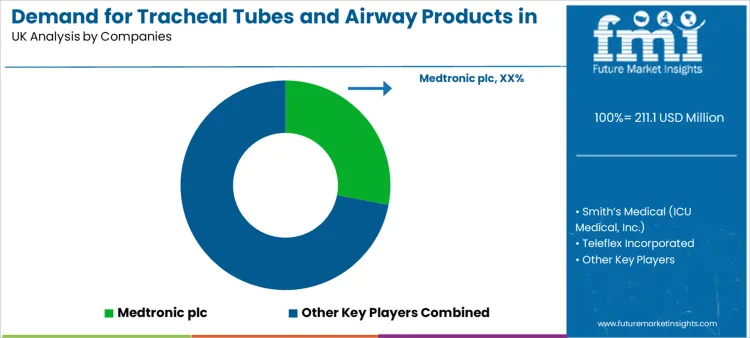
Competition centers on manufacturing reliability, material engineering, regulatory compliance, and distribution scale. Providers evaluate suppliers on batch consistency, cuff performance reliability, and delivery lead times.
Medtronic plc competes through broad respiratory care portfolios. Smith’s Medical maintains strong positioning through airway management specialization. Teleflex Incorporated emphasizes clinical workflow integration. Ambu A/S contributes through single-use device leadership. Vyaire Medical, Inc. strengthens competition through respiratory care portfolio depth.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Products; Accessories; Vacuums |
| Material | Polyvinyl Chloride; Thermoplastic Polyurethane; Silicone; Polyolefins; Others |
| End User | Hospitals; Surgical and Ambulatory Surgical Centers; Homecare Settings |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Medtronic plc; Smith’s Medical (ICU Medical, Inc.); Teleflex Incorporated; Ambu A/S; Vyaire Medical, Inc. |
How big is the demand for tracheal tubes and airway products in uk in 2026?
The demand for tracheal tubes and airway products in uk is estimated to be valued at USD 211.1 million in 2026.
What will be the size of tracheal tubes and airway products in uk in 2036?
The market size for the tracheal tubes and airway products in uk is projected to reach USD 366.9 million by 2036.
How much will be the demand for tracheal tubes and airway products in uk growth between 2026 and 2036?
The demand for tracheal tubes and airway products in uk is expected to grow at a 5.7% CAGR between 2026 and 2036.
What are the key product types in the tracheal tubes and airway products in uk?
The key product types in tracheal tubes and airway products in uk are products, accessories and vacuums.
Which material segment is expected to contribute significant share in the tracheal tubes and airway products in uk in 2026?
In terms of material, polyvinyl chloride (pvc) segment is expected to command 46.2% share in the tracheal tubes and airway products in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.