Demand for Instrument Cleaning Chemistries in USA
This report covers the Instrument Cleaning Chemistries demand landscape in USA through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, product type trends, process type trends, instrument type trends, end user analysis, supply chain developments, strategic growth opportunities.
Demand for Instrument Cleaning Chemistries in USA Market Size, Market Forecast and Outlook By FMI
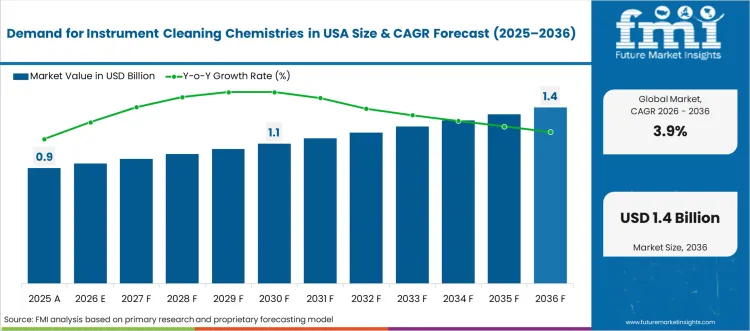
The Instrument Cleaning Chemistries in USA market was valued at USD 0.90 billion in 2025, projected to reach USD 0.94 billion in 2026, and is forecast to expand to USD 1.37 billion by 2036 at a 3.9% CAGR. FMI opines that FDA enforcement of validated reprocessing protocols and the Joint Commission's infection control accreditation requirements are sustaining mandatory procurement of specialised cleaning detergents and disinfection chemistries across USA healthcare facilities. As per FMI, the convergence of rising outpatient surgical procedure volumes and the expansion of ambulatory surgical centre networks is creating new procurement channels for instrument cleaning chemistry products that were historically concentrated within hospital central sterile processing departments.
Summary of Demand for Instrument Cleaning Chemistries in USA Market
- Market Overview
- The Instrument Cleaning Chemistries in USA market is valued at USD 0.90 billion in 2025 and is projected to reach USD 1.37 billion by 2036.
- The industry is expected to grow at a 3.9% CAGR from 2026 to 2036, creating an incremental opportunity of USD 0.43 billion.
- The market is a compliance-driven healthcare consumables category where FDA reprocessing validation mandates, Joint Commission accreditation requirements, and ambulatory surgical centre expansion define competitive positioning among cleaning chemistry suppliers in the United States.
- Demand and Growth Drivers
- Demand is sustained by FDA enforcement of validated reprocessing protocols and Joint Commission infection control accreditation requirements across all USA healthcare facilities.
- Rising outpatient surgical procedure volumes and ambulatory surgical centre network expansion are creating new procurement channels beyond hospital central sterile departments.
- Automated washer-disinfector system adoption is increasing per-cycle chemistry consumption by approximately 35% compared to manual cleaning protocols.
- West USA leads at 4.5% CAGR, followed by South USA at 4.0%, Northeast USA at 3.6%, and Midwest USA at 3.1%.
- Product and Segment View
- The market includes product type, process type, instrument type, end user as primary segmentation dimensions.
- Detergents leads by Product Type with 73.2% share in 2026.
- Disinfection leads by Process Type with 33% share in 2026.
- Surgical Instruments leads by Instrument Type with 36.6% share in 2026.
- Geography and Competitive Outlook
- West USA is the fastest-growing region at 4.5% CAGR, while South USA anchors the second-largest demand base.
- Competition is shaped by technical specialisation, regulatory compliance capability, and distribution depth, with key players including STERIS Plc., Getinge Group, Ecolab Inc., 3M, Dr. Weigert.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant, opines: 'FMI's analysis reveals that the instrument cleaning chemistries in usa market is entering a phase where procurement decisions are increasingly dictated by regulatory compliance timelines and measurable performance thresholds. Organisations that delay platform investment risk permanent lockout from competitive procurement tiers their counterparts are actively securing.'
- Strategic Implications / Executive Takeaways
- Solution providers must secure certified integration pathways with tier-1 enterprise systems to qualify for forthcoming institutional procurement contracts.
- Capital project directors should phase out legacy approaches and mandate native platform compatibility in all new equipment and software procurement specifications.
- Technical teams must develop specialised competencies in product type validation to demonstrate compliance parameters during the evaluation phase.
Demand for Instrument Cleaning Chemistries in USA Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.94 billion |
| Industry Value (2036) | USD 1.37 billion |
| CAGR (2026 to 2036) | 3.9% |
Source: Future Market Insights, 2026
FMI is of the opinion that regional demand patterns across the United States reflect varying concentrations of surgical volume and healthcare facility density. West USA leads at 4.5% CAGR, driven by California and Washington hospital system expansions and ambulatory surgical centre proliferation. South USA follows at 4.0%, supported by Texas and Florida healthcare facility construction generating greenfield instrument reprocessing chemistry demand. Northeast USA registers 3.6% growth anchored by aging hospital systems in New York and Massachusetts upgrading from manual to automated cleaning workflows. Midwest USA maintains 3.1% growth as regional health system consolidation standardises cleaning chemistry procurement across affiliated facilities. Future Market Insights analysis indicates that facilities transitioning to fully automated washer-disinfector systems increase cleaning chemistry consumption per reprocessing cycle by approximately 35% compared to manual cleaning protocols.
Demand for Instrument Cleaning Chemistries in USA Market Definition
Based on FMI's report, the Instrument Cleaning Chemistries in USA market covers detergents and cleaners formulated specifically for reprocessing reusable medical instruments including surgical instruments, endoscopes, ultrasound probes, and dental instruments across healthcare facilities within the United States.
Demand for Instrument Cleaning Chemistries in USA Market Inclusions
Future Market Insights analysis covers detergents and cleaners used in disinfection, presoak, manual cleaning, and automatic cleaning workflows for medical instruments. The scope encompasses regional market sizes across USA census regions, forecast projections from 2026 to 2036, and segmentation by product type, process type, instrument type, and end user.
Demand for Instrument Cleaning Chemistries in USA Market Exclusions
The scope excludes surface disinfectants not designed for instrument reprocessing, sterilisation agents such as ethylene oxide and hydrogen peroxide vapour, and disposable single-use medical devices that do not require reprocessing.
Demand for Instrument Cleaning Chemistries in USA Market Research Methodology
- Primary Research: Analysts engaged with hospital sterile processing directors, ambulatory surgical centre operations managers, and infection control consultants to map product selection criteria and FDA compliance requirements.
- Desk Research: Data collection aggregated FDA medical device reprocessing guidance documents, Joint Commission infection control survey results, and published healthcare supply chain expenditure data from the American Hospital Association.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of instrument cleaning chemistry volumes consumed within USA healthcare facilities, applying surgical procedure volume growth curves and automated reprocessing penetration rates.
- Data Validation and Update Cycle: Projections are cross-validated against quarterly healthcare consumable revenue guidance from major cleaning chemistry manufacturers and CMS hospital cost report data.
Why is the Demand for Instrument Cleaning Chemistries in the USA Growing?
Demand for instrument cleaning chemistries in the USA is increasing because laboratories, medical facilities and manufacturing plants require safe, effective solutions to decontaminate and sterilize reusable equipment. Clinical labs, hospitals and research centers rely on detergents, enzymatic cleaners and degreasers that can remove biological residues, grease or chemical contaminants without damaging sensitive materials. Growth in biotechnology, pharmaceutical manufacturing and diagnostic testing supports heavy use of cleaning chemistries to maintain compliance with hygiene standards and avoid cross-contamination. Cleanroom environments, analytical labs and quality control units adopt specialized chemistries compatible with stainless steel, glass, plastic and silicone instruments. Recurring use of these chemistries ensures consistent demand for both bulk and ready-to-use formats.
Manufacturers develop low-foaming, low-residue formulations that support automated washers and ultrasonic baths used in high throughput labs. Increased environmental and regulatory pressure encourages supply of biodegradable and less corrosive cleaners, which meet safety guidelines for disposal and worker exposure. Constraints include cost sensitivity among small practices or facilities with limited budgets. Some chemistries require careful handling and disposal protocols, which introduces training or compliance burden. Supply chain disruption for specialty ingredients may affect availability for high-grade cleaning solutions.
Which Product Types, Cleaning Processes, and Instrument Categories Lead Demand for Instrument Cleaning Chemistries in the USA?
Demand for instrument cleaning chemistries in the United States is driven by compliance with infection prevention standards, device compatibility requirements, and increased surgical and diagnostic procedure volumes. Hospitals and ambulatory care facilities prioritize formulations that provide residue-free cleaning, efficiency under high throughput, and material safety for sophisticated reusable instruments. Growth is reinforced by ongoing shifts from single-use to reprocessable medical devices, driving consistent demand for validated cleaning chemistries in centralized sterile processing departments.
By Product Type, Detergents Hold the Largest Share
Detergents command 73.2%, due to broad applicability across surgical and diagnostic instruments, including those with lumens, hinges, and polymeric materials. Enzymatic and multi-enzyme formulations remove blood, protein, lipids, and biofilm while supporting compatibility with automated washers and ultrasonic cleaners. Cleaners represent 26.8%, covering solvent-based and neutral agents used for targeted residue removal and surface protection. USA procurement trends favor detergent formulations with low foaming, rapid soil breakdown, pH balance, and validated antimicrobial performance under FDA, AAMI, and CDC guidelines. Adoption also aligns with workflow efficiency in sterile processing units and traceability using standardized dilution and monitoring systems.
Key Points:
- Detergents remain essential across hospitals and high-volume SPD workflows
- Cleaner Usage focuses on specialty residue and material-specific protection
- Compatibility and validated performance drive product selection decisions
By Cleaning Process, Disinfection and Presoak Lead Demand
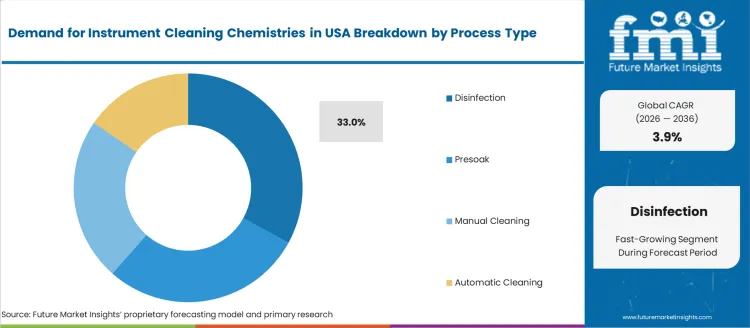
Disinfection processes hold 33.0%, driven by requirements to reduce microbial load pre-sterilization in accordance with USA infection control protocols. Presoak accounts for 28.5%, addressing dried bioburden removal during instrument transportation and holding times. Automatic cleaning contributes 25.0%, used in reprocessing automation for consistent outcomes and reduced labor dependency. Manual cleaning holds 13.5%, focused on delicate instruments and low-volume facilities. Hospital and ASC settings emphasize workflow standardization, device-specific procedure steps, and chemical compatibility to prevent corrosion, residue, or micro-damage under repeated cycles.
Key Points:
- Disinfection supports high-risk device reprocessing outcomes
- Presoak boosts soil removal efficiency before automated cycles
- Automation grows with labor optimization and performance repeatability
By Instrument Type, Surgical Instruments Hold the Largest Share
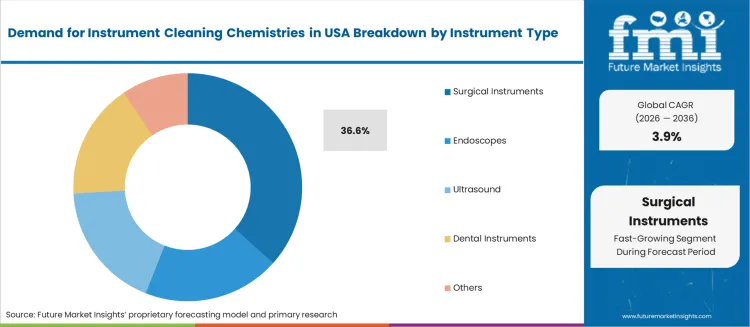
Surgical instruments represent 36.6%, reflecting extensive use across operating rooms and procedural care. Ultrasound devices contribute 28.0%, with demand driven by probe reprocessing requirements. Dental instruments hold 16.0%, linked to frequent turnover in clinical dentistry. Endoscopes represent 4.4%, with lower share based on dedicated high-level disinfectant categories rather than general chemistries. Other devices make up 15.0%, including orthopedic and reusable robotic instruments requiring validated cleaning action. Variation in geometry, material composition, and lumened structures drives product development focused on penetration, residue prevention, and bioburden control.
Key Points:
- Surgical instruments dominate due to high procedure volumes
- Ultrasound probe reprocessing supports strong secondary demand
- Device material and lumen complexity guide formulation choice
What are the Drivers, Restraints, and Key Trends of the Demand for Instrument Cleaning Chemistries in the USA?
Growth of surgical procedure volume, stricter infection-control standards and increased adoption of automated reprocessing drive demand.
In the United States, hospitals, outpatient surgical centers and dental clinics require validated cleaning chemistries for pre-cleaning, ultrasonic cleaning and washer-disinfector cycles to maintain sterility assurance. An aging population and expansion of specialty care drive high volumes of reusable medical and dental instruments, increasing consumption of enzymatic and alkaline formulations. Federal and accreditation-driven focus on reducing healthcare-associated infections supports procurement of chemistries proven to remove complex soils such as blood proteins, orthopedic debris and bioburden in minimally invasive instruments. Automated reprocessing equipment used in central sterile supply departments relies on compatible detergents, ensuring recurring demand across institutional healthcare networks.
Cost pressure on healthcare facilities, staff training requirements and shift toward single-use instruments restrain demand.
Budget-managed hospitals and clinics evaluate total reprocessing costs, which include detergents, utilities and labor, leading some facilities to limit premium products despite performance advantages. Effective use of cleaning chemistries requires training on dilution control, water quality and cycle timing, and inconsistent practices can reduce cleaning outcomes and discourage product switching. Growth of disposable instruments in specialties like ophthalmology and outpatient care reduces the number of reusable tools requiring multi-stage cleaning. These financial and operational constraints moderate demand growth in specific segments.
Shift toward neutral-pH enzymatic chemistries, increased compatibility for robotic and lumened instruments and rising adoption of monitoring indicators define key trends.
Manufacturers are developing low-foaming and neutral-pH formulas that are gentle on heat-sensitive device materials used in robotic surgery and minimally invasive instruments. Facilities adopt detergents with multi-enzyme blends that remove fats, biofilm and orthopedic cement to align with evolving instrument design. Reprocessing departments increasingly use indicators and dosing systems that validate concentration and soil breakdown, improving consistency and compliance. Sustainability goals encourage procurement of chemistries with lower toxicity profiles and reduced packaging waste. These trends reflect continued, patient-safety-driven demand for instrument cleaning chemistries across United States healthcare providers.
Analysis of the Demand for Instrument Cleaning Chemistries in the USA by Region
Demand for instrument cleaning chemistries in the United States is expanding as hospitals, diagnostic labs, ambulatory facilities, and dental centers reinforce infection-prevention compliance. Growth is linked to rising surgical procedure volumes, the shift to same-day care environments, and mandatory sterilization standards under national accreditation programs. Automated washers, ultrasound cleaners, and endoscope reprocessors require validated detergents, enzymatic cleaners, and disinfectants formulated for compatibility with high-performance tools. West USA leads with 4.5% CAGR, followed by South USA at 4.0%, Northeast USA at 3.6%, and Midwest USA at 3.1%, with expansion tied to healthcare infrastructure maturity and procurement standardization.
.webp)
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 4.5% |
| South USA | 4.0% |
| Northeast USA | 3.6% |
| Midwest USA | 3.1% |

How is the West USA shaping demand for instrument cleaning chemistries?
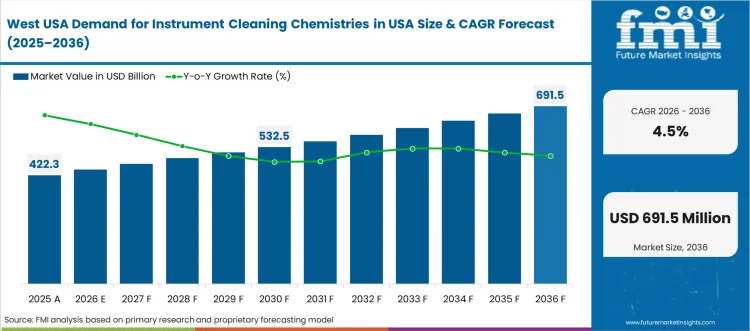
The West USA records 4.5% CAGR, driven by a high concentration of advanced surgical centers and research hospitals across California, Washington, and Colorado. These facilities require enzyme-based detergents and neutral pH formulations that protect delicate robotic and minimally invasive instruments. Endoscope reprocessing programs prioritize chemistries achieving validated microbial reduction outcomes aligned with strict procedural guidance. Procurement decisions emphasize compatibility with automated cleaning systems that prevent material corrosion and device failure. Growing outpatient orthopedic and ophthalmology volumes increase recurring usage of single-cycle cleaning products. Sustainability initiatives encourage low-foam and low-toxicity detergent choices, minimizing chemical discharge while supporting wastewater compliance.
- Robotic surgical adoption increasing enzymatic-detergent needs
- Strict endoscope-reprocessing standards driving validated SKUs
- Automation compatibility shaping detergent and disinfectant selection
- Sustainability compliance influencing product formulation
How is the South USA contributing to demand growth?
The South USA delivers 4.0% CAGR, supported by expanding regional healthcare networks in Texas, Florida, Georgia, and North Carolina. Hospitals depend on standardized chemistries to ensure repeatable disinfection outcomes in large-volume central sterile departments. Dental clinics and ambulatory surgical centers utilize multi-enzyme cleaners for lumen devices and hand instruments, reinforcing continuous procurement cycles. Climatic humidity conditions elevate contamination risks, prompting preventive cleaning workflows and high usage frequency. Distribution advantages through Gulf Coast logistics hubs help stabilize supply access for both private practices and regional health systems. Buyers prioritize value-driven SKUs demonstrating reliable soil breakdown for blood and tissue residues.
- High-volume sterile processing increasing recurrent demand
- Dental and ASC expansion sustaining enzyme-cleaner procurement
- Climate-driven infection-risk mitigation shaping protocols
- Logistics connectivity supporting product availability
How is the Northeast USA influencing purchasing behavior?
The Northeast USA posts 3.6% CAGR, influenced by academically affiliated hospitals and specialty care centers in New York, New Jersey, and Massachusetts. Procurement teams focus on chemistries backed by clinical evidence and validated performance against biofilm formation. Automated decontamination lines used in hospitals require concentrated detergents with controlled residue characteristics to support sensitive optical tools and powered surgical instruments. Regulatory oversight encourages documentation of chemical compatibility testing before approval. Compact healthcare environments in metro areas drive frequent equipment turnaround, increasing detergent consumption intensity. Sustainability compliance prompts selection of chemistries supporting reduced chemical exposure levels for sterile-processing staff.
- Academic medical centers prioritizing clinically proven products
- Compatibility assurance for delicate surgical devices
- High equipment turnover sustaining procurement volumes
- Worker-safety considerations shaping formulation choices
How is the Midwest USA supporting steady adoption?
The Midwest USA shows a 3.1% CAGR, driven by essential cleaning needs across hospital systems and community-based healthcare providers in Illinois, Michigan, and Ohio. Central sterile teams depend on long-standing protocols that use enzyme cleaners and non-corrosive detergents, aligned with equipment warranties. Multi-department usage, including laboratory specimen handling, supports predictable base-level consumption. Procurement practices lean toward cost-optimized replenishment cycles, emphasizing volume discounts and durable packaging formats. Training programs from equipment manufacturers guide correct dilution and dwell-time compliance to maintain cleaning consistency. Growth is moderate, reflecting established infrastructure and fewer high-complexity surgical installations compared to coastal regions.
- Cost-efficient procurement supporting consistent SKU rotation
- Multi-department utilization sustaining baseline demand
- Training-supported accuracy in cleaning procedures
- Stable healthcare infrastructure guiding gradual growth
What is the Competitive Landscape in the Instrument-cleaning Chemistries Industry in the United States?
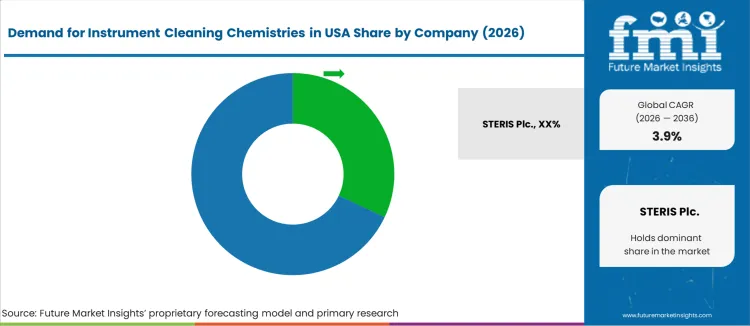
Demand for instrument-cleaning chemistries in the United States centers on supporting sterile-processing departments in hospitals, clinics, and surgical centers. Adoption depends on residue removal for complex reusable devices, compatibility with washer-disinfectors and ultrasonic baths, and validated performance under regulatory hygiene standards. Large providers require chemistry lines that protect stainless steel and maintain lumen cleanliness without compromising downstream sterilization. Purchasing teams prioritize cycle efficiency, dosing accuracy, and material-integrity outcomes to reduce manual cleaning variability.
STERIS plc is estimated near 32.3% based on its broad portfolio of enzymatic and alkaline detergents, neutral cleaners, and compliance support aligned with USA infection-prevention standards. Getinge Group earns share through integrated solutions tailored to automated reprocessing. Ecolab Inc. provides chemistry with linked staff-training programs to strengthen adherence in sterile-processing workflows. 3M participates through pre-treatment and device-care formulations leveraged across surgical-care accounts.
Dr. Weigert maintains a focused presence in precision cleaning where consistent chemical dosing and surface protection remain essential. Growth aligns with higher procedure volumes, reusable-device complexity, and stronger accountability for cleaning validation. The landscape supports innovation in low-foaming action, enzymatic performance against biofilms, and sustainability in healthcare cleaning chemistry.
Key Players in USA Instrument Cleaning Chemistries Demand
- STERIS Plc.
- Getinge Group
- Ecolab Inc.
- 3M
- Dr. Weigert
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.94 billion to USD 1.37 billion, at a CAGR of 3.9% |
| Market Definition | Based on FMI's report, the Instrument Cleaning Chemistries in USA market covers detergents and cleaners formulated specifically for reprocessing reusable medical instruments including surgical instruments, endoscopes, ultrasound probes, and dental instruments across healthcare facilities within the United States. |
| Segmentation | Product Type: Detergents, Cleaners; Process Type: Disinfection, Presoak, Manual Cleaning, Automatic Cleaning; Instrument Type: Surgical Instruments, Endoscopes, Ultrasound, Dental Instruments, Others; End User: Specialty Clinics, Hospitals, Ambulatory Surgical Centers, Diagnostic Centers, Others |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | STERIS Plc., Getinge Group, Ecolab Inc., 3M, Dr. Weigert |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up methodology combining installed base metrics with regulatory compliance adoption curves and primary research validation. |
USA Instrument Cleaning Chemistries Demand by Segments
Product Type:
- Detergents
- Cleaners
Process Type:
- Disinfection
- Presoak
- Manual Cleaning
- Automatic Cleaning
Instrument Type:
- Surgical Instruments
- Endoscopes
- Ultrasound
- Dental Instruments
- Others
End User:
- Specialty Clinics
- Hospitals
- Ambulatory Surgical Centers
- Diagnostic Centers
- Others
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Bibliography
- 1. USA Food and Drug Administration. (2025). Reprocessing Medical Devices in Healthcare Settings: Validation Methods and Labeling. FDA.
- 2. The Joint Commission. (2025). National Patient Safety Goals: Infection Prevention Standards. TJC.
- 3. American Hospital Association. (2025). Annual Survey of Hospital Supply Chain Expenditures. AHA.
- 4. Centers for Disease Control and Prevention. (2024). Guideline for Disinfection and Sterilization in Healthcare Facilities. CDC.
- 5. Association for the Advancement of Medical Instrumentation. (2024). ANSI/AAMI ST79: Comprehensive Guide to Steam Sterilization and Sterility Assurance. AAMI.
- 6. Organisation for Economic Co-operation and Development. (2025). Health at a Glance: Healthcare Quality Indicators. OECD Publishing.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the Instrument Cleaning Chemistries in USA market in 2026?
The Instrument Cleaning Chemistries in USA market is estimated to be valued at USD 0.94 billion in 2026.
What will be the market size of Instrument Cleaning Chemistries in USA by 2036?
Market size for Instrument Cleaning Chemistries in USA is projected to reach USD 1.37 billion by 2036.
What is the expected CAGR for Instrument Cleaning Chemistries in USA between 2026 and 2036?
The Instrument Cleaning Chemistries in USA market is expected to grow at a CAGR of 3.9% between 2026 and 2036.
Which Product Type leads the Instrument Cleaning Chemistries in USA market in 2026?
Detergents accounts for 73.2% share in 2026 by Product Type.
Which region is projected to grow fastest?
West USA is projected to grow at a CAGR of 4.5% during 2026 to 2036.
What does this report include in its scope?
The report covers market sizing, segmentation analysis, regional forecasts, competitive landscape evaluation, and forecast projections from 2026 to 2036.
How does FMI build and validate the Instrument Cleaning Chemistries in USA forecast?
Forecasting models apply a bottom-up methodology starting with installed base metrics and cross-validate projections against quarterly revenue volumes and regulatory compliance adoption data.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Detergents
- Cleaners
- Detergents
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Process Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Process Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Process Type, 2026 to 2036
- Disinfection
- Presoak
- Manual Cleaning
- Automatic Cleaning
- Disinfection
- Y to o to Y Growth Trend Analysis By Process Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Process Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Instrument Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Instrument Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Instrument Type, 2026 to 2036
- Surgical Instruments
- Endoscopes
- Ultrasound
- Dental Instruments
- Others
- Surgical Instruments
- Y to o to Y Growth Trend Analysis By Instrument Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Instrument Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Specialty Clinics
- Hospitals
- Ambulatory Surgical Centers
- Diagnostic Centers
- Others
- Specialty Clinics
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Process Type
- By Instrument Type
- By End User
- Competition Analysis
- Competition Deep Dive
- STERIS Plc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Getinge Group
- Ecolab Inc.
- 3M
- Dr. Weigert
- STERIS Plc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Process Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Instrument Type, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Process Type
- Figure 9: Global Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Instrument Type
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Type
- Figure 29: North America Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Process Type
- Figure 32: North America Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Instrument Type
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Product Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Process Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Instrument Type
- Figure 48: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by End User
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Product Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Process Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Instrument Type
- Figure 61: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by End User
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Process Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Instrument Type
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End User
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Product Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Process Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Instrument Type
- Figure 87: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by End User
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Process Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Instrument Type
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Process Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Process Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Process Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Instrument Type, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Instrument Type, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Instrument Type
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

