Zero Static Diaphragm Valve Market
Zero Static Diaphragm Valve Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Zero Static Diaphragm Valve Market Forecast and Outlook 2026 to 2036
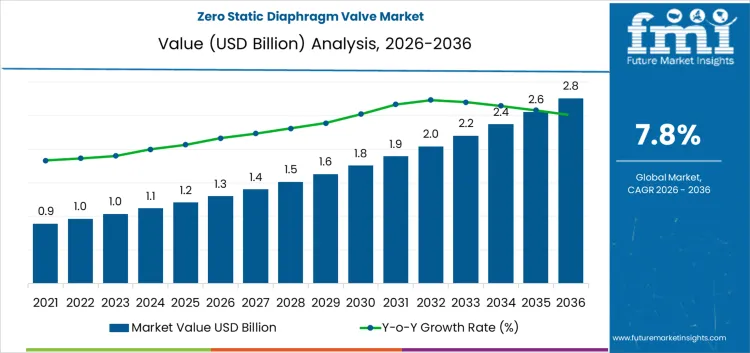
Process changeover requirements in biopharma and specialty chemical plants places the zero static diaphragm valve market at USD 1.3 billion in 2026 and extend it to USD 2.8 billion by 2036 at a 7.80% CAGR. These valves sit at points where dead legs create contamination or batch loss risk. Layout engineers position them inside transfer lines, sampling points, and drain paths during early piping design. Once a facility qualifies a piping class, component changes reopen validation work. Buyers focus on drainability, surface finish, and diaphragm life. Installation planning accounts for welding access, orbital weld schedules, and inspection windows.
Market behavior follows plant qualification cycles and reconfiguration projects rather than routine maintenance. Fabricators coordinate forging, machining, polishing, and cleaning around inspection capacity. Release requires surface roughness checks, material traceability, and leak testing. Integrators influence selection through skids and module packages. Maintenance teams value diaphragm change speed and spare availability. Documentation depth matters during audits. Engineering change control protects interchangeability across lines. Field issues surface as hold up volume or cleaning failures, not as simple leaks. Profit formation follows manufacturing yield, documentation discipline, and predictable project timing rather than price competition or frequent design refreshes.
Quick Stats for Zero Static Diaphragm Valve Market
- Zero Static Diaphragm Valve Market Value (2026): USD 1.3 billion
- Zero Static Diaphragm Valve Market Forecast Value (2036): USD 2.8 billion
- Zero Static Diaphragm Valve Market Forecast CAGR 2026 to 2036: 7.8%
- Leading Valve Type in Zero Static Diaphragm Valve Market: Zero static diaphragm valves
- Leading End Use Industry in Zero Static Diaphragm Valve Market: Pharmaceutical and biotech
- Key Growth Regions in Zero Static Diaphragm Valve Market: Asia Pacific, Europe, North America
- Top Players in Zero Static Diaphragm Valve Market: Emerson, Swagelok Company, ITT Inc., KSB SE & Co. KGaA
What Is the Growth Forecast for the Zero Static Diaphragm Valve Market through 2036?
Process purity requirements and piping design rules set the pace for the zero static diaphragm valve market more than general capital spending levels. In 2026, at about USD 1.3 billion, demand is concentrated in pharmaceutical production, biotechnology facilities, and high purity chemical lines where dead volume and residue carry direct contamination risk. Valve types are selected during process layout design and remain in place across long operating campaigns, which ties replacement demand to line upgrades and capacity changes rather than to routine maintenance. Expansion comes from additional process steps, higher segregation of fluid paths, and stricter internal cleanliness targets. The steady rise reflects broader adoption across critical fluid circuits rather than rapid turnover of existing installations.
Qualification depth and lifecycle cost control shape the later phase of the zero static diaphragm valve market. As value approaches roughly USD 2.8 billion by 2036, buyers focus on diaphragm life, surface finish stability, and repeatable sealing behavior. Product development effort shifts toward easier serviceability and more standardized connection formats. Manufacturing and testing loads increase as documentation and validation requirements grow. The move from the low one billions into the high two billion reflects accumulated build out of regulated production lines rather than a single capacity expansion wave. Suppliers that combine material consistency, reliable delivery schedules, and strong technical support tend to hold preferred positions in approved equipment lists.
Zero Static Diaphragm Valve Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.3 billion |
| Forecast Value (2036) | USD 2.8 billion |
| Forecast CAGR 2026 to 2036 | 7.8% |
How Is the Zero Static Diaphragm Valve Market Enabling Contamination Controlled Fluid Transfer?
Zero static diaphragm valves are designed so that no dead volume remains in the flow path after closure, which prevents product hold up and contamination. In biopharmaceutical and food processing lines, earlier valve designs left pockets that trapped media and complicated cleaning validation. Zero static layouts place the diaphragm directly against the process interface, allowing full drain and repeatable clean in place cycles. Engineers specify these valves by surface finish, elastomer compatibility, cycle life, and pressure rating rather than by size alone. Installation follows critical transfer points such as tank outlets, sampling ports, and filler inlets. Use expands where batch changeover time and microbial risk carry direct cost. Performance is judged through drainability tests, leak integrity, and stability under exposure.
Specification and purchasing are controlled by process engineering and quality groups because valve geometry affects validation data and change control filings. Earlier sourcing accepted mixed models across sites, which increased spares and documentation work. Current programs aim to standardize families across skids and rooms. Manufacturers compete on machining consistency, diaphragm material options, and traceability rather than on catalog breadth. Users compare total cost through cleaning time, downtime during batch change, and inspection findings, not unit price. Assemblers value short lead times and repeatable welding interfaces. Distributors support projects with kitting and local stock support. Over time, demand follows growth in single use systems and tighter audit expectations, where any trapped volume becomes a documented deviation requiring investigation and corrective action.
What Factors Are Shaping the Demand for Zero Static Diaphragm Valve Market in 2026?
Specification in Zero Static Diaphragm Valve Market is driven by fluid path integrity, cleanability requirements, and validation regimes rather than by routine valve replacement cycles. Valve selection is fixed during process design and becomes part of the approved piping and instrumentation documentation. Once a system is qualified, any change in valve configuration requires renewed cleaning validation and process verification. This ties demand to new line installations, capacity expansions, and major process requalification programs. Segment structure reflects differences in dead leg elimination, actuation method, and clean in place compatibility. Buyers prioritize surface finish quality, seal integrity, and traceable material certification because valve performance directly affects product safety and compliance status.
How Does Valve Type Selection Influence Process Control and Compliance Burden in the Zero Static Diaphragm Valve Market?
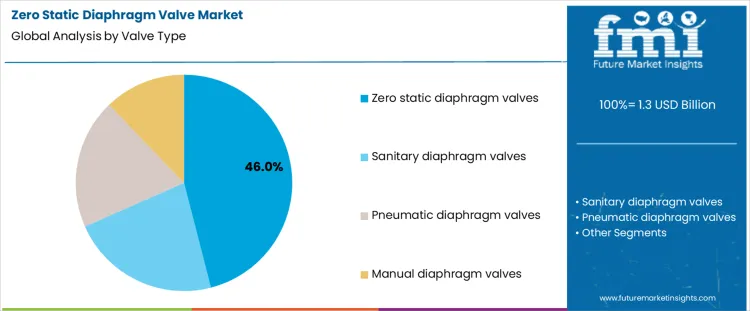
Zero static diaphragm valves represent about 46% of demand in the Zero Static Diaphragm Valve Market because they eliminate dead zones where residue can accumulate, which supports strict hygienic and sterile process requirements. Their design aligns with systems that require frequent cleaning and validated sterility. Sanitary diaphragm valves serve applications with lower sterility class but high hygiene expectations, such as food and beverage processing. Pneumatic diaphragm valves are specified where remote operation and integration into automated control systems are required, increasing the scope of control and validation. Manual diaphragm valves remain in use in utility and auxiliary lines, where simplicity and accessibility are prioritized over automation.
From a compliance management perspective, valve type choice establishes inspection and maintenance workload. Zero static and sanitary designs require routine surface and seal integrity checks as part of cleaning validation programs. Pneumatic systems add actuator and control loop verification into qualification protocols. Manual valves rely more heavily on procedural controls and operator discipline. Once a process line is validated with a given valve configuration, changes are avoided because requalification affects production schedules and regulatory status. Demand by valve type therefore follows long term process design standards and automation strategies rather than short term maintenance budgets.
Why Does End Use Industry Define Volume Concentration and Specification Rigor in the Zero Static Diaphragm Valve Market?
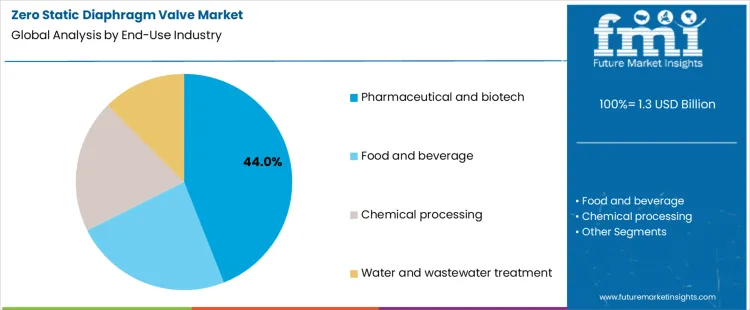
Pharmaceutical and biotech account for about 44% of demand in the Zero Static Diaphragm Valve Market because these facilities operate under strict regulatory oversight and require validated sterile or hygienic fluid handling systems. Food and beverage processing also requires hygienic design, though acceptable risk levels and validation depth differ by product category. Chemical processing applies diaphragm valves for corrosion resistance and containment, where material compatibility and actuator reliability dominate specification. Water and wastewater treatment uses manual and basic automated valves for isolation and control, where volume requirements are high but hygienic standards are lower. These differences explain why volume concentrates in regulated life science production while technical scrutiny varies across other sectors.
End use industry also shapes procurement and approval practice. Pharmaceutical manufacturers purchase through capital projects with formal design qualification and validation plans. Food processors align valve purchases with line upgrades and compliance audits. Chemical plants integrate valve selection into hazard and operability reviews and maintenance strategies. Water utilities follow budget cycles and infrastructure renewal programs. The resulting demand structure concentrates high specification products in life sciences, while other sectors contribute volume in lower specification categories. Segment shares therefore move with capacity expansion, regulatory inspection cycles, and process modernization programs rather than with short term production volume changes.
How Is The Zero Static Diaphragm Valve Market Being Defined By Contamination Control And Process Integrity Requirements?
The Zero Static Diaphragm Valve Market is shaped by how pharmaceutical, biotechnology, and high-purity process operators manage dead leg elimination and cleanability in fluid handling systems. In these environments, any trapped fluid can compromise batch quality and validation status. Buyers focus on valve geometry, surface finish, diaphragm reliability, and compatibility with cleaning and sterilization procedures. Selection decisions involve process engineering, quality, and validation teams rather than only procurement. This positions zero static diaphragm valves as process-critical components that directly influence yield protection and compliance confidence.
How Is Skid And Process Module Standardization Changing Valve Selection In The Zero Static Diaphragm Valve Market?
Process systems are increasingly built using repeatable skids and modules to simplify design, installation, and qualification. This is changing expectations in the Zero Static Diaphragm Valve Market toward valve designs that fit standardized piping layouts and consistent automation interfaces. Engineers prefer valve families that can be applied across multiple units without layout changes. This reduces engineering time and spare parts complexity. As a result, suppliers are evaluated on portfolio consistency, documentation quality, and long-term product availability rather than on highly customized solutions for single installations.
Where Is Repeat Installation Volume Being Built In The Zero Static Diaphragm Valve Market?
Repeat installation volume is being built in facilities that expand capacity in phases or replicate the same process line across multiple sites. Each new skid, line extension, or clean utility loop requires multiple zero static diaphragm valves. The Zero Static Diaphragm Valve Market also benefits from retrofit projects where older valve designs are replaced to reduce cleaning risk and improve process reliability. There is opportunity for suppliers that become part of approved vendor lists and standard design libraries used by engineering firms and plant owners, securing ongoing demand tied to capital project pipelines.
Why Do Qualification Burden And Higher Unit Cost Limit Wider Use In The Zero Static Diaphragm Valve Market?
Zero static diaphragm valves carry a higher unit cost than conventional valve types due to their specialized design and finishing requirements. In addition, once a valve type is qualified in a regulated process, changing it requires documentation updates and validation work. This makes users cautious about both adopting new designs and switching suppliers. In less critical parts of a plant, operators may accept standard valves to control costs. These factors limit broader use to high-purity and high-risk sections of processes and slow expansion into less sensitive applications.
What is the Demand for Zero Static Diaphragm Valve by Country?
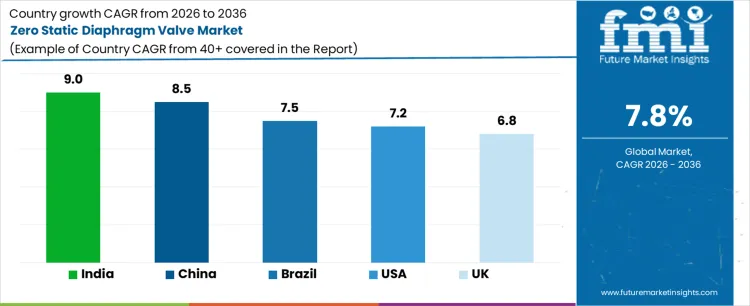
| Country | CAGR |
|---|---|
| USA | 7.2% |
| UK | 6.8% |
| China | 8.5% |
| India | 9.0% |
| Brazil | 7.5% |
Demand for zero static diaphragm valves is increasing as pharmaceutical, biotechnology, and high purity process industries tighten contamination control and cleanability requirements in fluid handling systems. India leads with a 9.0% CAGR, supported by rapid expansion of sterile drug manufacturing, vaccine production, and new cleanroom facilities. China follows at 8.5%, driven by growth in biologics, semiconductor chemicals, and regulated process plants that require low hold up volume valve designs. Brazil records 7.5%, reflecting capacity additions in pharmaceuticals and specialty chemicals. The USA grows at 7.2%, shaped by equipment upgrades in existing facilities and compliance driven retrofits. The UK, at 6.8%, reflects steady but mature investment in regulated manufacturing infrastructure.
How Do High Purity Process Standards Shape Demand in the United States?
Process control investment keeps the zero static diaphragm valve market in the United States on a 7.2% CAGR path. Semiconductor fabs, biopharmaceutical plants, and specialty chemical lines represent the main users. Engineering teams specify valves based on dead leg free flow paths, surface finish quality, and cleaning validation compatibility. Procurement aligns with tool installation programs and plant expansion schedules. Factory acceptance testing covers leakage, cycle life, and material traceability. Maintenance teams monitor diaphragm wear and actuator response. Replacement demand follows tool upgrades or contamination control audits. Distributors manage regional stock and service coordination. Documentation supports quality system reviews and customer inspections. Training programs guide correct installation and torque practices. Inventory planning follows equipment delivery timelines. Commercial position depends on approved vendor status and consistent field performance rather than competition for short cycle component orders.
Why Do Validation Protocols and Clean Utility Projects Guide Adoption in the United Kingdom?
Regulated production requirements place the zero static diaphragm valve market in the United Kingdom on a 6.8% CAGR trajectory. Biopharma sites, research laboratories, and fine chemical plants account for most installations. Project teams define valve specifications around drainability, clean in place performance, and surface finish standards. Procurement flows through framework agreements tied to utility system upgrades. Acceptance relies on material certificates and pressure testing records. Maintenance planning focuses on diaphragm condition and actuator reliability. Replacement occurs after audit observations or line reconfiguration. Distributors maintain spares near major clusters. Installation schedules follow shutdown and qualification windows. Documentation remains part of validation files. Training supports consistent assembly practices. Capital planning aligns with facility refurbishment cycles. Commercial access depends on framework inclusion and audit performance rather than competition for isolated valve purchases.
What Role Do Large Scale Fabrication Programs Play in China?
Industrial expansion programs place the zero static diaphragm valve market in China on an 8.5% growth path. Semiconductor fabs, battery material plants, and pharmaceutical facilities drive most demand. Design institutes publish standard piping modules specifying valve types and materials. Domestic manufacturing supports volume output and delivery stability. Procurement aggregates quantities through project packages. Installation teams fit valves during system build stages. Quality groups verify surface finish, pressure integrity, and functional response. Replacement activity follows line expansion or contamination control reviews. Regional warehouses stage spares for rapid support. Payment schedules align with construction milestones. Training programs guide handling and installation. Documentation supports acceptance inspections. Commercial success depends on inclusion in national project standards and consistent execution across contractors rather than fragmented sales to small independent plants.
How Do Capacity Buildout and Contract Manufacturing Shape Demand in India?
Manufacturing investment fixes the zero static diaphragm valve market in India at a 9.0% CAGR. Pharmaceutical plants, biologics facilities, and electronics material sites represent core customers. Engineering consultants specify valves based on cleanability, drainability, and corrosion resistance. Procurement often occurs through turnkey project contracts. Local assembly supports basic configurations while specialized units arrive through approved suppliers. Installation aligns with piping qualification schedules. Maintenance teams track diaphragm life and actuator performance. Replacement demand follows inspection findings or process modifications. Distributor networks manage regional service coverage. Training programs support correct installation practices. Inventory planning follows project pipelines. Documentation remains part of validation packages. Commercial results depend on inclusion in approved vendor lists and dependable execution rather than competition for one time line level component purchases.
How Do Process Modernization and Utility Upgrades Shape Spending in Brazil?
Facility upgrades keep the zero static diaphragm valve market in Brazil near a 7.5% CAGR. Pharmaceutical plants, specialty chemical units, and food ingredient facilities form the main user base. Engineering firms define valve selection based on hygiene standards and cleaning access. Procurement follows capital projects rather than incremental buying. Import procedures influence lead times for high specification units. Local distributors supply standard configurations. Acceptance relies on pressure testing and material documentation. Maintenance planning covers diaphragm replacement and actuator checks. Replacement appears after audit findings or piping changes. Distributors stock spares near industrial centers. Training supports correct handling. Documentation remains part of inspection records. Commercial position depends on approved supplier status and reliable delivery rather than competition for small batch orders.
How Do Valve Manufacturers Compete for Process Approval in the Zero Static Diaphragm Valve Market?
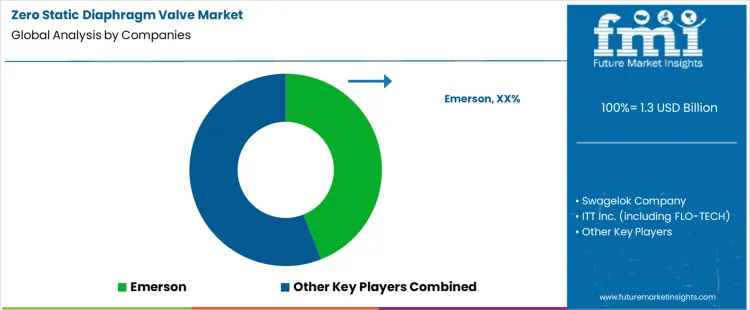
Zero static diaphragm valve selection sits inside process design reviews for chemical handling, semiconductor wet benches, and high purity fluid systems. Engineers define dead volume limits, cleanability targets, pressure classes, and actuation requirements before any brand appears in specifications. Emerson, Swagelok, ITT, KSB compete during that approval stage through different strengths. Once a valve family enters piping standards, replacement requires new flushing studies and validation records. Emerson aligns valves with broader control and instrumentation architectures. Swagelok focuses on system fit, tubing compatibility, and assembly practices. ITT supplies niche designs used in sampling and specialty flow control. KSB supports industrial plants that require standardized materials and long service intervals. Installed base and documentation depth anchor decisions across multiyear operating plans.
Competitive position follows reliability records, surface finish control, and maintenance procedures rather than list pricing. Process owners examine cavity geometry, diaphragm life, actuation repeatability, and particle generation during qualification. Construction teams value suppliers that deliver consistent port dimensions and drawing packages. Maintenance groups prefer designs that allow predictable diaphragm change schedules and simple leak checks. Emerson benefits from integration with plant control standards and procurement frameworks. Swagelok secures positions where tubing systems and valves share common specifications. ITT holds share in applications that require compact bodies and sampling oriented layouts. KSB competes in chemical and industrial plants that emphasize standardization and service coverage. Market positions vary by industry purity class, plant governance model, and internal change control rules.
Key Players of Zero Static Diaphragm Valve Market
- Emerson
- Swagelok Company
- ITT Inc. (including FLO-TECH)
- KSB SE & Co. KGaA
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Valve Type | Zero static diaphragm valves, Sanitary diaphragm valves, Pneumatic diaphragm valves, Manual diaphragm valves |
| End-Use Industry | Pharmaceutical and biotech, Food and beverage, Chemical processing, Water and wastewater treatment |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Emerson, Swagelok Company, ITT Inc. (including FLO-TECH), KSB SE & Co. KGaA |
| Additional Attributes | Dollar sales by valve type and end-use; demand driven by biopharma, chemicals, and high-purity lines; selection based on drainability, surface finish, diaphragm life, and validation needs; procurement via capital projects and skids; focus on documentation, traceability, inspection records, maintenance cycles, and repeat installations in regulated facilities. |
Zero Static Diaphragm Valve Market Segmentation
Valve Type:
- Zero static diaphragm valves
- Sanitary diaphragm valves
- Pneumatic diaphragm valves
- Manual diaphragm valves
End-Use Industry:
- Pharmaceutical and biotech
- Food and beverage
- Chemical processing
- Water and wastewater treatment
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- ASME BPE Committee. (2024). ASME BPE 2024: Bioprocessing Equipment — Standards for design and construction of components for use in bioprocessing, pharmaceutical, and high-purity applications. ASME.
- ASME Bioprocessing Equipment Committee. (2025). ASME BPE 2025: Bioprocessing equipment — Standards for design and construction of components used in bioprocessing, pharmaceutical, and high-purity applications. American Society of Mechanical Engineers.
- International Organization for Standardization. (2025). ISO 21003-1:2025 — Valves for the food industry — Hygienic design and aseptic processing. ISO.
Frequently Asked Questions
How big is the zero static diaphragm valve market in 2026?
The global zero static diaphragm valve market is estimated to be valued at USD 1.3 billion in 2026.
What will be the size of zero static diaphragm valve market in 2036?
The market size for the zero static diaphragm valve market is projected to reach USD 2.8 billion by 2036.
How much will be the zero static diaphragm valve market growth between 2026 and 2036?
The zero static diaphragm valve market is expected to grow at a 7.8% CAGR between 2026 and 2036.
What are the key product types in the zero static diaphragm valve market?
The key product types in zero static diaphragm valve market are zero static diaphragm valves , sanitary diaphragm valves, pneumatic diaphragm valves and manual diaphragm valves.
Which end‑use industry segment to contribute significant share in the zero static diaphragm valve market in 2026?
In terms of end‑use industry, pharmaceutical and biotech segment to command 44.0% share in the zero static diaphragm valve market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Valve Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Valve Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Valve Type , 2026 to 2036
- Zero static diaphragm valves
- Sanitary diaphragm valves
- Pneumatic diaphragm valves
- Manual diaphragm valves
- Zero static diaphragm valves
- Y to o to Y Growth Trend Analysis By Valve Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Valve Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End‑Use Industry
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End‑Use Industry, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End‑Use Industry, 2026 to 2036
- Pharmaceutical and biotech
- Food and beverage
- Chemical processing
- Water and wastewater treatment
- Pharmaceutical and biotech
- Y to o to Y Growth Trend Analysis By End‑Use Industry, 2021 to 2025
- Absolute $ Opportunity Analysis By End‑Use Industry, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Valve Type
- By End‑Use Industry
- By Country
- Market Attractiveness Analysis
- By Country
- By Valve Type
- By End‑Use Industry
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Valve Type
- By End‑Use Industry
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Valve Type
- By End‑Use Industry
- Competition Analysis
- Competition Deep Dive
- Emerson
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Swagelok Company
- ITT Inc. (including FLO‑TECH)
- KSB SE & Co. KGaA
- Emerson
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Valve Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End‑Use Industry, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Valve Type
- Figure 6: Global Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End‑Use Industry
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Valve Type
- Figure 23: North America Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by End‑Use Industry
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Valve Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by End‑Use Industry
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Valve Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by End‑Use Industry
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Valve Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End‑Use Industry
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Valve Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by End‑Use Industry
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Valve Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End‑Use Industry
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Valve Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Valve Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Valve Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End‑Use Industry, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End‑Use Industry, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End‑Use Industry
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

