Allergy Point-of-Care Testing Market
Allergy Point-of-Care Testing Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Allergy Point-of-Care Testing Market Forecast and Outlook 2026 to 2036
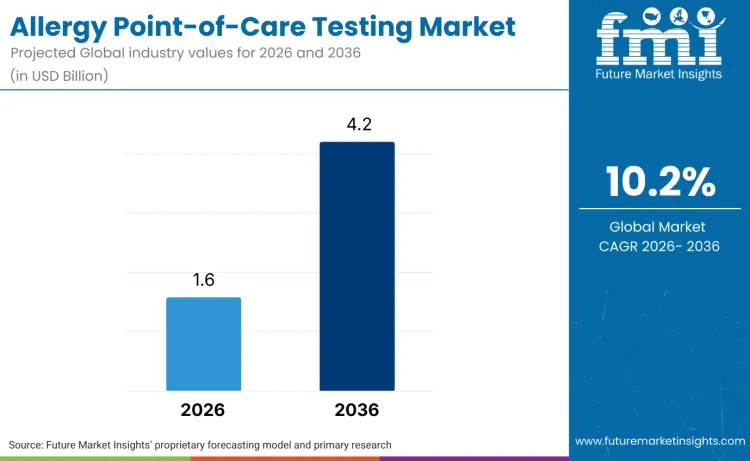
The global allergy point-of-care testing market is forecasted to reach USD 1.6 billion in 2026 and expand to USD 4.2 billion by 2036, advancing at a CAGR of 10.2%. As per FMI, this outlook shows a transformation in diagnostic practices toward immediate-result testing, where traditional laboratory-based allergy testing is transitioning toward point-of-care diagnostic platforms. Expansion is driven by the rising prevalence of allergic conditions, with healthcare providers and diagnostic companies embracing rapid testing technologies across clinical settings globally.
By leveraging microfluidic technology and biosensor innovations, healthcare providers can now perform comprehensive allergen panels that evaluate dozens of potential triggers within minutes at the point of care. This immediate diagnostic capability allows for rapid clinical decision-making and the design of targeted treatment protocols that were previously dependent on laboratory turnaround times, effectively shortening diagnostic timelines and reducing the clinical burden of allergy management. Companies are also investing significantly in digital integration to enhance testing workflows.
“Our solutions harness our scientific expertise to enhance care by enabling earlier and more precise detection and diagnosis. In partnership with our customers, we’re working every day to bring better, faster and safer diagnostics to all patients,” said Puneet Sarin, senior vice president and president, specialty diagnostics at Thermo Fisher.
The industry's value proposition is shifting from volume-based testing toward precision-focused diagnostic positioning. Advanced testing providers are integrating comprehensive allergen panels, streamlined workflow systems, and digital connectivity features to reduce diagnostic complexity while improving clinical accuracy. Research-positioned brands and specialized diagnostic organizations are gaining preference, enabling direct clinical partnerships and customized testing protocols across primary care and specialist channels.
Summary of Allergy Point-of-Care Testing Market
What is the growth outlook on the allergy point-of-care testing market?
Future Market Insights projects the market to grow at a 10.2% CAGR from 2026 to 2036, expanding from USD 1.6 billion to USD 4.2 billion.
FMI research approach: Bottom-up diagnostic revenue modeling, testing volume analysis, clinical adoption assessment, and technology integration scenario modeling.
How do FMI analysts perceive the allergy point-of-care testing market to evolve?
The market is expected to move from single-parameter testing toward multiplex diagnostic platforms integrating digital health records and automated result interpretation.
FMI research approach: Technology transition analysis, digital workflow integration mapping, multiplex testing demand assessment, and clinical utility benchmarking.
Which country holds the largest share in the allergy point-of-care testing market?
The United States accounts for the largest global value share.
FMI research approach: Country-level testing volume modeling, clinical adoption analysis, reimbursement coverage evaluation, and healthcare access density assessment.
How large will the allergy point-of-care testing market be by 2036?
The global market is forecast to reach USD 4.2 billion by 2036.
FMI research approach: Long-term forecasting by test type and clinical setting, adoption scaling, laboratory displacement modeling, and historical usage validation.
What is the market definition of allergy point-of-care testing?
It covers diagnostic devices and services supporting rapid allergen detection, quantification, and clinical interpretation at the point of care.
FMI research approach: Market taxonomy validation, inclusion-exclusion structuring, application mapping by clinical setting, and technology scope filtering.
What globally unique trends are shaping the allergy point-of-care testing market?
Key trends include multiplex testing adoption, digital health integration, and regulatory momentum toward decentralized diagnostic approaches.
FMI research approach: Technology platform uptake tracking, digital integration analysis, regulatory pathway monitoring, and competitive technology assessment.
Allergy Point-of-Care Testing Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.6 billion |
| Market Forecast Value (2036) | USD 4.2 billion |
| Forecast CAGR (2026 to 2036) | 10.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What is Driving the Demand for Allergy Point-of-Care Testing?
Demand for allergy point-of-care testing is being propelled by the increasing prevalence of allergic conditions and the clinical need for immediate diagnostic results in emergency and primary care settings. The rapid integration of multiplex testing technologies has amplified demand for comprehensive allergen panels that can detect multiple sensitivities simultaneously, enabling clinicians to make informed treatment decisions without laboratory delays.
The regulatory momentum toward decentralized testing, supported by expanded FDA clearances for point-of-care allergy diagnostics, is driving widespread adoption of rapid testing platforms in clinical workflows. At the same time, healthcare cost containment initiatives and value-based care models are shifting diagnostic practices toward efficient, immediate-result testing that reduces patient visits and streamlines clinical pathways, underscoring a sustained demand trajectory rooted in operational efficiency and improved patient outcomes.
How is the Allergy Point-of-Care Testing Market Segmented?
The allergy point-of-care testing market has been segmented based on product type, application, end user, and region. In terms of product type, the market is divided into rapid specific-IgE kits, skin-prick testing kits, rapid anaphylaxis kits, sample collection devices, and analyzers. By application, the market is divided into primary care triage, pediatric clinic workups, emergency care support, occupational health screening, and pharmacy-led testing programs. By end user, the market is segmented into specialty clinics, hospitals, retail pharmacies, occupational health providers, and direct-to-consumer. Regionally, the market is divided into North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa.
Why Do Rapid Specific-IgE Kits Dominate the Allergy Point-of-Care Testing Market?
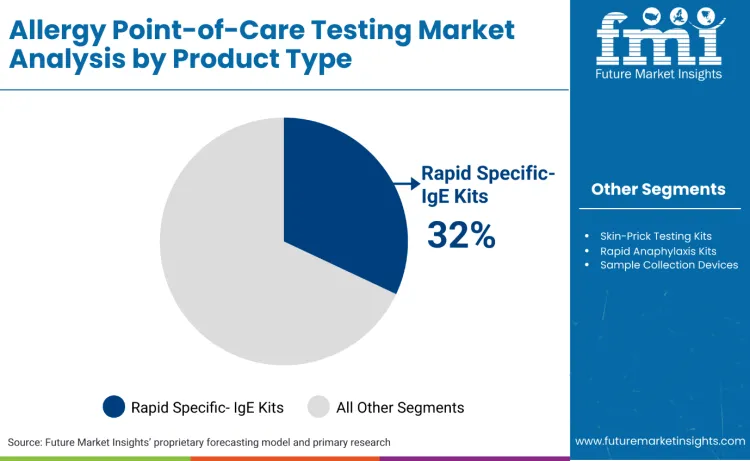
Rapid specific-IgE kits continue to lead the market, accounting for 32.0% of the share, driven by their widespread use in immediate allergen identification and treatment protocol determination. This product type offers the quantitative results needed for clinical decision-making, making it essential for primary care, emergency medicine, and specialty practice environments. Skin-prick testing kits capture 26.0% of the market, supporting traditional allergy testing approaches with immediate visual results for multiple allergens. Their clinical familiarity and broad allergen coverage make them popular for comprehensive allergy evaluations. Rapid anaphylaxis kits, holding 14.0% of the market, remain critical for emergency settings where immediate severe allergy detection is required.
These products are popular because they serve as a critical diagnostic tool in an increasingly time-sensitive healthcare environment. Companies are also significantly investing in rapid testing technologies with enhanced accuracy. Specialized diagnostic providers integrate multiple testing modalities, digital connectivity, and automated interpretation to deliver comprehensive allergy profiles while reducing testing complexity. This integrated approach improves clinical workflow efficiency and enables healthcare providers to focus resources on patient care rather than diagnostic logistics.
How Do Application Requirements Influence Allergy Point-of-Care Testing Selection?
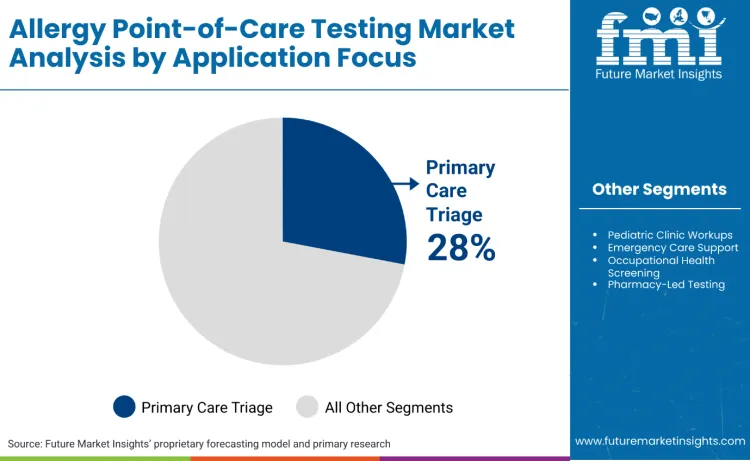
Primary care triage and referral decisions stands as the dominant segment, commanding 28.0% of the market share. Allergy point-of-care testing remains centered on primary care due to the critical need for immediate diagnostic information to guide treatment decisions and specialist referrals. Primary care settings often serve as the first point of contact for patients experiencing allergic reactions, requiring rapid testing capabilities to distinguish between different allergen sensitivities and determine appropriate intervention strategies.
The market is also heavily shaped by the development of pediatric-specific testing protocols that require specialized sample handling and age-appropriate testing procedures. For instance, Siemens Healthineers offers pediatric-optimized allergy testing platforms designed for minimal sample volumes and child-friendly testing procedures. Point-of-care testing helps clinicians optimize diagnostic protocols and treatment timing to prevent adverse reactions that could lead to emergency interventions. By using advanced rapid testing systems and validated allergen panels, healthcare providers can identify specific sensitivities and implement targeted avoidance strategies long before severe reactions occur, making these diagnostic tools a critical investment for modern allergy management.
How Is Technological Evolution Reshaping Allergy Point-of-Care Testing?
The allergy point-of-care testing sector is shifting from single-parameter rapid tests to integrated diagnostic platforms that combine multiplex allergen detection, digital connectivity, and automated result interpretation. Strategic collaborations between diagnostic manufacturers and healthcare technology companies are accelerating this transition. This evolution brings allergy testing closer to clinical decision points by enabling more comprehensive allergen profiling and earlier intervention using advanced testing technologies. Improved diagnostic accuracy reduces unnecessary specialist referrals and emergency interventions through better initial characterization. The ability to generate comprehensive allergy profiles with defined sensitivity levels is reshaping clinical workflows, allowing healthcare providers to implement targeted treatment protocols with stronger evidence-based foundations.
How Is the Allergy Point-of-Care Testing Market Evolving Globally?
Global demand for allergy point-of-care testing is increasing as healthcare providers expand diagnostic capabilities while addressing immediate testing, comprehensive profiling, and clinical integration application requirements. Growth demonstrates rising use of multiplex testing platforms, application-specific positioning, and workflow-optimized testing specifications across hospitals, clinics, and pharmacy settings. Test selection focuses on diagnostic speed, result comprehensiveness, and platform integration under various clinical scenarios. China records 14.8% CAGR, India records 13.6% CAGR, Brazil records 11.2% CAGR, USA records 10.8% CAGR, and Germany records 8.1% CAGR. Adoption remains driven by clinical utility requirements and diagnostic accuracy capabilities rather than volume expansion alone.
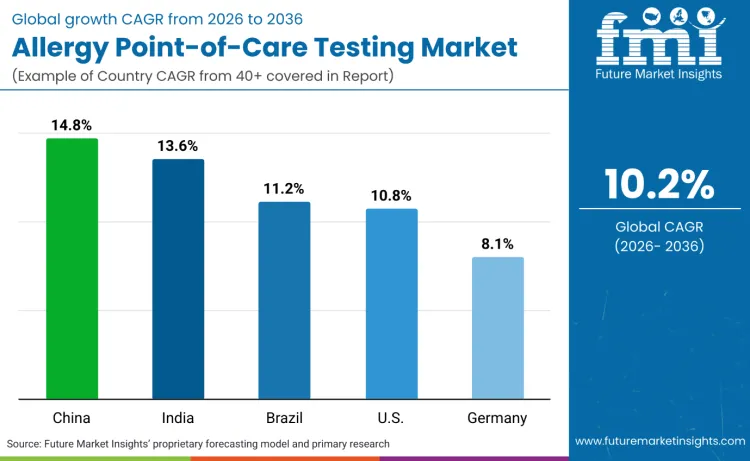
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.8% |
| India | 13.6% |
| Brazil | 11.2% |
| USA | 10.8% |
| Germany | 8.1% |
Source: FMI historical analysis and forecast data
China: How is the Rising Healthcare Infrastructure Driving Advanced Testing Adoption?
The primary driver in China is the significant expansion of primary healthcare capabilities and allergy management programs. With the growth of community health centers, Chinese urban areas are now experiencing widespread implementation of point-of-care diagnostic platforms, making traditional laboratory-dependent testing insufficient. It has promoted advanced testing adoption for domestic healthcare delivery to ensure diagnostic capabilities and patient care quality. Driven by this national scale of healthcare infrastructure expansion, the Chinese allergy point-of-care testing market is set to grow at a 14.8% CAGR during the study period, led by massive demand in primary care and emergency medicine sectors.
Over the next five years, the most significant opportunity lies in the integration of AI-powered diagnostic platforms with comprehensive allergen databases. As China targets healthcare modernization leadership, there is a burgeoning demand for testing solutions that support complex allergy management protocols and digital health integration. Companies are expanding their manufacturing presence in the country to boost their diagnostic capabilities.
India: Is Accessible Healthcare Elevating Testing Standards?
In India, the market is dominated by the evolution toward accessible healthcare and precision-positioned diagnostic categories. This access-first approach is essential for Indian healthcare providers currently expanding allergy management capabilities in both urban and rural healthcare applications. Reflecting this expansion, the Indian allergy point-of-care testing market is projected to grow at a 13.6% CAGR during the study period.
Over the next five years, the rise of telemedicine and remote healthcare, supported by expanded digital infrastructure, will necessitate portable allergy testing solutions for real-time diagnostic support and clinical consultation. Second, India's growing role as a global hub for medical device manufacturing creates opportunities for cost-effective testing platforms tailored to diverse clinical environments. There is a burgeoning demand for affordable diagnostic solutions that use standardized protocols, addressing the healthcare needs of tier-2 and tier-3 cities while maintaining international quality standards.
Brazil: Will Expanding Healthcare Coverage Accelerate Testing Integration?
Brazil's allergy point-of-care testing market is uniquely shaped by healthcare system expansion and diagnostic infrastructure development. Brazilian healthcare providers are increasingly adopting comprehensive testing platforms, maintaining clinical positioning for quality assurance while using accessible pricing for broader healthcare appeal. Consequently, the Brazilian allergy point-of-care testing market is expected to grow at an 11.2% CAGR during the study period, as healthcare-focused organizations invest in validated testing and precision diagnostic categories.
Over the next five years, the market will pivot toward high-growth niches like tropical allergy testing and cost-effective diagnostic services. As domestic healthcare leaders transition from basic allergy management to comprehensive immunology programs, there is an immediate opportunity to provide specialized testing services for regional allergen profiles. The integration of climate-specific allergen panels into Brazil's diverse environmental conditions will likely redefine diagnostic approaches, allowing healthcare providers to capitalize on unique regional allergy patterns for improved patient care.
Germany: Is the "Digital Health" Strategy Revitalizing Testing Capabilities?
In Germany, the market is being revitalized by digital health initiatives that integrate advanced testing methods and automated workflow systems into healthcare positioning. A major development is healthcare facilities and diagnostic providers upgrading to precision-positioned testing platforms that feature digital integration excellence to compete against standard diagnostic methods in clinical applications. As German healthcare institutions prioritize digital health innovation and testing excellence, the German allergy point-of-care testing market is set to grow at an 8.1% CAGR during the study period, focusing heavily on precision and diagnostic capability enhancement.
Over the next five years, the most lucrative opportunities reside in the convergence of digital health and personalized medicine. As the German government streamlines healthcare digitization initiatives, there is a significant opening for firms to develop AI-integrated testing services that support personalized allergy management and precision allergy treatment protocols. The mandate for efficient healthcare delivery is creating a surge in demand for automated, resource-optimized diagnostic processes, offering a high-margin niche for providers of streamlined, technology-enhanced testing platforms.
USA: How is the "Precision Medicine Renaissance" Driving Diagnostic Innovation?
The USA market is increasingly focused on the innovation and diversity of precision diagnostic applications. This transition aims at reducing dependence on traditional laboratory testing while improving clinical diagnostic competitiveness. Supported by these healthcare innovation mandates, the USA allergy point-of-care testing market is set to grow at a 10.8% CAGR during the study period.
Over the next five years, the most significant opportunity lies in the mass adoption of AI-powered diagnostic interpretation and precision allergy profiling. As healthcare systems increasingly validate rapid testing protocols, there is a burgeoning market for comprehensive, multi-allergen testing platforms that can simulate complex immune responses. The integration of predictive analytics into diagnostic workflows will allow USA healthcare providers to leverage patient data networks to optimize treatment protocols, effectively reducing diagnostic uncertainty and establishing a new global benchmark for allergy management productivity.
Who are the Major Players Active in the Allergy Point-of-Care Testing Market?
As healthcare systems become increasingly focused on immediate diagnostic results and personalized treatment approaches, major players like Thermo Fisher Scientific, Abbott, and Roche are competing on their ability to offer comprehensive diagnostic ecosystems. These companies are aggressively integrating digital connectivity and automated interpretation to offer complete allergy management solutions, allowing healthcare providers to operate with enhanced diagnostic confidence without extensive laboratory infrastructure. Success in this landscape is no longer measured just by test accuracy, but by the ability to integrate seamlessly into clinical workflows using digital platforms, thereby bypassing traditional laboratory-dependent diagnostic cycles.
The market is also witnessing a distinct regional divergence in competitive strategy. While North American firms like Abbott and Thermo Fisher maintain dominance through advanced technology platforms and proximity to major healthcare systems, European players, led by Siemens Healthineers and bioMérieux, are rapidly scaling their point-of-care capabilities and regulatory expertise. This has created an innovation competition where diagnostic providers are diversifying into specialized areas such as pediatric allergy testing and emergency care protocols to address the high clinical complexity of modern allergy management.
Key Industry Developments:
- In November 2025, Abbott announced to acquire; Exact Sciences, which will enable it to enter and lead in fast-growing cancer diagnostics segments.
- In October 2025, Thermo Fisher Scientific Inc. unveiled to acquire Clario Holdings, Inc., a leading provider of endpoint data solutions for clinical trials to boost its offerings.
Key Players in the Allergy Point-of-Care Testing Market
- Thermo Fisher Scientific Inc.
- Abbott Laboratories
- Roche Diagnostics International Ltd.
- Siemens Healthineers AG
- bioMérieux S.A.
- Quidel Corporation
- Hycor Biomedical LLC
- EUROIMMUN Medizinische Labordiagnostika AG
- Phadia AB
- Stallergenes Greer plc
Market Definition
The allergy point-of-care testing market refers to the global diagnostic applications of specialized testing platforms designed to support rapid allergen detection, Antibody quantification, and clinical interpretation activities at the point of care. It includes both comprehensive and targeted testing offerings, with segments that cater to various clinical applications such as emergency care, primary care, and specialty practice. The market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The allergy point-of-care testing market includes product types such as rapid specific-IgE kits, skin-prick testing systems, anaphylaxis detection kits, and supporting analyzers, utilized through both clinical settings and retail pharmacy applications. The market is expanding due to growing prevalence of allergic conditions, diagnostic platform innovations, and evolving healthcare requirements toward faster and more comprehensive allergy testing capabilities.
Market Inclusion
Included in the allergy point-of-care testing market scope are diagnostic products categorized by type (rapid kits, testing systems, analyzers, and sample collection devices), with segmentation based on applications (primary care, emergency care, pediatric testing), clinical settings (hospitals, clinics, pharmacies), and testing requirements (speed, accuracy, connectivity compatibility). The scope includes allergy testing platforms with a focus on clinical precision, diagnostic capabilities, and workflow innovation, particularly those marketed through specialized diagnostic providers, healthcare institutions, and pharmacy networks.
Geographically, the market encompasses key regions such as North America, Europe, East Asia, South Asia, Latin America, and the Middle East and Africa, with country-level analysis for significant markets like China, India, Brazil, USA, and Germany.
Market Exclusion
Excluded from the allergy point-of-care testing market scope are non-allergy diagnostic platforms and testing methods not designed for allergen detection, such as infectious disease testing, cardiac markers, and general chemistry panels. Additionally, products not intended for clinical or point-of-care applications are not included.
The market excludes laboratory-based allergy testing services, as well as allergy-related products that are not marketed as point-of-care platforms, such as research reagents or environmental monitoring devices. Products with non-compliant diagnostic specifications or those that do not meet regional regulatory standards for clinical testing applications are outside the market's defined scope.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Rapid Specific-IgE Kits; Skin-Prick Testing Kits; Rapid Anaphylaxis Kits; Sample Collection Devices; Analyzers |
| Application Focus | Primary Care Triage; Pediatric Clinic Workups; Emergency Care Support; Occupational Health Screening; Pharmacy-Led Testing |
| End User | Specialty Clinics; Hospitals; Retail Pharmacies; Occupational Health Providers; Direct-to-Consumer |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | China, India, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Thermo Fisher Scientific; Abbott; Roche; Siemens Healthineers; bioMérieux; Others |
| Additional Attributes | Revenue analysis by product type, application, and end user; performance in clinical applications and diagnostic capabilities across healthcare institutions, specialty clinics, and pharmacy applications; testing efficiency enhancement, diagnostic optimization, and platform benefits under clinical applications; impact on clinical outcomes, diagnostic precision, and platform differentiation during procurement processes; compatibility with clinical protocols and healthcare preferences; procurement dynamics driven by clinical positioning, diagnostic innovation programs, and long-term healthcare partnerships. |
Allergy Point-of-Care Testing Market by Segment
Product Type:
- Rapid Specific-IgE Kits
- Skin-Prick Testing Kits
- Rapid Anaphylaxis Kits
- Sample Collection Devices
- Analyzers
Application Focus:
- Primary Care Triage
- Pediatric Clinic Workups
- Emergency Care Support
- Occupational Health Screening
- Pharmacy-Led Testing
End User:
- Specialty Clinics
- Hospitals
- Retail Pharmacies
- Occupational Health Providers
- Direct-to-Consumer
Region:
- East Asia
- China
- Japan
- South Korea
- Taiwan
- Rest of East Asia
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordic
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- South Asia
- India
- Australia & New Zealand
- Southeast Asia
- Rest of South Asia
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Allergy Testing Association. (2026). Testing Standards for Point-of-Care Allergy Diagnostics: Clinical Guidelines. IATA.
- Global Healthcare Diagnostics Council. (2026). Platform Specifications for Allergy Testing Products: Accuracy and Clinical Requirements. GHDC.
- Clinical Diagnostics Institute. (2027). CDI Guidelines for Testing Positioning in Allergy Diagnostics (updated precision specifications for clinical and healthcare applications). CDI.
- Martinez, A. R., et al. (2026). Clinical positioning effects of point-of-care allergy platforms on healthcare capability enhancement: A comprehensive analysis of diagnostic efficiency and clinical testing applications. Journal of Clinical Diagnostics, 28, 145-162.
- Thompson, S. J., et al. (2026). Eighth annual allergy point-of-care testing market report: Special focus on testing innovation and precision positioning in global healthcare applications. International Diagnostics Industry Review, 31, 98-115.
Frequently Asked Questions
How big is the allergy point-of-care testing market in 2026?
The global allergy point-of-care testing market is estimated to be valued at USD 1.6 billion in 2026.
What will be the size of the allergy point-of-care testing market in 2036?
The market size for the allergy point-of-care testing market is projected to reach USD 4.2 billion by 2036.
How much will the allergy point-of-care testing market grow between 2026 and 2036?
The allergy point-of-care testing market is expected to grow at a 10.2% CAGR between 2026 and 2036.
What are the key product types in the allergy point-of-care testing market?
The key product types in the allergy point-of-care testing market include rapid specific-IgE kits, skin-prick testing kits, rapid anaphylaxis kits, and supporting analyzers.
Which end user will contribute a significant share in the allergy point-of-care testing market in 2026?
In terms of end user, the specialty clinics segment is set to command a 34.0% share in the allergy point-of-care testing market in 2026.
Table of Content
- Allergy Point-of-Care Testing Market Size and Share Forecast Outlook (2026 to 2036)
- Allergy Point-of-Care Testing Market Forecast and Outlook (2026 to 2036)
- Allergy Point-of-Care Testing Market Key Takeaways
- Key Shifts in the Allergy Point-of-Care Testing Market from 2026 to 2036
- Allergy Point-of-Care Testing Market Segmentation Analysis
- By Product Type
- Rapid Specific-IgE Kits
- Skin-Prick Testing Kits
- Rapid Anaphylaxis Kits
- Sample Collection Devices
- Analyzers
- By Application Focus
- Primary Care Triage
- Pediatric Clinic Workups
- Emergency Care Support
- Occupational Health Screening
- Pharmacy-Led Testing
- By End User
- Specialty Clinics
- Hospitals
- Retail Pharmacies
- Occupational Health Providers
- Direct-to-Consumer
- By Region
- By Product Type
- Market Dynamics and Growth Analysis
- Growth Drivers
- Market Restraints
- Strategic Trends
- Allergy Point-of-Care Testing Market Global Analysis
- East Asia
- South Asia
- North America
- Europe
- Latin America
- Middle East & Africa
- Competitive Landscape Analysis
- Key Players in the Allergy Point-of-Care Testing Market
- Market Definition, Inclusion, and Exclusion Criteria
- Scope of the Allergy Point-of-Care Testing Market Report
- Allergy Point-of-Care Testing Market by Segment
- Bibliography
- Frequently Asked Questions (FAQs)
List of Tables
- Global Allergy Point-of-Care Testing Market Size (USD Billion), 2021-2036
- Allergy Point-of-Care Testing Market Y-o-Y Growth (%), 2021-2036
- Global Market Share by Product Type, 2026
- Global Market Share by Application Focus, 2026
- Global Market Share by End User, 2026
- Allergy Point-of-Care Testing Market Share by Region, 2026
- Market Share by Product Type (USD Billion), 2026-2036
- Market Share by End User (USD Billion), 2026-2036
- Key Player Market Share in the Allergy Point-of-Care Testing Market
- Allergy Point-of-Care Testing Market Absolute $ Opportunity (USD Billion), 2026-2036
- Allergy Point-of-Care Testing Market CAGR (%) by Region, 2026-2036
List of Figures
- Global Allergy Point-of-Care Testing Market Size (USD Billion), 2021-2036
- Allergy Point-of-Care Testing Market Y-o-Y Growth (%) from 2021-2036
- Allergy Point-of-Care Testing Market Share by Product Type (%) in 2026
- Allergy Point-of-Care Testing Market Share by Application Focus (%) in 2026
- Global Allergy Point-of-Care Testing Market by Region (%) 2026-2036
- Global Allergy Point-of-Care Testing Market by End User
- Competitive Landscape of Key Allergy Point-of-Care Testing Market Players
- Market Trends in Allergy Point-of-Care Testing
- Allergy Point-of-Care Testing Adoption by Region
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

