Ascorbic Acid Market
Ascorbic Acid Market Forecast and Outlook 2026 to 2036
Ascorbic Acid Market Size and Share Forecast Outlook 2026 to 2036
The global ascorbic acid sector is on track to achieve a valuation of USD 3.0 billion by 2036, accelerating from USD 1.7 billion in 2026 at a CAGR of 5.7%. As per Future Market Insights, expansion is structurally underpinned by the growing demand for vitamin C across pharmaceutical formulations, functional food fortification, and cosmetic active ingredients, combined with the ongoing restructuring of global production capacity. The World Health Organization (WHO) includes ascorbic acid on its Model List of Essential Medicines, validating its status as a foundational healthcare input across 150+ national formularies. This essential medicine designation compels governments and manufacturers to maintain secure supply chains for pharmaceutical-grade ascorbic acid. Simultaneously the regulatory landscape is evolving as the European Food Safety Authority (EFSA) tightens health claim substantiation requirements and China's National Medical Products Administration (NMPA) enforces stricter GMP standards for vitamin API exports.
In March 2024, dsm-firmenich completed the sale and transfer of its 100% equity interest in DSM Jiangshan Pharmaceutical (Jiangsu) Co., Ltd. to Jingjiang Cosfocus Health Technology Co., Ltd. This divestiture was part of dsm-firmenich's broader strategy to restructure its vitamin portfolio, shifting away from standard commodity-grade production in China to focus on its Quali-C high-quality, low-carbon production site in Dalry, Scotland. FMI opines that this portfolio bifurcation will accelerate the premium-certification trend in Western markets, as pharmaceutical and cosmetic formulators increasingly specify Quali-C or equivalent certified-origin ascorbic acid to meet ESG procurement mandates and EU health claim substantiation requirements.
The competitive landscape in 2024, 2025, and 2026 is defined by capacity restructuring and downstream integration. Northeast Pharmaceutical Group signed a framework agreement in November 2024 to acquire a 70% equity stake in Beijing Dingcheng Peptide Source Biotechnology Co., Ltd., integrating peptide biotechnology with its existing vitamin and API production lines to enhance pharmaceutical-grade ascorbic acid capabilities. Shandong Luwei Pharmaceutical Group was awarded the High-quality Development Benchmark Enterprise Award in February 2025 for its pilot project on comprehensive utilisation of waste from Vitamin C fermentation, establishing a scientific standard for recycling production waste. SEIWA KASEI launched iVC 3GA-green in November 2025, a plant-derived sustainable anti-aging vitamin C ingredient for the cosmetics market. OnScent introduced Acerola Cherry Ferment in January 2025, a natural vitamin C-rich ingredient targeting botanical and organic skincare demand. dsm-firmenich reported in January 2026 that Chinese producers are shifting focus to value over volume, with pricing stabilisation and gradual upward movement after 18 months of volatility. As per FMI, this convergence of Western premium-certification, Chinese value-over-volume pricing, and downstream peptide integration confirms that the ascorbic acid market is transitioning from a commodity-driven price war toward a structurally bifurcated supply architecture.
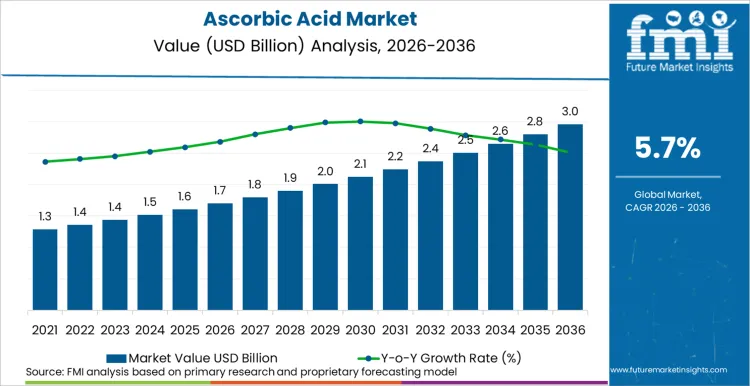
Summary of Ascorbic Acid Market
What Is Growth Outlook for Ascorbic Acid Market as per Future Market Insights Projection?
Future Market Insights projects the ascorbic acid industry to expand at a CAGR of 5.7% from 2026 to 2036, increasing from USD 1.7 Billion in 2026 to USD 3.0 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on WHO Essential Medicines List inclusion, EFSA health claim regulation tracking, and Chinese production capacity monitoring.
How Do FMI Analysts Perceive Ascorbic Acid Market to Evolve?
FMI analysts perceive the market evolving toward a bifurcated supply architecture where premium-certified Western production (dsm-firmenich Quali-C Dalry) serves pharma and cosmetics, while restructured Chinese producers pursue value-over-volume strategies in food-grade and commodity segments.
FMI Research Approach: dsm-firmenich Jiangshan divestiture and Quali-C retention strategy analysis.
Which Country Holds Largest Share in Global Ascorbic Acid Market?
China holds the largest share of the global ascorbic acid market by volume which is supported by the concentration of Northeast Pharmaceutical, Shandong Luwei, CSPC Pharmaceutical, and multiple fermentation-based producers that collectively supply over 80% of global production capacity.
FMI Research Approach: FMI country-level revenue modeling by Chinese API export data and NMPA production license tracking.
How Large Will Ascorbic Acid Market Be by 2036?
The global ascorbic acid market is projected to reach USD 3.0 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from WHO essential medicine consumption projections and premium-grade certification adoption curves.
What Is Definition of Ascorbic Acid Market?
The ascorbic acid market includes pharmaceutical-grade ascorbic acid (vitamin C API), food-grade ascorbic acid for fortification and preservation, cosmetic-grade ascorbic acid derivatives, and feed-grade ascorbic acid used across pharmaceutical, food and beverage, personal care, and animal nutrition applications.
FMI Research Approach: FMI market taxonomy aligned with WHO Essential Medicines classification, EFSA nutrition guidelines, and pharmacopoeial API standards (USP, EP, JP).
What Are Globally Unique Trends Shaping Ascorbic Acid Market?
Globally unique trends include Western premium-portfolio restructuring (dsm-firmenich Jiangshan divestiture/Quali-C retention), Chinese value-over-volume pricing pivot (Q1 2026), and peptide-vitamin integration for pharma-grade applications (Northeast Pharmaceutical/Dingcheng acquisition).
FMI Research Approach: dsm-firmenich Q1 2026 Vitamin Market Update and Northeast Pharmaceutical acquisition tracking.
Ascorbic Acid Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.7 Billion |
| Industry Value (2036) | USD 3.0 Billion |
| CAGR (2026 to 2036) | 5.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Top Highlights Driving Growth in the Ascorbic Acid Industry
Strict regulatory requirements combined with high profit margins make Pharmaceutical Grade Ascorbic Acid prominent
Standards of quality and regulatory compliance in the pharmaceuticals industry are non-negotiable and this makes Pharmaceutical Grade Ascorbic Acid a big segment. Certain categories relevant to pharmaceutical manufacturing, such as ethical manufacturing practices, extensive concomitant documentation, additional tests and compliance with various standards, set a high threshold for market entry.
However, after entry, such conditions are compensated by profits, which exceed the food-grade ones, hence making pharmaceutical-grade production more appealing for manufacturers. Measures for quality control such as testing for impurities, metal content and presence of microbiological contaminants, along with validation requirements for the production process, lead to high expenses, however, ensure a high-quality product.
Rising applications of Ascorbic Acid in Food & Beverages due to growing functional food & natural preservatives demand
The food and beverage sector has seen remarkable growth in ascorbic acid use in recent years, due to rising consumer demand for functional foods and natural preservatives. This development is in line with the growing demand for more nutritious foods among buyers. Manufacturers also use ascorbic acid in their products as a vitamin C source and regard it as a naturally derived substitute instead of chemical preservatives.
The wide functionalities of ascorbic acid from beverages and baked goods to meat products have made it an important component of modern food and beverage formulations. Furthermore, as Ascorbic acid helps in shelf life extension, color preservation and product quality enhancement, it has become a widely used ingredient in processed foods, dairy goods and functional beverages.
Granular Ascorbic Acid is preferred in F&B use due to its reduced caking & better dissolution properties
Granular Ascorbic Acid has become the preferred form in food and beverages due to its better handling characteristics. This addresses an important processing issue that manufacturers face with the traditional crystalline form. The granular form reduces caking issues which are involved in both storage and manufacturing processes, thus enabling the smooth production of goods.
Furthermore, its better dissolution properties improve the dispersion of ascorbic acid in different food formulations where evenness and quickness are needed. Granular ascorbic acid also improves dosing accuracy and mixing efficiency, which leads to more consistent final products. All of these benefits lead to reduced cycle times, lower manufacturing costs and better quality of the product. In addition to this, the granular shape form is also less likely to be affected by moisture, which improves storage stability.
Comparison of 2021 to 2025 Sales vs 2026 to 2036 Demand Forecast for Ascorbic Acid
The ascorbic acid industry went through many changes in the past few years as applications and consumer preferences evolved. In the early 2000s, Chinese manufacturers disrupted the industry using fermentation technology and building cost-competitive manufacturing plants. This, in turn, led to excess supply and rapid expansion thus leading to price wars. With consistent growth in demand across the pharmaceutical, food and personal care industries, the fluctuations in price have become lesser and stability has come back to the industry.
Moving forward, multiple trends are expected to increase growth. The pandemic has created a shift in demand with consumers paying more attention to preventive healthcare and consuming immune-boosting products. Furthermore, the industry also has the potential to grow with the food and beverage industry as an antioxidant and as a preservative. There is a focus on environmental sustainability, which is steadily becoming an area of concern, thus leading to manufacturers adopting cleaner production technologies, including bio-based processes.
Key Players and Market Concentration in the Ascorbic Acid Industry
With many manufacturers across the globe, especially in China, the Ascorbic acid market has a fragmented industry structure. The industry has been largely dominated by local manufacturers in China, with some international companies that target specific premium segments.
The presence of numerous manufacturers such as CSPC Pharmaceutical Group, Northeast Pharmaceutical Group, Shandong Luwei and Henan Huaxing indicates the fragmented nature of the industry. In the industrial and feed-grade sectors, these regional companies compete on low cost and high volumes. Due to globalization and the lack of any other major players, China remains the key player in the industry with significant market share.
There are some MNCs operating in the industry, which include DSM-Firmenich and BASF among others. While having a much smaller market share in comparison, they cater to an important segment of pharmaceutical-grade products. These MNCs are not price-driven in competition as they rely more on quality certifications and advanced technology. Food and beverage, pharmaceuticals, cosmetic as well as animal feed industries are fragmented, which add to the lack of concentration in the Ascorbic acid industry. Each of these has its evaluations of quality as well as competition which makes some producers of certain qualities thrive.
Overview of Prominent Companies in the Ascorbic Acid Sector
With CSPC being among the top ascorbic acid suppliers in China, they are working towards increasing their export market with more competitive pricing as their Hebei Province's vertically integrated production facilities have been successful in cost management. They have also installed fermentation devices to improve productivity and reduce pollution.
NEPG modernized its ascorbic acid manufacturing plant in order to retain its standing as one of the biggest suppliers in China. Their focus is on high-quality ascorbic acid for use in pharmaceuticals. They have also worked towards increasing their export operations and have successfully established their visibility outside China through mobile distribution resources in Southeast Asia and Europe market.
Analysis of Top Countries Producing and Consuming Ascorbic Acid
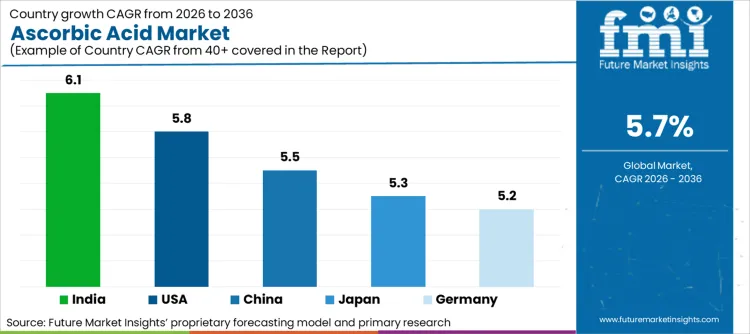
The following table shows the estimated growth rates of the top five markets. These countries are set to exhibit high consumption, recording high CAGRs through 2036.
| Countries | CAGR, 2026 to 2036 |
|---|---|
| China | 5.5% |
| USA | 5.8% |
| Germany | 5.2% |
| India | 6.1% |
| Japan | 5.3% |
China maintains its position of largest producer of Ascorbic acid along with its high domestic demand
The shift of dominance from other nations to China in the industry has been due to China's prominence in both the production and distribution of ascorbic acid. This has been achieved by government support in technology advancements along with low-cost provisioning capabilities. The broad consumer base within China, along with rising disposable income is also allowing for better export capability. With the government's support for the various chemical manufacturing industries, especially in Research & Development, China has been able to maintain this advantage at the international level.
Recently, however, Chinese manufacturers have strongly invested in quality control measures in order to shift the global standards towards the Chinese system. The fragmented nature of the industry which consists of multiple small players ensures healthy competition to increase efficiency and quality. The industry for Ascorbic Acid in the country is projected to exhibit a CAGR of 5.5% during the assessment period and revenue from the sales of the product in the country is expected to reach USD 866.8 million with a market share of 31% by 2035.
Advanced Pharmaceutical industry & manufacturing infrastructure bolstering growth in USA
The USA has advanced infrastructure that cater to the requirements of ascorbic acid manufacturers. The combination of high-quality standards, along with research and a high level of investment provides the environment for high-quality products. Leading firms within the region have continued to develop an edge within the ascorbic acid supply chain making them competitive.
Nutraceuticals and pharmaceuticals are in high demand, which has, in turn, led to the demand for ascorbic acid. Much of the demand is too high for western companies and so, a significant portion of the overall demand is exported from the various Chinese manufacturers who currently dominate the industry. Due to the attention being paid to drug improvement initiatives, the industry has been growing relatively quickly as well. These trends are leading to a projected value of USD 559.2 million by 2035 with Ascorbic Acid demand calculated to rise at a value CAGR of 5.8% during the forecast period (2025 to 2035) with a value share of 20%.
Japan’s strong personal care & nutraceuticals industries driving growth of Ascorbic Acid
The Japan Ascorbic acid market has a unique identity owing to its mature personal care and nutraceuticals sectors. With a rapidly aging population and the shift towards preventive medicine, the demand for vitamin C supplements and medicines has been rising constantly in Japan. Beauty and skincare products that have scientific backing are in high demand, which in turn is driving demand for Ascorbic Acid. Recent groundbreaking research regarding the usage of ascorbic acid in cosmetics alongside more complex drug delivery systems is also expected to bolster growth of market.
Given that the Japanese consumers are highly sensitive to quality, manufacturers are now focusing on producing high quality customized ascorbic acid grades and forms. These factors are responsible for the robust projected forecast CAGR of 5.3% from 2025 to 2035, with the country achieving a global value share of 7% in 2035 with a value of USD 195.7 million.
Ascorbic Acid Industry Analysis by Top Investment Segments
Ease of transportation, superior stability & longer shelf life are reasons for prominence of Powder / Crystal form
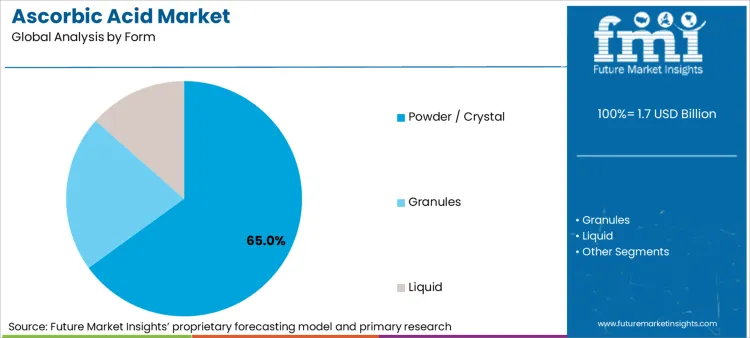
| Segment | Powder / Crystal |
|---|---|
| Main Segment | Form Type |
| Value Share (2036) | 65% |
The powder or crystal form of ascorbic acid has maintained its leadership in the segment due to practical advantages in the supply chain. This form's stability under various environmental conditions decreases degradation during transportation and storage, thus improving the quality of the end-product. Its extended shelf life reduces waste and logistical costs, while the ease of transportation makes it a very economical choice for both manufacturers and distributors.
The powder form's usage in various applications, from direct compression in tablets to easy dissolution in liquid formulations, makes it the preferred form of ascorbic acid. Its ability to be precisely measured and mixed with other ingredients are also reasons for the segment’s continued growth.
Reichstein Process is widely used for production due to its high yield & consistent quality
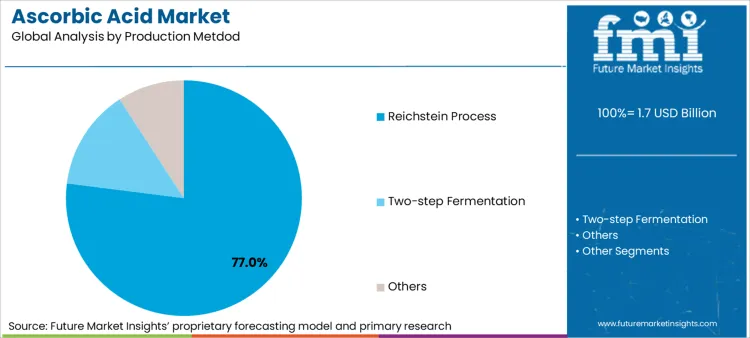
| Segment | Reichstein Process |
|---|---|
| Main Segment | Production Metdod |
| Value Share (2036) | 77% |
The Reichstein Process continues to maintain its position as the industry standard for ascorbic acid production, despite being developed decades ago. This process has been continuously refined and optimized, offering manufacturers the perfect balance of efficiency and quality. Its ability to produce consistent, high-quality ascorbic acid at a commercial scale, combined with well-understood process parameters and established quality control measures, makes it a very reliable production method.
The process's predictable outcomes have led to many investments in optimizing and automating the various stages, which has resulted in improved results and reduced production costs. The extensive industry experience with this process has also created a deep understanding of the possible errors and their troubleshooting procedures, making it a dependable choice for large-scale production.
Leading Suppliers in the Ascorbic Acid Industry
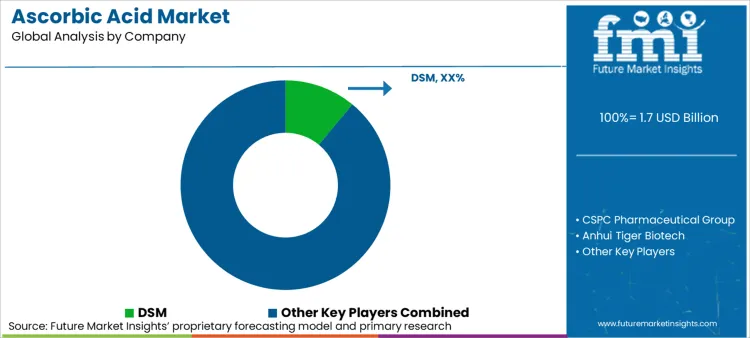
The industry of ascorbic acid has tough competition from both international and local players, with the latter dominating currently. Companies are investing in up-to-date and cutting-edge technologies in order to get an edge over their competitors. Research & Development of new product lines is being prioritized in order to deal with the changing consumer needs as well as changing manufacturing technologies.
The global shift towards sustainable production methods has also become a USP for companies wishing to separate themselves from the fragmented mass of companies fighting for market share.
Strategic and long-lasting partnerships with end-user B2B industries such as pharmaceuticals and food industries are proving to be highly useful in securing and improving the current competitive position of the companies.
Ensuring that the products are certified for quality and are compliant with the relevant regulations offers companies a chance to market their products as premium offerings, which has turned out to be one of the main competitive approaches, especially in developed regions.
Recent Developments
- In March 2024, dsm-firmenich completed the sale of DSM Jiangshan Pharmaceutical (Jiangsu) to Jingjiang Cosfocus, restructuring its portfolio toward premium Quali-C production in Dalry, Scotland.
- In November 2024, Northeast Pharmaceutical Group signed a framework agreement to acquire 70% of Beijing Dingcheng Peptide Source Biotechnology, integrating peptide technology with vitamin API production.
- In February 2025, Shandong Luwei Pharmaceutical was awarded the High-quality Development Benchmark Enterprise Award for its Vitamin C production waste recycling pilot.
Top Companies Shaping the Ascorbic Acid Competitive Landscape
- DSM
- CSPC Pharmaceutical Group
- Northeast Pharmaceutical Group (NEPG)
- North China Pharmaceutical Group
- Shandong Luwei Pharmaceutical
- Aland Nutraceuticals Group
- Anhui Tiger Biotech
- Bactolac Pharmaceuticals
- Hebei Welcome Pharmaceutical
- Shandong Tianli Pharmaceutical
- Zhengzhou Tuoyang
- Ningxia Qiyuan
- Henan Huaxing Pharmaceutical
- Foodchem International Corporation
- Zhejiang Hangzhou Xinfu Pharmaceutical
- Others
Market Definition
The ascorbic acid market represents revenue generated from the manufacture and sale of vitamin C (L-ascorbic acid) across pharmaceutical, food-grade, cosmetic, and feed-grade applications. The market measures the value of bulk ascorbic acid API, ascorbic acid derivatives (sodium ascorbate, calcium ascorbate, ascorbyl palmitate), and stabilised vitamin C actives sold to pharmaceutical manufacturers, food processors, cosmetic formulators, and animal feed producers.
Inclusions cover pharmaceutical-grade ascorbic acid API (USP, EP, JP grade), food-grade ascorbic acid for fortification and antioxidant preservation, cosmetic-grade stabilised vitamin C derivatives (ascorbyl glucoside, 3-O-ethyl ascorbic acid), feed-grade ascorbic acid for aquaculture and poultry, and certified-origin premium ascorbic acid (Quali-C and equivalent).
Exclusions include finished-dosage vitamin C supplements sold to consumers, multivitamin products where ascorbic acid is a minor component, synthetic intermediate chemicals used in ascorbic acid manufacturing, and natural fruit-derived vitamin C extracts sold as whole-food ingredients.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.7 Billion |
| Product Type | Pharmaceutical-Grade API, Food-Grade, Cosmetic-Grade Derivatives, Feed-Grade |
| End User | Pharmaceutical, Food and Beverage, Personal Care, Animal Nutrition |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | China, USA, India, Germany, Japan, Scotland/UK, and 30+ countries |
| Key Companies Profiled | dsm-firmenich, Northeast Pharmaceutical, Shandong Luwei, CSPC Pharmaceutical, SEIWA KASEI |
Top Segments Studied in the Ascorbic Acid Product Report
By Form Type:
As per Form Type, the industry has been categorized into Powder / Crystal, Granules and Liquid.
By Production Method:
As per Production Method, the industry has been categorized into Reichstein Process, Two-step Fermentation and Others.
By Grade Level:
As per Grade Level, the industry has been categorized into Food Grade, Feed Grade, Pharmaceutical Grade and Industry Grade.
By Application:
As per Application, the industry has been categorized into Food & Beverages, Animal Feed, Cosmetics & Personal Care, Pharmaceuticals and Industrial Applications.
By Region:
Industry analysis has been carried out in key countries of North America, Latin America, Western Europe Eastern Europe, Balkans & Baltic, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific and Middle East & Africa.
Bibliography
- World Health Organization. (2024). Model List of Essential Medicines: Ascorbic Acid Inclusion. WHO.
- dsm-firmenich. (2024, March). Sale of DSM Jiangshan Pharmaceutical to Jingjiang Cosfocus. dsm-firmenich.
- dsm-firmenich. (2026, January). Vitamin Market Update Q1 2026: Chinese value-over-volume pricing shift. dsm-firmenich.
- Northeast Pharmaceutical Group. (2024, November). Framework agreement to acquire Beijing Dingcheng Peptide Source Biotechnology. Northeast Pharmaceutical.
Frequently Asked Questions
What is the current global market size for Ascorbic Acid?
The global market is valued at USD 1.7 Billion in 2026, driven by pharmaceutical and cosmetic-grade demand growth and Chinese pricing stabilisation.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a CAGR of 5.7% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
Asia Pacific leads by production volume through Chinese manufacturing dominance, while Europe leads premium-grade growth through dsm-firmenich Quali-C certification and EFSA health claim compliance.
What are the primary market drivers?
WHO essential medicine designation, pharmaceutical and cosmetic-grade certification demand, Chinese value-over-volume pricing restructuring, and EFSA health claim substantiation requirements are the primary growth catalysts.
Who are the leading suppliers in the industry?
dsm-firmenich, Northeast Pharmaceutical, Shandong Luwei, and CSPC Pharmaceutical are key players, differentiating through premium certification (Quali-C), peptide-vitamin integration, and sustainable production processes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form , 2026 to 2036
- Powder / Crystal
- Granules
- Liquid
- Powder / Crystal
- Y to o to Y Growth Trend Analysis By Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Production Metdod
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Production Metdod, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Production Metdod, 2026 to 2036
- Reichstein Process
- Two-step Fermentation
- Others
- Reichstein Process
- Y to o to Y Growth Trend Analysis By Production Metdod, 2021 to 2025
- Absolute $ Opportunity Analysis By Production Metdod, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Form
- By Production Metdod
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By Production Metdod
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By Production Metdod
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Form
- By Production Metdod
- Competition Analysis
- Competition Deep Dive
- DSM
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CSPC Pharmaceutical Group
- Anhui Tiger Biotech
- Zhengzhou Tuoyang
- Henan Huaxing Pharmaceutical
- Ningxia Qiyuan
- DSM
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Production Metdod, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Form
- Figure 6: Global Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Production Metdod
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Form
- Figure 23: North America Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Production Metdod
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Form
- Figure 30: Latin America Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Production Metdod
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Form
- Figure 37: Western Europe Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Production Metdod
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Form
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Production Metdod
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Form
- Figure 51: East Asia Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Production Metdod
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Production Metdod
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Production Metdod, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Production Metdod, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Production Metdod
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

