The global asthma-COPD overlap treatment market is forecasted to reach USD 13.4 billion in 2026 and expand to USD 23.0 billion by 2036, advancing at a CAGR of 5.5%. As per FMI, this outlook shows a transformation in respiratory therapeutics toward precision medicine approaches, where traditional single-disease treatments are transitioning toward ACO-specific therapeutic platforms. Expansion is driven by the rising complexity of overlap syndrome management, with pharmaceutical companies and specialized respiratory centers embracing targeted interventions across healthcare institutions globally.
By leveraging biomarker-driven phenotyping and advanced diagnostics, clinicians can now perform comprehensive patient stratification that evaluates eosinophil counts, fractional exhaled nitric oxide levels, and inflammatory profiles before any treatment initiation occurs. This diagnostic precision allows for the rapid identification of ACO phenotypes and the design of complex, multi-mechanistic therapies that were previously considered challenging to implement, effectively improving patient outcomes and reducing healthcare burden. Companies are also investing significantly in digital health solutions to enhance treatment monitoring. In May 2024, Blackstone Life Sciences unveiled Uniquity Bio, a clinical-stage drug development company focused on immunology and inflammation.
“As a nimble startup with a proven leadership team, we believe Uniquity Bio is well-positioned to develop promising therapeutics in this field and partner with leading global pharmaceutical companies,” said Stephen Tullman, Executive Chairman of Uniquity Bio. “I’m confident that our work will deliver new and highly valuable medicines to people living with immune-mediated diseases.”
The industry's value proposition is shifting from volume-based treatment delivery toward precision-focused therapeutic positioning. Advanced treatment providers are integrating comprehensive biomarker analysis, streamlined diagnostic workflows, and flexible platform designs to reduce treatment standardization challenges while improving symptom control. Specialty-positioned brands and dedicated respiratory organizations are gaining preference, enabling direct clinical partnerships and customized treatment services across hospital and outpatient care channels.
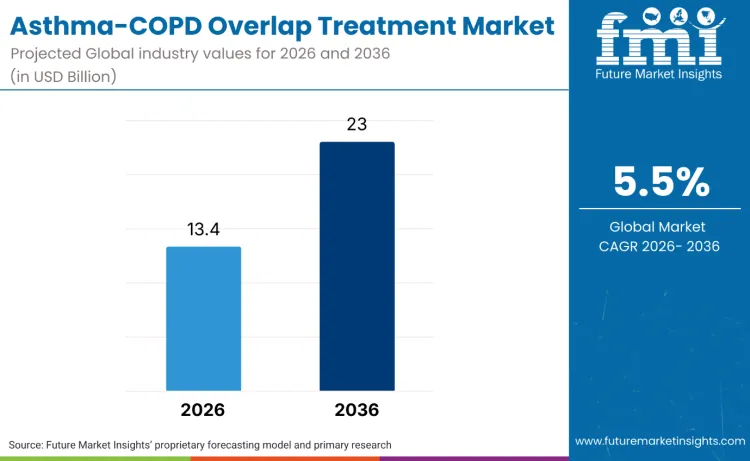
Future Market Insights projects the market to grow at a 5.5% CAGR from 2026 to 2036, expanding from USD 13.4 billion to USD 23.0 billion.
FMI research approach: Bottom-up therapeutic revenue modeling, phenotype-weighted treatment analysis, prescription intensity assessment, and biomarker-led diagnostic scenario modeling.
The market is expected to move from standardized treatment protocols toward AI-enabled precision platforms integrating phenotyping, biomarker assessment, and autonomous therapy optimization.
FMI research approach: Precision medicine transition analysis, AI workflow integration mapping, biologics and combination therapy demand assessment, and predictive phenotyping benchmarking.
The United States accounts for the largest global value share.
FMI research approach: Country-level pharmaceutical revenue modeling, respiratory care concentration analysis, adoption scoring for precision medicine methods, and healthcare infrastructure evaluation.
The global market is forecast to reach USD 23.0 billion by 2036.
FMI research approach: Long-term forecasting by therapeutic class and phenotype focus, precision medicine demand scaling, clinical revenue inclusion, and historical prescription validation.
It covers therapeutic interventions supporting ACO patient management, phenotyping, treatment optimization, monitoring, and clinical validation for respiratory overlap syndromes.
FMI research approach: Market taxonomy validation, inclusion-exclusion structuring, application mapping by treatment indication, and therapeutic scope filtering.
Key trends include precision phenotyping adoption, decentralized respiratory monitoring, and regulatory momentum toward personalized treatment protocols.
FMI research approach: Biomarker platform uptake tracking, remote monitoring expansion analysis, regulatory pathway monitoring, and algorithm-focused competitive assessment.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 13.4 billion |
| Market Forecast Value (2036) | USD 23.0 billion |
| Forecast CAGR (2026-2036) | 5.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Demand for asthma-COPD overlap treatment is being propelled by the broad therapeutic shift toward precision medicine paradigms and the escalating complexity of overlap syndrome management such as phenotype-specific biologics and combination therapies, which require advanced diagnostic, optimization, and monitoring platforms. The rapid integration of artificial intelligence and machine learning into patient stratification workflows has amplified demand for biomarker-driven screening and predictive treatment assessment, enabling clinicians to reduce therapeutic failures and optimize treatment timelines.
The regulatory momentum toward personalized medicine approaches, exemplified by revised FDA guidance on precision respiratory care, is driving widespread uptake of biomarker-guided treatment protocols and high-fidelity phenotyping systems in clinical workflows, increasing procurement of specialized therapeutic services. At the same time, pharmaceutical investment patterns, including expanded research infrastructure in Asia-Pacific respiratory hubs, are shifting innovation toward decentralized, precision-focused facilities, underscoring a sustained demand trajectory rooted in phenotype engineering and predictive medicine.
The asthma-COPD overlap treatment market has been segmented based on drug class, treatment use cases, end user, and region. In terms of drug class, the market is divided into triple therapy (ICS/LABA/LAMA), dual therapy (ICS/LABA or LABA/LAMA), biologics, and systemic therapies. By treatment use cases, the market is divided into exacerbation prevention in ACO phenotypes, severe asthma with fixed airflow limitation, COPD with eosinophilic inflammation, post-hospital discharge optimization, and comorbidity-driven management. By end user, the market is segmented into retail pharmacy channel, hospitals, pulmonology clinics, primary care centers, and others. Regionally, the market is divided into North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa.
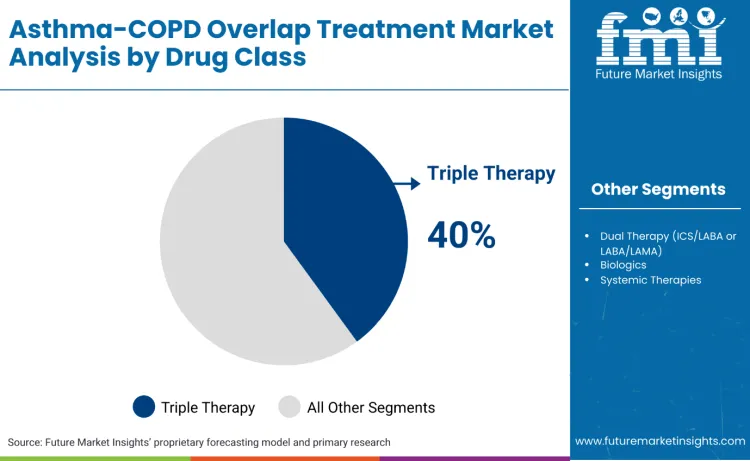
Triple therapy treatments continue to lead the market, accounting for 40.0% of the share, driven by their widespread use in complex ACO management, exacerbation prevention, and comprehensive symptom control. This therapeutic approach offers the foundation needed for managing both asthma treatment and COPD components simultaneously, making it essential for respiratory specialists, hospital systems, and primary care organizations. Dual therapy treatments capture 24.0% of the market, supporting patients with less severe overlap presentations while maintaining efficacy for specific phenotypes. Their precision is critical for reducing side effects while maintaining therapeutic benefits across different ACO presentations. Biologics, holding 20.5% of the market, remain a popular choice for treating Type-2 inflammation patterns, offering targeted solutions for precision medicine approaches where biomarker-guided therapy is required.
These treatments are popular because they serve as a critical therapeutic strategy in an increasingly complex and heterogeneous patient population. Companies are also significantly investing in combination therapy approaches with tailored formulations. Specialized pharmaceutical providers integrate advanced delivery mechanisms, biomarker-guided selection, and AI-driven monitoring to identify optimal therapeutic combinations while filtering out ineffective or harmful protocols early. This precision-first approach reduces costly treatment failures and improves patient outcomes, allowing healthcare systems to remain cost-efficient and focus resources on clinical optimization rather than trial-and-error approaches.
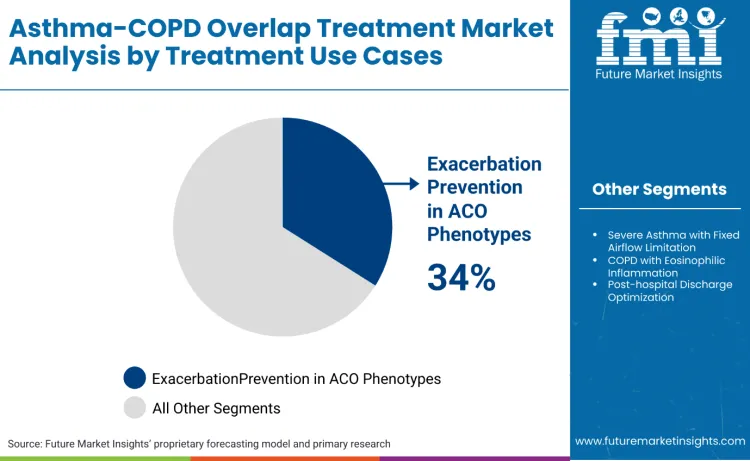
Exacerbation prevention in ACO phenotypes stands as the dominant segment, commanding 34.0% of the market share. Asthma-COPD overlap treatment remains centered on exacerbation prevention due to the exceptional clinical impact these interventions offer in reducing hospitalizations and emergency department visits. ACO patients often display complex inflammatory patterns that serve as specific targets for engineered combination therapies, allowing for preventive treatment that avoids the systemic complications caused by acute exacerbations. Clinical studies are essential in this field to validate how these treatments interact within the complex respiratory environment, ensuring they can address both asthmatic and COPD therapeutics effectively while recruiting appropriate anti-inflammatory responses.
The market is also heavily shaped by the development of next-generation modalities like targeted biologics and precision-guided combination therapies which require rigorous validation to establish optimal therapeutic windows. Clinical validation helps researchers optimize the stability of drug combinations and the efficacy of delivery mechanisms to prevent off-target effects that could lead to therapeutic failure. By using advanced monitoring systems such as remote spirometry and biomarker tracking, clinicians can predict patient responses and identify potential resistance patterns long before treatment optimization becomes necessary, making these services a critical investment for modern respiratory care.
Greater regulatory clarity around precision medicine approaches is stabilizing investment conditions for respiratory therapeutic developers and enabling more confident platform expansion. In May 2025, Sanofi secured authorization from the FDA to commence the phase 3 clinical trial of dupilumab combination therapy, an investigational treatment targeting IL-4/IL-13 pathways for severe ACO phenotypes. Verona Pharma is actively enrolling Phase 2 trials for Ohtuvayre in asthma, pursuing a strategy to cover both COPD (approved) and asthma indications.
Clearer regulatory expectations are lowering entry barriers for specialized pharmaceutical companies to deploy advanced respiratory technologies such as biomarker-guided selection, combination therapy optimization, and next-generation monitoring for phenotype analysis. Companies are increasingly adopting platform-based treatment approaches that can be validated once and reused across ACO populations, supporting faster clinical pathways and sustained innovation in complex respiratory therapeutic formats.
Global demand for asthma-COPD overlap treatment is increasing as clinicians expand therapeutic capabilities while addressing phenotype identification, treatment optimization, and monitoring application requirements. Growth demonstrates rising use of automated diagnostic platforms, phenotype-specific treatment positioning, and efficiency-optimized monitoring specifications across healthcare institutions, specialized respiratory facilities, and pharmaceutical companies. Treatment selection focuses on therapeutic efficacy, biomarker comprehensiveness, and platform performance under various clinical scenarios. China records 13.0% CAGR, India records 11.5% CAGR, Germany records 8.5% CAGR, USA records 6.7% CAGR, and France records 5.5% CAGR. Adoption remains driven by precision treatment requirements and monitoring capabilities rather than volume expansion alone.
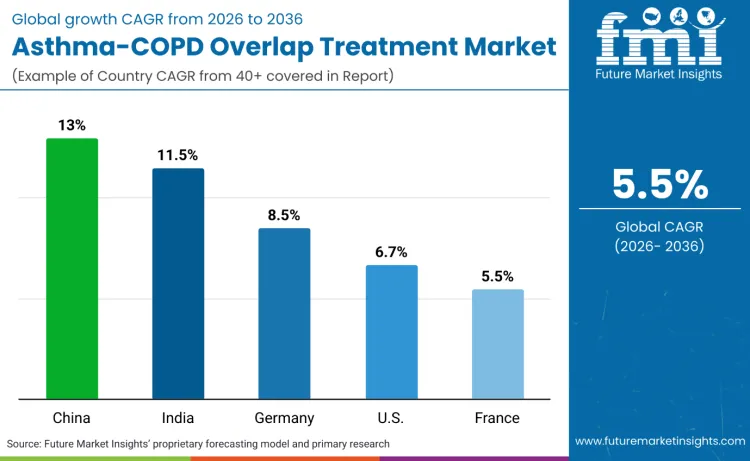
| Country | CAGR (2026-2036) |
|---|---|
| China | 13.0% |
| India | 11.5% |
| Germany | 8.5% |
| USA | 6.7% |
| France | 5.5% |
Source: FMI historical analysis and forecast data
The primary driver in China is the significant growth of domestic respiratory care capabilities and precision medicine research institutions. With the expansion of specialized respiratory facilities, Chinese metropolitan areas are now experiencing millions of new diagnostic platform implementations, making traditional single-drug treatment methods insufficient. This has promoted advanced therapeutic platform adoption for domestic healthcare development to ensure treatment capabilities and clinical innovation. Driven by this national scale of healthcare capability expansion, the Chinese asthma-COPD overlap treatment market is set to grow at a 13.0% CAGR during the study period, led by massive demand in hospital and outpatient sectors.
Over the next five years, the most significant opportunity lies in the integration of artificial intelligence-powered diagnostic platforms with high-throughput biomarker analysis hardware. As China targets leadership in precision respiratory medicine, there is a burgeoning demand for specialized treatments that support complex phenotypes like severe ACO and treatment-resistant overlap syndromes. Companies are expanding their clinical presence in the country to boost their treatment capabilities.
In India, the market is dominated by the evolution toward healthcare excellence and precision-positioned treatment categories. This precision-first approach is essential for Indian clinicians currently competing with international treatment standards in domestic respiratory applications. Reflecting this maturity, the Indian asthma-COPD overlap treatment market is projected to grow at a 11.5% CAGR during the study period.
Over the next five years, the rise of precision medicine and biomarker-guided treatments, supported by expanded healthcare infrastructure, will necessitate high-precision therapeutic monitoring services for real-time clinical assessment and quality control. Second, India's growing role as a global hub for pharmaceutical outsourcing creates a massive opening for high-throughput diagnostic services tailored to treatment optimization. There is a burgeoning demand for cost-effective therapeutic platforms that use standardized protocols, addressing the healthcare needs of tier-2 and tier-3 respiratory centers while maintaining international quality standards.
In Germany, the market is being revitalized by the deployment of advanced biomarker methods and automated workflow integration into therapeutic platform positioning. A major development is healthcare institutions and pharmaceutical companies upgrading to precision-positioned treatment platforms that feature innovation-heritage excellence to compete against standard therapeutic methods in domestic healthcare applications. As German institutions prioritize treatment innovation and healthcare excellence, the German asthma-COPD overlap treatment market is set to grow at a 8.5% CAGR during the study period, focusing heavily on precision and therapeutic capability enhancement.
Over the next five years, the most lucrative opportunities reside in the convergence of digital health and companion diagnostics. As the German government streamlines regulatory barriers for precision medicine development, there is a significant opening for firms to develop artificial intelligence-integrated services that support personalized respiratory care and complex phenotype management. The mandate for efficient healthcare delivery is creating a surge in demand for automated, resource-optimized treatment processes, offering a high-margin niche for providers of streamlined, eco-friendly therapeutic platforms.
The USA market is increasingly focused on the innovation and diversity of precision treatment applications. This transition aims at reducing dependence on standard therapeutic methods while improving domestic healthcare platform competitiveness. Supported by these healthcare development mandates, the USA asthma-COPD overlap treatment market is set to grow at a 6.7% CAGR during the study period.
Over the next five years, the most significant opportunity lies in the mass adoption of new biomarker methodologies and artificial intelligence-driven predictive treatment selection. As the FDA increasingly validates precision medicine models, there is a burgeoning market for high-fidelity, phenotype-based therapeutic services that can simulate optimal treatment responses. The integration of federated machine learning into respiratory care workflows will allow USA firms to leverage vast, secure healthcare networks to design optimized treatment protocols, effectively reducing traditional therapeutic timelines and establishing a new global benchmark for respiratory care and treatment productivity.
As respiratory pipelines become dominated by complex combination therapies and targeted biologics, major players like GSK, AstraZeneca, and Boehringer Ingelberg are competing on their ability to offer "comprehensive care" models. These giants are aggressively integrating artificial intelligence and precision diagnostics to offer respiratory therapies, allowing healthcare systems to operate with enhanced efficiency without extensive infrastructure investments. Success in this landscape is no longer measured just by a provider's drug portfolio size, but by their ability to "code" optimal treatment protocols using predictive analytics, thereby bypassing traditional, time-consuming trial-and-error therapeutic cycles.
The market is also witnessing a distinct regional divergence in competitive strategy. While North American firms like Regeneron and Pfizer maintain dominance through high-value intellectual property and proximity to major healthcare hubs, European players, led by Novartis and Roche, are rapidly scaling their biomarker-guided production and precision medicine capabilities. This has created an "innovation arms race" where therapeutic providers are diversifying into niche areas such as phenotype-specific biologics and advanced monitoring systems to mitigate the high failure rates of complex respiratory treatments.
The asthma-COPD overlap treatment market refers to the global healthcare applications of specialized therapeutic platforms designed to support ACO patient management, phenotyping, treatment optimization, monitoring, and clinical validation activities. It includes both precision and standard treatment offerings, with segments that cater to various clinical applications such as exacerbation prevention, severe asthma management, and eosinophilic COPD. The market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The asthma-COPD overlap treatment market includes therapeutic categories such as triple therapy combinations, dual therapy approaches, biologics, and systemic treatments, utilized through both hospital systems and outpatient healthcare applications. The market is expanding due to growing interest in precision medicine-based therapies, diagnostic platform innovations, and evolving clinical requirements toward higher-efficiency and more specialized phenotyping capabilities.
Included in the asthma-COPD overlap treatment market scope are therapeutic interventions categorized by class (triple therapy, dual therapy, biologics, systemic treatments), with segmentation based on treatment applications (exacerbation prevention, severe asthma management, eosinophilic inflammation), clinical focus (hospital, outpatient, specialty care), and treatment requirements (efficacy, monitoring, precision compatibility). The scope includes ACO treatment platforms with a focus on therapeutic precision, clinical capabilities, and treatment innovation, particularly those marketed through specialized healthcare providers, respiratory institutions, and pharmaceutical development facilities.
Geographically, the market encompasses key regions such as North America, Europe, East Asia, South Asia, Latin America, and the Middle East and Africa, with country-level analysis for significant markets like China, India, Germany, USA, and France.
Excluded from the asthma-COPD overlap treatment market scope are non-respiratory therapeutic platforms and treatment methods not designed for overlap syndrome management, such as cardiovascular treatments, diabetes care systems, and oncology-based therapeutic platforms. Additionally, treatments not intended for ACO or respiratory applications are not included.
The market excludes non-prescription respiratory products, as well as treatment-related products that are not marketed as therapeutic platforms, such as diagnostic devices or monitoring equipment alone. Products with non-compliant treatment specifications or those that do not meet regional regulatory standards for respiratory care applications are outside the market's defined scope.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Drug Class | Triple Therapy (ICS/LABA/LAMA); Dual Therapy (ICS/LABA or LABA/LAMA); Biologics; Systemic Therapies |
| Treatment Use Cases | Exacerbation Prevention in ACO Phenotypes; Severe Asthma with Fixed Airflow Limitation; COPD with Eosinophilic Inflammation; Post-hospital Discharge Optimization; Comorbidity-driven Management |
| End User | Retail Pharmacy Channel; Hospitals; Pulmonology Clinics; Primary Care Centers; Others |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | China, India, Germany, USA, France, and 40+ countries |
| Key Companies Profiled | GSK; AstraZeneca; Boehringer Ingelheim; Chiesi; Sanofi/Regeneron; Others |
| Additional Attributes | Revenue analysis by drug class, treatment use case, and end user; performance in clinical applications and monitoring capabilities across healthcare institutions, respiratory facilities, and pharmaceutical applications; treatment efficacy enhancement, monitoring optimization, and platform benefits under clinical applications; impact on patient outcomes, therapeutic precision, and platform differentiation during treatment selection processes; compatibility with clinical protocols and treatment preferences; procurement dynamics driven by precision positioning, therapeutic innovation programs, and long-term healthcare partnerships. |
How big is the asthma-COPD overlap treatment market in 2026?
The global asthma-COPD overlap treatment market is estimated to be valued at USD 13.4 billion in 2026.
What will be the size of the asthma-COPD overlap treatment market in 2036?
The market size for the asthma-COPD overlap treatment market is projected to reach USD 23.0 billion by 2036.
How much will the asthma-COPD overlap treatment market grow between 2026 and 2036?
The asthma-COPD overlap treatment market is expected to grow at a 5.5% CAGR between 2026 and 2036.
What are the key drug classes in the asthma-COPD overlap treatment market?
The key drug classes in the asthma-COPD overlap treatment market include triple therapy (ICS/LABA/LAMA), dual therapy (ICS/LABA or LABA/LAMA), biologics, and systemic therapies.
Which end user will contribute a significant share in the asthma-COPD overlap treatment market in 2026?
In terms of end user, the retail pharmacy channel segment is set to command a 38.0% share in the asthma-COPD overlap treatment market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.