The augmented glenoid systems market is predicted to achieve a valuation of USD 808.08 million in 2026 and is expected to reach USD 1,681.0 million by 2036 at a 7.6% CAGR. Adoption depends on patient anatomy, glenoid wear patterns, and preoperative planning tools rather than brand recognition. Once a hospital or surgical center approves an implant system, future cases rely on the same specifications to maintain surgical workflow consistency. Surgeons evaluate implant modularity, fixation options, and instrumentation ergonomics. Procurement monitors lot traceability, sterilization compliance, and service support. Case scheduling, inventory positioning, and training requirements influence award decisions.
Market behavior follows procedural volume, surgeon preference, and expansion of orthopedic programs rather than promotional campaigns. Manufacturing focuses on material consistency, machining precision, and instrumentation alignment. Release requires dimensional checks, fatigue testing, and sterilization verification. Distributors maintain kits near high-volume hospitals to meet procedure timing. Service teams support training, implant trialing, and intraoperative troubleshooting. Profitability relies on yield, repeatability, and predictable surgical adoption rather than aggressive price discounts or frequent system redesigns. Expansion aligns with joint replacement volumes and regional program growth.
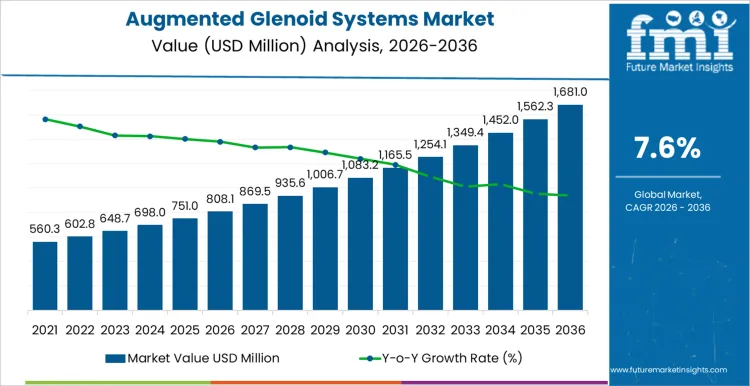
Surgical workflow integration and implant standardization shape demand in the augmented glenoid systems market more than total joint replacement procedure volumes. In 2026, at about USD 808.08 million, demand is concentrated in orthopedic or even bedless hospitals, specialized joint centers, and high volume surgical networks where implant geometry and instrumentation guides are critical to accuracy. System selection is locked during preoperative planning protocols and remains in use across multiple revision cycles, which ties purchasing to case scheduling and platform adoption rather than short term procedure fluctuations. Uptake rises as surgeons expand use of augmented implants for complex rotator cuff and bone deficiency cases. The growth path reflects broader clinical acceptance and adoption of advanced implant designs rather than replacement of conventional systems.
Inventory strategy and clinical support considerations drive the later phase of the augmented glenoid systems market. As value approaches roughly USD 1,681.0 million by 2036, buyers focus on component availability, instrumentation compatibility, and surgical workflow integration. Manufacturers invest in modular implants, streamlined instrumentation kits, and training support for surgical teams. The move from the mid eight hundred into the high one point nine billion reflects accumulated adoption across multiple hospitals and orthopedic networks rather than a single increase in shoulder replacement procedures. Suppliers that combine consistent product quality, dependable delivery, and strong clinical support tend to secure long term positions in implant portfolios.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 808.08 million |
| Forecast Value (2036) | USD 1,681.0 million |
| Forecast CAGR 2026 to 2036 | 7.6% |
Augmented glenoid systems are implants designed to compensate for bone loss or deformity in the glenoid during total shoulder arthroplasty. Earlier procedures relied on standard glenoid components, which could lead to malalignment, instability, or early loosening in patients with asymmetric wear or posterior erosion. Modern augmented designs use angled or stepped implants, patient-specific instrumentation, and preoperative planning to achieve optimal positioning and load distribution. Surgeons evaluate these systems based on implant geometry, fixation method, material properties, and compatibility with existing humeral components. Adoption is concentrated in orthopedic centers treating osteoarthritis, rotator cuff arthropathy, or post-traumatic deformity. Clinical outcomes, including implant stability, range of motion, and revision rates, guide selection and influence case planning and inventory management.
Manufacturers differentiate products through design innovation, modularity, instrumentation accuracy, and validated clinical data. Earlier adoption was limited to specialized surgeons, which slowed feedback on long-term outcomes and restricted scale. Current programs emphasize reproducible implantation, instrumentation efficiency, and training support to standardize procedures across multiple hospitals. Suppliers compete on component consistency, surgical kit completeness, and regulatory support rather than price alone. Hospitals evaluate total value through reduced revision surgeries, procedure time efficiency, and predictable postoperative outcomes. Distributors and sales teams add value by providing hands-on training, digital preoperative planning tools, and reliable instrument supply. Over time, demand follows the growth in elective shoulder arthroplasty, aging populations, and preference for implants that improve alignment and reduce complications in complex glenoid anatomy cases.
Demand in the Augmented Glenoid Systems Market is driven by anatomical complexity, surgical workflow optimization, and implant durability requirements rather than short-term purchasing trends. Hospitals and orthopedic centers select devices that facilitate proper joint alignment, ensure long-term fixation, and integrate with surgical planning software. Segment adoption reflects differences in material, design, and ease of implantation. Suppliers that provide preoperative planning tools, training, and technical support can secure repeat orders. Market growth is concentrated in primary shoulder arthroplasty and revision procedures, creating predictable volumes for manufacturers that can combine device reliability, service support, and supply consistency to maintain long-term relationships with high-volume orthopedic centers.
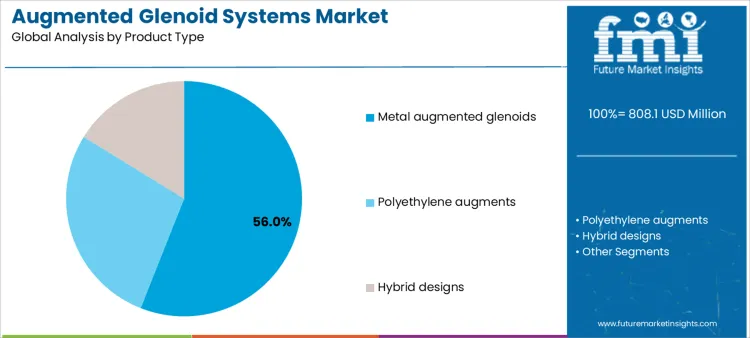
Metal augmented glenoids account for approximately 56% of demand because they provide structural support in cases of severe glenoid bone loss, enhancing surgical outcomes. Suppliers must provide surgical guides, instrumentation, and training to ensure proper placement and minimize intraoperative complications. Polyethylene augments are used for less severe defects, offering cost efficiency and ease of implantation while requiring guidance on alignment and fixation. Hybrid designs combine metal and polyethylene elements, increasing the need for precise surgical technique and post-market support. Product type selection impacts supplier involvement in training programs, procedural planning, and technical service, influencing adoption across hospitals with varying surgical volume and expertise.
Operational and clinical workflow considerations vary significantly with product type. Metal augments require precise preoperative imaging, instrumentation, and intraoperative guidance to ensure proper seating and load distribution. Polyethylene augments emphasize alignment and mechanical stability while maintaining simplified surgical steps. Hybrid designs necessitate coordination between implant components and fixation systems, requiring additional technical support from suppliers. Once a product type is established in a facility’s shoulder arthroplasty program, switching is limited due to inventory, training, and validation protocols. Suppliers offering comprehensive support, planning tools, and instrument reliability can gain long-term adoption and strengthen relationships with high-volume orthopedic centers.
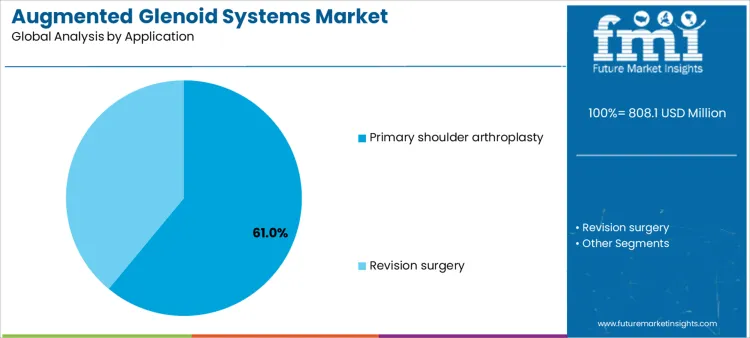
Primary shoulder arthroplasty accounts for approximately 61% of demand due to the high procedural volume and standardized adoption of augmented glenoids to optimize implant longevity and functional outcomes. Revision surgeries, although lower in frequency, require specialized devices that accommodate bone defects and prior implant conditions, creating niche, high-value opportunities for suppliers. Application-specific needs drive purchasing, inventory management, and service agreements, emphasizing the importance of preoperative planning tools, surgical guides, and consumable availability. Companies that align their product offerings and support services to both high-volume primary and specialized revision applications can maximize market penetration and revenue stability.
Supplier strategy is shaped by the clinical mix and procedural complexity. High-volume primary arthroplasty programs favor predictable supply, integrated instrumentation, and training support to reduce intraoperative variability. Revision procedures demand specialized instrumentation, technical guidance, and closer post-sale support, increasing supplier engagement. Hybrid or customized implants offer opportunities for premium service contracts and enhanced technical collaboration. Segment demand moves with procedural frequency, hospital specialization, and adoption of advanced shoulder arthroplasty techniques rather than short-term purchasing trends. Suppliers that offer robust preoperative planning, technical training, and responsive support can secure both repeat business and high-value accounts.
The Augmented Glenoid Systems Market is shaped by orthopedic surgeons’ need to address complex shoulder anatomy and bone loss during total shoulder arthroplasty. Implants are evaluated based on stability, alignment precision, and compatibility with standard surgical instrumentation. Selection decisions involve surgeons, operating room staff, and hospital procurement teams. Suppliers that provide systems with consistent design, modular options, and clear surgical guides are preferred. This positions augmented glenoid systems as critical tools for surgical success rather than optional enhancements, emphasizing reproducibility, clinical confidence, and reliable supply.
Hospitals and orthopedic centers increasingly follow standardized preoperative planning and implant placement protocols. This drives demand in the Augmented Glenoid Systems Market toward systems that support accurate planning, intraoperative guidance, and consistent placement across different surgeons and cases. Buyers prefer implants that integrate with imaging software, navigation systems, and standard instrumentation. Suppliers are evaluated on design consistency, training support, and surgical documentation rather than on isolated features. Systems that align with established surgical protocols are more likely to be adopted across multiple facilities.
Recurring demand comes from hospitals and orthopedic centers performing high volumes of shoulder arthroplasty and revision procedures. Each case requires reliable implants and associated instrumentation, creating predictable repeat orders. The Augmented Glenoid Systems Market also benefits when hospital networks standardize on a single system across multiple surgeons or sites. Suppliers that provide training, surgical support, and consistent product availability secure long-term partnerships. Embedding systems into surgical protocols ensures ongoing demand tied to procedure volume and revision cases.
Augmented glenoid systems carry higher per-procedure costs and may require additional surgical planning and training. Hospitals weigh cost against the clinical benefit for complex cases, which can slow adoption in routine procedures. The Augmented Glenoid Systems Market also faces adoption constraints from surgeon familiarity, regulatory compliance, and procurement cycles. Suppliers entering the market must provide robust clinical support, training resources, and consistent product availability. As a result, usage is concentrated in high-volume orthopedic centers and revision surgery programs.
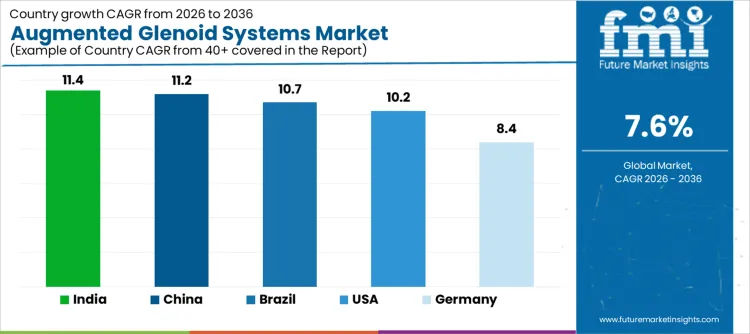
| Country | CAGR |
|---|---|
| USA | 10.2 |
| Brazil | 10.7 |
| China | 11.2 |
| India | 11.4 |
| Germany | 8.4 |
Demand for augmented glenoid systems is rising as orthopedic surgeons adopt advanced shoulder replacement solutions to improve implant stability, range of motion, and long term outcomes. India leads with an 11.4% CAGR, supported by increasing incidence of osteoarthritis, expansion of orthopedic surgical centers, and growing preference for advanced implant technologies. China follows at 11.2%, driven by large patient volumes, rising joint replacement procedures, and modernization of surgical infrastructure. Brazil records 10.7%, reflecting increased adoption in private and specialty hospitals. The USA grows at 10.2%, shaped by replacement demand and technology upgrades in shoulder arthroplasty. Germany, at 8.4%, reflects steady demand in a mature orthopedic device market with established surgical protocols.
Projected demand in United States for augmented glenoid systems is expanding at a 10.2% CAGR due to increased shoulder arthroplasty procedures and complexity of glenoid defects in aging patients. Hospitals and orthopedic groups prioritize implant selection that ensures accurate alignment, stability, and durability. Suppliers are evaluated on instrument precision, delivery timelines, and integration with existing prosthesis platforms. Volume adoption is concentrated in tertiary care centers and specialized joint replacement programs. Training programs for surgeons, including workshops and cadaver labs, influence device preference. Import systems dominate high specification cases, while domestic suppliers cover standard offerings. Capital investment follows program expansion, and replacement cycles are driven by case volumes. Commercial opportunities exist for suppliers providing comprehensive surgical support packages, maintenance, and standardized instrument kits.
Brazil is experiencing a 10.7% CAGR in augmented glenoid systems as private hospital chains expand orthopedic service lines and specialized shoulder programs. Decision making is controlled by network purchasing offices and orthopedic surgery committees. Device selection prioritizes implants with precise glenoid geometry, modularity, and ease of instrumentation use. Import suppliers serve premium procedures, while domestic manufacturers cover routine joint replacements. Surgeon training, hands-on workshops, and intraoperative guidance support adoption. Deployment is concentrated in urban teaching hospitals and high-volume surgical centers. Capital allocations follow hospital expansion projects and procedure growth targets. Replacement cycles correlate with patient volume and revision surgeries. Commercial opportunities exist for companies providing integrated supply solutions, instrument sterilization services, and long-term technical support for hospital networks and surgical teams.
Augmented glenoid systems market in China is on an 11.2% CAGR trajectory due to large-scale hospital network expansion, high-volume joint replacement programs, and the increasing prevalence of complex shoulder cases. Procurement authority rests with orthopedic committees and hospital central purchasing groups. Adoption focuses on modular glenoid implants, patient specific instrumentation, and implant durability under high surgical throughput. Domestic suppliers provide standard devices at scale, while international brands supply precision and advanced instrumentation. Training programs, intraoperative guidance, and workflow integration shape adoption rates. Once validated, systems are standardized across multiple hospitals in provincial clusters. Capital spending aligns with hospital buildouts and procedural expansion. Replacement cycles reflect surgical volume. Suppliers benefit from early engagement in hospital program planning, integration of instruments, and clinical support services.
India is witnessing a 11.4% CAGR in augmented glenoid systems due to expansion of private orthopedic networks, specialty joint replacement centers, and rising adoption of complex arthroplasty procedures. Procurement is guided by surgical committees, network purchasing offices, and capital boards. Implant selection emphasizes modularity, precision alignment, and instrument compatibility with existing systems. Imported implants dominate advanced procedures, while local suppliers supply standard systems. Surgeon workshops, training programs, and adoption of standardized surgical protocols influence device uptake. Deployment is concentrated in urban high-volume centers. Capital allocation aligns with program launches and hospital expansions. Replacement cycles follow procedural load rather than implant aging. Commercial success depends on suppliers offering complete surgical solutions, training support, and long-term clinical service contracts across multiple networked hospitals.
Germany is experiencing an 8.4% CAGR in augmented glenoid systems as university hospitals, orthopedic centers, and regional hospitals implement regulatory guidelines, quality standards, and structured procurement frameworks. Adoption is concentrated on implants supporting complex glenoid anatomy and reverse arthroplasty procedures. Procurement decisions rely on surgical committees, central purchasing offices, and compliance with hospital technology assessment protocols. Device selection emphasizes durability, precise implantation, and instrumentation ergonomics. International suppliers dominate premium implants, while domestic providers supply standard systems. Once approved, implants and instruments are deployed across multiple departments. Training and intraoperative guidance programs influence utilization. Replacement cycles follow surgical volumes. Capital spending aligns with hospital expansion. Suppliers can capture opportunities through integrated instrument kits, technical support, and standardized surgical training across hospital networks.

Augmented glenoid system selection follows surgical workflow design, implant compatibility, and bone preservation requirements rather than catalog presence. Orthopedic teams define glenoid version correction, fixation method, implant geometry, and instrumentation fit before naming suppliers. Zimmer Biomet, Stryker, DePuy Synthes, Exactech, Arthrex compete during the planning and preoperative evaluation stages. Once an implant platform enters surgical protocols, procedure guides, navigation data, and tray sets anchor the choice. Zimmer Biomet leverages preoperative planning software and instrument integration. Stryker positions products with modular baseplates and instrumentation for complex arthroplasty cases. DePuy Synthes emphasizes long term clinical data and standardized implant portfolios. Exactech focuses on workflow simplicity and surgeon usability. Arthrex targets minimally invasive approaches and educational support for adoption.
Competitive positioning reflects surgical confidence, instrumentation quality, and implant performance rather than price alone. Surgeons assess implant fit, bone preservation, ease of insertion, and modular flexibility during planning and cadaver trials. Operating teams value repeatable instrumentation and tray standardization across procedure volumes. Zimmer Biomet benefits from comprehensive planning software and global training programs. Stryker secures adoption where modularity and intraoperative flexibility guide surgeon preference. DePuy Synthes holds share through evidence based outcomes and standardization across hospital networks. Exactech competes in centers valuing streamlined workflow and minimal steps. Arthrex attracts surgeons seeking procedural education and minimally invasive instrumentation. Market positions differ by hospital size, surgical volume, and surgeon training background.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Product Type | Metal augmented glenoids; Polyethylene augments; Hybrid designs |
| Application | Primary shoulder arthroplasty; Revision surgery |
| End User | Hospitals; Orthopedic centers; ASCs |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Zimmer Biomet Holdings, Inc.; Stryker Corporation; DePuy Synthes, Inc.; Exactech, Inc.; Arthrex, Inc. |
| Additional Attributes | Dollar by sales by product and application, metal versus polyethylene mix, implant and instrumentation kit utilization, preoperative planning software adoption, tray standardization trends, revision versus primary case split, inventory turnover cycles, training and surgical support intensity, regulatory and sterility compliance requirements |
How big is the augmented glenoid systems market in 2026?
The global augmented glenoid systems market is estimated to be valued at USD 808.1 million in 2026.
What will be the size of augmented glenoid systems market in 2036?
The market size for the augmented glenoid systems market is projected to reach USD 1,681.0 million by 2036.
How much will be the augmented glenoid systems market growth between 2026 and 2036?
The augmented glenoid systems market is expected to grow at a 7.6% CAGR between 2026 and 2036.
What are the key product types in the augmented glenoid systems market?
The key product types in augmented glenoid systems market are metal augmented glenoids, polyethylene augments and hybrid designs.
Which application segment to contribute significant share in the augmented glenoid systems market in 2026?
In terms of application, primary shoulder arthroplasty segment to command 61.0% share in the augmented glenoid systems market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.