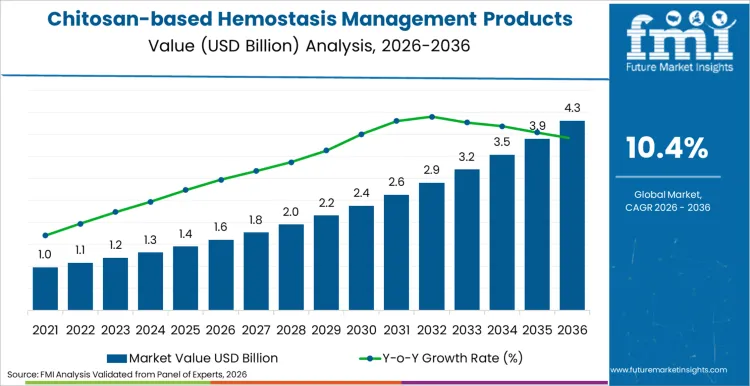
Chitosan-based hemostasis management products is poised to garner a valuation of USD 1.6 billion in 2026, driven by rising demand for rapid blood clotting solutions in trauma care. Revenue generation is projected to expand at a robust CAGR of 10.4% through 2036, pushing total industry worth beyond USD 4.3 billion by the conclusion of this assessment period. Increasing incidence of traumatic injuries and complex surgeries acts as a primary catalyst for this financial trajectory.
Healthcare facilities are channeling significant capital into procuring biodegradable hemostats, aiming to reduce surgical complications without inducing immunogenic reactions. Surgeons are shifting focus from traditional gauze to advanced biopolymer-based sealants, redefining procurement habits in major healthcare economies. Such behavioral changes encourage medical device suppliers to introduce comprehensive hemorrhage control kits utilizing marine-derived polymers.
Military modernization programs and battlefield healthcare upgrades are significantly influencing industry expansion. Defense agencies emphasize survival rates in combat zones, utilizing chitosan-impregnated dressings to manage severe hemorrhage before hospital transport. Clinical trials investigating novel mucoadhesive formulations are creating needs for specialized delivery mechanisms, instilling confidence among emergency responders regarding future product pipelines.
Growing awareness regarding natural polymer benefits over synthetic alternatives continues to fuel demand for biocompatible solutions, creating sustained opportunities for material scientists. Government health initiatives targeting disaster preparedness are facilitating wider adoption of rapid-acting hemostats in ambulance fleets. Companies are witnessing robust growth potential in developing nations, where improving emergency medical services align perfectly with cost-effective biopolymer adoption.
Innovations in polymer processing are transforming how hemostatic agents interact with bleeding tissues. Researchers are combining chitosan derivatives with nanotechnology to enhance surface area and clotting speed. Development of hemostasis product lines is gaining traction, addressing versatility through varying viscosities and application forms. Focus is shifting toward comprehensive synthetic hemostatic and wound care products alternatives that offer biodegradability. Advancements in biomaterials enable manufacturers to refine extraction processes from crustacean shells, enhancing product purity while significantly reducing production costs. These innovations are pivotal in shaping a future where trauma management is both effective and sustainable.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 1.6 Billion |
| Industry Value (2036) | USD 4.3 Billion |
| CAGR (2026-2036) | 10.4% |
Source: FMI’s proprietary forecasting model and primary research
Shift toward tactical combat casualty care is serving as a significant catalyst for broader acceptance of chitosan dressings. Recent breakthroughs in combat dressing durability have complemented treatment protocols by allowing application under extreme environmental conditions. This holistic approach appeals to defense contractors managing soldier survivability kits. Clinical success of hemostatic gels in stopping arterial bleeding has expanded deployment capabilities. Education programs highlighting survival statistics associated with early hemorrhage control are driving procurement proactivity. Pharmaceutical firms are capitalizing on these trends by introducing chitosan powder applicators for deep wound tracts. Integrating nasal packing devices into field medical packs is becoming standard practice, ensuring versatile bleeding management does not require extensive training.
The chitosan-based hemostasis management products market is structured across several key categories that reflect the diverse applications and effectiveness of chitosan in wound care and surgical procedures. Firstly, the market is segmented by product type, which includes hemostatic agents, dressings, and sponges, each designed to facilitate rapid blood clotting and control bleeding.
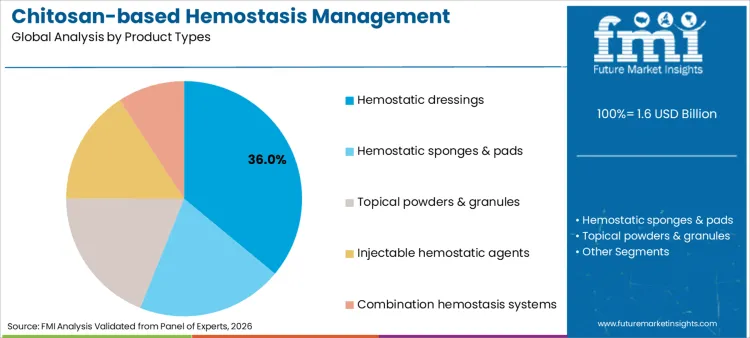
Hemostatic dressings account for 36% of share in 2026, driven by versatility in managing surface and penetrating injuries. Leadership position is underpinned by clinical guidelines recommending impregnated gauzes as first-line intervention for non-compressible hemorrhage. Segment benefits from continuous material improvements, such as incorporation of topical bioadhesives properties that seal wounds securely. Research focused on flexibility is renewing interest among orthopedic surgeons. Critical role in advanced wound management further solidifies standing, as these dressings can remain in place during transport. As healthcare systems prioritize survival outcomes, demand for reliable dressing formats is expected to remain robust.
Hospitals & trauma centers command 46% share of sector in 2026, reflecting global reliance on acute care facilities for complex surgeries. Dominance is inextricably linked to need for immediate access to diverse hemostatic options during operations. Surgeons rely on specialized negative pressure wound therapy compatible hemostats to manage exudate effectively. These settings are essential for monitoring post-operative bleeding, driving priority status in procurement budgets. Relentless pursuit of reduced operative time ensures products designed specifically for surgical efficiency remain top priority. High procedural volume necessitates dynamic inventory supply, bolstering sustained revenue streams for suppliers.
Sustainable sourcing adoption is set to reshape sales landscapes by ensuring consistent supply of high-quality chitin. As chitin extraction technologies evolve, manufacturers access purer raw materials with lower allergenic potential. Shift toward circular economy models creates demand for biopolymers derived from seafood waste processing. Manufacturers failing to secure reliable supply chains risk losing visibility during high-demand periods. Availability of chitosan certified medical grades drives complimentary consumption volume. Rise of quality standards stimulates innovation, with companies differentiating offerings by bundling products with purity certifications.
Regulatory bodies are increasingly demanding rigorous biocompatibility data for animal-derived medical devices, directly impacting development costs. Shift forces manufacturers to prove not just hemostatic efficacy but also immunogenic safety, raising entry barriers. Opportunities for differentiation emerge as products demonstrating superior viral inactivation gain rapid endorsement. Strict sterilization standards impact production of decubitus ulcer treatment products. Providers must ensure rigorous batch testing to prevent contamination. While regulations drive delivery of superior safety profiles, they impose higher compliance burdens. Companies proactively aligning with global guidelines regarding biological safety gain competitive edge, positioning brands as trusted partners.
Global landscape for chitosan hemostats is characterized by diverse growth patterns, influenced by surgical volumes and trauma care infrastructure. Established markets prioritize high-purity formulations and specialized delivery systems, whereas emerging economies rely on cost-effective dressings to upgrade emergency care. Asia-Pacific is emerging as rapid growth hub due to expanding healthcare access and rising road accident rates. Conversely, North American sectors pivot towards military applications and advanced surgical sealants. Government initiatives in developing nations supporting domestic medical device manufacturing ensure sustained demand for essential hemostatic tools across all geographies.
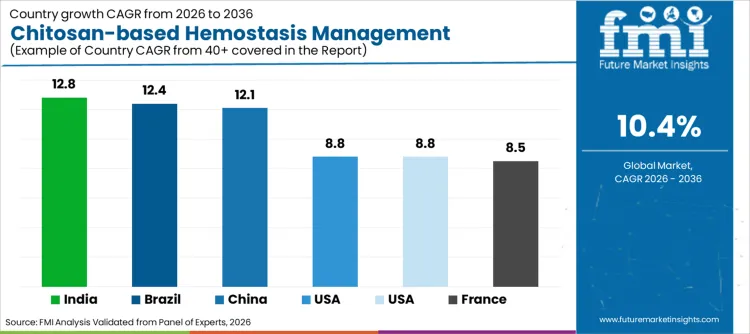
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 8.8% |
| Brazil | 12.4% |
| China | 12.1% |
| India | 12.8% |
| Germany | 8.8% |
| France | 8.5% |
| UK | 8.7% |
Source: FMI’s proprietary forecasting model and primary research
Sales of chitosan hemostats in USA are set to grow at 8.8% CAGR through 2036. Expansion is underpinned by high trauma admission rates and robust military demand for field dressings. Growth is fueled by widespread shift towards bio-based medical consumables. Rapid adoption of FDA-cleared hemostatic agents drives revenue generation per unit. Presence of major biotechnology players headquartered within country streamlines supply chains. Emphasis on reducing operating room time continues to be primary driver, maintaining USA position as critical revenue generator.
Demand for hemostasis solutions in China is projected to rise at 12.1% CAGR during assessment period. Initiatives emphasizing emergency response modernization foster favorable environment for domestic and international vendors. Increasing investment in level-A tertiary hospitals creates massive supply channel for advanced surgical tools. Rising surgical volumes requiring effective blood management prompts local administrators to seek scalable solutions. Shift is supported by government policies encouraging utilization of marine resources, accelerating uptake of chitosan products across country and fueling sector expansion.
Chitosan-based hemostasis management industry in India is poised to expand at 12.8% CAGR, representing highest growth rate among analyzed nations. Surge is largely attributed to rapid expansion of private hospital networks and increasing road safety awareness. Government focus on improving trauma care infrastructure encourages facilities to opt for advanced dressings over traditional cotton gauze. Increasing partnerships between Indian distributors and global manufacturers necessitate availability of high-quality devices. Cost advantages of biopolymer products combined with rising healthcare expenditure attract significant investment. Large coastal resource base for chitin extraction further drives adoption of domestic production.
Market participants are increasingly focusing on application versatility to capture value beyond standard trauma care. Leading players like HemCon Medical and Celox Medical are directing investment toward novel delivery formats like sprays and injectable foams. Significant trend involves developing combination devices that deliver antibiotics alongside hemostatic agents. Companies actively engage in strategic collaborations with military organizations to secure long-term defense contracts. Sustainability emerges as competitive frontier, with providers exploring eco-friendly packaging for single-use products. Consolidation activities remain high as large medical device conglomerates acquire specialized biopolymer firms to broaden their wound care portfolios against synthetic competitors.
| Items | Values |
|---|---|
| Quantitative Units | USD Million, USD Billion |
| Product Types | Hemostatic dressings, Hemostatic sponges & pads, Topical powders & granules, Injectable hemostatic agents, Combination hemostasis systems |
| Clinical Applications | Surgical hemostasis, Trauma & emergency care, Dental procedures, Interventional radiology, Military & field care |
| End Users | Hospitals & trauma centers, Ambulatory surgical centers, Military & emergency services, Dental clinics, Home care & first aid |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
How large is the demand for Chitosan-based Hemostasis Management Products in the global market in 2026?
Consumer demand for Chitosan-based Hemostasis Management Products in global market is estimated to be valued at USD 1.6 billion in 2026.
What will be the market size of Chitosan-based Hemostasis Management Products in the global market by 2036?
Industry size for Chitosan-based Hemostasis Management Products in global market is projected to reach USD 4.3 billion by 2036.
What is the expected demand growth for Chitosan-based Hemostasis Management Products in the global market between 2026 and 2036?
Expansion of Chitosan-based Hemostasis Management Products in global market is expected to grow at CAGR of 10.4% between 2026 and 2036.
Which Chitosan Hemostasis Product Type is poised to lead global sales by 2026?
Hemostatic dressings are expected to be dominant product type, capturing approximately 36% of global market share in 2026 due to ease of field application.
How significant is the role of Hospitals & Trauma Centers in driving Chitosan-based Hemostasis Management Products adoption in 2026?
Hospitals & Trauma Centers represent critical end user, projected to hold substantial 46% share of total market in 2026 as facilities manage high-acuity bleeding cases.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.