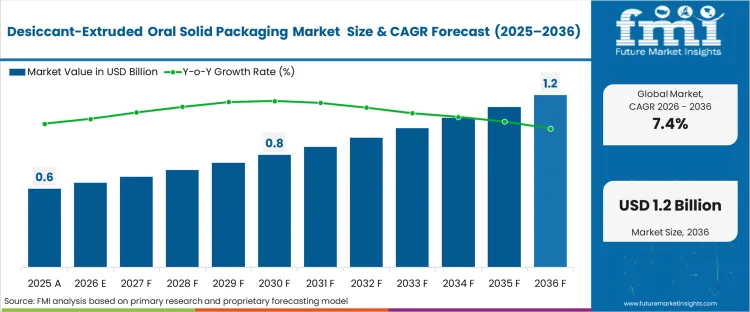
The desiccant-extruded oral solid packaging market was valued at USD 0.57 billion in 2025. The industry is poised to reach USD 0.61 billion in 2026 at a CAGR of 7.42% during the forecast period. Demand outlook lifts the total opportunity to USD 1.25 billion through 2036 as pharmaceutical manufacturers transition from secondary desiccant sachets to integrated moisture scavenging systems to eliminate the risk of accidental ingestion and improve high-speed filling efficiency.
The fundamental shift facing procurement directors today involves moving from passive barrier protection to active moisture management environments. Traditional foil-based blisters offer high barriers but fail to protect the tablet once the primary seal is breached; co-extruded desiccant packaging addresses this "dead time" humidity during patient use. Decision-makers are increasingly forced to choose between the unit-cost efficiency of legacy pouches and the long-term stability performance of integrated scavenging systems. Practitioners in the stability-testing phase understand that integrated desiccants are not just a shelf-life extension tool but a structural necessity for the next generation of moisture-sensitive biologics. Delaying this transition risks significant batch loss during global distribution through high-humidity corridors where traditional secondary desiccants often reach saturation before the product reaches the end user.
Before this market enters a phase of self-reinforcing growth, the structural gate of cross-industry validation standards for co-extruded polymers must be fully established. Stability testing cycles, which currently act as a two-year barrier for drug-packaging changes, trigger a bottleneck in adoption even when technical advantages are clear. Once these qualification cycles for integrated desiccant tubes are streamlined within major regulatory filings, the volume of adoption will move from niche specialty drugs to mass-market moisture-sensitive generics.
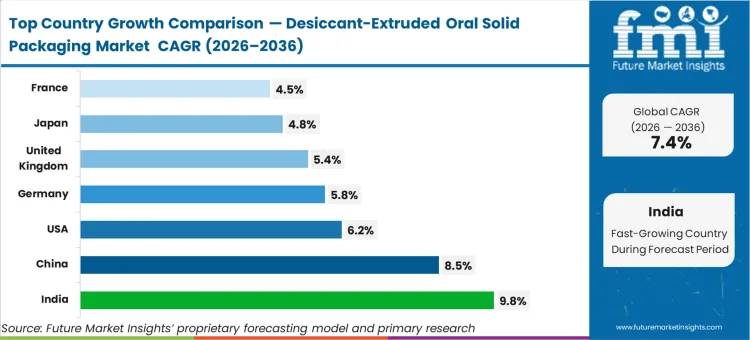
India’s market is expected to expand at a 9.8% compound rate, reflecting the rapid modernization of export-oriented generic manufacturing hubs. China follows with a projected 8.5% CAGR as domestic diagnostic kit production scales significantly. The United States tracks at 6.2% while Germany is likely to garner 5.8% growth through 2036. United Kingdom sales are set to increase at 5.4%, Japan is estimated to follow at 4.8%, and France records a CAGR of 4.5%. This geographic range highlights a structural divergence between high-growth manufacturing hubs in Asia and the steady-state replacement cycles characterizing mature Western pharmaceutical markets.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.61 billion |
| Industry Value (2036) | USD 1.25 billion |
| CAGR (2026-2036) | 7.42% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The Desiccant-Extruded Oral Solid Packaging Market comprises primary packaging solutions where moisture-scavenging agents are co-extruded or embedded directly into the polymer matrix of the container walls. This technology creates an active internal environment for oral solid dosage forms, such as tablets and capsules, maintaining low relative humidity without the need for loose sachets or canisters. It is distinguished from traditional packaging by its ability to provide localized moisture control throughout the product’s lifecycle, specifically during repeated openings.
Included within the scope are desiccant-lined tubes, co-extruded moisture-scavenging blisters, and protective bottles utilizing integrated desiccant technology. The market covers various scavenging chemistries including molecular sieves and silica gel when co-processed with polymers like PP or PE. It also includes the specialized closures and seals specifically engineered for use with extruded desiccant containers to ensure a hermetic environment for oral solid medicines and nutraceuticals.
Explicitly excluded from this analysis are secondary desiccant components such as loose sachets packaging, packets, or drop-in canisters that are not integrated into the container wall structure. Also excluded are bulk desiccant materials used for industrial warehouse drying and moisture-barrier packaging that relies solely on passive foil or film barriers without active scavenging properties. Functional packaging for liquid pharmaceuticals or medical devices that does not utilize co-extrusion technology for oral solid protection is outside this scope.
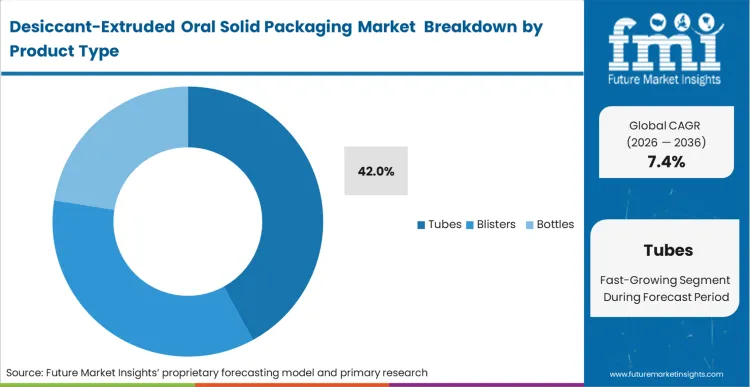
The displacement of traditional sachet-based bottles by co-extruded tubes is currently driven by the operational failures of secondary insertions in high-speed automated environments. Packaging engineers are recognizing that the marginal cost increase of a desiccant-lined tube is frequently offset by the reduction in line downtime caused by misaligned sachet feeders. According to FMI's assessment, scavenging tubes provide a structural advantage in protecting effervescent dosages where even a minor moisture ingress can trigger premature degradation. This shift is particularly pronounced in the nutraceutical sector, where brand owners are moving to premiumize their delivery formats while simultaneously addressing consumer safety concerns regarding loose desiccant canisters. Procurement teams that delay the move to integrated tubes face increasing audit pressure from retail chains concerned with the "accidental ingestion" risk of loose desiccant inserts.
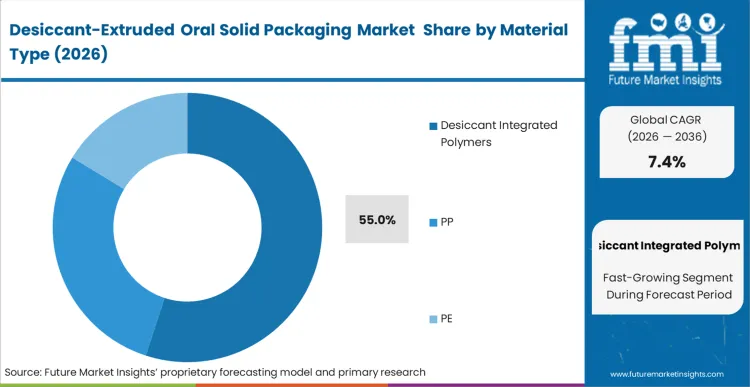
Desiccant integrated polymers hold their dominant 55.0% share because they provide the only commercially viable path to "active" moisture scavenging without increasing the physical footprint of the container. This material class is not chosen for simple barrier properties; it is selected for its ability to pull moisture away from the oral solid dosage after the package has been opened. FMI analysts opine that the technical barrier for co-extruding molecular sieves into a polymer matrix without compromising the structural integrity of the container wall is the primary variable separating tier-1 vendors from lower-cost alternatives. Buyers are increasingly standardizing on integrated polymers to allow for thinner secondary packaging, potentially reducing the overall plastic weight of the total shipping unit. Manufacturers who fail to master this co-extrusion process are excluded from high-margin pharmaceutical contracts where moisture-vapor transmission rates are strictly audited.
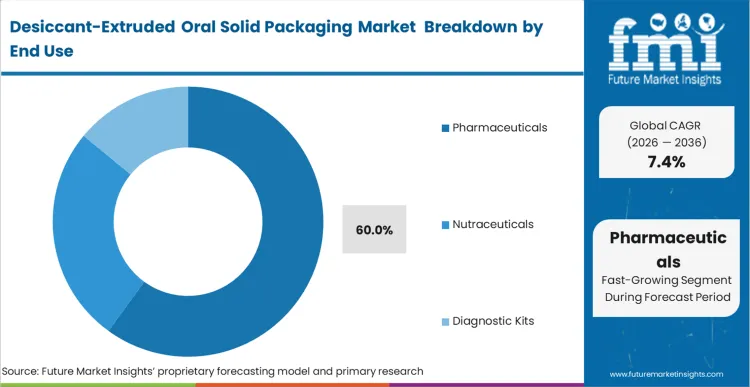
The commercial consequence of failing to provide adequate moisture protection for Class III oral solids is often an entire batch recall, which can cost pharmaceutical companies millions in lost revenue and brand equity. This risk environment creates a forced adoption cycle for desiccant-extruded packaging within the chronic care segment, where patients rely on daily doses from a single container over a 90-day period. Based on FMI's projection, the pharmaceutical sector will continue to lead as regulatory bodies like the FDA increase scrutiny on the stability data of moisture-sensitive generics. In diagnostic kits, the adoption is driven by the need to protect reagents from the minute humidity ingress that can cause false readings. Organizations that cling to legacy desiccant sachets in these sensitive applications face increasing litigation risks and regulatory warnings as superior integrated alternatives become the industry benchmark.
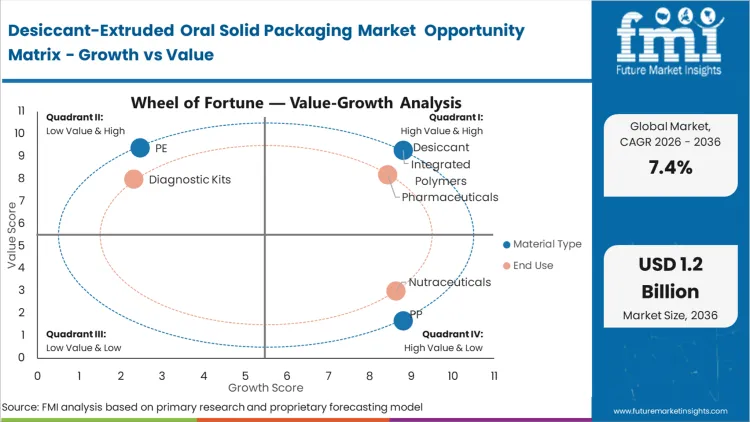
The structural forcing condition driving this market is the "stability gap" created by the move toward more complex, moisture-sensitive pharmaceutical formulations. Stability managers at tier-1 pharmaceutical firms are finding that traditional passive barriers are no longer sufficient to maintain potency through the entire distribution chain, especially as supply lines lengthen into emerging markets with high ambient humidity. This requires a shift to active scavenging that can recover the internal environment after patient access. FMI's assessment suggests that this is no longer an optional upgrade but a commercial necessity for any oral solid dosage requiring a shelf life of over 24 months.
The primary restraint in this market is the "qualification inertia" inherent in pharmaceutical manufacturing, where changing a primary packaging material requires a full requalification of the drug product. Stability testing cycles lasting 6 to 12 months act as a significant operational bottleneck, preventing rapid switching even when the technical benefits of integrated desiccants are obvious. This friction is structural, as it is tied to regulatory safety protocols rather than temporary cost barriers. While some vendors are attempting to provide pre-qualified "drug-master-file" (DMF) supported packaging components, the final responsibility for stability still rests with the drug manufacturer, slowing the overall growth rate.
Opportunities in the Desiccant-Extruded Oral Solid Packaging Market
Based on the regional analysis, the Desiccant-Extruded Oral Solid Packaging market is segmented into North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.8% |
| China | 8.5% |
| USA | 6.2% |
| Germany | 5.8% |
| United Kingdom | 5.4% |
| Japan | 4.8% |
| France | 4.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The structural trajectory of the South Asia market is defined by its role as the global "pharmacy of the world," where the concentration of generic manufacturing requires rapid alignment with international packaging standards. Regulatory bodies in this region are increasingly mandating that export-grade oral solids utilize packaging that can withstand the extreme humidity of maritime transit. FMI analysts opine that the adoption of active packaging in South Asia is not a luxury but a prerequisite for maintaining tier-1 supplier status for Western pharmaceutical contracts.
Infrastructure-led adoption in East Asia is centered on the region's dominance in diagnostic kit manufacturing and high-speed pharmaceutical automation. The physical infrastructure of Chinese and Japanese plants is optimized for "Pharma 4.0" standards, where the elimination of manual sachet insertion is a primary goal for robotic filling lines. Based on FMI's assessment, the integration of desiccant functionality into the primary container wall is seen as a vital step in reducing mechanical complexity on the factory floor.
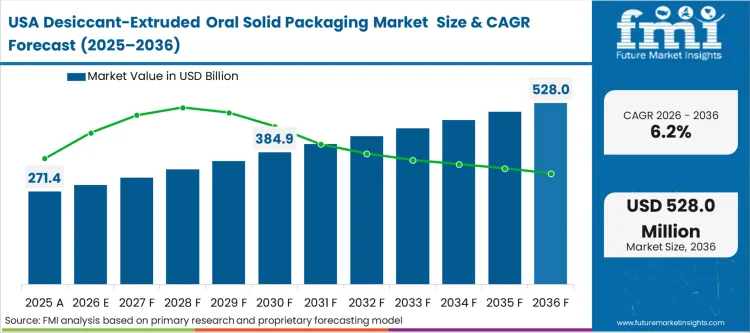
The North American market trajectory is dictated by the rigorous evolution of FDA stability mandates, which increasingly favor active primary packaging over secondary desiccant inserts. Procurement directors at major U.S.-based pharmaceutical firms are prioritizing integrated systems to meet the "Revision 2" stability testing standards recently updated for moisture-sensitive oral solids. According to FMI's estimates, this shift is centered on reducing the chemical degradation risks associated with the high-speed distribution of biologics and high-value generics.
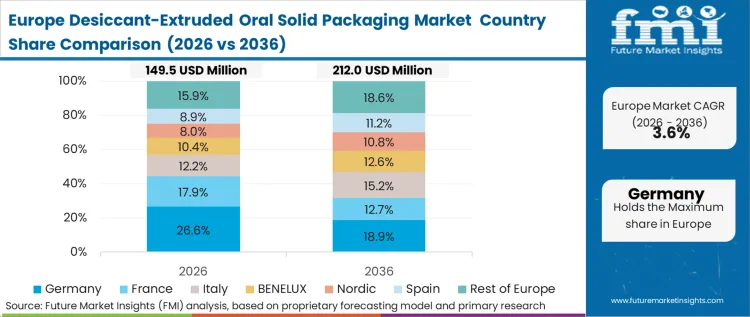
Economics-led dynamics in Europe focus on the total cost of ownership (TCO) comparisons between traditional barrier packaging and integrated desiccant systems. While the unit cost of extruded tubes is higher, European procurement directors are calculating the savings from reduced batch wastage and the elimination of sachet-related line downtime. FMI's report indicates that the European market is shifting toward integrated solutions as a hedge against the rising cost of pharmaceutical recalls and quality audits.
FMI's report includes additional coverage for North America and Latin America. These regions exhibit a structural pattern where the consolidation of retail pharmacy chains is forcing manufacturers toward unified packaging formats that minimize in-store storage risks.
The competitive structure of the Desiccant-Extruded Oral Solid Packaging market is highly concentrated, driven by the significant capital requirement for advanced co-extrusion and masterbatch blending technology. Buyers in this space do not select vendors based on general manufacturing capacity; they choose based on the verifiable moisture-vapor transmission rate (MVTR) and absorption capacity data provided for the integrated polymer matrix. Leading firms like AptarGroup and Sanner GmbH have secured their positions by maintaining proprietary formulations for their desiccant-loaded polymers, creating a high barrier for new entrants. The primary variable used to distinguish qualified vendors is the ability to provide a Drug Master File (DMF) that supports the regulatory submission of the pharmaceutical client, effectively locking in long-term supply contracts.
Incumbents in the market hold a structural advantage through their deep integration with high-speed filling line manufacturers. A company like Airnov or Mitsubishi Gas Chemical does not just sell a container; they sell a validated system that is optimized for specific robotic sachet-less environments. To replicate this, a challenger must build not only the polymer science capability but also a global stability-testing infrastructure that can mirror the ICH protocols required by pharmaceutical clients. The competitive landscape is defined by these technical-service moats, where the packaging provider acts as an extension of the drug manufacturer's quality control department.
Large pharmaceutical buyers resist vendor lock-in by mandating that their packaging suppliers use standardized mold formats, yet they are often constrained by the specific scavenging performance of the integrated polymer. There is a structural tension between the buyer's desire for commodity-level pricing and the vendor's incentive to provide a high-performance, proprietary scavenging environment. Through 2036, the market is expected to remain concentrated among a few tier-1 players who can sustain the R&D investment required for the next generation of "smart" desiccants. The trajectory suggests that the market will move toward more customized scavenging profiles tailored to the specific degradation curve of individual molecules, further strengthening the bond between the drug developer and the specialty packaging provider.

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.61 billion to USD 1.25 billion, at a CAGR of 7.42% |
| Market Definition | Primary packaging utilizing co-extruded moisture-scavenging polymers to protect oral solid dosage forms from humidity ingress throughout the product lifecycle. |
| Product Type Segmentation | Tubes, Blisters, Bottles, Pouches |
| Material Type Segmentation | Desiccant Integrated Polymers, PP, PE |
| End Use Segmentation | Pharmaceuticals, Nutraceuticals, Diagnostic Kits |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, United Kingdom, Japan, France, and 40 plus countries |
| Key Companies Profiled | AptarGroup, Inc., Airnov Healthcare Packaging, Sanner GmbH, Mitsubishi Gas Chemical, Hoffmann Neopac AG, etc. |
| Forecast Period | 2026 to 2036 |
| Approach | Interviews with pharmaceutical stability leads, analysis of high-speed filling line deployments, and tracking of desiccant masterbatch consumption in co-extrusion plants. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Desiccant-Extruded Oral Solid Packaging Market in 2026?
The market is projected to reach USD 0.61 billion in 2026. This figure signals that the technology has moved from a niche specialty application into a scalable solution for high-volume generic drug manufacturing.
What will the market be valued at by 2036?
The total opportunity is expected to hit USD 1.25 billion by 2036. This growth reflects the long-term replacement of secondary desiccant sachets as active packaging becomes the industry standard for oral solid dosages.
What CAGR is projected for the Desiccant-Extruded Oral Solid Packaging Market?
FMI forecasts a CAGR of 7.42% between 2026 and 2036. This rate is determined by the capital expenditure cycles of pharmaceutical manufacturers as they upgrade legacy filling lines to sachet-less automated environments.
Which product type segment leads the market?
The Tubes segment leads with 42.0% share in 2026. This leadership is sustained by the tube’s superior functional integration for effervescent tablets, where co-extruded lining provides the most efficient moisture protection.
Which material type is most dominant?
Desiccant Integrated Polymers lead with 55.0% share. This dominance is driven by the material’s ability to actively scavenge moisture after the package is opened, a performance mechanism that standard passive barriers cannot match.
Which end use drives the most demand?
The Pharmaceuticals segment accounts for 60.0% of the market. This is because pharmaceutical manufacturers face the strictest regulatory stability requirements and the highest financial risks associated with batch degradation.
What is the primary driver for rapid growth?
The move toward high-speed, automated sachet-less filling lines is the non-obvious driver. Manufacturers are adopting integrated desiccant packaging to eliminate the mechanical bottlenecks and downtime caused by secondary desiccant insertion stations.
What is the biggest restraint facing the market?
The primary restraint is the 12-month stability-testing requirement for primary packaging changes. This regulatory gate creates a structural delay in adoption, as pharmaceutical companies cannot switch formats without extensive and time-consuming validation data.
Which country is growing the fastest?
India’s 9.8% CAGR reflects a rapid infrastructure pivot toward export-oriented generic biologics, whereas China’s 8.5% is anchored to domestic diagnostic reagent scaling. India's growth is driven by compliance-led capital refresh cycles required for USFDA/EMA market access, creating a steeper adoption curve than China's internal standardization.
How does the ICH stability protocol impact packaging decisions?
The ICH protocols mandate that oral solids maintain potency under accelerated aging conditions (40°C/75% RH). Integrated desiccant packaging is often the only way to meet these thresholds for moisture-sensitive molecules without using expensive cold-chain logistics.
What is the technical challenge of co-extruded polymers?
The challenge lies in loading high percentages of molecular sieves into a polymer matrix without making the container wall brittle. Only tier-1 vendors with proprietary masterbatch blending technology can achieve the necessary balance of absorption and structural integrity.
How does active scavenging differ from passive barrier protection?
Passive barriers like foil blisters only slow the ingress of moisture. Active scavenging, found in co-extruded packaging, removes moisture already present in the headspace or moisture that enters during the patient’s repeated use of the product.
Why is sachet insertion considered a bottleneck for Pharma 4.0?
In fully automated environments, loose sachets can tear, jam feeding mechanisms, or be missed by sensors, leading to expensive line stops. Integrated desiccant packaging eliminates this station entirely, allowing for seamless high-speed production.
What role does the Drug Master File (DMF) play in vendor selection?
Pharmaceutical companies only select vendors who have a pre-filed DMF with regulatory agencies. This allows the client to reference the vendor’s manufacturing data in their own drug application, significantly shortening the packaging approval process.
How does the nutraceutical market use desiccant-extruded packaging?
Nutraceutical brands use desiccant-lined tubes to protect high-potency probiotics and enzymes that are highly moisture-sensitive. This active protection allows them to guarantee product efficacy to the consumer through the entire use-cycle.
Is there a sustainability benefit to integrated desiccants?
Yes, by integrating active scavenging, manufacturers can often use thinner external polymer walls or eliminate the need for secondary foil over-wrapping. This reduces the total plastic footprint of the pharmaceutical shipment while maintaining safety.
How do point-of-care diagnostic kits use this technology?
Diagnostic kits utilize co-extruded desiccant strips inside the test device housing to prevent moisture from degrading the chemical reagents. This ensures that the test remains accurate even when stored in non-controlled environments.
What is the 'in-use stability period' and why does it matter?
In-use stability refers to the time after a patient first opens a multi-dose container. Integrated desiccants are unique in their ability to protect the remaining tablets from the moisture introduced every time the cap is removed.
How concentrated is the competitive landscape?
The market is highly concentrated among five to seven global leaders due to the specialized polymer science required. These incumbents maintain their share through proprietary formulations and deep partnerships with machinery providers.
What changes are expected in regional adoption by 2036?
By 2036, Southeast Asia and India will transition from low-cost sachet systems to integrated desiccants as their domestic quality regulations align with Western export standards. This will make the Asia-Pacific region the primary volume driver for the market.
Can integrated desiccants be used with biologics?
Yes, as oral biologics become more common, the need for precise micro-environment control increases. Integrated scavenging provides a more stable humidity profile than sachets, which is critical for maintaining the three-dimensional structure of sensitive proteins.
How does FMI validate its market sizing for this sector?
FMI triangulates its data by analyzing desiccant masterbatch production volumes and comparing them against the growth of high-speed filling line shipments that do not feature sachet-insertion stations.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.