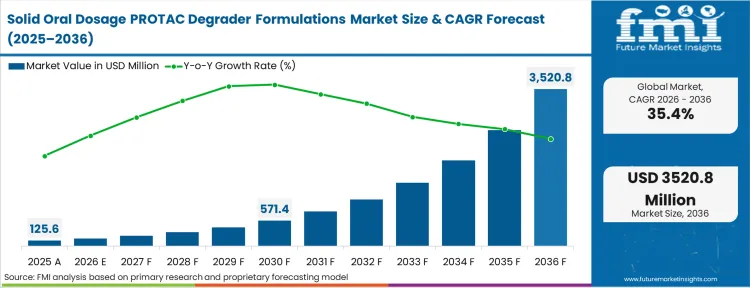
The solid oral dosage PROTAC degrader formulations market was valued at USD 130 million in 2025. Sales are expected to reach USD 170 million in 2026 at a CAGR of 35.40% during the forecast period. Consistent investment propels the valuation to USD 3.49 billion through 2036 as massive molecular weight and poor solubility of these targeted degraders force innovators to adopt advanced solid dispersion technologies to achieve systemic exposure, removing reliance from intravenous routes.
Biopharmaceutical developers are no longer evaluating whether target protein degradation works, but whether it can be delivered outside an infusion clinic. This pivot forces early-stage innovators to integrate oral solid dosage pharmaceutical formulation design during lead optimization rather than pre-clinical scale-up. Relying on traditional crystalline approaches for these complex molecules guarantees clinical failure due to negligible bioavailability. Companies delaying complex formulation integration risk losing their primary competitive advantage to fast-followers who master the solid-state architecture first.
The gate unlocking this progression is the consistent achievement of human oral bioavailability above the ten per cent threshold. Once a specific polymer matrix demonstrates reproducible pharmacokinetic profiles across a patient cohort, scaling drug formulation from clinical to commercial batches becomes an engineering exercise rather than a biological gamble. Contract manufacturers successfully crossing this threshold establish immediate lock-in with asset owners, fundamentally altering the outsourcing landscape.
India is expected to advance at 40.2%, driven by its dense concentration of specialized contract manufacturers capable of scaling complex molecules. China follows closely at 39.6%, while South Korea is estimated to register 35.9%. Japan tracks at 33.5% as its established pharmaceutical sector pivots toward advanced degradation mechanisms. The United States is poised to expand at 32.8%, anchored by primary discovery investments. Germany and the United Kingdom are likely to garner 31.1% and 30.7% respectively. The stark divergence across these regions reflects a transition from Western-led basic research to Asian-dominated complex formulation scale-up.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 170 Million |
| Industry Value (2036) | USD 3.49 Billion |
| CAGR (2026-2036) | 35.40% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The solid oral dosage PROTAC degrader formulations space encompasses specialized matrices, excipient systems, and processing technologies engineered specifically to deliver proteolysis-targeting chimeras through the gastrointestinal tract. Functionally, it is defined by the absolute requirement to overcome the "beyond rule of five" physicochemical properties of bifunctional degraders, converting highly lipophilic and high-molecular-weight active pharmaceutical ingredients into systemically available solid oral formats.
Scope includes amorphous solid dispersions, lipid-based solidified formulations, hot-melt extruded matrices, and spray-dried intermediate powders specifically applied to targeted protein degraders. It covers the formulation services, enabling excipients, specialized coating systems, and complete final dosage forms formatted as tablets, capsules, or multiparticulates. The market specifically tracks revenue generated by high potent oral solid dosage contract manufacturing networks dedicated to these unique bifunctional molecules.
Intravenous, subcutaneous, and other parenteral formulations of PROTACs are strictly excluded, as they bypass the gastrointestinal permeability challenges that define this market. Traditional small-molecule kinase inhibitor formulations are excluded because their lower molecular weights allow conventional crystalline blending. Early-stage unmodified PROTAC APIs without an enabling drug delivery system applied are excluded, as the boundary isolates the formulation phase rather than basic discovery.
Standard liquid-filled capsule approaches fail to deliver the massive drug loading required for PROTAC efficacy, forcing a total displacement of early-stage formulation strategies.
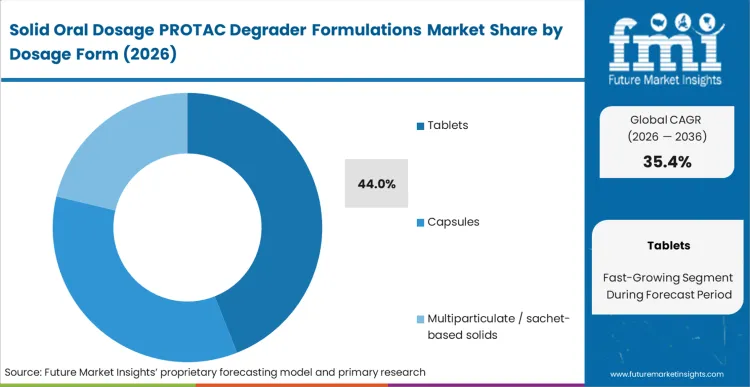
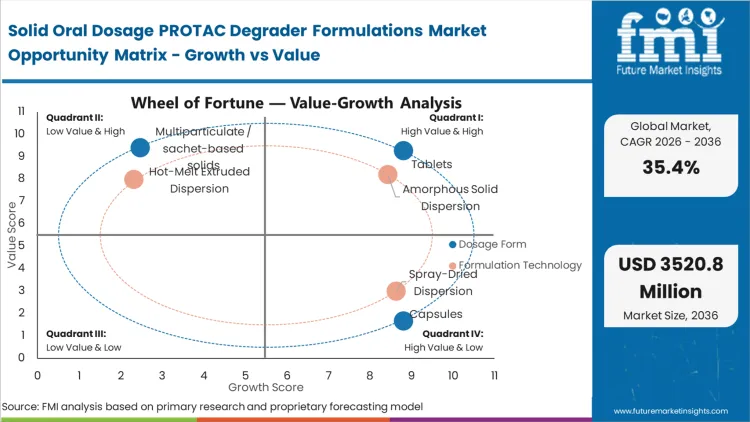
Based on FMI's assessment, tablets command 44.0% of the market because they effectively handle the bulky stabilizing polymers required to keep PROTACs in an amorphous state without requiring patients to swallow an unmanageable number of pills. Innovators leveraging drug delivery technology platforms optimized for tableting avoid critical adherence drop-offs in chronic outpatient settings. Sponsors who resist this transition routinely face costly bridging studies when forced to switch formats for commercial viability, as stabilizer excipients behave differently under high-pressure compression than in loose powder fills.
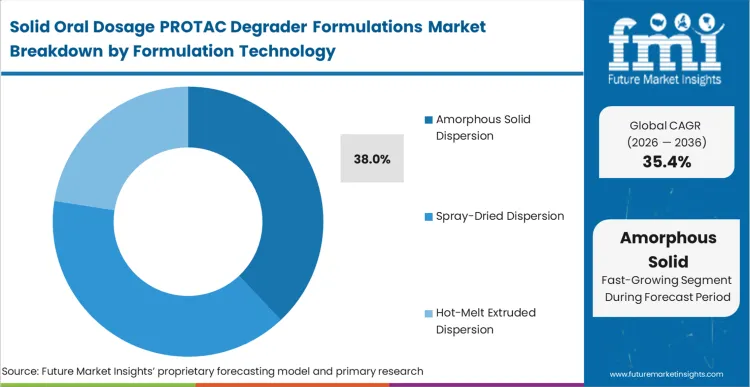
The reason amorphous solid dispersion holds 38.0% of the market is that PROTACs exist in a chemical space where traditional salt formation simply cannot generate the necessary free energy for absorption.
According to FMI's estimates, this technology isolates individual degrader molecules within a polymer matrix, bypassing the catastrophic lattice energy barriers that prevent crystalline uptake. Formulators rely heavily on advanced pharmaceuticals excipients to maintain this high-energy state throughout the product shelf life. The financial penalty for poor physical stability is extreme, requiring total batch destruction if recrystallization occurs during transit. Relying on basic lipid vehicles without advancing to true solid dispersions limits clinical exploration, restricting the asset's total addressable patient population. The integration of permeation enhancers like pharmaceutical excipient snac further amplifies this architectural advantage.
Patient enrollment stalls completely when advanced therapies require daily intravenous infusions for chronic maintenance.
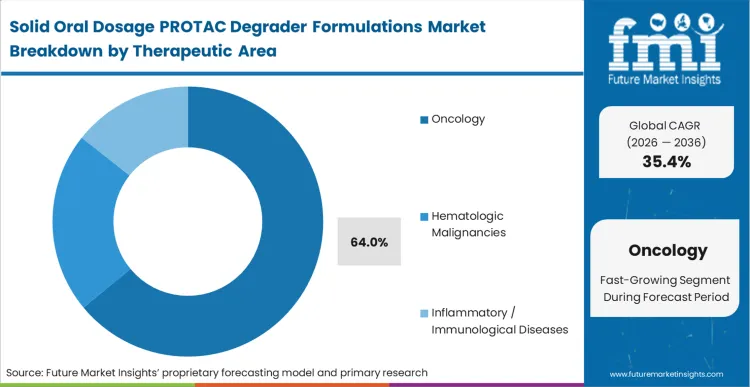
In FMI's view, this commercial consequence forces oncology, which holds 64.0% share, to aggressively pursue solid oral formats. Medical directors treating advanced solid tumors recognize that outpatient oral administration transforms the health economics of degradation therapies by removing infusion clinic bottlenecks. The transition from clinical supervision to home dispensing allows treatment protocols to span months rather than weeks, opening access to vast new patient pools. Asset managers who fail to secure viable pharmaceutical drug delivery solutions for their oncology pipelines face steep valuation discounts during licensing negotiations, as large pharma partners refuse to absorb the commercial limitations of needle-based regimens.
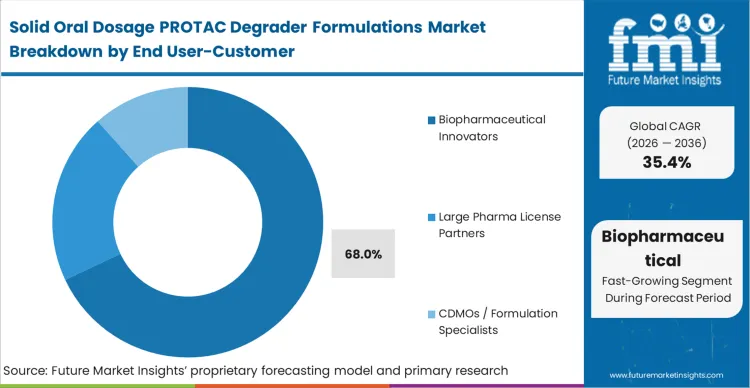
Venture-backed biotechnology firms possess the proprietary degrader chemistry but fundamentally lack the capital-intensive infrastructure to manufacture complex solid dispersions in-house.
As per FMI's projection, biopharmaceutical innovators capture 68.0% share by aggressively outsourcing this tension. These agile firms contract biopharmaceutical contract manufacturing networks the moment a lead molecule is selected, prioritizing speed to the clinic over internal capacity building. The friction lies in securing dedicated spray-drying suites from top-tier partners who typically reserve capacity for established commercial products rather than novel assets moving into early clinical trials. Innovators who hesitate to lock down outsourcing agreements find their clinical timelines entirely gated by external manufacturing waitlists.
The absolute requirement to move targeted degradation from acute inpatient settings to chronic outpatient management compels clinical development directors to qualify solid oral forms. The commercial viability of degrading established targets relies entirely on matching the convenience of existing kinase inhibitors, forcing a rapid evolution in the procurement of complex small molecule api processing capacity. Delaying this transition limits an asset to niche hematology indications, cutting off access to the vastly larger solid tumor market, where outpatient adherence dictates commercial success.
Formulators face a severe operational bottleneck in maintaining the physical stability of amorphous PROTACs under commercial storage conditions. Unlike standard active pharmaceutical ingredients, degraders possess high molecular flexibility that actively promotes recrystallization over time, destroying bioavailability. This instability requires intense environmental controls and highly specialized pharmaceutical solvents during processing. While advanced polymeric wrapping offers a partial solution, the fundamental thermodynamics of these massive molecules restrict the shelf life of early iterations, elevating supply chain risk.
Opportunities in the Solid Oral Dosage PROTAC Degrader Formulations Market
From a regional perspective, the market stretches across North America, Asia Pacific, and Europe, representing more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 40.2% |
| China | 39.6% |
| South Korea | 35.9% |
| Japan | 33.5% |
| United States | 32.8% |
| Germany | 31.1% |
| United Kingdom | 30.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research

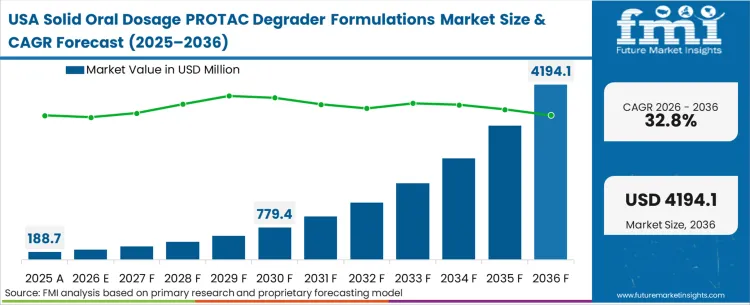
Early-stage discovery capital shapes the adoption pattern across North America, driving the deepest pipeline of novel degrader targets globally. FMI analysts opine that procurement behavior here favors proximity and speed during the initial technology transfer phase, heavily utilizing domestic early phase clinical trial outsourcing hubs. The ecosystem is distinctly weighted toward venture-backed innovators requiring high-touch formulation design rather than pure high-volume commercial production.
The regional dynamic is driven by aggressive investments in high-potency containment infrastructure and complex chemical synthesis capabilities. According to FMI's estimates, local contract manufacturers leverage their established cmo cdmo networks to absorb the scale-up phases that Western innovators cannot afford to run domestically, shifting the center of gravity for commercial volumes.
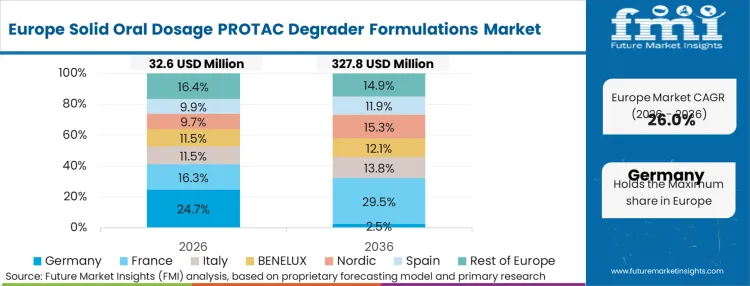
Complex procurement practices and highly fragmented national clinical trial regulations drive the European landscape. In FMI's view, biopharmaceutical sponsors operating here must build formulation strategies capable of passing stringent, diverse stability audits across multiple jurisdictions before initiating multi-center studies.
FMI's report includes extensive coverage of adjacent markets executing similar high-potency solid dispersion strategies. The capacity to securely manage and distribute highly sensitive clinical drug supply chain services fundamentally separates the specialized service providers from generalized pharmaceutical contractors across these advanced regulatory environments.
The competitive landscape is heavily concentrated among a small tier of specialized formulation houses capable of managing extreme potency alongside complex amorphous solid dispersions. Large pharmaceutical companies evaluating degraders do not utilize basic contract manufacturers; they rigorously screen partners for proven experience handling "beyond rule of five" molecules under high containment. The primary variable separating qualified vendors from the broader market is the verifiable demonstration of high-containment spray drying and hot-melt extrusion operating simultaneously under strict current Good Manufacturing Practices.
Incumbents like Pfizer and specialized developers such as Arvinas and C4 Therapeutics possess a deep advantage through their proprietary understanding of PROTAC physical chemistry behavior. They have established robust internal predictive models that map how specific polymer matrices stabilize massive molecular weights. To compete, emerging investigational new drug cdmo partners must build end-to-end analytical suites capable of tracking trace crystallization events in real-time. The ability to integrate advanced clinical trial packaging solutions that protect these moisture-sensitive dispersions is no longer optional but a baseline requirement for entry.
Through 2036, the tension exists between biopharmaceutical innovators demanding dedicated, flexible manufacturing suites and contract organizations requiring guaranteed volumes to justify capital expenditures. Large buyers resist lock-in by validating secondary suppliers early in Phase 2, forcing primary vendors to compete strictly on scale-up yield and advanced drug delivery solutions rather than resting on basic formulation capability. The market is programmed to remain concentrated, as the extreme technical barriers to handling potent solid dispersions prevent generalist manufacturers from participating in commercial-stage production.
| Metric | Value |
|---|---|
| Quantitative Units | USD 170 Million to USD 3.49 Billion, at a CAGR of 35.40% |
| Market Definition | The space encompasses specialized matrices, excipient systems, and processing technologies engineered specifically to convert highly lipophilic, high-molecular-weight proteolysis-targeting chimeras into systemically available solid oral dosage forms. |
| Dosage Form Segmentation | Tablets, Capsules, Multiparticulate / sachet-based solids |
| Formulation Technology Segmentation | Amorphous Solid Dispersion, Spray-Dried Dispersion, Hot-Melt Extruded Dispersion, Lipid-Based Solidified Formulations |
| Therapeutic Area Segmentation | Oncology, Hematologic Malignancies, Inflammatory / Immunological Diseases, Other Indications |
| End User / Customer Type Segmentation | Biopharmaceutical Innovators, Large Pharma License Partners, CDMOs / Formulation Specialists, Academic / Translational Centers |
| Regions Covered | North America, Asia Pacific, Europe |
| Countries Covered | United States, China, India, Germany, United Kingdom, Japan, South Korea, and 40 plus countries |
| Key Companies Profiled | Arvinas, Pfizer, Nurix Therapeutics, C4 Therapeutics, Kymera Therapeutics, Monte Rosa Therapeutics, BeOne Medicines |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted bioprocessing engineers and clinical supply chain managers handling complex molecular scale-ups. Baseline valuation anchored to the progression rate of oral degraders from Phase 1 to Phase 2, validated against R&D expenditures of pure-play targeted protein degradation firms. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Solid Oral Dosage PROTAC Degrader Formulations in 2026?What is the primary restraint?
The market is valued at USD 170 million in 2026. This figure reflects the immediate outsourcing capital deployed by biopharmaceutical innovators moving their leading degradation candidates into Phase 2 trials.
What will it be valued at by 2036?
The market will reach USD 3.49 billion by 2036. This massive expansion signals a change where complex solid dispersions become the industry baseline, entirely replacing early-stage liquid-filled capsule bridges.
What CAGR is projected?
A compound annual growth rate of 35.40% is expected. This aggressive rate maps directly to the anticipated commercial approvals of the first wave of oral degraders, triggering heavy commercial-scale manufacturing contracts.
Which Dosage Form segment leads?
Tablets hold 44.0% share because they provide the necessary compression density. Formulators cannot fit the required high ratio of stabilizing polymer to active ingredient into standard capsules without demanding unmanageable patient pill burdens.
Which Formulation Technology segment leads?
Amorphous Solid Dispersion leads by functionally trapping the massive PROTAC molecules in a high-energy state. Without this specific technology, the molecules crystallize, rendering them completely incapable of crossing the gastrointestinal boundary.
Which Therapeutic Area segment leads?
Oncology dominates with 64.0% share due to the intense pressure to transition targeted cancer therapies to outpatient settings. Intravenous administration severely limits patient enrollment and degrades the commercial value of solid tumor treatments.
What drives rapid growth?
Clinical development directors recognize that advanced degrader therapies cannot compete commercially if they require hospital infusion. The requirement for chronic, at-home administration forces immediate investment in complex formulation services.
What is the primary restraint?
The inherent physical instability of these massive molecules constantly risks recrystallization. This forces manufacturers to maintain intense environmental controls and utilize highly specialized solvent systems, severely limiting available global capacity.
Which country grows fastest?
India expands at 40.2%, outpacing China's 39.6%. The difference stems from India's established dense networks of contract manufacturers actively repurposing high-potency API synthesis suites to handle Western clinical scale-up demands, rather than relying solely on state-backed mega-facilities.
How does the clinical scale-up environment dictate technology choice?
Innovators are forced to select spray drying early because bridging studies from lipid solutions are too costly. Choosing the final commercial architecture during lead optimization prevents catastrophic delays during Phase 3 scale-up.
What specific capability separates qualified formulation vendors?
The ability to seamlessly integrate high-containment API handling with hot-melt extrusion lines. Vendors lacking dedicated suites for highly potent amorphous intermediates are immediately disqualified from commercial requests for proposals.
How do large biopharma companies avoid outsourcing lock-in?
They actively qualify secondary spray-drying facilities the moment an asset clears Phase 2. This creates intense tension, forcing primary vendors to compete fiercely on overall yield rather than resting on initial process development success.
Why are biopharmaceutical innovators dominating the end-user segment?
Venture-backed pure-plays hold the core degrader intellectual property but lack internal manufacturing capacity. They are required to outsource the entirety of their solid dispersion requirements.
How does Amorphous Solid Dispersion compare to lipid formulations?
Lipid-based systems serve as quick bridges for early toxicology studies, but they fail to accommodate high commercial dosing requirements. Amorphous dispersions provide the long-term stability essential for global distribution chains.
What is the hidden cost of handling PROTAC formulations?
The extreme moisture sensitivity of the amorphous state requires specialized packaging and cold-chain-like tracking. These stringent controls add a permanent operational premium to the final drug product lifecycle.
Why is the United States market heavily weighted toward formulation design?
The massive concentration of early discovery capital in US biotech hubs funds iterative prototyping. Clinical supply managers prioritize speed to Phase 1 over bulk commercial production, contracting local boutique formulation labs.
What role do stabilizers play in the final tablet form?
They prevent the "spring and parachute" dissolution profile from crashing. Without perfectly matched polymeric stabilizers, the degrader precipitates out of solution before it can be absorbed in the intestinal tract.
How do European stability requirements alter formulation timelines?
Highly fragmented national regulations force formulators to over-engineer physical stability protocols. A solid dispersion matrix must prove viable across a wider range of climatic testing zones before securing multi-center trial approval.
Why is South Korea's growth trajectory unique?
South Korea is actively transitioning its massive biologics quality systems to handle potent small molecules. The market advances as innovators leverage these hubs for rapid, strictly compliant execution rather than pure cost savings.
What defines a failure in the solid dispersion scale-up phase?
Even minor variations in spray dryer inlet temperatures can induce trace crystallinity. A failure is absolute, requiring the total destruction of the batch, as the clinical bioavailability drops to near zero.
Why are traditional kinase inhibitor formulators struggling with PROTACs?
Kinase inhibitors are small enough to utilize conventional salt forms or simple blending. PROTACs represent a distinct class of "beyond rule of five" chemistry that destroys standard tableting equipment assumptions.
How does the shift to outpatient administration impact procurement?
It forces a total overhaul of the vendor qualification process. Sourcing directors must now evaluate CDMOs based entirely on their proprietary understanding of amorphous state thermodynamics rather than simple tablet pressing capacity.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.