Herbal Medicinal Products Market
The Herbal Medicinal Products Market is segmented by Product Type (Tablets & Capsules, Powders, Extracts, Syrups), Indication (Digestive Health, Respiratory Care, Pain Management, Skin Care), Distribution Channel (Pharmacies, Hospital Pharmacies, Online Stores, Retail Stores), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Herbal Medicinal Products Market Size, Market Forecast and Outlook By FMI
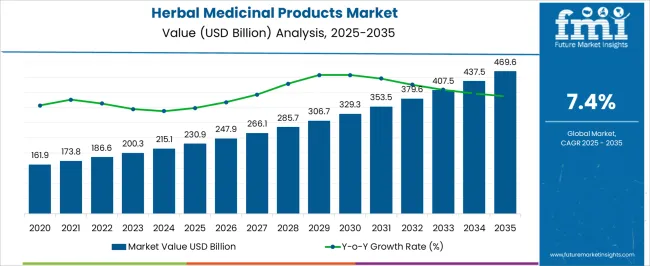
Herbal medicinal products market value is placed at USD 271.1 billion in 2026. FMI forecasts the market to move toward USD 606.3 billion by 2036, with growth placed at 8.4% CAGR from 2026 to 2036. Demand is rising as herbal products shift from loose supplement use toward validated phytomedicines with clearer quality proof.
Summary of the Herbal Medicinal Products Market
- Demand and Growth Drivers
- Clinical validation is increasing trust in plant-based therapeutic products.
- Traditional medicine integration is improving formal healthcare acceptance.
- Quality audits are pushing suppliers toward verified raw material sourcing.
- Digestive care demand is creating repeat use across daily wellness routines.
- Product and Segment View
- Tablets and capsules lead due to precise dosing and easier compliance.
- Digestive health holds the largest indication share due to frequent use.
- Pharmacies lead distribution because buyers trust pharmacist-backed product guidance.
- Extracts and syrups gain use where targeted formulations need clear dosage control.
- Geography and Competitive Outlook
- China leads growth as TCM moves toward stronger regulatory standardization.
- India benefits from Ayush-linked exports and stronger manufacturing upgrades.
- Germany keeps a strong evidence-based phytomedicine base.
- Competition centers on traceability, clinical data, and reliable branded distribution.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Analyst for Healthcare at FMI, opines, “Herbal medicinal product demand is moving toward evidence-backed formulations and consistent dosage formats. Suppliers with verified sourcing, quality documentation, and strong pharmacy access should hold the clearest advantage through 2036.”
- Herbal Medicinal Products Market Value Analysis
- Herbal medicinal products are moving from informal wellness use toward regulated care pathways.
- Value stays concentrated in tablets and capsules due to dosage reliability.
- Pharmacies account for the strongest channel share due to trust-based selection.
- AI-based botanical discovery can improve product precision and evidence depth.
- Stronger traceability will decide supplier strength in regulated markets.
Summary of Herbal Medicinal Products Market
- The herbal medicinal products market comprises plant-derived therapeutic formulations including tablets, capsules, powders, and syrups used for digestive health, respiratory care, and pain management across pharmacy and retail channels.
- Scope boundaries explicitly include standardized phytopharmaceuticals, Ayurvedic medicines, Traditional Chinese Medicine (TCM), and clinically validated herbal extracts, while excluding raw unbranded commodities and home-remedy ingredients sold without therapeutic labeling.
- According to FMI's estimates, the market value will climb from USD 271.1 billion in 2026 to USD 606.3 billion by 2036, compounding at an 8.4% CAGR due to the premiumization of evidence-based botanical drugs.
- Operating models are shifting from simple extraction and bulk supply toward vertically integrated "farm-to-pharmacy" chains that secure verified raw material sources to meet tighter quality audits.
- Competition is concentrating among scaled incumbents like Himalaya Wellness, Dabur, and Swisse who control distribution, alongside biotech challengers like Enveda Biosciences that utilize AI for compound discovery.
- India represents the fastest-growing market at 9.0% CAGR driven by government-backed export incentives, while China remains the largest value share holder supported by deep integration of TCM into its national medical infrastructure.
Herbal Medicinal Products Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 271.1 billion |
| Industry Value (2036) | USD 606.3 billion |
| CAGR (2026-2036) | 8.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why Clinical Validation Is Rewriting Procurement?
Procurement teams in hospital networks and pharmacy chains are increasingly treating herbal medicinal products as regulated pharmaceuticals, creating a compliance-driven gate that favors suppliers with robust clinical dossiers. FMI analysts opine that this shift is forcing manufacturers to abandon volume-based ingredient sourcing in favor of "seed-to-shelf" traceability models that can withstand rigorous safety audits. In 2024, the Ministry of Ayush reported that India's export value of herbal products reached USD 651 million, a figure achieved largely because certified exporters could prove adherence to global quality standards. For distributors, the binding constraint is no longer just price, but the ability to provide documentation that guarantees batch-to-batch consistency and freedom from contaminants. Consequently, the profit pool is migrating toward players who can bundle their products with verified certificate of analysis data, effectively shutting out gray-market suppliers who cannot meet these elevated technical specifications.
How Will Segment Mix Evolve By 2036?
The segmentation landscape, covering product types like tablets and powders, indications such as digestive and respiratory care, and channels ranging from pharmacies to online stores, is pivoting toward formats that offer precise dosing and higher bioavailability. FMI analysts opine that by 2036, the market will heavily favor standardized solid oral dosage forms over loose powders, as consumers and prescribers demand consistent therapeutic outcomes. This transition is supported by supply chain data from Nestlé, which reported 7.2% organic growth in 2023, driven partly by pricing power in their scientifically formulated nutrition portfolios. Value is concentrating in segments that solve chronic lifestyle issues through convenient formats, while traditional raw formats face margin compression due to their commoditized nature. This structural upgrade implies that future growth will rely on converting herbal supplements users into consumers of clinically targeted therapies.
Why Do Tablets & Capsules Dominate Revenue?
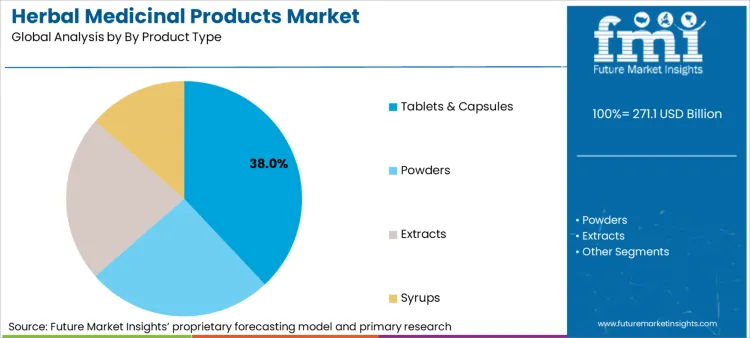
Tablets and capsules capture 38% of the market because they solve the critical "compliance and convenience" equation that modern consumers and pharmacy benefit managers require. This dominant share exists because solid dosage forms allow for precise quantification of active ingredients, addressing the dosage variability risks that historically plagued liquid or powder herbal formats. For manufacturers, the transition to these formats creates a defensible moat, as producing stable, standardized capsules requires significantly higher capital expenditure and formulation expertise than simple grinding. Official filings show that during 2024, China's CDE concluded 1,907 technical reviews for traditional medicines, predominantly for modernized formats that meet strict pharmaceutical standards. This regulatory preference for standardized delivery systems forces smaller players to upgrade their manufacturing capabilities or exit the formal market, further consolidating share among technologically advanced incumbents.
Why Is Digestive Health The Leading Indication?
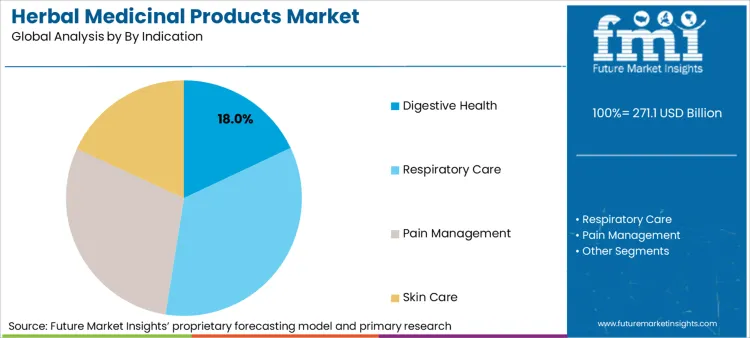
Digestive health applications account for 18% of market value, driven by a global surge in functional gastrointestinal disorders that consumers increasingly self-manage with herbal nutraceuticals solutions. This segment leads because the mechanism of action for herbs like ginger, peppermint, and senna is widely accepted by both traditional practitioners and modern gastroenterologists, lowering the skepticism barrier for adoption. For retailers, this category offers high velocity and repeat purchase rates, as digestive issues are often chronic and require continuous management rather than acute, one-off treatment. Recent corporate filings from Dabur indicate an improvement in Operational Equipment Effectiveness (OEE) to 77.7% in FY 2024-25, a capacity expansion largely targeted at meeting high-volume demand for daily wellness staples like digestive aids. Consequently, shelf space for digestive health products is expanding, pushing out slower-moving categories that lack immediate, tangible symptom relief.
Why Are Pharmacies The Preferred Channel?
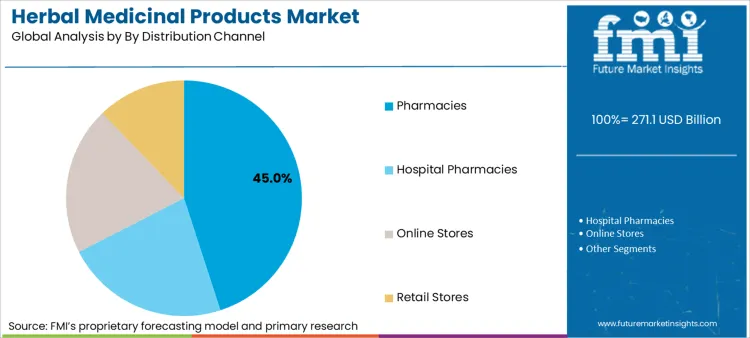
Pharmacies command 45% of distribution because they provide the "trust anchor" and professional guidance that consumers seek when navigating the complex landscape of therapeutic botanical claims. Unlike supermarkets or unverified online marketplaces, pharmacies act as a validation gate where products are vetted for safety and efficacy before reaching the shelf. This channel advantage is amplified by the growing trend of pharmacists recommending herbal personal care products and remedies alongside conventional drugs to manage side effects or treat minor ailments. In August 2024, the American Botanical Council reported a 10% increase in USA herbal supplement sales for the prior year, a growth curve heavily supported by mainstream retail pharmacy chains expanding their wellness assortments. For suppliers, winning in this channel requires investing in pharmacist education programs and packaging that clearly communicates clinical benefits, rather than relying solely on heritage or tradition.
What Role Will AI Play In Discovery?
Artificial intelligence is fundamentally altering the "hit rate" for botanical drug discovery, transforming the sector from trial-and-error tradition into a high-throughput computational science. FMI analysts opine that the integration of AI platforms allows companies to decode complex plant chemistries and identify bioactive compounds that were previously invisible to standard screening methods. This technological leap addresses the industry's historic inability to patent natural products by enabling the creation of novel, standardized extracts with defined mechanisms of action. A clear signal of this shift is the $130 million Series C funding raised by Enveda Biosciences in 2024 to advance its AI-driven pipeline of ten development candidates. For investors, this signals that the botanical ingredients is moving toward a biotech-like valuation model, where intellectual property and clinical data assets outweigh physical inventory. Consequently, legacy players will likely pursue M&A strategies to acquire these AI-native startups, ensuring they remain relevant in an era of precision phytomedicine.
How Are Regional Regulations Shaping Trade?
The global market is bifurcating into two distinct regulatory zones: "modernized traditional" markets in Asia and "clinical botanical" markets in the West. FMI analysts opine that while Asia focuses on integrating traditional medicine into national health coverage to drive volume, Western markets are prioritizing safety data and standardization to drive value. This divergence forces multinational companies to operate dual supply chains, one optimized for mass-market traditional products in the East and another for high-purity, documented extracts in the West. Trade flows are heavily influenced by these standards, with India and China investing billions to upgrade their export quality to meet European and American pharmacopeial requirements. The herb and spice extracts dynamics illustrate this split, as raw material exports increasingly face non-tariff barriers related to purity and sustainability certifications.
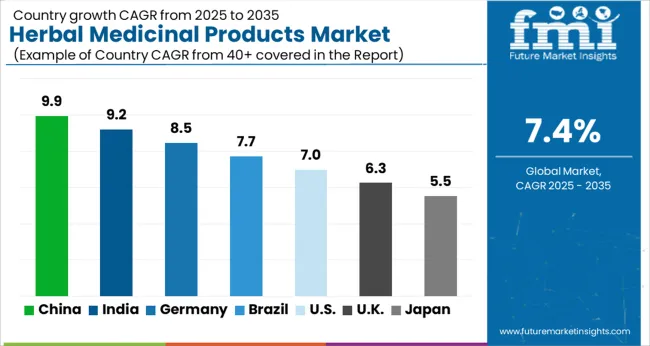
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 9.50% |
| India | 9.00% |
| Brazil | 8.10% |
| USA | 7.80% |
| Germany | 7.20% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Why Is China Aggressively Modernizing TCM?
Sales of herbal medicinal products in China are set to rise at 9.50% CAGR, driven by a state-mandated strategy to elevate Traditional Chinese Medicine (TCM) to the same regulatory status as Western pharmaceuticals. The central government is deploying massive resources to standardize TCM manufacturing processes, aiming to make these products exportable as high-value drugs rather than low-value commodities. Between 2013 and 2024, regulators approved 77 new TCMs for marketing, a clear indicator that the innovation pipeline is active and government-supported. This policy push compels local manufacturers to invest in modern extraction and quality control technologies, fundamentally changing the cost structure of the industry. For global competitors, the implication is significant: China is preparing to flood international markets with clinically validated, standardized herbal medicines that could undercut less sophisticated botanical products.
Why Is India Prioritizing Export Quality?
Demand for herbal medicinal products in India is anticipated to grow at 9.00% CAGR, fueled by the government's "Ayush" initiative which seeks to position Indian Ayurveda as a global healthcare solution. This growth is not merely domestic; it is structurally linked to an export strategy that rewards companies for achieving international quality certifications. Mohit Malhotra, CEO, Dabur India Ltd (March 2025): "In FY 2024-2025, Dabur transitioned to a Double Materiality approach, recognizing that sustainability issues not only affect our business performance (financial materiality) but are also shaped by the impact our operations have on the environment and society (impact materiality)". This strategic shift implies that leading Indian players are proactively aligning with global ESG and quality standards to secure shelf space in regulated Western markets. Consequently, the medicinal and aromatic plant in India is professionalizing, with contract farming replacing wildcrafting to ensure raw material consistency.
Why Is Brazil Cleaning Up Its Backlog?
Herbal medicinal products in Brazil are poised to register a 8.10% CAGR, a trajectory unlocked by the national health regulator's decisive moves to clear administrative bottlenecks. Anvisa has implemented new resolution frameworks specifically designed to accelerate the review of phytotherapeutic products, effectively removing the regulatory uncertainty that previously stalled investment. In 2024, Anvisa reported a backlog of 2,200 processes, but introduced measures to make 50% of these suitable for optimized analysis, signaling a massive release of pent-up commercial energy. For international brands, this regulatory "unclogging" creates an immediate opportunity to enter Latin America's largest market with herbal beauty products and medicinal lines. The implication is that Brazil will serve as the primary entry point and manufacturing hub for the broader South American botanical sector.
Why Is The USA Pivoting To Biotech?
The USA herbal medicinal products industry is projected to expand at 7.80% CAGR, characterized by a distinct shift from retail supplements toward "botanical drugs" developed using biotechnology. Investors and startups are increasingly treating plants as data repositories for drug discovery rather than just ingredients, aiming for FDA approval rather than just DSHEA compliance. Viswa Colluru, Ph.D., CEO, Enveda Biosciences (November 2024): "We developed our platform to rapidly expand access to nature’s chemistry to find therapeutics at roughly four times the speed–and it’s already delivering results in the form of a deep and differentiated pipeline. This funding will help us advance multiple candidates to exciting clinical catalysts in the next year, confirming our guiding vision that life’s chemistry is an excellent source for new medicines". This tech-led approach implies that the future USA market will be defined by high-margin, patent-protected therapies that bridge the gap between adaptogens trends and mainstream medicine.
Why Is Germany Focusing On Evidence?
Herbal medicinal products activity in Germany is projected to expand at 7.20% CAGR, driven by a deeply entrenched medical culture that demands rigorous clinical proof for phytomedicines. Unlike other markets where herbals are viewed as supplements, German consumers and physicians expect these products to perform with the reliability of synthetic drugs. Bionorica SE, a market leader, generated €471 million in revenue in 2023, underscoring the massive scale of the evidence-based phytomedicine sector in the country. This demand for efficacy forces suppliers to invest heavily in "phytoneering", the combination of nature and engineering, to produce extracts with consistent bioactive profiles. Consequently, Germany acts as the global quality benchmark, where success requires passing the highest technical hurdles in the dill oil market and broader botanical extract sectors.
Why Scale and Specs Decide Winners in Competitive Outlook?
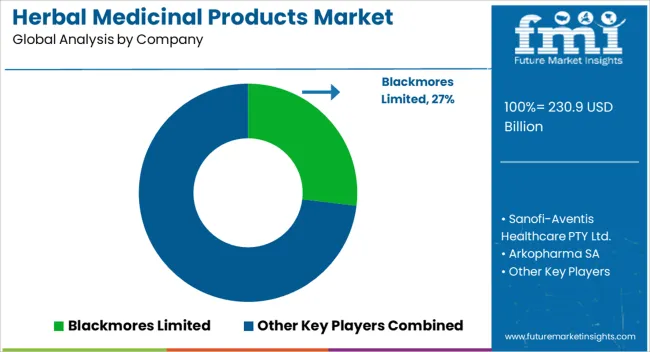
Profit pools are increasingly concentrating around vertically integrated incumbents who can control the entire value chain from "seed to patient," ensuring the traceability that modern procurement demands. FMI analysts opine that companies like Himalaya and Dabur are building defensive moats not just through brand equity, but through massive upstream investments in sustainable agriculture and downstream investments in automated manufacturing. For instance, H&H Group (Swisse's parent) issued USD 120.2 million in senior notes in 2024 to refinance and stabilize its capital structure, illustrating the scale of financial engineering required to maintain global operations. This capital intensity creates a barrier to entry for smaller players who cannot afford the compliance costs associated with the ayurvedic products and treatment. Consequently, the market is bifurcating: scaled giants capture the volume in regulated channels, while fragmented players are pushed into lower-margin, less regulated informal markets.
How Is Technology Disrupting Formulation?
Differentiation is moving from "heritage stories" to "molecular proof," as challenger biotech firms utilize artificial intelligence to unlock new therapeutic values from traditional plants. These new entrants are bypassing traditional retail battles by focusing on high-value, patentable botanical drugs that address specific unmet medical needs. Ayana Bio received a $300,000 grant from the NIH in 2024 to study plant-cell cultivated saffron, validating the shift toward lab-grown botanicals that solve supply chain sustainability issues. This technological wedge allows startups to offer ingredients with guaranteed purity and potency, attributes that traditional agricultural supply chains struggle to deliver consistently. For the ayurvedic supplement, the implication is clear: future leaders will be those who can digitize and bio-engineer nature, turning variability into standardized precision.
Recent Developments:
- In February 2025, Apotex acquired CanPrev, a leading Canadian natural health company specializing in premium herbal and vitamin supplements. This move marks Apotex's entry into the growing health and wellness sector, expanding its portfolio with evidence-based herbal medicinal products focused on natural ingredients for immune support, stress relief, and overall vitality.
- In May 2025, Traditional Medicinals launched the Herbs for All initiative, aimed at expanding access to herbal wellness and education through grants and partnerships with community organizations. This program includes the introduction of new organic herbal teas, supporting underserved communities while reinforcing the brand's commitment to ethical sourcing and plant-based medicinal solutions.
- In September 2025, Traditional Medicinals expanded its wellness offerings with the dual launch of Rosy Mood and Stress Ease Calm teas, the company's first Fair for Life-certified products. These herbal blends feature organic ingredients for mood support and relaxation, highlighting sustainable farming practices and advancing fair trade standards in the herbal medicinal products market.
Key Players in Herbal Medicinal Products Market
- Himalaya Wellness
- Dabur India Ltd
- Patanjali Ayurved
- Nature's Bounty
- Swisse (H&H Group)
- Bionorica SE
- Enveda Biosciences
- Ayana Bio
- Blackmores
- Ricola
- Bio-Botanica
- Gaia Herbs
Market Definition
the herbal medicinal products market refers to the entire commercial ecosystem involved in producing plant‑based therapeutic substances that are processed, packaged, and labeled for medicinal or health‑supporting purposes. This means the market includes every stage, from sourcing medicinal plants, extracting their active compounds, ensuring quality control, and manufacturing finished products, to distributing them through regulated channels. These products are not simply raw herbs; they undergo defined processes that make them suitable for safe, consistent consumer or clinical use.
This scope includes registered herbal medicines (phytopharmaceuticals), traditional systems of medicine (Ayurveda, TCM, Kampo) sold as finished products, and clinically validated botanical extracts used for specific health indications. It covers various dosage forms such as tablets, capsules, liquids, and topical preparations distributed through pharmacies, hospitals, and authorized retail channels.
Excluded from this market are raw, unprocessed herbs sold in bulk without therapeutic packaging, homeopathic preparations (which rely on dilution rather than botanical actives), and culinary herbs or spices used primarily for flavoring rather than health. It also excludes synthetic drugs that mimic natural compounds but do not contain actual plant extracts.
Report Scope
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 271.1 billion |
| Product Type | Tablets & Capsules, Powders, Extracts, Syrups |
| Indication | Digestive Health, Respiratory Care, Pain Management, Skin Care |
| Distribution Channel | Pharmacies, Hospital Pharmacies, Online Stores, Retail Stores |
| Regions covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries covered | China, India, USA, Germany, Brazil, and other key markets |
| Key companies profiled | Himalaya Wellness, Dabur, Patanjali, Nature's Bounty, Swisse |
| Additional attributes | Revenue analysis by segments, adoption trends across settings, regulatory and compliance landscape (as relevant), pricing and reimbursement considerations (when relevant), channel mix economics, supply chain exposure, and competitive positioning analysis |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Herbal Medicinal Products Market by Segments
By Product Type:
- Tablets & Capsules
- Powders
- Extracts
- Syrups
By Indication:
- Digestive Health
- Respiratory Care
- Pain Management
- Skin Care
By Distribution Channel:
- Pharmacies
- Hospital Pharmacies
- Online Stores
- Retail Stores
By Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- Ministry of Ayush (Govt of India). (2024). Export Value of Ayush and Herbal Products Report FY 2023-24.
- Anvisa (Brazilian Health Regulatory Agency). (2024). Management Report and Resolution Summaries.
- Ministry of Health and Family Welfare, Government of India. (2024). National Health Accounts Estimates 2021-22 Update. Government of India.
- National Medical Products Administration (NMPA), China. (2024). Annual Report on Drug Review and Approval Statistics. Government of China.
- European Medicines Agency (EMA). (2024). Annual Report 2024: Herbal Medicinal Products Committee Activities. European Union.
- USA Food and Drug Administration (FDA). (2024). Dietary Supplement Ingredient Advisory List and Regulatory Update. USA Government.
- Pan American Health Organization (PAHO). (2024). Traditional and Complementary Medicine Regulatory Landscape in the Americas. PAHO.
- Department of Health, Republic of South Africa. (2024). Regulations Relating to Complementary Medicines and Health Supplements. Government of South Africa.
- Therapeutic Goods Administration (TGA), Australia. (2024). Listed Complementary Medicines Annual Performance Statistics. Government of Australia.
Frequently Asked Questions
What is the current global market size for the Herbal Medicinal Products market?
The global Herbal Medicinal Products market is valued at USD 271.1 billion in 2026, reflecting strong demand for natural therapeutic alternatives.
What is the projected Compound Annual Growth Rate (CAGR) for the Herbal Medicinal Products market over the next 10 years?
The market is projected to grow at a robust 8.4% CAGR from 2026 to 2036, driven by regulatory modernization and clinical validation.
Which high-priority therapeutic areas or applications are the primary demand drivers?
Digestive Health is the leading indication, capturing 18% of the market as consumers increasingly seek natural remedies for chronic gut issues.
Who are the top 3-5 leading suppliers, and what is the basis of their market leadership?
Himalaya Wellness, Dabur, and Swisse lead the market, leveraging extensive distribution networks and vertically integrated supply chains to ensure quality.
What are the primary market risks, supply chain constraints, or regulatory hurdles affecting the Herbal Medicinal Products market globally?
Key risks include stricter regulatory compliance for purity and efficacy, which can act as a significant barrier for smaller, non-standardized manufacturers.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Tablets & Capsules
- Powders
- Extracts
- Syrups
- Tablets & Capsules
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Digestive Health
- Respiratory Care
- Pain Management
- Skin Care
- Digestive Health
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Pharmacies
- Hospital Pharmacies
- Online Stores
- Retail Stores
- Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Himalaya Wellness
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Dabur
- Patanjali
- Nature's Bounty
- Swisse
- Himalaya Wellness
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by By Indication, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by By Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by By Product Type
- Figure 6: Global Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by By Indication
- Figure 9: Global Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by By Distribution Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by By Product Type
- Figure 26: North America Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by By Indication
- Figure 29: North America Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by By Distribution Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by By Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by By Indication
- Figure 39: Latin America Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by By Distribution Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by By Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by By Indication
- Figure 49: Western Europe Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by By Distribution Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by By Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by By Indication
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by By Distribution Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by By Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by By Indication
- Figure 69: East Asia Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by By Distribution Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by By Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by By Indication
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by By Distribution Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by By Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by By Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by By Indication, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by By Indication, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by By Indication
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by By Distribution Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by By Distribution Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by By Distribution Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

