Hydrolyzed Egg Membrane Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
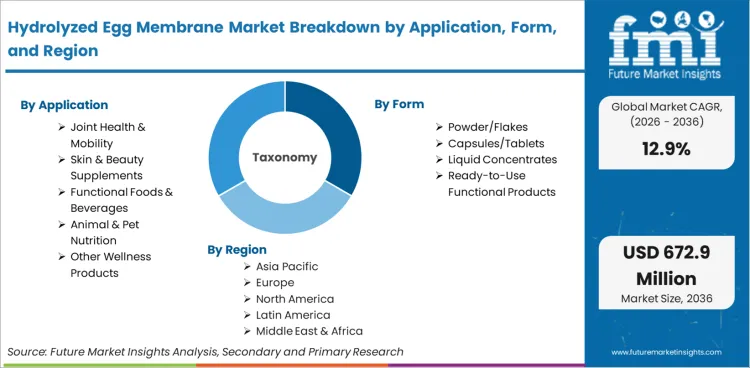
The Hydrolyzed Egg Membrane Market is segmented by Application (Joint Health & Mobility Supplements, Skin & Beauty Supplements, Functional Foods & Beverages, Animal & Pet Nutrition, Other Wellness Products), Form (Powder/Flakes, Capsules/Tablets, Liquid Concentrates, Ready-to-Use Functional Products), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Hydrolyzed Egg Membrane Market Size, Market Forecast and Outlook By FMI
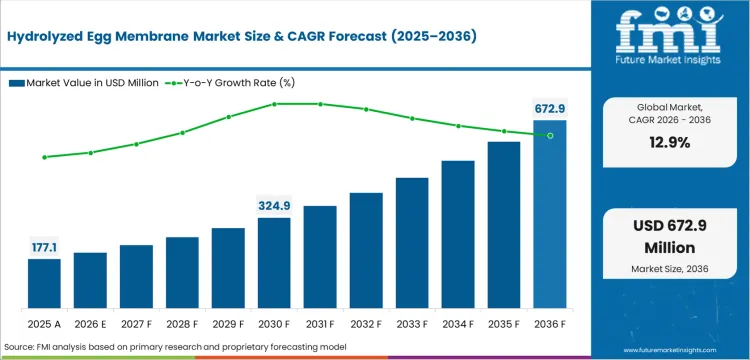
In 2025, the hydrolyzed egg membrane market crossed a valuation of USD 180.0 million and is projected to reach USD 200.0 million in 2026 and USD 675.0 million by 2036, reflecting a CAGR of 12.9%, with momentum tied to clinical adoption curves upgrading joint formulations. As per FMI's projection, efficacy data narrows procurement shortlists toward certified bio-inductive ingredients. Formulators source standardized fractions to meet rising consumer expectations for active nutrition. Buyers prioritize raw material suppliers maintaining strict traceability across global networks.
Absolute dollar growth of USD 475.0 million between 2026 and 2036 indicates a rapid shift from commodity peptides toward specialized regenerative extracts. Raised safety thresholds reset minimum capability within nutricosmetic supply chains. Regulators cap ingestion limits, forcing formulators to specify exact active concentrations before advancing to procurement. A 2025 FDA notice calculated daily background membrane intake at 46 mg/day, establishing baseline safety metrics for novel approvals [1]. Procurement teams transition toward verified compounds with defined biological yields.
Summary of Hydrolyzed Egg Membrane Market
- Hydrolyzed Egg Membrane Market Definition
- The industry covers bioactive nutraceutical ingredients produced through enzymatic or controlled hydrolysis of avian eggshell membranes, commercialized as standardized powders or soluble extracts and utilized primarily in joint health, mobility, and beauty-from-within supplement formulations.
- Demand Drivers in the Market
- Clinical superiority versus traditional collagen sources shifts procurement behavior.
- Broad regulatory approvals under GRAS notifications de-risk enterprise integration.
- Rising pet humanization trends expand veterinary application formats.
- Key Segments Analyzed in the FMI Report
- Form: Powder/flakes (52%) versus capsules/tablets, liquid concentrates, and ready-to-use functional products.
- Application: Joint health & mobility supplements (44% of total volume) versus skin & beauty supplements, functional foods, animal nutrition, and other wellness products.
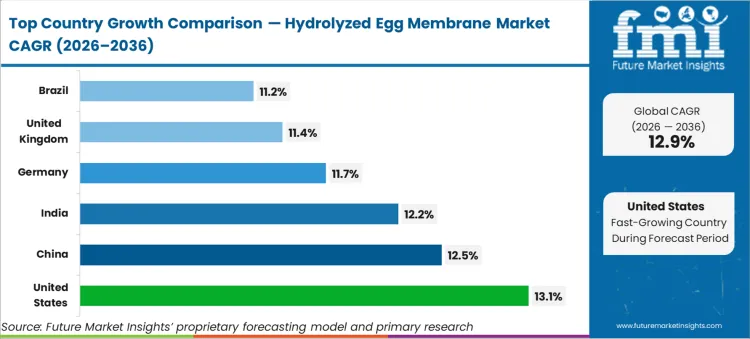
- Geography: High-growth North America and Asia-Pacific (United States 13.1%, China 12.5%, India 12.2%) versus mature Europe and Latin America (Germany 11.7%, United Kingdom 11.4%, Brazil 11.2%).
- Analyst Opinion at FMI
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “In our latest Hydrolyzed Egg Membrane Market evaluation, my team and I have identified category maturation as the next strategic shift. Early growth was driven by novelty and collagen adjacency, but long-term expansion will depend on mechanism clarity and outcome-based positioning. Brands must move beyond generic joint-support messaging and quantify improvements in mobility, stiffness reduction, and recovery timelines. Suppliers that combine standardized extraction, low-allergen validation, and robust clinical communication will secure durable differentiation in a crowded mobility-health landscape.”
- Strategic Implications/Executive Takeaways
- Lock localized feedstock contracts to decouple from global supply volatility.
- Reposition portfolios toward multi-functional nutricosmetics.
- Validate product safety through independent clinical certification bodies.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
As noted by Md Abul Hasnat, CEO of Alchem Enterprise, “We are very thrilled to announce our new partnership with Stratum, a trailblazer in the nutraceutical industry and the best in class when it comes to high-quality, science-backed nutraceutical products. This collaboration represents a powerful synergy between Alchem’s expertise in marketing & distribution and Stratum’s unparalleled innovation in product development and regulatory compliances.” [2]
Country dispersion skews toward regulatory-approved wellness corridors across the United States at 13.1% CAGR, China at 12.5% CAGR, India at 12.2% CAGR, Germany at 11.7% CAGR, the United Kingdom at 11.4% CAGR, and Brazil at 11.2% CAGR. Procurement clusters around framework agreements favoring incumbents with verified clinical safety records. Suppliers align capacity with local tender windows to capture regional wellness spending.
Hydrolyzed Egg Membrane Market Key Takeaways
| Metric | Details |
| Industry Size (2026) | USD 200.0 million |
| Industry Value (2036) | USD 675.0 million |
| CAGR (2026-2036) | 12.9% |
Hydrolyzed Egg Membrane Market Definition
The Hydrolyzed Egg Membrane market represents the global trade of bioactive ingredients derived from avian eggshell membranes through controlled enzymatic or chemical hydrolysis. It measures the commercial exchange of water-soluble membrane extracts standardized for collagen, hyaluronic acid, and glycosaminoglycan content. Market valuation captures ingredient-level supply and finished nutraceutical product revenues where hydrolyzed egg membrane functions as the primary active component across joint health, mobility, and beauty-from-within applications.
Market Inclusions
The scope includes revenues generated from partially and fully hydrolyzed eggshell membrane formats supplied as powders, liquid concentrates, and encapsulated preparations. It accounts for business-to-business ingredient transactions serving dietary supplement brands, functional beverage manufacturers, and animal nutrition formulators. Pre-formulated joint care and nutricosmetic applications containing standardized hydrolyzed membrane inputs are included within revenue calculations.
Market Exclusions
The scope excludes whole intact eggshells and non-hydrolyzed agricultural by-products without bioactive extraction. Topical cosmetic products lacking oral ingestion positioning are not considered. Adjacent protein powders, egg white derivatives, and synthetic collagen substitutes fall outside defined market boundaries.
Hydrolyzed Egg Membrane Market Research Methodology Market
- Primary Research: Direct interviews with extraction facility managers, nutraceutical formulators, and regulatory compliance officers operating across primary supply zones.
- Desk Research: Output reconciliation against USDA production bulletins, GRAS notice filings, and patent registry databases detailing enzymatic protocols.
- Market-Sizing and Forecasting: Bottom-up capacity modelling verified against import-export trade codes for specialized biological extracts.
- Data Validation and Update Cycle: Multi-stage peer review filtering anomalies through strict variance checks prior to finalizing regional growth projections.
Segmental Analysis
Hydrolyzed Egg Membrane Market Analysis by Application
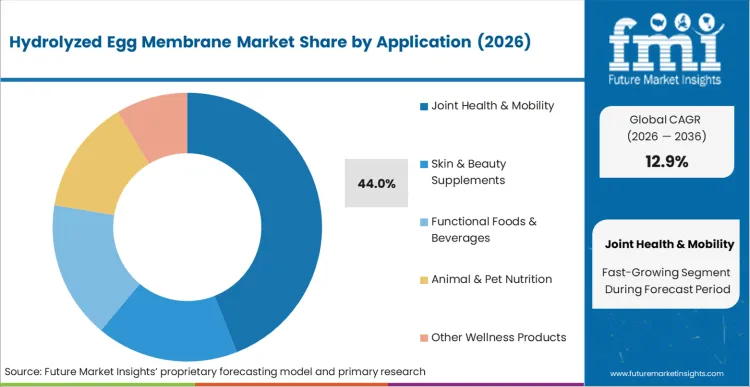
Joint health & mobility supplements command 44% of total market revenue in 2026. According to FMI's estimates, aging demographics mandate preventive biological restoration, expanding patient pools beyond traditional osteoarthritis cases. Formulators specify exact glycosaminoglycan profiles to satisfy clinical endpoints. Buyers integrate pre-validated blends to accelerate product commercialization timelines across eggshell membrane powder networks. Formulators seek high-purity fractions to replace complex multi-component stacks.
- Procurement trigger: Clinical substitution accelerates where single-ingredient formulations replace bulky regimens. Formulators transition purchasing orders toward high-yield extracts following trial validations, heavily leveraging new hydrolyzed egg variants. A 2025 GRAS submission calculated an all-person mean intake of 308 mg/day for proposed food uses [3].
- Regulatory signal: Safety certifications elevate ingredient viability within enterprise portfolios. Manufacturers fund toxicology assessments to clear stringent international customs barriers and secure label claims. Recent EFSA evaluations demonstrated a 2,000 mg/kg bw/day NOEL for membrane hydrolysate [4].
- Supply-side move: Facility expansions stabilize volume availability for major pharmaceutical buyers. Processors invest capital into dedicated extraction lines to eliminate cross-contamination risks. Stratum Nutrition opened a high-capacity production plant in Carthage, Missouri in September 2025 [5].
Hydrolyzed Egg Membrane Market Analysis by Form
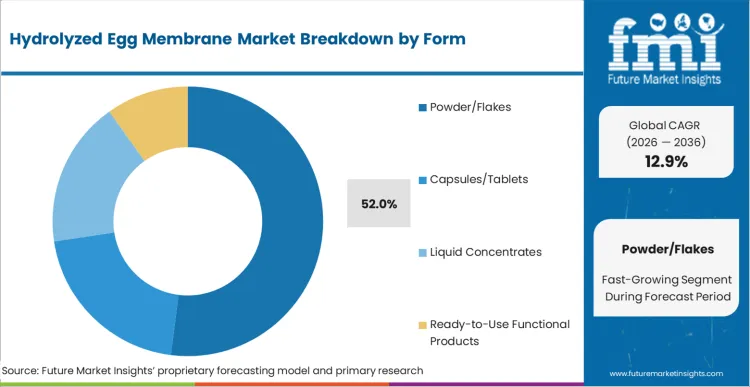
Processing facilities continue to favor powder and flake formats because their low moisture content improves shelf stability and reduces transportation risk. Soluble powders can be easily incorporated into a wide range of delivery systems, including gummies, functional bars, and other fortified products. During early-stage R&D, formulation flexibility often drives ingredient selection, making adaptable formats more attractive. Standardized bulk packaging further streamlines storage and handling, while high-grade egg powder streams ensure consistent active content across production batches, positioning powder and flakes to capture 52% of ingredient demand in 2026.
- Buyer priority: Freight efficiency mandates concentrated active compounds over diluted liquid suspensions. Procurement managers specify low-hygroscopicity grades to prevent caking during cross-border transport. Advanced processing yields up to 44.60 μg/mg of glycosaminoglycans from raw materials [6].
- Technology shift: Refined enzymatic hydrolysis methods lower molecular weights to improve cellular absorption rates. Suppliers recalibrate reaction parameters to preserve native peptide structures. Controlled trials from 2025 proved 450 mg/day interventions significantly increased hair density among aging demographics [7].
- Format conversion: Capsule manufacturers shift lines toward dense powders to maximize active payloads per serving. Operations teams re-tool encapsulation equipment to handle fine biological particulates. NOW Foods launched novel joint support capsules in February 2025 to expand category reach [8].
Hydrolyzed Egg Membrane Market Drivers, Restraints, and Opportunities
Clinical efficacy data drives rapid procurement adoption across functional wellness categories. Formulators replace bulky glucosamine regimens with low-dose biological alternatives to improve patient compliance. Purchasing teams mandate supplier transparency regarding extraction methods to guarantee active compound preservation. Operations managers integrate water-soluble grades to prevent nozzle blockages during continuous beverage production. Documented dermatological improvements validate higher retail price points for premium egg protein extracts. Double-blind studies in 2025 confirmed 300 mg/day of fresh membrane yields superior skin mechanical parameters versus 8,000 mg of hydrolyzed collagen [9].
Raw material supply volatility constrains aggressive global capacity expansions. Extraction facilities depend entirely upon commercial breaking operations for consistent feedstock deliveries. Avian disease outbreaks disrupt poultry inventories, causing cascading material shortages across the liquid egg and shell supply chains. Procurement managers stockpile dried stock to mitigate unexpected quarterly pricing spikes. Manufacturers face severe margin compression when attempting to absorb agricultural inflation. Official USDA data reported a 4% decline in total 2025 egg production, falling to 105 billion units [10].
- Veterinary Expansion: Pet food manufacturers formulate high-value joint treats to capture affluent animal owners. Formulators integrate proven human-grade extracts into premium kibble lines to guarantee safety profiles.
- Nutricosmetic Convergence: Beauty brands combine structural proteins with vitamins to command premium cosmetic valuations. Formulators specify clinically-tested fractions to validate label claims before advancing to clinical trials.
- Geographic Penetration: Regional distribution partnerships unlock localized enterprise accounts across emerging Asian economies. Suppliers establish localized warehousing to reduce cross-border transit times and secure robust distribution channels.
Regional Analysis
Based on the regional analysis, the hydrolyzed egg membrane market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 13.1% |
| China | 12.5% |
| India | 12.2% |
| Germany | 11.7% |
| United Kingdom | 11.4% |
| Brazil | 11.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Hydrolyzed Egg Membrane Market Analysis
North America dictates global innovation parameters through stringent safety protocols and aggressive clinical research funding. Formulators demand comprehensive toxicology data before committing capital to novel active ingredients. Regulatory bodies enforce tight specification limits on residual moisture and microbial counts. Suppliers must navigate complex GRAS notification pathways to legitimize commercial food inclusions. As per FMI's projection, established brand loyalty secures baseline volumes across domestic supply networks. Regional players actively refine specialty egg extractions to meet premium buyer criteria. Post-marketing surveillance data from 2025 revealed an exceptionally low adverse event rate of 2.16 per 1,000,000 servings [11].
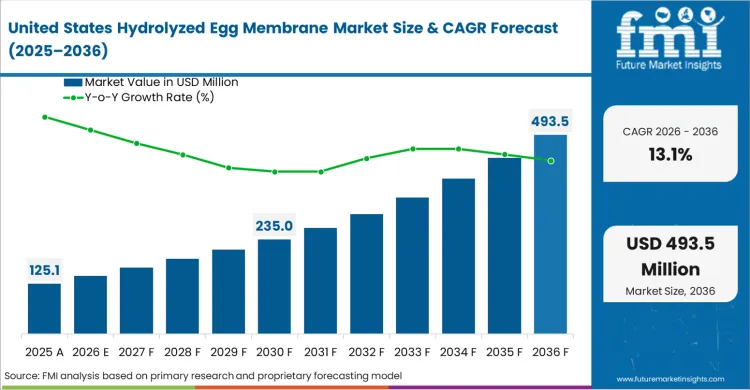
- United States: Demand for hydrolyzed egg membrane in the United States is expected to grow at 13.1% CAGR through 2036, supported by aging demographics seeking preventive alternatives before procurement cycles translate into higher retail volumes. Buyers prioritize ingredients carrying proven safety dossiers against a backdrop of domestic table egg production dropping to 632.2 million dozen in August 2025 [12].
FMI's report includes detailed analysis validating regional purchasing patterns across mature economies like Canada and Mexico. Procurement networks operating in these countries actively expand pet nutrition portfolios using premium structural proteins. Formulators monitor cross-border regulatory harmonization efforts to streamline future product launches.
Europe Hydrolyzed Egg Membrane Market Analysis
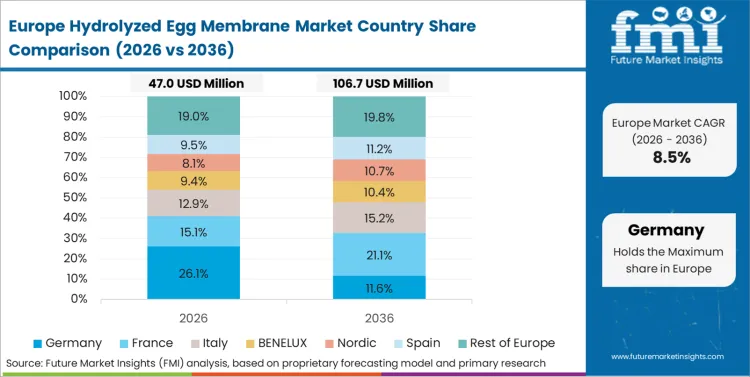
Europe operates as a strict regulatory gatekeeper, elevating minimum purity standards for imported biological compounds. Enterprise buyers require exhaustive EFSA compliance documentation prior to initiating supply tenders. Processing facilities invest heavily in traceable extraction methodologies to satisfy regional sustainability mandates. Procurement teams source verified non-GMO feedstocks to align with premium brand positioning. According to FMI's estimates, transparent supply chains command higher contract valuations. Active egg substitute formulations frequently leverage plant alternatives, forcing avian-derived compounds to compete purely on clinical superiority.
- Germany: Sales in hydrolyzed egg membrane in Germany are projected to rise at 11.7% CAGR from 2026 to 2036 as aging consumer bases seek scientifically validated nutricosmetics, creating a clearer pathway from clinical data to purchase orders. Formulators replace generic collagens with patented membrane fractions following strict EFSA compliance clearances.
- United Kingdom: The United Kingdom hydrolyzed egg membrane is poised to expand at 11.4% CAGR during 2026 to 2036 because an active aging population increases preventative healthcare spending, which lifts utilization rates and justifies formulation upgrades. Retailers allocate shelf space based upon proven clinical endpoints validated by ongoing independent safety audits.
FMI's report includes precise modeling of shifting ingredient specifications across European regulatory jurisdictions like France and Italy. Regional buyers across these zones leverage local clinical trials to validate premium cosmetic claims. Procurement networks negotiate localized warehousing to mitigate cross-border transit delays.
Asia Pacific Hydrolyzed Egg Membrane Market Analysis
Asia Pacific registers aggressive expansion trajectories fueled by rising disposable incomes and shifting consumer health paradigms. Local manufacturers aggressively integrate imported active compounds to upgrade domestic product lines. Distribution networks bypass traditional retail structures using direct-to-consumer digital channels. Processors scale production capacities to capture vast underserved demographic cohorts. FMI analysts opine that localized pricing strategies determine ultimate retail penetration depths. Domestic operators scale plant-based eggs across commodity lines, keeping functional membrane extracts positioned strictly as premium nutraceuticals.
- China: China is expected to rise 12.5% CAGR in functional food demand over 2026 to 2036 as expanding middle-class demographics prioritize active nutrition, tightening the link between domestic health trends and import-led investment. Buyers demand strict batch traceability to guarantee ingredient authenticity against broad food application guidelines.
- India: In India, hydrolyzed egg membrane is forecast to grow at 12.2% CAGR through 2036, with growth anchored in rising awareness of bio-available proteins that converts into measurable retail budget allocation. Formulators pivot toward high-yield ingredients following successful pilot extraction tests.
FMI's report includes extensive mapping of domestic supply chains and formulation preferences across Japan and South Korea. Operators within these borders dictate premium market dynamics through rigorous functional beverage innovations. Suppliers focus capital expenditure on scaling local formulation expertise to support enterprise clients.
Latin America Hydrolyzed Egg Membrane Market Analysis
Latin America scales functional ingredient consumption as multinational brands introduce affordable clinical formats. Regional formulators blend membrane extracts with native botanicals to create unique hybrid products. Supply chains rely heavily on established import corridors to secure standardized biological compounds. Procurement managers balance currency volatility against necessary clinical upgrades. Reliable feedstock availability dictates localized manufacturing viability. As per FMI's projection, consistent quality standards secure long-term vendor contracts across the frozen egg and dried powder import sectors.
- Brazil: Brazil is likely to witness structural protein revenues increase at 11.2% CAGR during 2026 to 2036, given rising consumer interest in active lifestyles and the resulting push for joint-support upgrades. Formulators integrate active ingredients into localized beverage formats while tracking raw material metrics from core agricultural hubs.
FMI's report includes detailed modeling evaluating import dependencies across expanding retail networks within Argentina and Chile. Buyers located in these zones increasingly source premium active nutrition compounds to differentiate domestic portfolios. Market penetration relies upon educating regional consumers regarding specific biological efficacy mechanisms.
Competitive Aligners for Market Players
Market leaders prioritize vertical integration to stabilize volatile biological feedstock pipelines. Processors negotiate multi-year supply contracts directly with major breaking operations to guarantee uninterrupted shell deliveries. Companies invest heavily in continuous enzymatic extraction technologies to lower production unit costs while protecting fragile peptide bonds. Operations managers scale localized processing hubs to reduce regional freight liabilities.
Regulatory compliance acts as a strict competitive barrier limiting new market entrants. Established suppliers fund extensive clinical trial networks to build proprietary safety dossiers for exclusive ingredient marketing. Brands leverage GRAS notifications and EFSA safety approvals to bypass enterprise procurement bottlenecks. Compliance teams conduct rigorous batch testing to verify specific glycosaminoglycan concentrations. Documented clinical superiority validates premium ingredient pricing structures against lower-cost commodity collagens.
Portfolio diversification allows ingredient suppliers to capture adjacent revenue streams beyond traditional joint health. Technical teams formulate specialized grades optimized for liquid dispersion, enabling integration into functional beverages and cosmetics. Marketing departments reposition specific fractions as premium beauty-from-within compounds. Companies engage localized distribution partners to accelerate geographic expansions. Strategic portfolio segmentation maximizes volumetric output across diverse manufacturing client bases while adhering to established clinical safety thresholds.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In November 2026, Stratum Nutrition announced that its clinically validated, upcycled collagen ingredient OVOLUX™ won the 2025 “Collagen Ingredient of the Year” award, recognizing its sustainable, beauty-from-within nutricosmetic innovation. [13]
- In May 2025, IQI Trusted Petfood Ingredients secured an exclusive North American distribution partnership for Ovopet brands, locking down critical supply routes and expanding reach across premium animal nutrition formulations [14].
Key Players in Hydrolyzed Egg Membrane Market
- Stratum Nutrition
- Biova LLC
- Eggnovo SL
- Kewpie Corporation
- Ecovatec Solutions
- Certified Nutraceuticals Inc.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 200.0 million (2026) to USD 675.0 million (2036), at a CAGR of 12.9% |
| Market Definition | Hydrolyzed egg membrane refers to an active nutraceutical ingredient derived from avian eggshells through enzymatic or chemical hydrolysis. |
| Application Segmentation | Joint Health & Mobility Supplements, Skin & Beauty Supplements, Functional Foods & Beverages, Animal & Pet Nutrition, Other Wellness Products |
| Form Segmentation | Powder/Flakes, Capsules/Tablets, Liquid Concentrates, Ready-to-Use Functional Products |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Brazil, Canada, Mexico, Argentina, France, Italy, Japan, South Korea, and 40 plus countries |
| Key Companies Profiled | Stratum Nutrition, Biova LLC, Eggnovo SL, Kewpie Corporation, Ecovatec Solutions, Certified Nutraceuticals Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up capacity modeling validated through hydrolysis plant throughput audits, cross-checked with import–export trade codes for processed egg derivatives to align membrane output volumes with verified global trade flows and supply availability |
Hydrolyzed Egg Membrane Market Analysis by Segments
Application
- Joint Health & Mobility Supplements
- Skin & Beauty Supplements
- Functional Foods & Beverages
- Animal & Pet Nutrition
- Other Wellness Products
Form
- Powder/Flakes
- Capsules/Tablets
- Liquid Concentrates
- Ready-to-Use Functional Products
Region
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] FDA. (2025). GRAS Notice Assessment Background Intake.
- [2] Stratum Nutrition. (2025). Distribution partnership with Alchem Enterprise.
- [3] FDA. (2025). GRAS Notice Assessment Proposed Intake.
- [4] EFSA/PMC. (2025). Toxicology/safety data supporting novel food use.
- [5] Stratum Nutrition. (2025). New Carthage Missouri facility announcement.
- [6] PMC. (2025). GAG extraction yields and upcycling data.
- [7] ClinicalTrials.gov. (2025). RCT hair/skin efficacy data summary.
- [8] WiseGuy Reports. (2025). Industry report referencing NOW Foods launch.
- [9] Frontiers in Nutrition. (2025). Clinical trial results on skin mechanical parameters.
- [10] USDA. (2026). Chicken and Eggs Annual Summary.
- [11] FDA. (2025). Post-marketing surveillance safety data update.
- [12] USDA. (2025). Livestock/Dairy/Poultry Outlook.
- [13] Stratum Nutrition. (2026, November 13). Stratum Nutrition’s OVOLUX™ named 2025 “Collagen Ingredient of the Year” by Beauty Innovation Awards.
- [14] Petfood Industry. (2025). IQI Trusted Petfood Ingredients & Eggnovo partnership.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across mature and emerging functional ingredient economies.
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated extraction capacity benchmarks.
- Growth opportunity mapping across powder, capsule, and liquid forms with emphasis on nutricosmetic transitions.
- Segment and regional revenue forecasts covering joint health, beauty supplements, and pet nutrition applications.
- Competition strategy assessment including integration models, supply survivability positioning, and compliance capability benchmarking.
- Product reformulation and clinical compliance tracking including GRAS and EFSA safety parameters.
- Regulatory impact analysis covering novel food mandates, import thresholds, and export driven specification upgrades.
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use.
Frequently Asked Questions
How large is the demand for Hydrolyzed Egg Membrane in the global market in 2026?
Demand for Hydrolyzed Egg Membrane in the global market is estimated to be valued at USD 200.0 million in 2026.
What will be the market size of Hydrolyzed Egg Membrane in the global market by 2036?
Market size for Hydrolyzed Egg Membrane is projected to reach USD 675.0 million by 2036.
What is the expected demand growth for Hydrolyzed Egg Membrane in the global market between 2026 and 2036?
Demand for Hydrolyzed Egg Membrane in the global market is expected to grow at a CAGR of 12.9% between 2026 and 2036.
Which Application is poised to lead global sales by 2026?
Joint Health & Mobility Supplements are expected to be the dominant application, capturing approximately 44% of global market share in 2026 due to clinical efficacy proving biological restoration.
How significant is the role of Powder/Flakes in driving Hydrolyzed Egg Membrane adoption in 2026?
Powder/Flakes represent a critical segment, projected to hold a substantial 52% share of the total market in 2026 as processing facilities prioritize stable, low-moisture formats for formulation flexibility.
What is driving demand in the United States?
Aging demographics seeking preventive alternatives and procurement cycles mandating ingredients with proven safety dossiers drive growth.
What compliance standards or regulations are referenced for the United States?
FDA GRAS notifications are referenced as key compliance benchmarks for food and supplement inclusions.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 13.1% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Growth is constrained by elevated safety compliance costs and strict EFSA purity standards that reward transparent, vertically integrated suppliers.
What type of demand dominates in Europe?
Replacement-driven demand for premium-grade, clinically-validated regenerative ingredients dominates regional consumption.
What is China growth outlook in this report?
China is projected to expand at a CAGR of 12.5% during 2026 to 2036.
Does the report cover India in its regional analysis?
Yes, India is included within Asia Pacific.
What are the sources referred to for analyzing India?
Company partnership disclosures detailing distribution agreements and localized nutricosmetic formulation trends are cited as primary reference sources.
What is the main demand theme linked to India in its region coverage?
Asian demand is associated with rising awareness of bio-available proteins and a shift toward vegetarian-adjacent compounds.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe.
What is the main Germany related demand theme in its region coverage?
Scientific validation for nutricosmetics and patented membrane fractions upgrading standard collagen formulations is emphasized.
Which product formats or configurations are strategically important for North America supply chains?
Standardized powders and capsules are prioritized due to lower moisture risk and reduced logistics constraints across domestic retail networks.
What is Hydrolyzed Egg Membrane and what is it mainly used for?
Hydrolyzed egg membrane is an active nutraceutical ingredient primarily used in supplements for joint health, skin firmness, and animal nutrition.
What does Hydrolyzed Egg Membrane mean in this report?
Hydrolyzed egg membrane refers to global production, trade, and industrial consumption of enzymatically processed biological eggshell extracts.
What is included in the scope of this Hydrolyzed Egg Membrane report?
Scope covers active membrane extracts by form, key applications such as joint supplements, and retail distribution channels.
What is excluded from the scope of this report?
Intact eggshells, crude agricultural membranes, and adjacent non-membrane protein powders are excluded unless part of specific hybrid blends.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined capacity and adoption assumptions for strategic planning purposes.
How does FMI build and validate the Hydrolyzed Egg Membrane forecast?
Forecast is developed using bottom-up capacity modeling validated through import-export trade codes and clinical trial reviews.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2026 to 2036
- Joint Health & Mobility Supplements
- Skin & Beauty Supplements
- Functional Foods & Beverages
- Animal & Pet Nutrition
- Other Wellness Products
- Joint Health & Mobility Supplements
- Y to o to Y Growth Trend Analysis By Application , 2021 to 2025
- Absolute $ Opportunity Analysis By Application , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder/Flakes
- Capsules/Tablets
- Liquid Concentrates
- Ready-to-Use Functional Products
- Powder/Flakes
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Form
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Form
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Form
- Competition Analysis
- Competition Deep Dive
- Stratum Nutrition
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Biova LLC
- Eggnovo SL
- Kewpie Corporation
- Ecovatec Solutions
- Certified Nutraceuticals Inc.
- Stratum Nutrition
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Application
- Figure 23: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Form
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Application
- Figure 30: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Form
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Application
- Figure 37: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Form
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Application
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Form
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Application
- Figure 51: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Form
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

