Infant Medicine Syringes Market
Infant Medicine Syringes Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Infant Medicine Syringes Market Forecast and Outlook 2026 to 2036
The infant medicine syringes market is valued at USD 651.6 million in 2026 and is projected to reach USD 1,071.5 million by 2036, expanding at a CAGR of 5.1% over the forecast period. Growth quality is being determined less by headline pediatric demand and more by where dosing events actually occur and how concentrated those channels are. With hospitals and NICUs representing a large institutional anchor, volumes tend to be steady but pricing is negotiated, specification-driven, and sensitive to procurement frameworks. Home-care demand expands the addressable base, yet it is more fragmented and influenced by pharmacy bundling practices, caregiver education, and the inclusion of syringes with liquid prescriptions.
Application concentration around oral dosing creates scale advantages for suppliers that can standardize barrel markings, adapters, and safety features across multiple drug formats while keeping unit economics tight. End-use dependency also shapes risk: when demand is tied to clinical protocols and dispensing habits, switching is slower, but supplier qualification and regulatory expectations raise the cost of entry. Between 2026 and 2036, the market rewards manufacturers that secure repeat institutional contracts while maintaining broad pharmacy reach for everyday pediatric dosing.
Quick Stats for Infant Medicine Syringes Market
- Infant Medicine Syringes Market Value (2026): USD 651.6 Million
- Infant Medicine Syringes Market Forecast Value (2036): USD 1,071.5 Million
- Infant Medicine Syringes Market Forecast CAGR 2026 to 2036: 5.1%
- Leading Product Type in Infant Medicine Syringes Market: Oral Dosing Syringes
- Leading End User in Infant Medicine Syringes Market: Hospitals & NICUs
- Key Growth Regions in Infant Medicine Syringes Market: Asia Pacific, Latin America, Middle East & Africa
- Top Key Players in Infant Medicine Syringes Market: Becton Dickinson (BD), B. Braun, Terumo, Nipro, Cardinal Health, Baxter, ICU Medical, Medline Industries, Henke-Sass, Wolf, Vygon
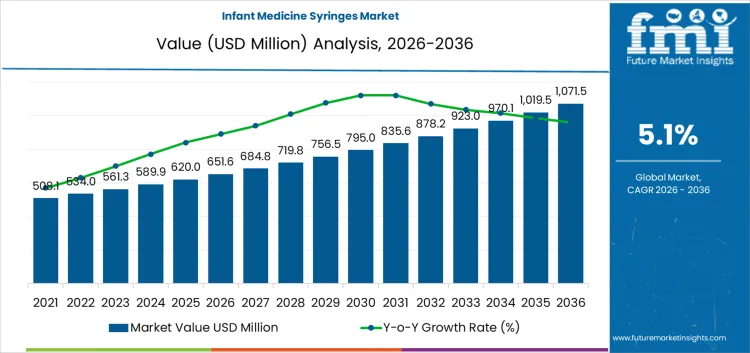
Infant Medicine Syringes Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 651.6 Million |
| Forecast Value (2036) | USD 1,071.5 Million |
| Forecast CAGR 2026 to 2036 | 5.1% |
How Is the Global Infant Medicine Syringes Market Advancing?
The global infant medicine syringes market is advancing at a steady pace, supported by increasing emphasis on accurate medication dosing and safe administration practices in pediatric care. Infant medicine syringes are widely used for delivering liquid medications to infants, ensuring precise dosage control and reducing the risk of dosing errors compared to traditional measuring tools.
A key driver supporting market growth is the growing awareness among caregivers and healthcare professionals regarding the importance of dosing accuracy for infant medications. Pediatric guidelines increasingly recommend the use of oral dosing syringes due to their clear measurement markings, ease of handling, and compatibility with commonly prescribed liquid formulations. These factors are improving caregiver confidence and adherence to prescribed dosing regimens.
Ongoing improvements in syringe design and materials are further supporting market adoption. Enhanced plunger smoothness, improved tip sealing, and the use of medical-grade, BPA-free materials are increasing product reliability and safety. In addition, wider availability through pharmacies, hospitals, and online retail channels is expanding market reach. As pediatric healthcare continues to focus on medication safety and ease of use, the infant medicine syringes market is expected to maintain stable growth over the forecast period.
What Is the Segment-Wise Overview of the Infant Medicine Syringes Market?
The infant medicine syringes market is driven by demand for accurate, safe, and easy-to-use dosing solutions across pediatric care settings. Oral dosing syringes lead with a 54% share, reflecting their widespread use for administering liquid medications to infants due to precise measurement, needle-free safety, and caregiver convenience. On the end-user side, hospitals and NICUs account for 38% of demand, supported by high patient volumes and stringent dosing accuracy requirements in neonatal and infant care. These settings prioritize sterile, standardized syringes to ensure medication safety for vulnerable patients. Together, the dominance of oral dosing syringes and institutional healthcare use highlights a market focused on dosing precision, patient safety, and consistent clinical performance.
Why Do Oral Dosing Syringes Lead the Infant Medicine Syringes Market?
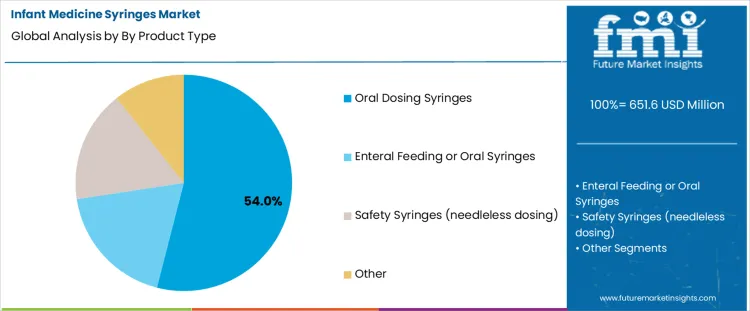
Oral dosing syringes account for 54% of the infant medicine syringes market due to their accuracy, ease of use, and suitability for administering liquid medications to infants. These syringes allow precise measurement of small doses, which is critical in pediatric care where dosing errors can pose significant risks. Oral dosing syringes are widely used for antibiotics, antipyretics, and nutritional supplements, both in clinical settings and at home. Their needle-free design improves safety and reduces discomfort for infants, while clear graduations support caregiver confidence. Compatibility with standard medicine bottles and dispensing adapters further enhances convenience. As pediatric healthcare emphasizes dosing accuracy and safe administration, oral dosing syringes continue to dominate product demand.
Why Do Hospitals and NICUs Dominate End-User Demand in the Infant Medicine Syringes Market?
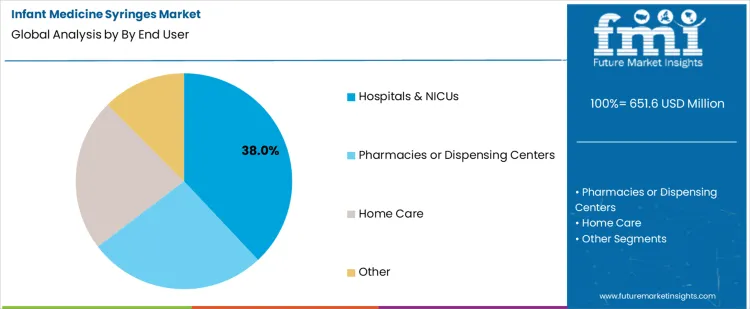
Hospitals and NICUs represent 38% of end-user demand in the infant medicine syringes market, driven by high patient volumes and strict clinical protocols for neonatal and infant care. These settings require reliable, sterile, and accurately calibrated syringes to administer medications, nutritional supplements, and oral solutions to vulnerable patients. In NICUs, precise dosing is essential due to low body weights and sensitive physiological conditions. Hospitals also prioritize standardized medical devices that comply with regulatory and safety requirements, reinforcing preference for clinically validated syringe products. Continuous demand from inpatient pediatric wards and neonatal units sustains large procurement volumes. As institutional focus on medication safety and neonatal care quality remains high, hospitals and NICUs continue to be the leading end-user segment.
What Are the Key Dynamics for the Infant Medicine Syringes Market?
The infant medicine syringes market is driven by the critical need for accurate, safe, and easy-to-use medication delivery tools for infants and young children. Infant medicine syringes are specifically designed to provide precise dosing of liquid medications, reducing the risk of dosing errors and improving treatment outcomes. Key market dynamics include rising pediatric healthcare visits, increased focus on caregiver and healthcare professional education about safe dosing practices, and expanding availability of pediatric pharmaceutical formulations that require precise delivery. Innovations in syringe ergonomics and measurement accuracy are also encouraging greater adoption.
Why Is the Infant Medicine Syringes Market Growing?
The market is growing as caregivers and healthcare providers emphasize accurate dosing to avoid under- or overdosing in infant patients. Increasing incidence of common childhood illnesses and chronic conditions that require liquid medication support demand for dedicated dosing devices. Enhanced access to healthcare services and rising awareness about proper medicine administration techniques are contributing to broader use of infant-specific syringes. Product features such as clear gradation markings, soft safety tips, and ergonomically designed barrels improve usability and caregiver confidence.
How Are the Key Drivers Shaping the Infant Medicine Syringes Market?
Key drivers shaping the market include the expanding pediatric population and heightened concern about medication safety in early childhood. Guidelines and recommendations from healthcare agencies and professional associations promote the use of purpose-built dosing devices, further encouraging adoption. Partnerships between pharmaceutical manufacturers, healthcare providers, and pharmacies to distribute dosing syringes with liquid medications enhance market reach. Continuous product development such as syringes with anti-slip grips, tamper-resistant designs, and compatibility with a wide range of bottle types is also improving user experience and fueling demand.
What Is the Growth Outlook for the Infant Medicine Syringes Market by Country?
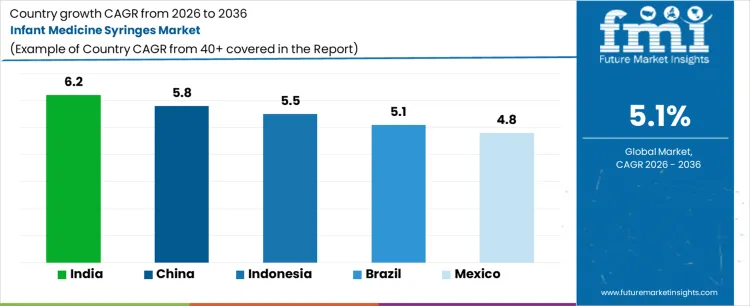
| Country | CAGR (%) |
|---|---|
| India | 6.2% |
| China | 5.8% |
| Indonesia | 5.5% |
| Brazil | 5.1% |
| Mexico | 4.8% |
The infant medicine syringes market is expanding steadily across emerging and developing regions as pediatric healthcare access improves. India leads with a 6.2% CAGR, driven by high birth rates and expanding child healthcare programs. China follows at 5.8%, supported by improved healthcare infrastructure and rising awareness of dosing accuracy. Indonesia grows at 5.5%, benefiting from public health initiatives and increasing healthcare access. Brazil expands at 5.1%, driven by growing pediatric services and pharmacy networks. Mexico grows at 4.8%, supported by steady improvements in child healthcare coverage. As accurate infant medication administration becomes increasingly important, demand for medicine syringes is expected to rise across these regions.
What Is Driving the Growth of the Infant Medicine Syringes Market in India?
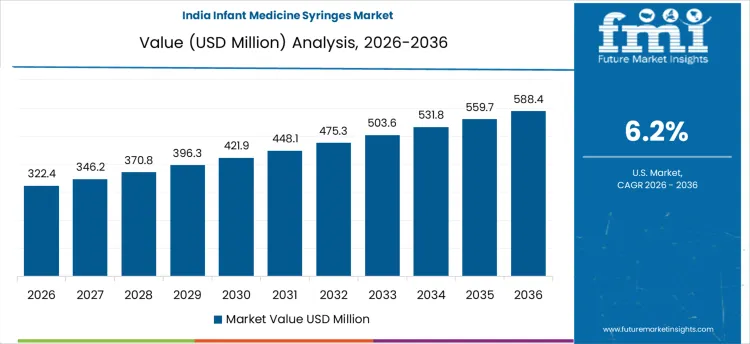
The infant medicine syringes market in India is growing at a CAGR of 6.2%, supported by high birth rates and expanding pediatric healthcare access. Increasing awareness among parents and caregivers about accurate infant dosing is encouraging the use of medicine syringes instead of household spoons. Growth in pediatric prescriptions, vaccination programs, and treatment of common childhood illnesses is driving steady consumption. Expansion of hospitals, clinics, and retail pharmacy chains across urban and semi-urban areas is improving product availability. Government-led maternal and child healthcare initiatives are strengthening early diagnosis and treatment coverage, indirectly supporting syringe demand. Manufacturers are also introducing BPA-free, disposable, and easy-to-read syringe designs to improve safety and hygiene. Rising focus on infant safety, combined with improving healthcare penetration in rural regions, is expected to sustain long-term demand for infant medicine syringes across India.
How Is the Infant Medicine Syringes Market Expanding in China?
The infant medicine syringes market in China is expanding at a CAGR of 5.8%, driven by rising healthcare spending and growing focus on pediatric treatment accuracy. Increased use of liquid formulations for infant medicines is supporting demand for precise dosing devices such as oral syringes. China’s extensive hospital network and strong pharmaceutical manufacturing base enable widespread distribution of infant healthcare products. Parents are increasingly aware of dosing errors and medication safety, leading to higher preference for calibrated syringes. Improvements in product quality standards and clearer measurement markings are also influencing purchasing decisions. Growth of e-commerce and retail pharmacy platforms is further enhancing accessibility. In addition, urbanization and improved healthcare literacy are strengthening demand across cities and emerging regions. As pediatric care quality continues to improve, adoption of infant medicine syringes in China is expected to grow steadily.
What Is Supporting Growth of the Infant Medicine Syringes Market in Indonesia?
The infant medicine syringes market in Indonesia is growing at a CAGR of 5.5%, supported by a growing infant population and gradual expansion of healthcare infrastructure. Increased emphasis on vaccination programs and treatment of childhood illnesses is driving demand for liquid medicines and accurate dosing tools. Rising awareness among parents regarding safe medicine administration is encouraging adoption of oral syringes. Expansion of primary healthcare centers and improved access to pharmacies are supporting product availability, particularly in urban areas. Public health initiatives and international healthcare support programs focused on maternal and child health are further increasing utilization. Cost-effective and disposable syringe designs are important for adoption across diverse income groups. As healthcare access improves and education around infant care strengthens, demand for infant medicine syringes in Indonesia is expected to rise consistently.
How Is the Infant Medicine Syringes Market Developing in Brazil?
The Infant Medicine Syringes Market in Brazil is expanding at a CAGR of 5.1%, driven by increased investment in public healthcare and pediatric services. Government healthcare programs and hospital networks distribute large volumes of infant medicines, increasing demand for reliable dosing devices. Parents and healthcare professionals increasingly prefer medicine syringes to reduce dosing inaccuracies and improve treatment outcomes. Expansion of retail pharmacy chains and improved distribution networks are supporting steady product availability across regions. Rising awareness of hygiene and infection control is encouraging the use of disposable syringe formats. In addition, improved access to pediatric care in underserved areas is increasing healthcare utilization. As Brazil continues to strengthen child healthcare delivery and improve medication safety practices, demand for infant medicine syringes is expected to grow gradually.
How Is the Infant Medicine Syringes Market Expanding in Mexico?
The infant medicine syringes market in Mexico is growing at a CAGR of 4.8%, supported by steady improvements in pediatric healthcare access and public health programs. Expansion of vaccination initiatives and infant treatment protocols is increasing use of liquid medicines that require accurate dosing. Caregivers are increasingly adopting medicine syringes due to ease of use and better dosing control compared to traditional methods. Growth of pharmacy chains and healthcare clinics in urban regions is improving product availability. Educational efforts by healthcare professionals are also raising awareness about correct infant medication practices. While affordability remains important, availability of low-cost syringe options supports wider adoption. As infant healthcare awareness continues to improve, demand for infant medicine syringes in Mexico is expected to grow at a stable pace.
How Are Companies Competing in the Infant Medicine Syringes Market?
Competition in the infant medicine syringes market is driven by dosing accuracy, safety features, material quality, and regulatory compliance for neonatal and pediatric use. Infant syringes must ensure precise low-volume dosing, smooth plunger action, and minimal risk of leakage or misadministration. As hospitals, clinics, and home caregivers prioritize medication safety for newborns and infants, manufacturers differentiate through clear graduation markings, oral-specific designs, latex-free materials, and compatibility with feeding adapters and oral dispensers.
Becton Dickinson (BD), B. Braun, and Terumo compete through clinical-grade design standards and strong hospital presence. BD emphasizes highly accurate, low-dead-space syringe designs with clear markings and consistent plunger resistance, supporting safe medication delivery in neonatal and pediatric care settings. B. Braun differentiates through quality-focused manufacturing, smooth-glide plungers, and syringe designs aligned with medication safety protocols in hospitals. Terumo competes by leveraging precision engineering and polymer expertise to deliver syringes with reliable dosing control and patient-safe materials, widely used in clinical and institutional environments.
Nipro, Cardinal Health, Baxter, ICU Medical, Medline Industries, Henke-Sass, Wolf, and Vygon compete through portfolio breadth, supply reliability, and application-specific designs. Nipro focuses on cost-effective, high-quality syringes suitable for large-volume healthcare systems. Cardinal Health and Medline Industries emphasize dependable supply chains and private-label offerings for hospitals and pharmacies. Baxter and ICU Medical integrate infant syringes into broader medication delivery and safety ecosystems. Henke-Sass, Wolf and Vygon differentiate through specialized pediatric and neonatal syringe designs tailored for precise oral dosing. Across the market, competitive advantage is shaped by dosing precision, safety-oriented design, regulatory compliance, and consistent availability across clinical and home-care channels.
Key Players in Infant Medicine Syringes Market
- Becton Dickinson (BD)
- B. Braun
- Terumo
- Nipro
- Cardinal Health
- Baxter
- ICU Medical
- Medline Industries
- Henke-Sass, Wolf
- Vygon
Scope of Report
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Million |
| Product Type | Oral Dosing Syringes, Enteral Feeding or Oral Syringes, Safety Syringes (Needleless Dosing), Other |
| End User | Hospitals & NICUs, Pharmacies or Dispensing Centers, Home Care, Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Becton Dickinson (BD), B. Braun, Terumo, Nipro, Cardinal Health, Baxter, ICU Medical, Medline Industries, Henke-Sass, Wolf, Vygon |
| Additional Attributes | Dollar sales by product type and end user; regional market size and forecast analysis; growth outlook across major regions; adoption trends for infant medicine syringes in clinical and home-care settings; assessment of dosing accuracy requirements, safety feature penetration, and demand patterns across hospitals, pharmacies, and home-based pediatric care. |
Infant Medicine Syringes Market by Key Segments
By Product Type
- Oral Dosing Syringes
- Enteral Feeding or Oral Syringes
- Safety Syringes (needleless dosing)
- Other
By End User
- Hospitals & NICUs
- Pharmacies or Dispensing Centers
- Home Care
- Other
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization (WHO). (2023). WHO Model Formulary for Children 2023: Guidance on Safe Oral Medication Dosing in Infants and Young Children. World Health Organization.
- U.S. Food and Drug Administration (FDA). (2024). Best Practices for Dosing Devices for Oral Liquid Medications: Guidance for Industry. U.S. Department of Health and Human Services.
- European Medicines Agency (EMA). (2023). Guideline on Pharmaceutical Development of Medicines for Paediatric Use (EMA/CHMP/QWP/805880/2012 Rev. 2). European Medicines Agency.
- International Organization for Standardization (ISO). (2024). ISO 7886-4:2024 — Sterile Hypodermic Syringes for Single Use – Part 4: Syringes for Special Medical Purposes, Including Oral and Enteral Dosing. International Organization for Standardization.
- United Nations Children’s Fund (UNICEF). (2023). Child Health Supplies and Safe Medication Administration: Technical Note on Oral Dosing Syringes for Pediatric Use. UNICEF Supply Division.
Frequently Asked Questions
How big is the infant medicine syringes market in 2026?
The global infant medicine syringes market is estimated to be valued at USD 651.6 million in 2026.
What will be the size of infant medicine syringes market in 2036?
The market size for the infant medicine syringes market is projected to reach USD 1,071.5 million by 2036.
How much will be the infant medicine syringes market growth between 2026 and 2036?
The infant medicine syringes market is expected to grow at a 5.1% CAGR between 2026 and 2036.
What are the key product types in the infant medicine syringes market?
The key product types in infant medicine syringes market are oral dosing syringes, enteral feeding or oral syringes, safety syringes (needleless dosing) and other.
Which by end user segment to contribute significant share in the infant medicine syringes market in 2026?
In terms of by end user, hospitals & nicus segment to command 38.0% share in the infant medicine syringes market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By Product Type , 2026 to 2036
- Oral Dosing Syringes
- Enteral Feeding or Oral Syringes
- Safety Syringes (needleless dosing)
- Other
- Oral Dosing Syringes
- Y to o to Y Growth Trend Analysis By By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By By End User, 2026 to 2036
- Hospitals & NICUs
- Pharmacies or Dispensing Centers
- Home Care
- Other
- Hospitals & NICUs
- Y to o to Y Growth Trend Analysis By By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By By Product Type
- By By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By By Product Type
- By By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By By Product Type
- By By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By By Product Type
- By By End User
- Competition Analysis
- Competition Deep Dive
- B. Braun
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Terumo
- Nipro
- Cardinal Health
- Baxter
- ICU Medical
- Medline Industries
- Henke-Sass, Wolf
- Vygon
- B. Braun
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by By End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by By Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by By End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by By Product Type
- Figure 6: Global Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by By End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by By Product Type
- Figure 23: North America Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by By End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by By Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by By End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by By Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by By End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by By Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by By End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by By Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by By End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by By Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by By End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by By Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by By Product Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by By Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by By End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by By End User, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by By End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

